Abstract
Floodplain wetlands remain important habitats for most macrophytes, macroinvertebrates, birds, fish, amphibians, wildlife and in particular large mammals. They are dynamic in nature and provide many ecosystem services even to humans. The present study was undertaken to assess water and sediment chemistry as drivers of macroinvertebrates and fish communities in Makuleke floodplain wetlands in north Kruger National Park, South Africa. Water, sediments, macroinvertebrates and fish samples were collected across different hydroperiods (i.e., Low water period and high water period) from six floodplain pans. Macroinvertebrates were dominated by (Notonectidae, Libellulidae, Gerridae, Chironomidae larvae, Belostomatidae, gomphidae, dytiscidae and Baetidae, while fish were dominated byTilapia sparminii, Gambusia affinis, Coptodon rendali, Oreochromis hybrid, Oreochromis mossambicus, Enteromius palludinosus and Clarais gariepinus. Generally, fish and macroinvertebrate abundances and diversity were elevated during high water levels as compared to low water levels, suggesting that hydroperiod plays a significant role in structuring aquatic faunal communities. Redundancy and canonical– correlation analysis identified salinity, TDS (water) and Zn, C and B concentrations (sediment) as the major drivers of macroinvertebrate community structure, while pH, TDS (water), and K, Ca and Mg concetrations (sediment) were the major drivers of fish communities. In addition, pelagic chlorophyll –a was strongly positively associated with fish, particularly Gambusia affinis, during the high water level period. The results of this study provide important baseline information on the ecology of the Makuleke pans.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Floodplain wetlands pans are considered important ecosystems hosting fish, macroinvertebrate, plant and microbial communities which cumulatively facilitate the provision of various ecosystem services (Acosta et al. 2020; Dube et al. 2020; Petsch et al. 2022). Within these temporal floodplain pans, macroinvertebrates are good predictors of wetland health and potential to sustain other components of biodiversity, such as fish and birds (Kärnä et al. 2019; Guareschi et al. 2015). As such, macroinvertebrates and fish can be used as bio–monitoring agents for water quality and ecological status of aquatic environments (Kaban et al. 2018; Pandiarajan et al. 2019; Kunwar et al. 2022). Environmental conditions such as water temperature, salinity, and habitat structure all determine which types of fish and macroinvertebrate taxa occur in floodplain wetlands systems. Less sensitive macroinvertebrate taxa in such habitats include chironomid midges and oligochaete worms, while less sensitive fish taxa include catfish (Clarias gariepinus) and numerous cichlid species among others (Kanu et al. 2022; Raphahlelo et al. 2022; Edeh et al. 2023).
Numerous factors operating at multiple spatial scales influence the distribution and abundance of fauna in floodplain pans systems (Nhiwatiwa et al. 2017; Sarkar et al. 2020). Predator–prey dynamics have been identified as an important biological driver of fish assemblages, but competition for food resources also plays a significant role (Svanbäck et al. 2008; Munyai et al., 2022). These biotic interactions operate together with environmental conditions such as water and sediment chemistry to drive macroinvertebrates communities in aquatic environments (Florencio et al. 2014; Dalu et al. 2017; Farooq et al. 2022). Numerous studies (e.g., Ward et al. 1999; Frisch et al., 2005; Pilière et al. 2014) have demonstrated that aquatic organisms’ groups such as zooplankton, macroinvertebrates, fish, amphibians, and macrophytes, respond differently to changes in ecological and hydrological conditions of aquatic ecosystems.
The hydrological regime of a river floodplain ecosystem plays a significant role in determining habitat connectivity between the river channel and its floodplain, with implications for the system’s ecological functioning and biodiversity patterns (Nhiwatiwa et al. 2011; Hayes et al. 2018; Dube at al., 2019). Floodplain pans strongly rely on natural patterns such as the hydrological connection with the river which can provide water through surface or subsurface flow, sustainig biota and ecosystem processes such as organic matter decomposition and nutrient cycling (Heath and Plater 2010; Smit et al. 2016; Gordon et al. 2020). According to Dube et al. (2019), the exchange of organisms in river–floodplain ecosystems is heavily influenced by the distance of the floodplain from the river and the existence of hydrological linkages between the river and the floodplain pans. This implies that wetlands that are closer to main rivers are more likely to be influenced by exchanging organisms, nutrients and sediments and vica–versa (Tockner et al. 1999; Weigelhofer et al. 2015; Paillex et al. 2015). Compromised catchments, however, have implications for rivers and how they interact with their associated floodplains.
The present study aims to determine the relationships between fish and macroinvertebrates assemblages and various environmental variables in floodplain pans on the Limpopo river catchment in South Afirca. Aquatic biodiversity is under severe stress all over the world, and many river systems in the southern African region are no exception. Habitat loss is widely acknowledged as a major contributor to decreased biodiversity, particularly in freshwater ecosystems (Ayyad 2003; Laurance 2007; Gulzar et al. 2023). In many instances habitat is altered or lost, reducing diversity and complexity. Freshwater ecosystems are no exception. According to Hussain (2010), an estimated 39% of all freshwater species of the global aquatic ecosystem are extinct, endangered, or vulnerable due to habitat disturbances. uman–mediated alterations of environmental conditions can have implications for population and community structure and functioning of the food web in floodplain pans (Rask et al. 2020; Verma and Prakash 2020; Volkoff and London 2018). Recent environmental changes, declining water quality, and fishing pressure have, for example, made freshwater fish species more vulnerable. Numerous fish species found in floodplains, rivers, and estuaries have consequently become threatened or extinct due to the latter pressures (Boelee et al. 2019; Chen et al. 2020). HMacroinvertebrates are similarly negatively affected both directly and indirectly by changes to aquatic ecosystems, with implications for theircontributions to ecosystem functioning (Höök et al., 2020).
The main aim of this study was to determine the macroinvertebrates and fish community structure within the selected Makuleke pans across hydroperiods. In achieving this objective, the study focused on identifying the potential causes of macroinvertebrates and fish communities by assessing species diversity metrics and abundances as well as other drivers such as water and sediment quality. We hypothesized that (i) the abundance and diversity of macroinvertebrates and fish in the floodplains pans are driven by hydroperiod (their dependence on receiving enough water during the summer rainfall season), whereby fluctuation in water level affect the diversity metrics and abundance of species within the pans and (ii) differences in the richness and diversity of macroinvertebrate and fish species are attributed to variations in water and sediment chemistry, since changes in these variables can have a significant impact on communities within pans.
Materials and Methods
Ethical Clearance and Permits
Fish and macroinvertebrates samples were collected in accordance with the Kruger National Park permit (KNP Permit Reference no. DALT1635) and ethical clearance for the present study was approved by the University of Venda Research committee (Ethical clearance No. SES/20/ERM/14/1611).
Site Description
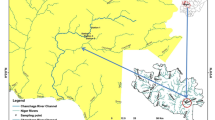
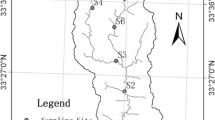
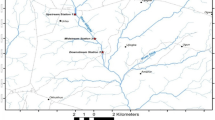
Six floodplain pan wetlands systems namely; Nyavadi, Mapimbi, Mambvumbvanyi, Nghila, Jachacha and Nwambi, were studied within the Makuleke Ramsar site at Pafuri, North of Kruger National Park (Fig. 1). From each pan, three sites were chosen as sampling points for environmental variables with sampling taking place in September 2020 (LW1), April 2021 (HW1), December 2021 (LW2) and March 2022 (HW2). September 2020 and December 2021 were considered low–water (LW) periods, while April 2021 and March 2022 were considered high–water (HW) periods. The Makuleke Wetland Concession is a region of land with an extent of 20 000 ha and is found between the Luvuvhu and Limpopo rivers in the northern and eastern parts of the Limpopo Province, South Africa (Malherbe 2018). The area receives annual rainfall of around 500–800 mm and is dominated by woodland. The underlying geology of the pans is composed of sedimentary rocks such as sandstone, several acidic, intrusive granites, and gneisses, with a majority of the pans covered by sandy soils and a clay soil lens. In 1969, the Makuleke area was proclaimed as part of the KNP to conserve fauna and flora that are found in the northern region of the park. In 2007, the Makuleke wetlands received the Ramsar recognition status as wetlands of international importance (Pretorius 2013; Dyamond 2017). This area was the first Ramsar site to be owned and co–managed by a local community (Makuleke community) and the Kruger National Park in South Africa. The area has prominent floodplain features including flood pans, floodplains and river channels, which are intermittently filled from floods and rain that support a high diversity of birds and wildlife (Dzurume et al. 2022; De Necker et al. 2023).
Environmental Variables of Water
During each sampling event, at each site environmental parameters such as pH, oxygen redox potential (ORP), electrical conductivity (EC), total dissolved solids (TDS), salinity, resistivity and temperature (°C) (n = 3 per site) were measured by immersing a portable multi–parameter probe (PCTestr 35, Eutech/Oakton Instruments, Singapore) into the water and results recorded in–situ at each sampling site.
Determination of Pelagic Chlorophyll–a
For determination of pelagic chlorophyll–a (chl–a) in the laboratory, 250 mL water samples from each pan (n = 3 samples) were filtered (vacuum < 5 cm Hg) through GIC Scientific glass fiber filters with a pore size of 0.7 μm (47 mm diameter). After filtration, chl–a was extracted by placing the filter in a 15 mL centrifuge tube containing 10 mL of 90% acetone solution and keeping it in the freezer for 24 h. After 24 h, the samples were centrifuged at 5 000 rpm for 5 min. Approximately 2 mL was extracted from each sample to measure absorbance at 665 and 750 nm using a SPECTRO star NANO (BMG LabTech GmbH, Ortenberg) following Human et al. (2018). To measure the absorbance, a 1 cm quartz cuvette was used before and after acidification with two drops of 0.01 N hydrochloric acid. Chlorophyll–a concentration was calculated based on Lorenzen (1967) using the following formulas:
Calculating pelagic chlorophyll–a;
where “a” in fraction is the quantity of acetone used for extraction in µg L− 1, v is the quantity of filtered water in µg L− 1, Fo is the chl–a reading before acidification with 1 N HCl (hydrochloric acid), Fa is the chl–a reading after acidification with 1 N HCl (hydrochloric acid), and C is the constant value (0.325).
Sediment Chemistry Variables
The benthic sediments (n = 2 per pan) were collected from each pan using acid–washed wooden splints and each integrated sample was placed in new polyethylene Ziplock bags to avoid cross contamination. The composite sediment samples were immediately packed in a cooler box with ice and transported to the University of Venda pollution laboratory for processing within 24 h. In the laboratory, the samples were oven–dried at 60 °C for 72 h to constant weight before being disaggregated in a porcelain mortar. The dried sediment samples were then homogenized using a riffle splitter, and thereafter a sediment subsample of 0.5 kg was separated and sent to BEMLAB, Cape Town, for further analysis. The pH, phosphorus (P), ammonium (NH4+), potassium (K), calcium (Ca), magnesium (Mg), sodium (Na), copper (Cu), zinc (Zn), Manganese (Mn), boron (B), iron (Fe), Carbon (C), Sulphur (S), SOM concentrations were quantified in sediment for each pan and hydroperiod. Briefly, for each sediment core, elements such as K, Mg, Na Ca, Cu, Zn, Mn, B, Fe and S were measured using an Inductively coupled plasma atomic emission spectroscopy (ICPMS) instrument (see Rice, 2012 for detailed methodology), while the ammonia and phosphorus were analysed using a SEAL AutoAnalyzer 3 high resolution and Bray–2 extract as described by Bray and Kurtz (1945).
Determination Benthic Chlorophyll‒a
A Perspex sediment corer of 20 mm internal diameter was used to collect wet sediments in the littoral zone by inserting a hand into the sediment (n = 3 sites per pan). About 10 mL of 90% acetone was inoculated into the sediment sample, swirled in the vortex, and frozen at − 20 °C for 24 h for chlorophyll–a extraction. After 24 h, samples were removed from the freezer and were covered with a dark cloth and centrifuged at 3000 rpm for 10 min. All chl–a samples were handled inside a dark room. The 2 ml elutes were then put into a vial to measure absorbance at 665 and 750 nm using a SPECTRO star NANO (BMG LabTech GmbH, Ortenberg). The following formula was used to calculate the concentration of chlorophyll–a:
Calculating benthic chlorophyll–a;
Where, L – cuvette light–path (cm) (1 cm), Ve – extraction volume (mL) (10 mL), Vf – filtered volume (L) (250 mL) and K = 2.43.
Macroinvertebrates Sampling
Macroinvertebrates were sampled quantitatively using a nylon handheld kick net (dimension, 30 × 30 cm; mesh size, 500 μm) with an aluminum rim and a 1.5 m rod handle following a procedure by Dalu et al. (2021). At each sampling pan (water depth 0.1–0.6 m), macroinvertebrates were collected (n = 2 samples) by submerging the sampling kick net, kicking benthic substratum to dislodge any attached taxa in sediment and/or rocks, sweeping and dragging the net through aquatic vegetation, along the 10 m length transect inside the pan. The kick net was then carefully lifted out of the water to prevent the escape of macroinvertebrates. The macroinvertebrates were transferred into 250 mL plastic jar containers and preserved in 70% ethanol, then transported to the laboratory for further identification, according to Gerber and Gabriel (2002). Macroinvertebrates samples in 70% ethanol were poured into a tray and identified to family level, and all species were recorded.
Fish Sampling
Two techniques (i.e., Seine net and cast net) were used to sample fish communities depending on the habitat characteristics of the floodplain pan. In shallow pans, a 5 m long seine net was deployed into the pan in a straight–line motion and hauled by swiping all fish found in the pan (n = 2). Following Malherbe et al. (2016), in deep pans a cast net (diameter = 1.85 m, mesh size = 2.5 cm) was thrown into the pan (n = 5) and pulled back in a slow motion to allow smooth removal of fish from the pan and avoid kills. Different fish species were identified on site following Skelton (2001), and species names were recorded; then fish were released back into the pans.
Data Analysis
Significant differences in environmental variables among floodplain pans (i.e., Nyavadi, Jachacha, Mambvumbvanyi, Nghila, Mapimbi and Nwambi) and across hydroperiods (i.e., low–water and high–water period) were assessed using a two–way ANOVA in SPSS v25.0 (SPSS Inc., 2007). This was done after the data was found to meet all the parametric tests, i.e., homogeneity of variances and normality. To assess for differences in species diversity among floodplain pans and hydroperiods, diversity metrics (species richness, species evenness, Simpson and Shannon–Weiner) were calculated per site, using the macroinvertebrate community dataset in PAST version 2.0 (Hammer et al. 2001). A non–parametric Kruskal–Wallis test was used to test for differences in diversity metrics among study pans and hydroperiods using SPSS v16.0 (SPSS Inc., 2007). We further examined the differences in macroinvertebrates and fish communities and identified the primary species that contributed to the differences using the analyses of similarities (ANOSIM) and similarity percentages–species contributions (SIMPER) in PRIMER 5.0.
We used canonical–correlation analysis (CCA) to determine the major environmental and sediment variables that drive fish communities across floodplain pans and hydroperiods. Consequently, redundancy analysis (RDA) was used to determine the major environmental and sediment variables that drive macroinvertebrate communities across floodplain pans and hydroperiods. We used the forward selection method with the Monte Carlo permutation test (9999 permutations) for both CCA and RDA, where water and sediment parameters were put as explanatory variables.
Results
Environmental Variables
The results of this study indicate high levels of conductivity, salinity and resistivity and high concentrations of sediment parameters such as P, K, Fe and S during high water periods (Table 1). Most of the water and sediment quality assessed using two–way ANOVA indicated significant differences across pans (p < 0.05) and between hydroperiods (p = 0.05) with the exception of pelagic chl–a, which was non–significant (p > 0.05) across pans, hydroperiods and their interaction (pans × hydroperiod) (Table 2).
Macroinvertebrate Communities
Twenty–three macroinvertebrate families were identified within Makuleke floodplain pans (see Table S1). Macroinvertebrates (i.e., Notonectidae, Libellulidae, Gerridae, Chironomidae larvae, Belostomatidae, Gomphidae, Dytiscidae and Baetidae) were found within all floodplain pans and were dominant during both hydroperiods (low– and high–water period). However, macroinvertebrates families such as Dryopidae, Noteridae, Ceratopogonidae, Tabanidae and Hydropsychidae had a low relative abundance, with most of these macroinvertebrates dominating during low water period than during high water period (Table S1). When looking at individual pans, Mambvumbvanyi pan had the highest species diversity, followed by Nwambi pan, both of which had a slarge species diversity which varied between hydroperiods (Table S1).
Macroinvertebrate taxa richness increased with changes in hydroperiods (from low to high water level). The diversity indices (i.e., abundance, Shannon–Wiener, Simpsons), were low especially during low water, however, increased during high water period in all pans except in Nyavadi, Jachacha and Mambvumvanyi which were zero for Simpson’s diversity (Table 3). Based on macroinvertebrates diversity matrices on two–way ANOVA, significant differences were observed across sites and hydroperiods (p < 0.001) (Table 4). We further observed significant differences in macroinvertebrates diversity metrics over a combination of sites and hydroperiods.
Analysis of similarities (ANOSIM) showed significant differences across all pans and hydroperiods in the macroinvertebrates communities (Global test R = 0.56, p < 0.05) (Table 5). Similarity percentages (SIMPER) revealed that the average dissimilarity across pans and hydroperiods in macroinvertebrates communities was 45.57%. The SIMPER results indicated that macroinvertebrates such as Libellulidae, Notonoctidae, and Chironomidae larvae were the main dissimilarity contributing taxa across pans and hydroperiods (Table 5).
Macroinvertebrate Communities in Relation to Environmental Variables
The differences in environmental variables as driving factors for the macroinvertebrate communities were combined for both hydroperiods (low and high–water period) and represented in Fig. 2. Based on RDA and CCA analysis, environmental variables (two water parameters, i.e., salinity and TDS; and three sediment metals – Zn, C and Boron) were found to be the major drivers in explaining the variation of macroinvertebrate community structure across all sample pans and hydroperiods. According to the results of RDA Axis 2, salinity and TDS in water were the main drivers for the communities of Aeshnidae, Corixidae and Gyrinidae, while metals Zn – C – and B were the major drivers of macroinvertebrates (i.e., Gerridae, Libellulidae – Notonectidae, gomphidae, belastomatidae – Beatidae, Velidae, dyctiscidae, respectively) (Fig. 2a).
(a) Redundancy analysis (RDA) of major environmental and sediment variables in relation to macroinvertebrate communities across floodplain pans and hydroperiod. (b) Canonical– correlation analysis (CCA) of major environmental and sediment variables in relation to fish communities across floodplain pans and hydroperiod. Abbreviations: TDS – total dissolved solids, K – Potassium, Ca – Calcium, Mg – Magnesium, Zn – Zinc, C – Carbon, B – Boron
Fish Communities
A total of sixteen fish species were found in six sampled floodplain pans of the Makuleke wetlands (Table S2). The results from this study indicate that high fish abundance was found during the high–water period compared to a very low abundance during the low water period. We further observed that, in most cases, during low–water period (which is considered dry season due to low rainfall), most pans were found with little or no water resulting in the absence of fish. During the low–water period, pans such as Nghila were found with no water at all, whereas Nyavadi pan was found with very little turbid water and dominated by only one fish species (catfish – Clarias gariepinus). In contrast, when the pans were full of water (during the high–water period), we observed high diversity of fish species across all the pans. The fish diversity indices generally showed an increased trend from low to high water period, however, Nghila pan had zero fish diversity indices in the low water period. High fish abundances were found across all the pans, whereas high species richness (mean = 7.5) was found in two pans (Jachacha and Mambvumbvanyi) and high species evenness (mean = 0.8) was found in Jachacha and Nyavadi during the low and high–water period, respectively (Table 3). Shannon–Wiener diversity for fish highlighted a steady decline from low water to high water period, with the highest recorded mean of 1.6 in Jachacha and the lowest mean of 0 in Nghila (Table 3).
ANOSIM further revealed a significant difference across all pans and hydroperiods in the fish communities (Global test R = 0.681, p < 0.05) (Table 6). The average dissimilarity across pans and hydroperiods in fish communities was 61.95%. Fish species such as Enteromius paludinosus, Tilapia sparrmanii and Oreochromis mossambicus were the main dissimilarity contributing taxa across pans and hydroperiods (Table 6).
Fish Communities in Relation to Environmental Variables
Both water and sediment variables were found to have an influence on fish communities with floodplain pans systems. The results from CCA axis 1 indicated that pelagic chlorophyll–a had a greater influence on one fish species – Gambusia affinis, during high–water period across pans (Fig. 2b). Other water parameters such as pH and TDS were the major drivers of fish species such as Oreochromis mossambicus and Enteromius palludinosus, which were the two species that were generally abundant in all six sampled pans (Fig. 2b). Sediment metals such as K, Ca and magnesium were the major drivers of fish species (i.e., Labeo rosae, Cyprinus carpio and Enteromius unitaeniatus) across hydroperiods (Fig. 2b).
Discussion
The results support our first hypothesis, which stated that the abundance and diversity of macroinvertebrates and fish in the floodplains pans are driven by hydroperiod. In this study, high fish abundances were associated with high macroinvertebrates abundances facilitating high biodiversity, especially during the high water periods. Furthermore, water chemistry variables (i.e., pH, TDS, salinity) and metals (i.e., K, Ca, Zn, C, B) accumulated in benthic sediments were found to be the major drivers of macroinvertebrates and fish community structures within the floodplains pans. It can be noted that macroinvertebrate community structure followed the observed water quality changes among the floodplain pans and hydroperiods, with the effects of sediment chemistry being integrated into the overall macroinvertebrate community structuring (Bere et al., 2016). Therefore, the present study showed that macroinvertebrate variability with regard to environmental tolerances and preferences within the different taxa were sufficient to disguise any patterns of spatiotemporal structuring, as shown by ordination results (Fig. 2). Different macroinvertebrate taxa respond differently to hydroperiods (low and high–water period), resulting in community structure shifts (Batzer et al., 2015).
Water and sediment quality varied from one pan to another, with high concentrations recorded during the high water period. The sources of nutrients and metals are, however, unclear and require further tracing. Nutrient dynamics in systems engineered by megaherbivores are particularly complex. For example, elephants and other wildlife regularly utilise the pans’ water facilitating allochthonous nutrient transfer from terrestrial to aquatic environments, with implications for productivity (Wasserman and Dalu, 2023). These nutrients tend to dissolve in water and settle in benthic sediment, accumulating and changing the pans’ functionality. Previous studies (i.e., Gereta et al. 2004a, b; Kühn et al. 2013) have similarly observed the same results in wetlands whereby nutrients within the pans originated from animal droppings and urine instead of anthropogenic inputs. With regards to metals concentrations, source tracing is difficult in river systems as far–removed catchment activites often have implications for downstream concentrations. High concentrations of metals (i.e., B, Mn, Zn, Ca) can, however, also occur naturally as is observed in the igneous and sedimentary rocks found within the Pafuri region. Jiang et al. (2017), for example, observed that metals such as Mn recorded within wetlands sediments, originated from natural processes such as weathering of soils and bedrock within the local vicinity where the pans are located.
Water quality had a high implication for macroinvertebrates’ community structure as there were significant differences among various study pans and hydroperiods. Macroinvertebrates abundances declined with water changes resulting in high concentrations of metals and nutrients as the pans dried out. Parameters such as conductivity and TDS were high in the low water period, which seriously impacted the macroinvertebrates’ taxa richness. Similar trends were also observed with fish species, where the decline in fish abundance was associated with water chemistry variable changes.Demeke and Tassew (2016) similarly observed that fish populations in aquatic environments declined with changes in water variables, however, in most instances, some fish were highly tolerant to the changes and thus could survive the harsh conditions. Therefore, the macroinvertebrates and fish taxa in the present study can be used to determine water quality, on the other hand, the results presented here indicated that water quality could also be used to determine macroinvertebrates and fish community structure and assemblages.
The results of the present study showed that chl‒a concentrations significantly changed with hydroperiod for both the pelagic and benthic samples. From the CCA analysis, only pelagic chl‒a concentration significantly affected the fish community structure within the pans as it could potentially affect the dissolved oxygen concentration, thereby affecting fish diversity, and also it could have been a source of food for some fish species such as O. mossambicus, Enteromius spp., C. carpio and C. rendali (Franklin 2014; Poole; Kuang et al. 2020). From both RDA and CCA results, TDS was found as a major parameter driving fish and macroinvertebrates community structure, respectively. It is evident that TDS can be used as a predictor for aquatic biomass (Prepas 1983), thus, in this study, it was observed that an increase in TDS resulted in high primary productivity and reduced fish populations within the pans. The results further highlighted that the pans had high TDS and chl‒a concentration during the high–water period, and this was likely due to increased nutrients which came from runoff, wildlife and natural processes. Previous studies (i.e., McFarland et al. 2019; Müller et al. 2020) have shown that in shallow water bodies, rainfall runoff is considered the primary vehicle for nutrient additions.
Previous studies on the Makuleke wetlands highlight their variability in community structure. For example, a recent study by De Necker et al. (2023), revealed large differences in macroinvertebrate community structure across temporal scales with high diversity among pans. A study on aquatic invertebrates community structure (i.e., macroinvertebrates and zooplankton) in the Makuleke floodplain pans by Dyamond (2017) highlighted that the most dominant macroinvertebrate family was the notonectids (83%) in all pans during high water periods. In the present study, Shannon–Weiner index indicated that macroinvertebrates communities were heterogeneous within the pans. Taxa richness for both macroinvertebrates and fish was found to be high during high water periods suggesting that taxa were more likely to be reproducing and dispersing during these periods (Lyon et al. 2010). Macroinvertebrates taxa richness varied among the six floodplain pans, being generally high in Mambvumbvanyi and Jachacha pans, which may have been due to low number of predators observed during sampling. The relatively low diversity in the Nghila and Nyavadi could possibly be explained by low or no water found in those pans during both hydroperiods which reduced habitats and increased predation and competition for the available niches. High abundance and taxa richness of fish and macroinvertebrates also demonstrate that the area is well conserved as most of the ecological processes have not been altered as compared to Nylsvlei Ramsar site where the diversity of macroinvertebrates was low (Dalu et al., 2021). Based on ANOSIM, we found that macroinvertebrates and fish were significantly different in all pans and across hydroperiods, although in macroinvertebrates such as Libellulidae, Notonoctidae, and Chironomidae and fish species such as Enteromius paludinosus, Tilapia sparrmanii and Oreochromis mossambicus were the main dissimilarity contributing taxa across pans and hydroperiods. These results may imply that such species are the major drivers of community structure within floodplain pans since they also responded to changes in environmental variables as per the results of RDA and CCA analysis.
Conclusion
In conclusion, the macroinvertebrates and fish community matrices in the Makuleke floodplain pans showed high taxa diversity, suggesting that the pans are still minimally impacted by direct human activities, facilitated by falling within a protected area. It was further identified that the assessed biotic and abiotic variables in this study were important in structuring macroinvertebrate and fish communities, highlighting the importance of water quality integrity through sound cathment management. The results of this study are important as they can be used as baseline information to inform decision–makers on monitoring and conservation measures wetlands in protected areas like Kruger National Park. We further recommend future studies on the assessment of allochthonous and autochthonous contributions tofoodweb structures of these largely pristine wetlands systems.
Data Availability
All the data collected during this research is presented in the manuscript.
Change history
06 December 2023
A Correction to this paper has been published: https://doi.org/10.1007/s13157-023-01758-4
References
Acosta AA, Netherlands EC, Retief F, de Necker L, du Preez L, Truter M, Alberts R, Gerber R, Wepener V, Malherbe W, Smit NJ (2020) Conserving freshwater biodiversity in an african subtropical wetland: South Africa’s lower Phongolo River and floodplain. Managing wildlife in a changing world. IntechOpen, London, UK
Ayyad MA (2003) Case studies in the conservation of biodiversity: degradation and threats. J Arid Environ 54(1):165–182
Boelee E, van der Geerling G, Blauw A, Vethaak AD (2019) Water and health: from environmental pressures to integrated responses. Acta Trop 193:217–226
Brazner, J. C., & Beals, E. W. (1997). Patterns in fish assemblages from coastal wetland and beach habitats in Green Bay, Lake Michigan: a multivariate analysis of abiotic and biotic forcing factors. Canadian Journal of Fisheries and Aquatic Sciences, 54(8), 1743-1761.
Chen Y, Qu X, Xiong F, Lu Y, Wang L, Hughes RM (2020) Challenges to saving China’s freshwater biodiversity: Fishery exploitation and landscape pressures, vol 49. Ambio, pp 926–938
Dalu T, Wasserman RJ, Tonkin JD, Mwedzi T, Magoro ML, Weyl OL (2017) Water or sediment? Partitioning the role of water column and sediment chemistry as drivers of macroinvertebrate communities in an austral south african stream, vol 607. Science of the total environment, pp 317–325
De Necker L, Dyamond K, Greenfield R, van Vuren J, Malherbe W (2023) Aquatic invertebrate community structure and functions within a Ramsar wetland of a premier conservation area in South Africa. Ecol Ind 148:110135
Demeke A, Tassew A (2016) A review on water quality and its impact on fish health. Int J Fauna Biol Stud 3(1):21–31
Dube T, Pinceel T, De Necker L, Wepener V, Lemmens P, Brendonck L (2019) Lateral hydrological connectivity differentially affects the community characteristics of multiple groups of aquatic invertebrates in tropical wetland pans in South Africa. Freshw Biol 64(12):2189–2203
Dube T, De Necker L, Wepener V, Smit NJ, Pinceel T, Mwaijengo GN, Lemmens P, Brendonck L (2020) A comparison of aquatic macroinvertebrate and large branchiopod community composition between temporary pans of a conservation area and surrounding communal area in South Africa. Afr Zool 55(1):67–77
Dyamond KS (2017) Macro–invertebrate diversity within the Makuleke Wetlands in the Pafuri Region of Kruger National Park. University of Johannesburg (South Africa)
Dzurume T, Dube T, Shoko C (2022) Remotely sensed data for estimating chlorophyll–a concentration in wetlands located in the Limpopo Transboundary River Basin, South Africa, vol 127. Physics and Chemistry of the Earth, Parts A/B/C, p 103193
Edeh IC, Nsofor CI, Iheanacho SC, Ikechukwu CC, Nnachi IA, Nwankwo CG (2023) Behavioural and haematological alterations in the african catfish (Clarias gariepinus) exposed to varying concentrations of glyphosate. Asian J Fisheries Aquat Res 21(1):32–42
Farooq M, Li X, Li Z, Yang R, Tian Z, Tan L, Fornacca D, Li Y, Cili N, Ciren Z, Liu S (2022) The joint contributions of environmental filtering and spatial processes to macroinvertebrate metacommunity dynamics in the alpine stream environment of Baima Snow Mountain, Southwest China. Diversity 14(1):28
Florencio M, Díaz-Paniagua C, Gómez‐Rodríguez C, Serrano L (2014) Biodiversity patterns in a macroinvertebrate community of a temporary pond network. Insect Conserv Divers 7(1):4–21
Franklin PA (2014) Dissolved oxygen criteria for freshwater fish in New Zealand: a revised approach. N Z J Mar Freshwat Res 48(1):112–126
Frisch D, Libman BS, D’Surney SJ, Threlkeld ST (2004) Diversity of floodplain copepods (Crustacea) modified by flooding: species richness, diapause strategies and population genetics. Arch Hydrobiol 162(1):1–18
Gerber A, Gabriel MJM (2002) Aquatic invertebrates of south african rivers: field guide. Department of Water Affairs and Forestry, Resource Quality Services
Gereta E, Mwangomo E, Wolanski E (2004a) The influence of wetlands in regulating water quality in the Seronera River, Serengeti National Park, Tanzania. Wetlands Ecol Manage 12:301–307
Gereta E, Meing’ataki O, Mduma GE, Wolanski S, E (2004b) The role of wetlands in wildlife migration in the Tarangire ecosystem, Tanzania. Wetlands Ecol Manage 12:285–299
Gordon BA, Dorothy O, Lenhart CF (2020) Nutrient retention in ecologically functional floodplains: a review. Water 12(10):2762
Guareschi S, Abellán P, Laini A, Green AJ, Sánchez–Zapata JA, Velasco J, Millán A (2015) Cross–taxon congruence in wetlands: assessing the value of waterbirds as surrogates of macroinvertebrate biodiversity in Mediterranean Ramsar sites, vol 49. Ecological Indicators, pp 204–215
Gulzar R, Hassan T, Gulzar A (2023) Developmental Activities and Freshwater Biodiversity. Biodiversity of Freshwater Ecosystems. Apple Academic Press, pp 133–146
Hammer Ø, Harper DAT, Ryan PD (2001) PAST–palaeontological statistics, ver. 1.89. Palaeontol electron 4(1):1–9
Hayes DS, Brändle JM, Seliger C, Zeiringer B, Ferreira T, Schmutz S (2018) Advancing towards functional environmental flows for temperate floodplain rivers. Sci Total Environ 633:1089–1104
Heath SK, Plater AJ (2010) Records of pan (floodplain wetland) sedimentation as an approach for post–hoc investigation of the hydrological impacts of dam impoundment: the Pongolo river, KwaZulu–Natal. Water Res 44(14):4226–4240
Human LRD, Magoro ML, Dalu T, Perissinotto R, Whitfield AK, Adams JB, Deyzel SHP, Rishworth GM (2018) Natural nutrient enrichment and algal responses in near pristine micro–estuaries and micro–outlets. Sci Total Environ 624:945–954
Hussain MG (2010) Freshwater fishes of Bangladesh: fisheries, biodiversity and habitat. Aquat Ecosyst Health Manag 13(1):85–93
Jiang X, Xiong Z, Liu H, Liu G, Liu W (2017) Distribution, source identification, and ecological risk assessment of heavy metals in wetland soils of a river–reservoir system, vol 24. Environmental Science and Pollution Research, pp 436–444
Kaban S, Armanto ME, Ridho MR, Hariani PL (2018) Rapid Assessment of Water Quality in Teluk Lake by Using Macroinvertebrates (A Family Level Biotic Index). In E3S Web of Conferences (Vol. 68, p. 04006). EDP Sciences
Kanu KC, Otitoloju AA, Amaeze NH (2022) Survival analysis of african catfish and Nile tilapia briefly exposed to complex pesticide mixtures. J Basic Appl Zool 83(1):1–15
Kärnä OM, Heino J, Laamanen T, Jyrkänkallio–Mikkola J, Pajunen V, Soininen J, Tolonen KT, Tukiainen H, Hjort J (2019) Does catchment geodiversity foster stream biodiversity? vol 34. Landscape Ecology, pp 2469–2485
Kuang L, Shi P, Hua C, Chen B, Zhu H (2020) An enhanced extreme learning machine for dissolved oxygen prediction in wireless sensor networks. Ieee Access 8:198730–198739
Kühn M, Maier U, Herbig C, Ismail–Meyer K, Le Bailly M, Wick L (2013) Methods for the examination of cattle, sheep and goat dung in prehistoric wetland settlements with examples of the sites Alleshausen–Täschenwiesen and Alleshausen–Grundwiesen (around cal 2900 BC) at Lake Federsee, south–west Germany. Environ Archaeol 18(1):43–57
Kunwar PS, Sapkota B, Badu S, Parajuli K, Sinha AK, De Boeck G, Sapkota K (2022) Chlorpyrifos and dichlorvos in combined exposure reveals antagonistic interaction to the freshwater fish Mrigal, Cirrhinus mrigala. Ecotoxicology 31(4):657–666
Laurance WF (2007) Have we overstated the tropical biodiversity crisis? Trends Ecol Evol 22(2):65–70
Lorenzen CJ (1967) Determination of chlorophyll and phaeophytin. Spectrophotometric equations. Limnol Oceanogr 12:343–346
Lyon J, Stuart I, Ramsey D, O’Mahony J (2010) The effect of water level on lateral movements of fish between river and off–channel habitats and implications for management. Mar Freshw Res 61(3):271–278
Malherbe W (2018) Ramsar wetlands in South Africa: historic and current aquatic research. S Afr J Sci Technol 38(1):1–13
Malherbe W, Wepener V, Van Vuren JHJ (2016) The effect of a large–scale irrigation scheme on the fish community structure and integrity of a subtropical river system in South Africa, vol 69. Ecological Indicators, pp 533–539
McFarland AR, Larsen L, Yeshitela K, Engida AN, Love NG (2019) Guide for using green infrastructure in urban environments for stormwater management. Environmental science: Water research & technology, 5(4), pp.643–659
Müller A, Österlund H, Marsalek J, Viklander M (2020) The pollution conveyed by urban runoff: A review of sources. Science of the Total Environment, 709, p.136125
Nhiwatiwa T, Brendonck LUC, Waterkeyn A, Vanschoenwinkel B (2011) The importance of landscape and habitat properties in explaining instantaneous and long-term distributions of large branchiopods in subtropical temporary pans. Freshw Biol 56(10):1992–2008
Nhiwatiwa T, Brendonck L, Dalu T (2017) Understanding factors structuring zooplankton and macroinvertebrate assemblages in ephemeral pans, vol 64. Limnologica, pp 11–19
Paillex A, Castella E, zu Ermgassen PS, Aldridge DC (2015) Testing predictions of changes in alien and native macroinvertebrate communities and their interaction after the restoration of a large river floodplain (french Rhône). Freshw Biol 60(6):1162–1175
Pandiarajan S, Thambiratnam S, Sivaruban IRB (2019) Bio–monitoring and detection of water quality using Ephemeroptera, Plecoptera and Trichoptera (EPT) complex in Karanthamalai Stream of Eastern Ghats. Indian J Ecol 46(4):818–822
Petsch DK, Cionek VDM, Thomaz SM, dos Santos NCL (2022) Ecosystem services provided by river–floodplain ecosystems. Hydrobiologia, pp.1–22
Pilière A, Schipper AM, Breure AM, Posthuma L, de Zwart D, Dyer SD, Huijbregts MA (2014) Comparing responses of freshwater fish and invertebrate community integrity along multiple environmental gradients, vol 43. Ecological indicators, pp 215–226
Poole AS, Koel TM, Thomas NA, Zale AV (2020) Benthic suffocation of invasive lake trout embryos by fish carcasses and sedimentation in Yellowstone lake. North Am J Fish Manag 40(5):1077–1086
Prepas EE (1983) Total dissolved solids as a predictor of lake biomass and productivity. Can J Fish Aquat Sci 40(1):92–95
Pretorius E (2013) Image classification towards mapping of vegetation structure: a practical approach. In International Cartographic Conference, Dresden, Germany
Raphahlelo ME, Addo–Bediako A, Luus–Powell WJ (2022) Distribution and diversity of benthic macroinvertebrates in the Mohlapitsi River, South Africa. J Freshw Ecol 37(1):145–160
Rask M, Malinen T, Olin M, Peltonen H, Ruuhijärvi J, Vesala S, Hietala J (2020) Responses of the fish community in a eutrophicated lake to long–term food web management assessed by multiple sampling methods, vol 847. Hydrobiologia, pp 4559–4573
Sarkar UK, Bakshi S, Lianthuamluaia L, Mishal P, Das Ghosh B, Saha S, Karnatak G (2020) Understanding enviro–climatological impact on fish biodiversity of the tropical floodplain wetlands for their sustainable management. Sustainable Water Resources Management 6:1–12
Skelton PH (2001) A complete guide to the Freshwater fishes of southern Africa, 2nd edn. Struik, Cape Town, South Africa
Smit NJ, Vlok W, Van Vuren JHJ, Du Preez L, Van Eeden E, O’Brien GC, Wepener V, Socio–ecological system management of the lower Phongolo River and floodplain using relative risk methodology. Water Research Commission (WRC), Report (2016) (2185/1), p.16
Sonone SS, Jadhav S, Sankhla MS, Kumar R (2020) Water contamination by heavy metals and their toxic effect on aquaculture and human health through food chain. Lett Appl NanoBioScience 10(2):2148–2166
Svanbäck R, Eklöv P, Fransson R, Holmgren K (2008) Intraspecific competition drives multiple species resource polymorphism in fish communities. Oikos 117(1):114–124
Tockner K, Pennetzdorfer D, Reiner N, Schiemer F, Ward JV (1999) Hydrological connectivity, and the exchange of organic matter and nutrients in a dynamic river–floodplain system (Danube, Austria). Freshw Biol 41(3):521–535
Verma AK, Prakash S (2020) Limnological studies of Semara Taal, a wetland of district Siddharthnagar, Uttar Pradesh, India. J Fisheries Life Sci 5(1):15–19
Volkoff H, London S (2018) Nutrition and reproduction in fish. Encyclopedia of Reproduction, 2, pp.1–6
Ward JV, Tockner K, Schiemer F (1999) Biodiversity of floodplain river ecosystems: ecotones and connectivity1. River Res Appl 15(1–3):125–139
Weigelhofer G, Preiner S, Funk A, Bondar-Kunze E, Hein T (2015) The hydrochemical response of small and shallow floodplain water bodies to temporary surface water connections with the main river. Freshw Biol 60(4):781–779
Funding
We are greatful for the financial support of the University of Venda Niche Grant (FSEA/21/GGES/02) received by Farai Dondofema, the NRF Thuthuka Grant (138206) received by Tatenda Dalu, and a grant from the South African Institute for Aquatic Biodiversity received by Ryan Wasserman. We further acknowledge funding from the National Research Foundation Postgraduate Bursary (UID: 129098) received by Linton Munyai.
Author information
Authors and Affiliations
Contributions
LFM: Conceptualization, Investigation, Data curation, Formal analysis, Writing – original draft, review & editing. LM: Investigation, Data curation, Supervision, Writing – review & editing. RJW: Conceptualization, Investigation, Supervision, Writing – original draft, review & editing. FD: Conceptualization, Investigation, Resources, Data curation, Writing – original draft, review & editing. TD: Conceptualization, Investigation, Resources, Data curation, Formal analysis, Supervision, Writing – original draft, review & editing.
Corresponding author
Ethics declarations
Conflict of Interest
All co-authors have seen and agree with the contents of the manuscript and there is no financial interest to report. Thus, all authors have declared that no competing interests exist.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Munyai, L.F., Mugwedi, L., Wasserman, R.J. et al. Assessing Fish and Macroinvertebrates Assemblages in Relation to Environmental Variables in Makuleke Floodplain Pans: Implications for Biodiversity Conservation. Wetlands 43, 93 (2023). https://doi.org/10.1007/s13157-023-01738-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13157-023-01738-8