Abstract
Invasive cattails (Typha angustifolia and Typha × glauca) pose a problem for many Laurentian Great Lakes wetlands, especially sedge/grass meadows. In western Lake Superior, early signs of invasion into sedge-dominated peatlands along the Kakagon and Bad Rivers, owned and managed by the Bad River Band of Lake Superior Chippewa, were noticed in the early 1990s. In 1998, we began tracking expansion of Typha patches and assessing causes and ramifications. Perimeters of patches were delineated with GPS, with repeat delineations in 2005 and 2013. At the Kakagon site, permanent transects were established tangential to the 1998 patch perimeters and perpendicular to those transects, extending into the cattails and outward into sedges. Plant communities were sampled along transects in all years. Transects across stands of non-invasive Typha latifolia were sampled for comparisons but showed little change. In 2005, area encompassed by the Kakagon River patches increased by 66% from 1998; increase was 143% by 2013. The Bad River patches increased by 25% in 2005; further analyses were not possible because some patches had coalesced. Across years, mean Importance Value (IV) for invasive Typha increased for all tangential and perpendicular transects, reflecting expansion into adjacent sedge meadow. Among changes in other species, mean IV for dominant sedge Carex lasiocarpa also decreased on both transects. Typha invasion seems to be directional and not cyclical in relation to lake-level changes, which might have future influence. Given the tendency for early Typha expansion, land managers should scout for new patches and begin treatment soon after discovery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Wetlands in the Laurentian Great Lake of North America have been studied in detail by many researchers, providing the foundation for understanding ecological processes, especially related to water-level fluctuations and invasive plant species (e.g., Keddy and Reznicek 1986; Wilcox 2004; Boers and Zedler 2008; Wilcox et al. 2008; Wilcox and Nichols 2008; Lishawa et al. 2010; Smith et al. 2021). These studies have broader applications for wetlands of large lakes elsewhere.
Lake Superior coastal wetlands are often ecologically different from mainland interior marshes and peatlands, in part because they are subject to quasi-periodic, large fluctuations in water levels exceeding one meter (Argyilan et al. 2005; Johnston et al. 2012, 2014). Coupled with flooding and dewatering on a lesser scale of water-level changes, the result is pulse stability of plant communities (Odum 1971) that endures shifts in vegetation; maintains health, diversity, and viability by flushing out accumulated detritus; periodically exposes and oxidizes sediments; and prevents dominance by any one species (e.g., Woo and Zedler 2002; Wilcox 2004, Wilcox and Nichols 2008; Farrer and Goldberg 2009; Larkin et al. 2012). Even as the wetlands change in species abundances in the short term in response to extreme lake levels, over longer time scales, they remain stable as specific types of wetland. This pulse stability can exist as long as water quality and climate do not change drastically and invasive species do not become dominant.
In many wetlands around the Great Lakes, a number of invasive plants, including narrow-leaf (Typha angustifolia) and hybrid cattail (Typha × glauca), common reed (Phragmites australis), purple loosestrife (Lythrum salicaria), and at higher elevations, reed canarygrass (Phalaris arundinacea), have invaded (e.g., Galatowitsch et al. 1999; Albert and Minc 2004; Tulbure et al. 2007; Wilcox et al. 2008; Wilcox 2012). The changes do not seem to be cyclic (or pulse stable) but directional, with the abundance of smaller, native plant species eventually being reduced.
Although Typha latifolia is not considered to be invasive (Bansal et al. 2019), T. angustifolia and T. × glauca (hybrids of T. angustifolia and T. latifolia) are considered to be invaders in many different freshwater wetlands across North America, including the Great Lakes, throughout the Midwest, the Prairie Pothole Region, large Canadian prairie lakes and the Pacific Northwest (e.g., Shay et al. 1999; Wilcox et al. 2008; Bansal et al. 2019; Pieper et al. 2020; Stewart 2021; Tangen et al. 2022). Invasion in the Laurentian Great Lakes is commonly associated with physical, chemical, and hydrologic disturbances (e.g., McDonald 1955; Galatowitsch et al. 1999; Thiet 2002; Woo and Zedler 2002; Albert and Minc 2004; Wilcox et al. 2008; Smith et al. 2021). Typha is an obligate wetland plant, is reasonably flood-tolerant, and is often favored by moderate flooding (e.g., Harris and Marshall 1963; Bedish 1967; Grace and Harrison 1986; Squires and van der Valk 1992; Ellison and Bedford 1995; Boers et al. 2007). Typha angustifolia can invade by wind-blown seed (van der Valk and Davis 1978), while hybrid T. × glauca typically expands by vegetative growth from small colonies, often in response to stable water levels (e.g., Waters and Shay 1990, 1992, Shay et al. 1999; Seabloom et al. 2001; Boers and Zedler 2008; Wilcox et al. 2008; Lishawa et al. 2010). However, T. angustifolia can also expand vegetatively, and T. × glauca can be fertile and spread by seed (Kirk et al. 2011; Pieper et al. 2020). Levels of sexual and asexual reproduction are overall likely to be comparable between parent Typha species and hybrids (Pieper et al. 2020). There are many potential drivers of Typha invasion (Bansal et al. 2019). For example, Typha is more able to make use of excess nutrients than native sedge species (Woo and Zedler 2002; Larkin et al. 2012), and prolonged inundation increases P availability in wetland sediments via internal eutrophication (Boers and Zedler 2008). Low water conditions may also be conducive to Typha invasion (McDonald 1955; Frieswyk and Zedler 2007; Tulbure et al. 2007; Lishawa et al. 2010).
Typha invasion is already prominent in many Great Lakes wetlands subject to disturbance. This invasion has nearly eliminated sedge/grass meadow marsh in all Lake Ontario wetlands as a result of water-level regulation, where T. × glauca is most prominent in the higher elevation remnant sedge/grass meadow and T. angustifolia often forms floating mats at lower elevations (Wilcox et al. 2008, 2018; Vaccaro et al. 2009; Wilcox and Bateman 2018; Smith et al. 2021). At Ottawa National Wildlife Refuge on Lake Erie, T. angustifolia became prominent following glyphosate treatment to control Phragmites australis (Carlson et al. 2009). In Lake St. Clair, T. angustifolia was found at lower elevations when water receded following flooding by 1986 high lake levels, while T. × glauca was found on higher ridges that were not flooded (Wilcox 2012). In Saginaw Bay of Lake Huron, T. angustifolia became prominent, presumably from seed, on a shoreline exposed when lake levels receded from the 1986 high (Wilcox and Nichols 2008). Both T. angustifolia and T. × glauca became established in a drowned river mouth wetland of Lake Michigan when water levels receded from highs in 1986 and 1997 (Wilcox et al. 2022).
A transition from peatland vegetation to marsh vegetation is progressing in several Lake Superior coastal wetlands (Meeker and Fewless 2008), and we saw evidence in the early 1990s that Typha invasion had begun in the Kakagon/Bad River wetlands. We thus began a long-term study in these wetlands to delineate patches of invasive Typha, track changes over time, and assess causes and ramifications. We also collected data on changes in non-invasive T. latifolia to ensure that conclusions regarding invasive Typha were not due to environmental conditions that could affect all cattails and associated native vegetation. Studies such as this that include pre- and post-invasion data at wetland, transect, and plot scales are not common.
Methods
Study Area
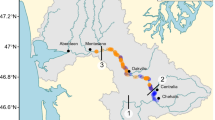
The study area included sedge-dominated peatlands along the Kakagon River and Bad River, which are drowned river mouth wetlands (Albert et al. 2005) that flow into Lake Superior in Ashland County, Wisconsin (46°39’N 90°41’W; Fig. 1). Open water channels (sloughs) weave through this 4,350 ha wetland complex owned and managed by the Bad River Band of Lake Superior Chippewa (“Bad River Band”). Expansive stands of manoomin (wild-rice, Zizania palustris var. palustris) grow along the sloughs and in coastal lagoons and are a key focus of the tribe’s stewardship of these wetlands. Deeper water zones are characterized by submergent and floating-leaved aquatic communities that intergrade into marsh, shore fen, poor fen, conifer swamps, and lowland hardwood forests (Meeker 1998; Epstein et al. 1997). The diverse wetland complex provides important fish spawning habitat and critical habitat for migratory birds. Described as the largest, least-impaired coastal wetland in Lake Superior (Epstein et al. 1997), these unique wetlands are a National Natural Landmark and were designated as a Ramsar Wetland of International Importance in 2012.
The Bad River Band has designated the Kakagon-Bad River Sloughs as a Conservation Area that prioritizes management for ecological and cultural values . Management activities have included removal of vegetation invading the margins of channels within the wild-rice habitat zones, including hand-cutting of invasive cattails (T. angustifolia and T. × glauca) and airboat mowing of pickerelweed (Pontederia cordata), a native perennial that can out-compete annual rice plants. Cattail removal efforts have generally not extended beyond the emergent rice zone and did not affect our sampling.
We targeted sampling peatland areas that included dense Typha patches dominated by a mix of T. × glauca and T. angustifolia (initially identified in the field by physical characteristics). Most common was T. × glauca, which is more prevalent than T. angustifolia in the western Great Lakes (Travis et al. 2010) and elsewhere (Kirk et al. 2011; Freeland et al. 2013; Pieper et al. 2020), often displacing both parental species. These two taxa were not identified individually in quadrat sampling and were combined as Typha spp. For comparison purposes, we also sampled adjacent peatland areas with “diffuse” Typha, characterized by scattered, low density T. latifolia that had obvious physical features of that species. Initial sampling took place in late June and July 1998, and repeat sampling took place in July 2005 and July 2013. Sampling was conducted at two spatial scales: delineating patch sizes at both the Kakagon and Bad River Slough sites and sampling Typha abundance and community composition in quadrats along permanently marked transects at the patches of the Kakagon site only.
Patch Delineation
Kakagon Sloughs
In 1998, five patches of Typha spp. were randomly chosen from a pool of 22 riverine sites where Typha was present and presumably recently invading along a 1600-m stretch of the upper portion of the Kakagon River and associated Northeast and Big Sloughs (Fig. 2). Each chosen site had a discrete patch margin adjacent to the water’s edge that bordered an aquatic vegetation zone dominated by northern wild-rice (Z. palustris). Landward, all of the chosen patch sites were bordered by sedge meadow.
During all three survey years, the areal extent of each patch was delineated in the field using a Trimble Juno SB Global Positioning System unit with an average real-time differential correction accuracy of 4 m (using Terrasync v3.2 software). A length of PVC pipe was placed in the ground at the edge of the Typha population to act as a start/finish point. The patch perimeter was walked with the GPS unit until the starting point was reached. The border of each patch was defined as encompassing all ramets (stems) less than 2 m from each other; for example, if a single Typha ramet was greater than 2 m from the perceived border, it was not included as part of the patch. In most cases, however, the patch margins were very discrete, and there were no Typha ramets visible outside the patch being delineated.
Bad River Sloughs
In 1998, eight Typha patches were delineated in the Bad River system similarly to those at the Kakagon site. They were located in areas with predictably high sediment input, as well as areas thought to receive less sediment. In 2005, the eight patches were re-sampled and 10 more were delineated for the first time. In 2013, we intended to re-delineate all patches marked in 1998 and 2005. However, many of the original patches expanded and sometimes coalesced, so only 11 discrete patches remained to delineate in 2013. No community sampling along transects was conducted.
Plant Community Sampling Associated with Patches
In 1998, two permanent transects were established at each of the five patches in the Kakagon Sloughs with the intention of identifying baseline conditions while also capturing areas where the Typha patches may expand. ‘Tangential transects’ were created tangential to the patch along the sedge meadow/Typha border and extended the width of the patch at the edge where placed (ranging from 10 to 20 m in length). ‘Perpendicular transects’ were created perpendicular to the Typha border—resulting in a survey line that extended from within the Typha-dominated patch to beyond the influence of Typha in the surrounding sedge meadow. Perpendicular transects for patches 1 and 4 were 20-m long and shorter for smaller patches 2 (16 m), 3 (10 m), and 5 (12 m). Both start and end points of all transects were marked with PVC pipe and located using a GPS unit. At every meter along one side of a taut transect line, a 1 m x 1 m quadrat frame was placed. Sampling during summer high lake levels of July took place on the same side of each transect in all years. All plants were identified, generally to species level, and percent cover of each was estimated by single digits to 20 and then by increments of five. Taxa covering less than 1% of a quadrat were systematically recorded as 0.1%. Scientific nomenclature follows Flora of North America (http://www.Efloras.org). Due to difficulty in discrete morphological separation of T. angustifolia and T. × glauca in field identification of thousands of specimens, these taxa were combined as Typha spp. All living and previous-year dead ramets of Typha in each quadrat were also counted and water depth measured at each quadrat when sampled.
Plant Community Sampling Associated with Diffuse Typha
Six ‘diffuse transects’ were also established at the Kakagon site (only) in 1998 and re-sampled in 2005 and 2013 in sedge meadows with moderate concentrations of T. latifolia ramets. As with the invasive patches, the transect locations were randomly chosen but from a larger pool of potential sites with moderate T. latifolia abundance. The diffuse transects were located along the same stretch of the river and associated sloughs as the patches but were at least 100 m away from patches (Fig. 2). As with the patch transects, the terminal points of each 20-m diffuse transect were marked with PVC pipe, and GPS coordinates were recorded. Plant community, ramet count, and water depth assessments were conducted as described for the patch transects.
Data Analyses
Changes in Patch Size
We transferred GPS data from Trimble TerraSync software to Trimble GPS Pathfinder Office and applied post-processing tools for differential correction to improve accuracy to 1–3 m. We exported data from Pathfinder to ArcGIS and overlaid polygon layers onto aerial imagery to compute the area (m2) of each polygon, representing the size of the Typha patch during each of the three sample years. Changes in patch size were calculated as the difference in m2 area between time periods.
Changes in Transect Diversity and Species Abundances
By design, each of the tangential, perpendicular, and diffuse transects was treated as an experimental unit, with the quadrats as subsamples of the transects rather than units for analyses. Species richness was calculated from quadrat data for each of the 16 transects at the Kakagon Sloughs in each year. Percent cover, frequency of occurrence, and Importance Value (IV) were also calculated from those quadrat data. Percent cover was the total cover over all the quadrats for each taxon at each transect. Frequency was calculated as the number of quadrats in which a taxon occurred on a transect divided by total number of quadrats on that transect. Importance Value was calculated as the sum of relative mean cover and relative frequency. Mean IV was used to track species changes over time because it encompasses both forms of abundance for a particular plant species. Mean counts for live and dead ramets of Typha spp. and mean water depth were calculated for each transect in each year.
Changes in Transect Communities (Multivariate Ordinations)
To summarize the initial floristic differences (1998) among the 16 transects and then to investigate their changes in 2005 and 2013, taxa x transect matrices were used to create non-metric multidimensional scaling (NMS) ordinations. The ordinations were performed in PCORD v.7 (McCune and Mefford 2016) using IV of taxa occurring across the sites. The initial ordination was conducted using IV for only the 45 taxa observed in the 1998 sampling of the 16 transects to describe baseline conditions and allow for comparisons with sampling seven and fifteen years later. A second ordination was conducted for the 16 transects at each of the three sampling times, including 60 taxa. Because the first two axes explain the majority of variation in these data in both ordinations, we do not present the third axis here. We applied successional vectors (McCune and Mefford 2016) to illustrate the direction of change in the wetland plant communities among perpendicular and tangential transect across the three time periods.
Results
Kakagon Sloughs: Changes in Typha Patch Size
In the Kakagon Sloughs, size of the non-native Typha patches more than doubled in the 15-year period. In 1998, the five patches occupied 6,150 m2 of potential sedge meadow habitat. The total was 10,205 m2 in 2005 and by 2013 had increased to 14,926 m2 (Fig. 3). In 2013, the patches covered much of the tangential and perpendicular transect lines, with the exception of Patch 4.
Bad River Sloughs: Changes in Typha Patch Size
In the Bad River Sloughs, Typha patches also expanded in size. In 1998, the original eight patches occupied 4,574 m2 and by 2005 had increased to 5,709 m2. All but one of the eight patches delineated in 1998 had coalesced with neighboring patches by 2013, so analyses of further changes in patch size were not as straightforward as in the Kakagon Sloughs and required combining areas for calculations. The addition of 10 new patches in 2005 raised the total in that year to 10,637 m2, with an increase to 23,690 m2 by 2013 (Fig. 4).
Kakagon Sloughs: Floristics
Sixty species were observed over all transects (including both patch and diffuse) during the three sampling periods at the Kakagon Sloughs, with 20 species accounting for 91.1% of the total IV (Tables 1, 2 and 3, S1, S2, S3). The most abundant taxa included typical sedge meadow species such as wiregrass sedge (Carex lasiocarpa), lake sedge (Carex lacustris), purple marshlocks (Comarum palustre), and bluejoint grass (Calamagrostis canadensis). Additionally, emergent marsh taxa were observed, including burreed (Sparganium eurycarpum) and arrowhead (Sagittaria latifolia), as well as several submergent bladderwort species (Utricularia). Typha spp. (T. × glauca and T. angustifolia) were prominent on the patch transects and T. latifolia on the diffuse transects.
Kakagon Sloughs: Species Changes
Un-invaded diffuse transects supported a greater number of species in all years (21.0, 22.2, 18.5) than transects tangential to invasive Typha patches (17.8, 17.2, 14.4) and perpendicular to patches (18.4, 19.6, 14.8) (Tables 1, 2 and 3, S1, S2, S3). Taxa number decreased among all transects in 2013.
In 1998, the tangential patch transects were dominated by C. lasiocarpa, with C. lacustris and S. eurycarpum prominent in Patches 1, 2, and 3, along with C. palustre and S. latifolia in Patch 3 (Table 1, S1). Menyanthes trifoliata (buckbean) was co-dominant in Patch 4, and non-Sphagnum moss was co-dominant in Patch 5, with Lysimachia thyrsiflora (swamp loosestrife) and Utricularia intermedia prominent. Mean IV for C. lasiocarpa for all tangential patch transects decreased from 25.0 to 19.2 to 14.9 across years despite an increase in Patch 4. Where present, mean IV for marsh emergents S. latifolia, S. eurycarpum, and the aquatic submergent Utricularia decreased across years. Mean IV for Typha spp. for all tangential transects increased from 5.0 to 19.3 to 27.1 across sampling years as Typha in the patches expanded into the adjacent sedge meadow (Table 1). However, mean Typha IV decreased in Patch 5 in 2013. Ramet counts of live Typha spp. increased from 1998 to 2005 across all patches but decreased in Patch 5 in 2013 (mean ramets/m2 = 1.2, 10.7, 8.1 for all patches across years) (Table 1). Mean ramet counts of dead Typha increased across years in all patches. Total number of taxa varied by transect and year but was less in 2013 than 1998 in all but Patch 4 (Table 1).
Carex lasiocarpa was also dominant on the perpendicular patch transects in 1998, with C. lacustris prominent in Patch 1, S. latifolia and S. eurycarpum prominent in Patch 3, and L. thyrsiflora in Patch 5 (Table 2, S2). Menyanthes trifoliata was dominant in Patch 4. The perpendicular patch transects laid out in 1998 had portions in the Typha patch and portions in the adjacent sedge meadow. As Typha began appearing along the sedge meadow portions of the transects in 2005 and 2013, mean IV for C. lasiocarpa for all perpendicular patch transects decreased from 20.2 to 18.9 to 12.0, although not in Patch 4 alone. Where present, M. trifoliata, S. latifolia, S. eurycarpum, and two species of Utricularia were also reduced greatly by 2013 on perpendicular patch transects. Mean IV for C. canadensis increased across years in Patches 2 and 3. In Patch 5, a decrease in L. thyrsiflora accompanied an increase in non-Sphagnum moss and the shrubs Chamaedaphne calyculata (leatherleaf) and Myrica gale (sweet gale). As the patches expanded into the sedge meadow (Fig. 3), the mean IV for Typha spp. across all perpendicular transects increased from 12.6 to 19.9 to 27.7 but decreased in Patch 5 alone in 2013. Ramet counts for live Typha increased from 1998 to 2005 in all patches but had a substantial decrease in Patch 5 in 2013 (mean ramets/m2 = 7.8, 11.6, 10.3 for all patches across years) (Table 2). Mean ramet counts for dead Typha increased across years in all but Patch 5, which had a decrease in 2013. There were fewer taxa in 2013 than in 1998 in all but Patch 4 (Table 2).
In 1998, the six diffuse transects were dominated by C. lasiocarpa, with C. lacustris co-dominant on transects D5 and D6 and Carex stricta (tussock sedge) co-dominant on D5 (Table 3, S3). Comarum palustre was co-dominant on transects D2, D3, and D6; S. latifolia was co-dominant on D5. Other prominent species were C. lacustris on D1, C. palustre on D4, and S. eurycarpum on D1 and D6. Many of the notable changes across years differed by transect. Increases in mean IV in 2005 included C. canadensis on D5; C. lasiocarpa on D1 and D2; C. lacustris on D1, D2, D4, and D6; and C. stricta on D5. A decrease in mean IV occurred for S. latifolia on D5. In 2013, C. lasiocarpa decreased on all diffuse transects, while two species of aquatic submergent Utricularia that had decreased in 2005 were nearly gone on all transects. Notable increases in 2013 were for C. lacustris on D1 and D4, as well as C. stricta on D5. Mean IV for T. latifolia decreased from 1998 to 2005 and then did not change in 2013 (6.3 to 3.4 to 3.4). Mean ramet counts for both live and dead T. latifolia remained low on all transects in all years (Table 3). No patterns were observed for total number of taxa, although D4 and D6 had more taxa in 2005 (Table 3).
Changes in Kakagon Sloughs Plant Communities
Axis 1 of the NMS ordination of 1998 vegetation data accounted for 79.1% of total variation and Axis 2 for 14.7% (Fig. 5). The ordination showed separation of the six diffuse transects from tangential and perpendicular patch transects, which were closely paired by patch except for Patches 2 and 4. Both transects for Patches 1 and 3 (P1P, P1T, P3P, P3T) and the tangential transect for Patch 2 (P2T) grouped near each other, as they were dominated by C. lasiocarpa (Tables 1 and 2), with lesser influences by other taxa. The community of the perpendicular transect of Patch 2 (P2P) was influenced by non-Sphagnum moss, which also influenced the ordination placement of P5T. Lysimachia thyrsiflora was prevalent on both P5P and P5T but not in other patches. Both P4P and P4T had communities distinct from other transects due to the high IV for M. trifoliata. Carex stricta and S. latifolia caused transect D5 to be an outlier from other diffuse transects that were otherwise influenced greatly by C. lasiocarpa (Table 3).
Plot of five tangential (T) and perpendicular (P) transects for Typha patches (P) (circled) and six diffuse (D) transects derived from NMS ordination of plant communities sampled at the Kakagon Sloughs in 1998. Manual settings, six initial axes, Sorenson distance, no species weighting, final stress = 7.53, final instability = 0.00004, number of iterations = 250. Axis 1 accounted for 79.1% of total variation and Axis 2 = 14.7%
Axis 1 of the NMS for 1998, 2005, and 2013 vegetation data accounted for 52.1% of total variation and Axis 2 for 29.3% (Fig. 6). The ordination showed pairing of tangential and perpendicular transects in all years. Changes in plant communities resulted in increased Axis 2 scores from year to year for all but Patch 4. Relatively lower density of invasive Typha spp. and influence of species typical of wetter conditions, such as Utricularia spp., Equisetum fluviatile (water horsetail), and M. trifoliata (Tables 1 and 2), caused Patches 4 and 5 to have greater Axis 1 scores and thus plot to the right in the ordination figure.
Plot of five tangential (T) and perpendicular (P) transects for Typha patches (P) (circled) and six diffuse (D) transects derived from NMS ordination of plant communities sampled at the Kakagon Sloughs in 1998 (a), 2005 (b), and 2013 (c). Successional vectors connect transects sampled across years. Increases on Axis 2 shown by vectors are related to increased Typha. Manual settings, six initial axes, Sorenson distance, no species weighting, final stress = 9.65, final instability = 0.0004, number of iterations = 250. Axis 1 accounted for 52.1% of total variation and Axis 2 = 29.3%
Increases in Axis 2 scores across years for Patches 1, 2, and 3, as shown by arrows in Fig. 6, were driven by increases in Typha spp. and decreases in C. lasiocarpa, S. latifolia, and S. eurycarpum,; increases in C. canadensis in Patches 2 and 3; and decreases in C. palustre and increases in C. lacustris and C. utriculata in Patch 3. Minor changes in Axis 2 scores for Patch 4 transects in 2013 were related to increased Typha spp. and E. fluviatile, as well as decreased Utricularia spp. By 2013, Patch 4 transects were more similar to the composition of the 1998 community of Patch 5. Changes in Axis 2 scores for Patch 5 were substantial from 1998 to 2005 and less so in 2013. Chamaedaphne calyculata and non-Sphagnum moss increased in both sampling years, while C. lasiocarpa, L. thrysiflora, and S. latifolia decreased. Myrica gale increased greatly and Utricularia spp. decreased in 2013. Although Typha spp. increased from 1998 to 2005, the decrease in 2013 had little effect on plotting in Fig. 6.
Diffuse transects again grouped together in 1998, except for D5a, and with that D5 exception mostly grouped and shifted together in 2005 and 2013 (Fig. 6). The native T. latifolia varied little among these transects over time. In addition to C. stricta, increased C. canadensis also affected plotting of transect D5c in 2013 (Table 3). Changes from 1998 to 2013 on the other diffuse transects that moved plottings from 1998 included increased C. lacustris and C. palustre.
Changes in Water Depth
Mean water depths, as measured in quadrats during sampling, were greater on Patch 1 than on most other tangential transects in 1998 and 2005, although Patch 4 depths were also greater in 2005 (Table 1). In 2013, mean water depths were smaller than 2005 on all tangential transects but in Patch 5. Mean water depths on perpendicular transects for Patches 2 and 3 were less than for other patches in all years (Table 2). No notable changes in mean water depth occurred across years on perpendicular transects except for decreases in Patches 4 and 5 in 2013. Diffuse transect D5 had lower mean water depths than other transects in 1998 and 2005. Increased mean water depths on D4 and D6 in 2005 were followed by much smaller depths in 2013.
Discussion
Mechanism of Typha Invasion
Typha angustifolia may get established in uninvaded areas from seed (van der Valk and Davis 1978; Keddy and Reznicek 1986), and with T. latifolia present, the various hybrids of T. × glauca can become established (Travis et al. 2010, 2011). Typha × glauca may also be fertile and expand by seed (Kirk et al. 2011; Pieper et al. 2017, 2020), although clonal expansion has been shown to be important (Pieper et al. 2020). Typha angustifolia and especially T. × glauca can expand clonally by vegetative growth from rhizomes; Boers and Zedler (2008) found clone diameters of T. × glauca to expand about 3.9 m/year and T. angustifolia to expand 2.5 m/year in Wisconsin wetlands. Our results show that vegetative expansion of patches is occurring in the sedge-dominated Kakagon Slough peatlands of Lake Superior, as new Typha ramets in later years were generally found in close proximity to patches consistent with vegetative growth. However, new patches were observed in 2005 and 2013, suggesting that seed-sourcing still occurs; extremely low water levels in 2007 exposed mud flats along the river, where Typha seedlings were later observed (Erickson 2010). Rhizomes may also have been distributed elsewhere during powerful storm and flooding events. As small patches expanded through time, the invasion front also lengthened and accelerated invasion. Mitchell et al. (2011) found that clones of Typha spp. in a dune and swale wetland along the western shore of Lake Michigan expanded from 1955 to 2007, but increases in stem density and litter mass occurred mostly within the first ten years. In comparison, most increases in ramet density along our patch transects at the Kakagon Sloughs occurred in early years 1998 to 2005, and density was often reduced in 2013. Increases in patch size at the Bad River Sloughs were not great between 1998 and 2005 but increased greatly from 2005 to 2013. Based on the Mitchell et al. (2011) conclusions, this suggests that Typha invasion at Bad River was just being initiated in 1998, and the boom in expansion did not begin until 2005.
Alteration of Native Plant Communities
Typha invasion may result in loss of less competitive species and decreases in diversity (Wisheu and Keddy 1992, Boers et al. 2007). Mean number of taxa was greater on diffuse transects in all years than among transects tangential and perpendicular to patches. A decrease in taxa number occurred among tangential and perpendicular transects in 2013. The exception was Patch 4, where taxa may have decreased in abundance as a result of invasion by Typha spp. but were not lost. The native T. latifolia changed little among diffuse transects over the 15-year period of study.
Typha spp. increased on tangential and perpendicular transects of the patches delineated in 1998, but the question is whether or not this was at the expense of the relative abundance of other taxa. The smaller-statured, narrow-leaf sedges may be in jeopardy from cattail invasion, as has been documented elsewhere (Wilcox et al. 1984; Woo and Zedler 2002; Frieswyk and Zedler 2007; Wilcox et al. 2008; Boers and Zedler 2008; Meeker and Fewless 2008). Our data show that increases in mean IV of Typha spp. were accompanied by decreases in mean IV of C. lasiocarpa across years on tangential patch transects (25.0 to 19.2 to 14.9) and perpendicular patch transects (20.2 to 18.9 to 12.0) but decreased only slightly in 2013 on diffuse transects (18.5 to 19.9 to 14.5). Thus, competition is likely a determining factor. In addition to competition for light, increased ramet density on patch transects may have resulted in a reduction in space for occupancy by sedges. Another component of competition may be litter from dead Typha, which has noted effects on sedges (Tuchman et al. 2009; Vaccaro et al. 2009; Farrer and Goldberg 2009; Lishawa et al. 2010; Larkin et al. 2012). Our dead Typha ramet counts, as a proxy for litter, increased substantially on tangential and perpendicular patch transects and exceeded counts for live stems on all transects in both 2005 and 2013.
Despite general trends of increases in Typha spp. and decreases in C. lasiocarpa across years, Patches 4 and 5 defied those trends. Numbers of taxa increased on both tangential and perpendicular transects of Patch 4 across years. Carex lasiocarpa IV increased in 2005 and then decreased slightly in 2013. Typha was much less dominant on the tangential transect of Patch 4 than for other patches in 2005 and 2013 (both IV and ramet count), so perhaps Patch 4 had not yet reached maximum expansion rate. Greater ground cover of M. trifoliata may also have been sufficient to suppress Typha expansion and give C. lasiocarpa a competitive edge. Water depths were greater on the Patch 4 tangential transect in 2005 but not deep enough to impact Typha (e.g., Grace and Harrison 1986). Although management cutting of Typha was initiated adjacent to Patch 4 by the Bad River tribe in 2006, it was not near the Patch 4 transects.
On both tangential and perpendicular transects of Patch 5, Typha spp. IV decreased from 2005 to 2013, and C. lasiocarpa IV did not decrease on the tangential transect. Changes in water depth were not sufficient to cause vegetative changes, so perhaps competition from C. calyculata, M. gale, and non-Sphagnum moss affected Typha growth in 2013.
Sagittaria latifolia, S. eurycarpum, and Utricularia spp. decreased in tangential and perpendicular patch quadrats across years, while only Utricularia showed changes in diffuse quadrats where there is no competition from Typha. This suggests that loss of the two emergent species is due to competition from Typha (see Tulbure et al. 2007), while the submergent species were affected by lake-level changes.
Vegetation Response to Lake-Level Changes
Water-level changes are a significant driver of vegetative changes in wetlands (Keddy and Reznicek 1986; Wilcox 2004; Keddy 2010), and fluctuations on Lake Superior have exceeded one meter. We measured water depths in all of our quadrats at the time of sampling in July. Differences across years were not consistent for all transects, so they were likely related to minor topographic differences rather than lake levels, which were similar during sampling years. At the time of sampling in 1998, Lake Superior peak water levels (183.43 m IGLD 1985) had dropped 0.3 m from a moderate high in 1997 (183.73 m). They were relatively stable until sampling in 2005 (183.43 m), and 2013 lake levels were similar (183.39 m). However, lake levels in the three years prior to 2013 sampling were as much as an additional 0.19 m lower, and 2007 lake levels (183.01 m) were 0.38 m lower than during 2013 sampling. The extreme 2007 low may have induced establishment of Typha seedlings on river-edge mudflats (Erickson 2010), and along with dewatering during the 2010–2012 low lake levels, resulted in loss of submergent Utricularia (Gathman et al. 2005). Sagittaria latifolia and S. eurycarpum would not have been affected by low lake levels, as they can survive in moist soils, as can Carex and Typha (Swink and Wilhelm 1979; Voss and Reznicek 2012).
Prognosis for Future
One year following our last sampling, Lake Superior water levels entered a longer-term high period (Argyilan et al. 2005; Johnston et al. 2012, 2014), with peak summertime levels averaging about 183.75 m from 2014 to 2019 and a 2020 peak of 183.86 m. Water depths of 0.36 to 0.49 m greater than in 2013 could flood both sedge meadow species and cattails to depths greater than 0.5 m. Typha may withstand this flooding until the next low-lake-level period arrives (Harris and Marshall 1963; Bedish 1967; Grace and Harrison 1986; Kercher and Zedler 2004; Boers et al. 2007), but there could be a change in species composition in sedge meadows (Kercher and Zedler 2004; Gathman et al. 2005; Boers et al. 2007), although perhaps an increase in C. lasiocarpa (Gathman et al. 2005). Higher lake levels have been shown to increase Typha invasion in Lake Ontario wetlands at the expense of sedge meadows (Wilcox et al. 2008; Wilcox and Bateman 2018; Smith et al. 2021) and have had similar effects elsewhere, especially if water levels are stabilized (Waters and Shay 1990, 1992, Shay et al. 1999, Boers and Zedler 2008). On the other hand, extended low lake levels may result in a return of sedge meadows (Quinlan and Mulamoottil 1987; Wilcox et al. 2008); with no invasive species present, this cyclical vegetative change has been occurring in Great Lakes wetlands for centuries (Keddy and Reznicek 1986; Wilcox 2004; Keddy 2010). However, invasive Typha is now present, so the future of sedge meadows in peatlands of Lake Superior could be dependent on lake-level cycles, the frequency and severity of extreme flood events like the July 2016 event (Fitzpatrick et al. 2017), and further human management actions.
Delineating perimeters and sampling on transects of patches over a 15-year period helps provide information about the dynamics of Typha invasion of relatively un-degraded peatlands of Lake Superior. It is clear from these data that Typha spp. is responding much differently than T. latifolia, which behaves as “one of many” taxa in these complex sedge meadows, much as in the Long Island wetland complex of Lake Superior (Meeker 1998). In that study, T. latifolia in swales decreased slightly as water levels in Lake Superior drew down and increased slightly as water levels increased—a cyclic response. It appears, however, that Typha spp. is not responding cyclically and is undergoing a directional change (steadily increasing). This poses questions about how the sedge meadow and coastal fen communities will look in years to come, and additionally, how these changes in the sedge meadow communities will reverberate through the system. How much of the total sedge meadow community is in this changing status? That is, how much invasive cattail is out there among Lake Superior coastal wetlands? The answers to this question require a broader aerial photographic analysis. F1 hybrids of T. × glauca tend to have greater fitness and are more prevalent than later backcrossed hybrids (Freeland et al. 2013), making them more likely to invade. However, creation of F1 hybrids may be limited in the future if T. angustifolia is less common as a result of both genetic and demographic swamping. Given the tendency for fast Typha expansion in the first decade after establishment (Mitchell et al. 2011), land managers of Lake Superior coastal wetlands should scout for new patches on a regular basis and, if feasible or desired, begin treatment soon after discovery (see Bansal et al. 2019).
Data Availability
All data produced from this study are provided in this manuscript.
References
Albert DA, Minc LD (2004) Plants as regional indicators of great lakes coastal wetland health. Aquat Ecosyst Health Manag 7:233–247
Albert DA, Wilcox DA, Ingram JW, Thompson TA (2005) Hydrogeomorphic classification for great lakes coastal wetlands. Journal of Great Lakes Research 31(Suppl 1):129–146
Argyilan EP, Forman SL, Johnston JW, Wilcox DA (2005) Optically stimulated luminescence dating of late Holocene raised strandplain sequences adjacent to southern Lake Superior, Upper Peninsula, Michigan, USA. Quaternary Research 63:122–135
Bansal S, Lishawa S, Newman S, Tangen B, Wilcox DA, Albert DA, Anteau M, Chimney M, Cressey R, DeKeyser S, Elgersma K, Finkelstein SA, Freeland J, Grosshans R, Klug P, Larkin D, Lawrence B, Linz G, Marburger J, Noe G, Otto C, Reo N, Richards J, Richardson CJ, Rogers L, Schrank A, Svedarsky D, Travis S, Tuchman N, van der Valk AG, Windham-Myers L (2019) Typha (cattail) invasion in north american wetlands: biology, regional problems, impacts, desired services, and management. Wetlands 39:645–684
Bedish JW (1967) Cattail moisture requirements and their significance to marsh management. The American Midland Naturalist 78:288–300
Boers AM, Veltman RLD, Zedler JB (2007) Typha × glauca dominance and extended hydroperiod constrain restoration of wetland diversity. Ecological Engineering 29:232–244
Boers AM, Zedler JB (2008) Stabilized water levels and Typha invasiveness. Wetlands 28:676–685
Carlson ML, Kowalski KP, Wilcox DA (2009) Promoting species establishment in a Phragmites-dominated great lakes coastal wetland. Natural Areas Journal 29:263–280
Ellison AM, Bedford BL (1995) Response of a wetland vascular plant community to disturbance: a simulation study. Ecological Applications 5:109–123
Epstein E, Smith W, Judziewicz E (1997) Priority wetland sites of Wisconsin’s Lake Superior basin. Wisconsin Department of Natural Resources - Bureau of Endangered Resources, Madison
Erickson S (2010) Cattail problems: hybrid cattail crowds out manoomin. Mazina’igan; a Chronicle of the Lake Superior Ojibwe. Eau Claire Company, Eau Claire, WI, p 5
Farrer EC, Goldberg DE (2009) Litter drives ecosystem and plant community changes in cattail invasion. Ecological Applications 19:398–412
Fitzpatrick FA, Dantoin ED, Tillison N, Watson KM, Waschbusch RJ, Blount JD (2017) Flood of July 2016 in Northern Wisconsin and the Bad River Reservation. U.S. Geological Survey Scientific Investigations Report 2017–5029. https://doi.org/10.3133/sir20175029
Freeland J, Ciotir C, Kirk H (2013) Regional differences in the abundance of native, introduced, and hybrid Typha spp. in northeastern North America influence wetland invasions. Biological Invasions 15:2651–2665
Frieswyk CB, Zedler JB (2007) Vegetation change in great lakes coastal wetlands: deviation from the historical cycle. Journal of Great Lakes Research 33:366–380
Galatowitsch SM, Anderson NO, Ascher PD (1999) Invasiveness in wetland plants in temperate North America. Wetlands 19:733–755
Gathman JP, Albert DA, Burton TM (2005) Rapid plant community response to a water level peak in northern Lake Huron coastal wetlands. Journal of Great Lakes Research 31(Suppl 1):160–170
Grace JB, Harrison JS (1986) The biology of Canadian weeds.: 73. Typha latifolia L., Typha angustifolia L. and Typha × glauca Godr. Canadian Journal of Plant Sciences 66:361–379
Harris SW, Marshall WH (1963) Ecology of water-level manipulations on a northern marsh. Ecology 44:331–343
Johnston JW, Argyilan EP, Thompson TA, Baedke SJ, Lepper K, Wilcox DA, Forman SL (2012) A sault-outlet-referenced mid- to late-holocene paleohydrograph for Lake Superior constructed from strandplains of beach ridges. Canadian Journal of Earth Sciences 49:1263–1279
Johnston JW, Thompson TA, Wilcox DA (2014) Paleohydrographic reconstructions from strandplains of beach ridges in the Laurentian Great Lakes. In: Martini IP, Wanless HR (eds) Sedimentary Coastal Zones from High to Low Latitudes: Similarities and Differences. Geological Society, London, UK. Special Publication 388, pp 213–228
Keddy PA (2010) Wetland Ecology: principals and conservation, second. Cambridge University Press, Cambridge
Keddy PA, Reznicek AA (1986) Great lakes vegetation dynamics: the role of fluctuating water levels and buried seeds. Journal of Great Lakes Research 12:25–36
Kercher SM, Zedler JB (2004) Flood tolerance in wetland angiosperms: a comparison of invasive and noninvasive species. Aquatic Botany 80:89–102
Kirk H, Connolly C, Freeland JR (2011) Molecular genetic data reveal hybridization between Typha angustifolia and Typha latifolia across a broad spatial scale in eastern North America. Aquatic Botany 95:189–193
Larkin DJ, Freyman MJ, Lishawa SC, Geddes P, Tuchman NC (2012) Mechanisms of dominance by the invasive hybrid cattail Typha × glauca Biological Invasions 14:65–77
Lishawa SC, Albert DA, Tuchman NC (2010) Water-level decline promotes Typha × glauca establishment and vegetation change in great lakes coastal wetlands. Wetlands 30:1085–1096
McCune B, Mefford MJ (2016) PC-ORD: multivariate analysis of ecological data. Version 7. MjM Software Design, Gleneden Beach
McDonald ME (1955) Cause and effects of a die-off of emergent vegetation. J Wildl Manage 19:24–35
Meeker J (1998) Wetlands of Long Island, Apostle Islands National Lakeshore. Report to the Apostle Island National Lakeshore, Bayfield
Meeker J, Fewless G (2008) Change in Wisconsin’s coastal wetlands. In: Waller D, Rooney T (eds) Vanishing Present, Wisconsin’s changing Lands, Water, and Wildlife. University of Chicago Press, Chicago, pp 183–192
Mitchell ME, Lishawa SC, Geddes P, Larkin DJ, Treering D, Tuchman NC (2011) Time-dependent impacts of cattail invasion in a great lakes coastal wetland complex. Wetlands 31:1143–1149
Odum EP (1971) Fundamentals of ecology, 3rd edn. W.B. Saunders, Philadelphia
Pieper S, Dorken M, Freeland J (2020) Genetic structure of hybrids and progenitors provides insight into processes underlying an invasive cattail (Typha × glauca) hybrid zone. Heredity 124:714–725
Pieper SJ, Nicholls AA, Freeland JR, Dorken ME (2017) Asymmetric hybridization in cattails (Typha spp.) and its implications for the evolutionary maintenance of native Typha latifolia The Journal of Heredity 108:479–487
Quinlan C, Mulamoottil G (1987) The effects of water-level fluctuations on three Lake Ontario shoreline marshes. Canadian Water Resources Journal 12:64–77
Seabloom EW, Moloney KA, van der Valk AG (2001) Constraints on the establishment of plants along a fluctuating water-depth gradient. Ecology 82:2216–2232
Shay JM, de Geus PMJ, Kapinga MRM (1999) Changes in shoreline vegetation over a 50-year period in the Delta Marsh, Manitoba in response to water levels. Wetlands 19:413–425
Smith IM, Fiorino GE, Grabas GP, Wilcox DA (2021) Wetland vegetation response to record-high Lake Ontario water levels. Journal of Great Lakes Research 47:160–167
Squires L, van der Valk AG (1992) Water-depth tolerances of the dominant emergent macrophytes of the Delta Marsh, Manitoba. Canadian Journal of Botany 70:1860–1867
Stewart D (2021) Undetected but widespread: the cryptic invasion of non-native cattail (Typha) in the Fraser River Estuary. Ph.D. dissertation, University of British Columbia, Vancouver
Swink F, Wilhelm G (1979) Plants of the Chicago Region. The Morton Arboretum, Lisle
Tangen BA, Bansal S, Freeland JR, Travis SE, Wasko JD, McGonigle TP, Goldsborough LG, Gow K, Marburger JE, Meier JA (2022) Distributions of native and invasive Typha (cattail) throughout the Prairie Pothole Region of North America. Wetlands Ecology and Management 30:1–17
Thiet RK (2002) Diversity comparisons between diked and undiked coastal freshwater marshes on Lake Erie during a high-water year. Journal of Great Lakes Research 28:285–298
Travis SE, Marburger JE, Windels S, Kubátová B (2010) Hybridization dynamics of invasive cattail (Typhaceae) stands in the Western Great Lakes Region of North America: a molecular analysis. Journal of Ecology 98:7–16
Travis SE, Marburger JE, Windels SK, Kubátová B (2011) Clonal structure of invasive cattail (Typhaceae) stands in the upper midwest region of the US. Wetlands 31:221–222
Tuchman NC, Larkin DJ, Geddes P, Wildova R, Jankowski K, Goldberg DE (2009) Patterns of environmental change associated with Typha × glauca invasion in a great lakes coastal wetland. Wetlands 29:964–975
Tulbure MG, Johnston CA, Auger DL (2007) Rapid invasion of a great lakes coastal wetland by non-native Phragmites australis and Typha Journal of Great Lakes Research 33(Suppl 3):269–279
Vaccaro LE, Bedford BL, Johnston CA (2009) Litter accumulation promotes dominance of invasive species of cattails (Typha spp.) in Lake Ontario wetlands. Wetlands 29:1036–1048
van der Valk AG, Davis CB (1978) The role of seed banks in the vegetation dynamics of prairie glacial marshes. Ecology 59:322–335
Voss EG, Reznicek AA (2012) Field manual of Michigan Flora. University of Michigan Press, Ann Arbor
Waters I, Shay JM (1990) A field study of the morphometric response of Typha glauca shoots to a water depth gradient. Canadian Journal of Botany 68:2339–2343
Waters I, Shay JM (1992) Effect of water depth on population parameters of a Typha glauca stand. Canadian Journal of Botany 70:349–351
Wilcox DA (2004) Implications of hydrologic variability on the succession of plants in great lakes wetlands. Aquatic Ecosystem Health & Management 7:223–232
Wilcox DA (2012) Response of wetland vegetation to the post-1986 decrease in Lake St. Clair water levels: seed-bank emergence and beginnings of the Phragmites australis invasion. Journal of Great Lakes Research 38:270–277
Wilcox DA, Apfelbaum SI, Hiebert RD (1984) Cattail invasion of sedge meadows following hydrologic disturbance in the Cowles Bog Wetland Complex, Indiana Dunes National Lakeshore. Wetlands 4:115–128
Wilcox DA, Bateman JA (2018) Photointerpretation analysis of plant communities in Lake Ontario wetlands following 65 years of lake-level regulation. Journal of Great Lakes Research 44:1306–1313
Wilcox DA, Bateman JA, Kowalski KP, Meeker JE, Dunn N (2022) Extent of sedge-grass meadow in a Lake Michigan drowned river mouth wetland dictated by topography and lake level. Wetlands 42:34
Wilcox DA, Buckler K, Czayka A (2018) Controlling cattail invasion in sedge/grassmeadows. Wetlands 38:337–347
Wilcox DA, Kowalski KP, Hoare HL, Carlson ML, Morgan HN (2008) Cattail invasion of sedge/grass meadows in Lake Ontario: photointerpretation analysis of sixteen wetlands over five decades. Journal of Great Lakes Research 34:301–323
Wilcox D, Nichols SJ (2008) The effect of water-level fluctuations on plant zonation in a Saginaw Bay, Lake Huron wetland. Wetlands 28:487–501
Woo I, Zedler JB (2002) Can nutrients alone shift a sedge meadow towards dominance by the invasive Typha × glauca? Wetlands 22:509–521
Acknowledgements
Funding for fieldwork in 1998, 2005, and 2013 was provided by the Bad River Band of Lake Superior Chippewa to J. Meeker (and S. Johnson co-PI in 2013). Staff of the Bad River Band that provided logistical support include E. Soulier and J. Strand. The Band’s seasonal staff provided boat transportation and guide support. Dale Soltis and Jeremy Ridlbauer assisted in the 1998 field sampling, V. Hofman shared sampling in 2005, and J. Elias and M. Hogfeldt helped sample in 2013. T. King generated the GIS figures in 2005, and J. Mead generated the GIS figures and conducted spatial analysis in 2013. Manuscript figures were created with assistance from A. Graham and G. Lawrence. We thank R. Schultz for a helpful review of a draft of the manuscript. We also thank Joan Elias for accessing and providing some data sets that were determined to be necessary during preparation of this paper after the passing of her husband, senior author Jim Meeker. The scientific community, especially wetland scientists in the Great Lakes, suffered a great loss when this kind, passionate, and insightful naturalist was taken from us.
Funding
Funding for fieldwork in 1998, 2005, and 2013 was provided by the Bad River Band of Lake Superior Chippewa.
Author information
Authors and Affiliations
Contributions
Meeker and Wilcox conceived study. Meeker and Johnson conducted data collection and did data analyses. Tillison provided critical site information and coordinated field efforts. Wilcox wrote much of the manuscript with assistance from other authors on selected parts.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Consent provided by Mike Wiggins, Chairman, Bad River Band of Lake Superior Chippewa.
Conflicts of Interest/Competing Interests
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
James E. Meeker is deceased.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Meeker, J.E., Wilcox, D.A., Johnson, S.E. et al. Tracking Vegetation Transitions Due to Invasion of Cattail (Typha) in Lake Superior Coastal Peatlands. Wetlands 43, 18 (2023). https://doi.org/10.1007/s13157-023-01664-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13157-023-01664-9