Abstract
Mangrove ecosystems have high carbon storage and sequestration rates and become substantial sources of greenhouse gases when disturbed by land-use change. Thus, they are extremely valuable for inclusion in climate change mitigation and adaptation strategies. However in Kerala, a west coast state of India, has lost 95% of its mangroves in the last three decades, posing a serious threat to global climate. The regional carbon stock data of mangroves that are at risk of depletion are rarely reported, despite the fact that they are crucial for mitigating and managing climate change impacts. In response, the study estimated the ecosystem carbon stocks and soil organic carbon sources of three different estuarine mangrove habitats of Kerala. The mean total ecosystem carbon stock of Kerala mangroves was estimated to be 218.98 ± 169.86 Mg C ha− 1 which is equivalent to 803.66 ± 621.47 Mg CO2 ha− 1, contributing a substantial amount of carbon to the global ecosystem carbon. Further 88% of the estimated ecosystem carbon stock was represented by vegetation biomass and 22% by the soil carbon stock. The stable isotopic signatures revealed that the accumulated autochthonous mangrove source attributed to the organic carbon in the soils of site 1 (Munroe island) and site 3 (Vypin) while the suspended organic matter in tidal water contributed to the soil organic carbon of site 2 (Ayiramthengu) mangroves. Mangrove structure, salinity, soil pH and bulk density were found to be the correlating factors for the carbon stock variations across the study sites. Hence, the understanding of the amount of carbon stocks in the mangroves of Kerala coupled with other ecosystem services they offer highlights their importance in the creation of conservation, restoration and climate change mitigation plans in the country.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mangrove ecosystems occupy only 0.17% of Earth’s continental area (∼137,600km2) (Bunting et al. 2018), but they are among the most carbon-rich forests on the planet (Bouillon et al. 2008a; Donato et al. 2011; Atwood et al. 2017). Mangroves, unlike terrestrial forests, are capable of storing vast quantities of carbon in their soils over centuries as these unique ecosystems have higher carbon burial rates and a thousand times slower soil C turnover rates than terrestrial forests due to the complex root structures, high sedimentation rates, and anoxic soils (Mcleod et al. 2011; Alongi 2012). This long-term capacity to store significant quantities of soil carbon (5–10.4 Pg globally) (Duarte et al. 2013; Jardine and Siikamaki 2014) for centuries makes them essential carbon sinks. Furthermore, reducing or preventing greenhouse gas (GHG) emissions caused by the depletion of these soil carbon reserves is considered as a low-cost alternative for mitigating climate change (Siikamaki et al. 2012; Murdiyarso et al. 2015; Howard et al. 2017).
Studies revealed that 8–20% of global anthropogenic carbon dioxide emission was contributed by land-use change and deforestation, next to the combustion of fossil fuels (van der Werf et al. 2009; Arifanti et al. 2019). In this context, Reduced Emissions from Deforestation and Degradation (REDD+) has been highlighted in the recent international climate agreements as a crucial and reasonably cost-effective alternative for climate change mitigation and adaptation (Adame et al. 2021). The major objective of this program is to conserve terrestrial carbon stocks by providing financial incentives for the protection and conservation of forest ecosystems. However, REDD + and other CCMA programs need a regular evaluation of carbon stocks and emissions (IPCC, 2007), highlighting the relevance of robust carbon storage valuation for various forest types, especially those having high C density coupled with extensive land-use change (Keith et al. 2009).
Mangrove forests, found along the coasts of most major oceans in 124 tropical and subtropical countries (Bunting et al. 2018) are facing a multitude of anthropogenic threats such as coastal development, aquaculture expansion, and pollution which in turn resulting in large scale global destruction (Alongi 2002; Polidoro et al. 2010; Giri et al. 2011; Murdiyarso et al. 2015; Kauffman et al. 2018). Rapid sea-level rise in the twenty-first century has also been identified as a major threat to mangroves (Gilman et al. 2008), which have adapted to past sea-level rise by migrating landward or upward (Alongi 2008; Lovelock et al. 2015). Over the last 60 years, more than one-third of the world’s mangroves have been disappeared (Alongi 2002; Hamilton and Casey 2016). Although many countries have adopted several conservation initiatives, mangroves continue to be lost at a global pace of about 0.2% each year (Hamilton and Casey 2016). This loss of mangroves around the world creates uncertainty about the fate of the huge amounts of carbon deposited in their soils since the degradation and loss of coastal vegetation may lead to the disruption of soil carbon down to 1 m depths, causing it to remineralize to CO2 (Pendleton et al. 2012). Further, the remineralization of mangrove soil carbon may considerably contribute to the part of anthropogenic GHG emissions labeled as ‘land-use change’ which is currently not documenting in the carbon estimations across the globe (IPCC Climate change 2007).
The International Panel on Climate Change (IPCC) release the guidelines for quantifying and reporting stocks and emissions includes those arising from mangroves and other blue carbon habitats (IPCC 2014). Enhancing the data collection of carbon stock at regional level and conservation initiatives of these carbon stocks is also encouraged by the Paris Agreement for increasing natural C sinks to prevent climate change. This reflects the growing awareness of the importance of mangrove ecosystem conservation and restoration in GHG emissions reduction strategies. However, data are scarce on the full extent of ecosystem carbon stocks that are vulnerable to depletion. Even though mangroves are recognized for their high carbon assimilation and flux rates (Kristensen et al. 2008; Komiyama et al. 2008; Bouillon et al. 2008a), information on the total ecosystem carbon stock, the amount that will be released with land-use change, is surprisingly scarce. While only a few components of carbon stock have been reported, the most notable of which is vegetation biomass (Twilley et al. 1992; Komiyama et al. 2008), but evidence of deep organic-rich soils (Eong 1993; Matsui 1998; Fujimoto et al. 1999) indicates that these inventories ignore the vast majority of total ecosystem carbon.
Kerala, located on the west coast of India, has 44 rivers as well as a vast network of estuaries and backwaters with tidal action, once had 700 km2 of mangroves along its coast (Ramachandran et al. 1986) but now declined to 9 km2 (FSI 2019) indicating that 95% of the mangrove vegetation has declined over the last three decades (Sreelekshmi et al. 2021). Altogether 18 species of true mangrove species were reported from Kerala of which Avicennia officinalis and Rhizophora mucronata are the most common species whereas Ceriops tagal, Avicennia alba and Sonneratia alba are rare (Sreelekshmi et al. 2018). In addition, the Kerala mangroves have the potential to contribute a substantial amount of carbon to the global ecosystem carbon reserve (Rani et al. 2021). Further, to be a part of a land-based GHG emission reduction activity, information on C storage and its dynamics is necessary. Considering these facts, our objectives are to estimate the carbon stocks in various compartments of mangrove ecosystems of Kerala and to characterize the historical source of organic carbon in these ecosystems. We hypothesized that Kerala mangroves have a high carbon storage potential in biomass and soil compared to other mangrove systems, and this potential would vary significantly among study sites based on the environmental (soil) characteristics and forest structure.
Materials and Methods
Study Area
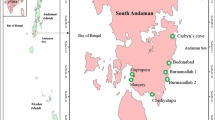
Kerala’s physiographic setting is unique since it is a tiny strip wedged between the Lakshadweep Sea and the Western Ghats, comprising a sequence of lagoons and estuaries. It extends between the latitudes 8º18′ and 12º48′ N and longitudes 71º53′ and 77º24′E, with a total area of 38,864 km2, of which the coastal wetlands make up a quarter i.e., 937.3 km2 (Nair and Sankar 2002). The coastline stretches for around 590 km, with the northern end at Manjeswaram (Kasargod district) and the southern end at Pozhiyar (Thiruvananthapuram district). Asymmetrical landscape, typified by undulating subdued hills and steep slopes, characterizes the shoreline, with altitude ranging from below mean sea level (MSL) to 2694 m above MSL (Jagtap et al. 2004).
Field samplings were conducted on a seasonal basis in three different estuarine mangrove forests in Kerala (Fig. 1) during 2019–2020. The study sites were, Site 1, Munroe island (8° 59’ 27’’ N 76° 36’ 47 ‘’ E), a degrading mangrove forest owing to excessive tourism activities fringing the Ashtamudi estuary which is a Ramsar site in Kerala. The mangrove species seen in this site are Excoecaria agallocha, Avicennia officinalis and Rhizophora mucronata. Site 2, Ayiramthengu (9° 6’ 58’’ N 76° 28’ 49’’ E), a lush mangrove ecosystem that borders the Kayamkulam estuary, a part of Vembanad lake which is also a Ramsar site in Kerala. Avicennia officinalis, Avicennia marina, Avicennia alba, Bruguiera cylindrica, Excoecaria agallocha and Lumnitzera racemosa are some of the primary species found here. Site 3, Vypin/Valappu (10° 0’ 45’’ N 76° 13’ 22’’ E), an island in Cochin estuary, is also a part of Vembanad lake. Bruguiera gymnorhiza, Bruguiera cylindrica, Avicennia officinalis and Excoecaria agallocha made up this mangrove forest.
Environmental Parameters
Soil samples (upto 1 m depth) were collected with a corer (4.6 cm diameter and 120 cm length) in each site and divided into four sections with depth intervals of 0–15, 15–30, 30–50, 50–100. The pH, Eh, and temperature of each section were measured insitu using a portable pH meter (Systronics make), Eh meter (Systronics make) and thermometer (AOAC 2000). The salinity and pH of the porewater were also measured in-situ using a hand held refractometer (make:Atago S/Mill-E) and pH meter (Systronics make). The textural analysis (Folk 1980) and bulk density measurement were carried out according to standard procedure. A known volume of the soil sample was dried to constant weight at 1050 C in a hot air oven for determining the bulk density. Bulk density (g cm− 3) = Dry soil weight (g) / Soil volume (cm3). The total nitrogen content of the soil was analyzed using the Kjeldahl digestion method and nitrogen distillation equipment (Anderson and Ingram 1993).
Structural Analysis
Fixed-area plot measurement was used to characterize structural attributes of the mangrove vegetation, as described by Cintrón & Schaffer-Novelli (1984). Four transects perpendicular to the waterline were put at 50 m intervals in each site and five quadrats (10 × 10 m) were laid at 50 m intervals in each transect, taking into account mangrove diversity and accessibility. Using a measuring tape, the dbh (diameter at breast height) of each species in the quadrats were determined. As suggested by Cintrón and Schaffer-Novelli (1984) trunk density, basal area and species dominance (% basal area) were estimated.
Biomass Stock Estimation
Living Biomass
The aboveground and belowground vegetation biomasses were calculated using the species-specific allometric equations established by Komiyama et al. (2005). Both formulae are based on Diameter at Breast height (DBH) and wood density of mangrove species. For belowground biomass calculations, species-specific wood density (g cm− 3) was derived from the wood density database (Zanne et al. 2009). The mangrove biomass was transformed into carbon stock by multiplying it by a factor of 0.47 for aboveground biomass and 0.39 for belowground biomass (Kauffman and Donato 2012).
Dead Biomass Stock
Depending on the state of decay, dead trees were classified into one of three classes (I, II and III) (Kauffman and Donato 2012). Class I dead trees are those that have recently died but still have the bulk of their primary and secondary branches, whereas Class III dead trees have only the main trunk and have lost all of their branches. The dead trees with primary branches attached to the main trunk were allocated to the Class II category. The biomass of dead trees was computed depending on their decay class. Status I dead trees were estimated to be 97.5% of a live tree’s biomass, status II dead trees were expected to be 80% of a live tree’s biomass, and status III trees were projected to be 50% of a live tree’s biomass (Kauffman and Donato 2012). Biomass of downed wood was converted to carbon mass using biomass to carbon conversion ratios.
Soil Carbon Stock
A PVC corer (4.6 cm diameter and 120 cm length) was used to collect samples from a depth of 100 cm for soil carbon analysis from five quadrats in each transect and the samples were dried and pulverized. Total Carbon (TC), Total Organic Carbon (TOC) were determined from these samples using the TOC analyzer HT 1300 solid module (Analytik Jena make).
Soil Carbon stock (MgC ha− 1) = C con (%) x Bulk density (g cm− 3) x depth (cm).
Source Characterisation of Organic Carbon and Nitrogen
The stable carbon (δ13C) and nitrogen (δ15N) isotopes were determined in each core sample from the three study sites. Air-dried subsamples were acidified with dilute HCl (5%) and then oven-dried at 40 °C to remove the carbonates. The samples were encapsulated in tin capsules and analyzed with an elemental analyzer coupled with an isotope ratio mass spectrometer (EA-IRMS) (Kauffman and Donato 2012) and the stable isotopic composition was reported in δ notation [per mil (‰) units].
Total Ecosystem Carbon Stock and Economic Valuation
The total ecosystem carbon stock was estimated by adding the carbon stock in above-ground biomass, belowground biomass, dead biomass, and soil together. The carbon dioxide (CO2) equivalents, or CO2e was calculated by multiplying the total ecosystem carbon stock with a factor of 3.6 (IPCC, 2007). The social cost of carbon was calculated by multiplying the CO2e with 86 as a ton of CO2 costs US $ 86 (Ricke et al. 2018).
Statistical Analyses
The Kolmogorov–Smirnov (K-S) test was used to determine the normality of the variables and Levene’s test for homogeneity of variance. The variations in the vegetation biomass and carbon stocks between study sites were assessed using two way-ANOVA and if the variation appeared significant (p < 0.05), the Tukey post hoc test was done to evaluate the variation. To analyze the relationships of major environmental characteristics with C stocks, Pearson’s correlation coefficients were calculated in all the study sites. All the statistical analyses were done using SPSS v16 software.
Results and Discussion
Community Structure and Vegetation Biomass
The study stations comprised a total of 8 true mangrove species (Table 1). The density data revealed that site 1 was dominated by Excoecaria agallocha (2100 ± 843 ha− 1), site 2 by Avicennia marina (22,000 ± 3202 ha− 1) while Bruguiera gymnorhiza (4000 ± 3219 ha− 1) dominated site 3. Among the three sites, the tree density was highest in site 2 (4475 ± 8597 ha− 1) followed by site 3 (1950 ± 1843 ha− 1) and site 1 (1233 ± 901 ha− 1). The highest basal area was represented by Rhizophora mucronata (39 ± 53.6 m2 ha− 1) in site 1, Bruguiera cylindrica (56.7 ± 38.7m2 ha− 1) in site 2 and Avicennia officinalis (12.3 ± 17.7 m2 ha− 1) in site 3 (Table 1). The mean density in the study sites ranged from 200 to 22,000 ha− 1and the mean basal area was 0.6–49.3m2 ha− 1 (Table A1) which fell within the range reported from tropics (Trettin et al. 2016, Sreelekshmi et al. 2018; Satyanarayana et al. 2002; Das et al. 2014 and Hinrichs et al. 2009). The mean aboveground,belowground and total biomass of mangroves of Kerala were 130.43 ± 163.88 Mg ha− 1, 423.55 ± 496.96 Mg ha− 1and 553.98 ± 660.75 Mg ha− 1respectively. Further Rhizophora mucronata (595.56 Mg ha− 1) had the maximum total biomass in site 1 and Avicennia marina (1913.31 Mg ha− 1) in site 2 and Avicennia officinalis (120.27 Mg ha− 1) in site 3. Relatively higher mean DBH for Avicennia marina (24.8 ± 4.44 cm) and Bruguiera cylindrica (23.66 ± 13.90 cm) were measured at site 2, resulting in higher above-ground and below-ground biomasses (p < 0.05) than the other two sites (Table 1). Earlier, Suresh et al. (2017) recorded 132.83 ± 97.5 Mg ha− 1biomass from central Kerala, while Vinod et al. (2018) reported 236.56 t ha− 1 biomass from Kadalundi mangroves (North Kerala).
Environmental Parameters
The study stations exhibited relatively low temperatures, neutral pH, highly reducing and mixo-mesohaline soil conditions (Table 2), which matched the findings of Rani et al. (2021); Sreelekshmi et al. (2020a). The soil temperature varied with the seasons ranging from 280 to 310 C with a mean of 29.47 ± 1.040 C. This pattern of temperature variation was due to the seasonal influences of freshwater owing to rainfall, wind force, high intensity of solar radiation and lower atmospheric air temperature (Sahu et al. 2012). The peak salinity was recorded during the pre-monsoon season in all the study sites owing to the low rainfall while lower salinity during monsoon season. The high and low salinity values recorded varied from 4 PSU (Mon, site 3) to 35 PSU (pre mon, Site 2). The high pH value (7.8) was recorded in site 3 during pre monsoon season, and the low value (6) was recorded in site 2 during post monsoon season. These alterations in pH could be attributed to freshwater influx, fluctuations in salinity and temperature (Rajasegar et al. 2002). The granulometric composition revealed silty sand in all the sites. The bulk density varied from 0.35 to 0.85 gcm− 3 and organic carbon content from 11.50 to 52.54 mgg− 1 (Table A2). While the organic carbon and total nitrogen content in all the sites appeared low owing to the sandy nature of the soil. Further site 2 exhibited relatively lower organic carbon (15.78 ± 3.63 mgg− 1) attributed to the continuous tidal flushing and increased salinity. The organic carbon concentrations in the study sites were comparable with the values reported from other mangrove ecosystems like Sundarbans (Rahman et al. 2014; Sreelekshmi et al. 2020b) and Pichavaram (Kathiresan et al. 2021). As indicated in Table 2, all parameters except soil temperature and total nitrogen exhibited significant variations between stations (p < 0.05), whereas all parameters except soil texture and bulk density showed significant differences across seasons (p < 0.05). In comparison to the other two sites, site 2 had higher salinity (Table 2), however in sites 1 and 3 the decrease in salinity resulted in an increase in organic carbon input through mangrove litter (Zhu 2001; Bandyopadhyay et al. 2003; Rahman et al. 2014).
Vegetation Carbon Stock
The mean total vegetation carbon stock in the mangrove ecosystems of Kerala was 194.03 ± 177.51 Mg C ha− 1. The belowground vegetation C pools (142.61 ± 127.67 Mg C ha− 1) were significantly higher (p < 0.05) than the aboveground C pools (51.42 ± 49.87 Mg C ha− 1) in all the sites. The findings matched the vegetation carbon stock reported from Vietnam (Tue et al. 2014), Indonesia (Donato et al. 2011) and Bhittarkanika, India (Banerjee et al. 2020). In comparison to the other two sites, site 2 showed significantly higher total vegetation carbon (p < 0.05). The significant differences in the vegetation carbon stock between sites could be attributable to the variability in the structural characteristics of mangrove stands (Kasawani et al. 2007). The contribution of vegetation to total ecosystem C stocks in site 1, site 2 and site 3 were 90.59%, 95.01% and 45.89% respectively. The study revealed a significant negative correlation between pH and vegetation carbon (r = -0.625, p < 0.05), while a positive correlation between pore water salinity and vegetation carbon (r = 0.709, p < 0.05). Higher vegetation carbon stock was observed in site 2 (Table 2) which showed higher salinity (17.33 ± 15.5psu) and lower vegetation carbon was found in site 3 with lower salinity(7.67 ± 3.79psu).
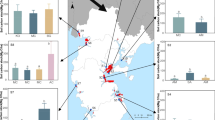
In site 1, Rhizophora mucronata had the highest vegetation carbon stock (242.20MgCha− 1) and Avicennia officinalis (74.39 MgCha− 1) had the lowest, whereas in site 2, Avicennia marina had the highest vegetation carbon stock (781.21 MgCha− 1) and Lumnitzera racemosa (18.77 MgCha− 1) had the lowest. While, in site 3, maximum carbon stock was represented by Avicennia officinalis(48.66 MgCha− 1), and minimum by Bruguiera cylindrica (5.05 MgCha− 1) as shown in Table 1; Fig. 2.
Downed Wood Carbon Stock
The mean downed wood carbon stock in the mangrove ecosystems of Kerala was 3.03 ± 3.26 Mg C ha− 1. Maximum downed wood carbon was recorded in site 2 (5.02 ± 4.75 Mg C ha− 1) followed by site 1 (3.04 ± 1.91 Mg C ha− 1) and site 3 (1.04 ± 1.08 Mg C ha− 1) as shown in Table 3. However, there were no significant variations in downed wood carbon between stations (Table 4). The contribution of downed wood carbon to the total ecosystem carbon stock appeared negligible, (1.68% in site 1, 1.24% in site 2, and 1.49% in site 3). Large rotten wood made up 46% of the downed wood carbon stock whereas medium and small wood only constituted 30% and 24% of the carbon pool, respectively.
Soil Carbon Stock
The mean depth of the mangrove soils across the sites was 1 m. However, the soil organic carbon stock ranged from 9.98 to 66.20 Mg C ha− 1 with a mean value of 22.52 ± 13.70 Mg C ha− 1 (Table 4). The amount of carbon is determined by the size of the soil particles. Fine silt and clay particles have greater carbon retention capacity due to their larger surface area than sand particles (Kauffman and Bhomia 2017; Kathiresan et al. 2021). Thus, the sandy nature of the soil in all the sites attributed to the relatively low carbon stock. A significant positive correlation (r = 0.648, p < 0.05), was found between the bulk density and organic carbon stock in the study sites while a significant negative correlation was observed for pH (r = -0.693, p < 0.05) and salinity (r = -0.715, p < 0.05) with the soil organic carbon stocks demonstrating that relatively low pH and salinity benefits the accumulation of organic matter.
Earlier reports revealed that mangrove ecosystems are important carbon sinks and that the soil OC pool accounts for the majority of ecosystem OC stock (Twilley et al. 1992; Donato et al. 2011; Murdiyarso et al. 2015; Rovai et al. 2018). However, in this investigation, samples were obtained from the top 1 m of the soil, resulting in much lower estimates of soil carbon stocks. Murdiyarso et al. (2015) found that soil OC density did not vary significantly with depth in mangrove habitats and that the drop in soil OC concentration with depth was compensated by the increased bulk density. Further, the top 1 m soil OC stock in this study (9.98–66.20 Mg C ha− 1), was within the range recorded in other mangrove wetlands of India (Table 5). The mean soil organic carbon content recorded in the present study (23.26 mgg− 1) was found to be greater than the global mean value (20 mgg− 1) for mangroves (Kristensen et al. 2008), demonstrating that Kerala mangroves have a significant soil carbon stock. Further, Donato et al. (2011) found that estuarine sites have relatively lower soil carbon content (with a mean of 7.9%) than oceanic sites (with a mean of 14.6%) when comparing the soil OC stock of different mangrove ecosystems in the tropics.
Stable Isotopic Signatures of Soil Organic Carbon Sources
Earlier reports revealed that the majority of organic matter in undisturbed mangrove soils comes from autochthonous sources, as determined by carbon (δ13C) and nitrogen (δ15N) stable isotope signatures (Ranjan et al. 2011; Stringer et al. 2016; Xiong et al. 2018; Li et al. 2018) as well as the ratio of total organic carbon to total nitrogen (C/N ratio) (Lamb et al. 2006), but if considerable rates of input from riverine or tidal sources are present, allochthonous organic matter may become more important (Jennerjahn and Ittekkot 2002). C3 plants such as mangroves have a δ13C signature ranging from − 32 to − 21‰ (Bouillon et al. 2008b), whereas C4 plants, as well as marine sources such as seagrass, and algae have a δ13C signature ranging from − 25 to − 8‰ (Lamb et al. 2006; Kennedy et al. 2010). The typical δ15N signature of mangrove biomass ranges from 0 to 11‰ (outliers: −10 to 20‰, Bouillon et al. 2008a; Ranjan et al. 2011), whereas the δ15N values of seagrass and marine microalgae are 6–12‰ and 0–4‰ respectively.
In the present study, site 1 and 3 had the lowest δ 13 C values (˗26.81 ± 2.14 and ˗28.47 ± 0.29 respectively) and higher C/N ratios (14.34 ± 0.07 and 14.98 ± 0.35 respectively) indicating mangrove litter as the potential organic carbon sources (Table 5). However, site 2 exhibited the highest δ 13 C value (˗23.09 ± 0.91), and the lowest C/N value (7.07 ± 0.008) indicating a marine input as this site was found close to the Arabian sea. The exchange of organic matter and nutrients in this site is attributed to the frequent tidal flushing. The δ 15 N values (1.11 ± 0.05) were also quite low in this site and appeared to be similar to the isotopic signatures of phytoplankton. The mean C/N, δ13C and δ15N values for phytoplankton were 6.67 ± 0.33, -25.83 ± 1.37‰ and 1.57 ± 1.18‰, respectively (Costa et al. 2021).
Total Ecosystem Carbon Stock
The total ecosystem carbon stock was found to be highest in site 2 (404.63 Mg C ha− 1) and lowest in site 3 (71.42Mg C ha− 1) as shown in Table 4; Fig. 3. The mean ecosystem carbon stock of Kerala mangroves was estimated to be 218.98 ± 169.86 Mg C ha− 1, which is higher than that of other mangrove habitats of India (Table 6). A wide range of total ecosystem carbon stocks in mangrove ecosystems of different nations has been reported, with values ranging from 54.3 MgCha− 1 in Cochin, Kerala to 1396.9 MgCha− 1 in Bintuni, Indonesia. On a global scale, mangrove forests in tropical countries at low latitudes have significantly more biomass than those in temperate zones (Komiyama et al. 2008). In the present study, the contribution of vegetation and soil carbon stocks to the total ecosystem carbon stock was 88% and 22% respectively. Furthermore, the belowground carbon stock accounted for 65% of the ecosystem carbon stock. Our results corroborate with the findings of Sanders et al. (2016) and Kauffman et al. (2020) that the belowground C stocks accounted for ~ 85% of the total ecosystem carbon stock in mangroves especially in medium and low-stature mangroves. However, the average estimated CO2 equivalents of the ecosystem carbon stocks was 805.78 ± 621.47 Mg CO2 ha− 1 and the social cost of carbon contributed by the mangroves of the study area was US $53447.05.
Conclusions
Indian mangroves, particularly Kerala mangroves are under tremendous development pressures despite the fact that sustainable mangrove management could contribute substantially to reduce national GHG emissions. However, as compared to other mangroves across the world, Kerala’s mangroves had a substantial amount of ecosystem carbon stock. Site 1 has a carbon stock of 180.90 Mg C ha− 1, while site 2 showed the highest carbon stock (404.63 Mg C ha− 1) whereas a carbon stock of 71.42 Mg C ha− 1 was estimated from site 3. The findings suggested that in site 2, the trapping of particulate organic matter from the adjacent coastal waters contributed more to the mangrove carbon sinks than the actual production of the mangrove trees, whereas the carbon sinks in the other two sites received the organic matter from autochthonous sources. The continuous tidal flushing in site 2, resulted in higher vegetation carbon stock but lower soil organic carbon stock, as demonstrated by stable isotopic studies and salinity range. The study also revealed that structural characteristics, salinity, soil pH and bulk density were the major factors influencing the carbon stock in the study sites.Therfore, considering the importance of mangroves as global carbon sinks, the disproportionate GHG emissions they produce when disturbed, and other vital ecological services they offer, they should be conserved, restored, and included in climate change adaptation and mitigation strategies. Being a state with a high risk of flooding and sea level rise, Kerala can adopt mangrove carbon sequestration as a key ‘climate change mitigation program’ in the future. Further, the estimation of the large C stocks of Kerala mangroves is important for policy formulation as per the guidelines of The United Nations Framework Convention on Climate Change (UNFCCC) to develop appropriate adaptive measures to deal with the climate change trends and to generate country- or region-specific data on C stocks and emission factors from various land-use activities in mangroves.
Data Availability
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
References
Adame MF et al (2021) Future carbon emissions from global mangrove forest loss. Global Change Biology 27:2856–2866
Alongi DM (2002) Present state and future of the world’s mangrove forests. Environmental Conservation 29:331–349
Alongi DM (2008) Mangrove forests: Resilience, protection from tsunamis, and responses to global climate change. Estuarine, Coastal and Shelf Science 76:1–13
Alongi DM (2012) Carbon sequestration in mangrove forests. Carbon Management 3:313–322
Alongi DM (2020) Global significance of mangrove blue carbon in climate change mitigation. Science 2(3):57. https://doi.org/10.3390/sci2030057
Anderson JM, Ingram JSI (1993) Tropical soil biology and fertility: a handbook of methods. CAB International, London, p 171
AOAC (2000) Official methods of analysis. Association of Official Analytical Chemists 805 International. Maryland, USA
Arifanti VB, Kauffman JB, Hadriyanto D, Murdiyarso D, Diana R (2019) Carbon dynamics and land use carbon footprints in mangrove- converted aquaculture: the case of the Mahakam Delta, Indonesia. Forest Ecology and Management 432:17–12
Atwood TB, Connolly RM, Almahasheer H et al (2017) Global patterns in mangrove soil carbon stocks and losses. Nature Climate Change. https://doi.org/10.1038/NCLIMATE3326
Bandyopadhyay BK, Maji B, Sen HS, Tyagi NK (2003) Coastal Soils of West Bengal dtheir Nature, Distribution and Characteristics. Bulletin No. 1/2003. CSSRI, RRS, Canning, pp 1–62
Banerjee K, Sahoo CK, Bal G, Mallik K, Paul R, ·Mitra A (2020) High blue carbon stock 829 in mangrove forests of Eastern India. Tropical Ecology. https://doi.org/10.1007/s42965-020-00072-y
Bouillon S, Connolly RM, Lee SY (2008a) Organic matter exchange and cycling in mangrove ecosystems: Recent insights from stable isotope studies. The Journal of Sea Research 59:44–58. https://doi.org/10.1016/j.seares.2007.05.001
Bouillon S et al (2008b) Mangrove production and carbon sinks: a revision of global budget estimates. Global Biogeochemical Cycles 22, GB2013
Bunting P, Rosenqvist A, Lucas RM, Rebelo LM, Hilarides L, Thomas N, Hardy A, Itoh T, Shimada M, Finlayson CM (2018) The global mangrove watch—A new 2010 global baseline of mangrove extent. Remote Sensing 10:1669
Cintron G, Schaffer Novelli YS (1984) Methods for studying mangrove structure. In: Samuel CS, Jane GS (eds) The mangrove ecosystem: research methods. UNESCO Publication, Paris, pp 91–113
Costa BVM, Moreira MZ, Bastos RF, Yogui GT (2021) Distribution and origin of sedimentary organic matter in an eutrophic estuary: Pina Sound-NE Brazil. Anais da Academia Brasileira de Ciências 93(1):e20190638. https://doi.org/10.1590/0001-3765202120190638
Das AK, Jha DK, Prashanthi Devi M, Sahu BK, Vinithkumar NV, Kirubagaran R (2014) Post-tsunami mangrove evaluation in coastal vicinity of Andaman Islands, India. Journal of Coastal Conservation. https://doi.org/10.1007/s11852-014-0312-5
Donato DC et al (2011) Mangroves among the most carbon-rich forests in the tropics. Nature Geoscience 4:293–297
Duarte CM, Losada IJ, Hendriks IE, Mazarrasa I, Marbà N (2013) The role of coastal plant communities for climate change mitigation and adaptation. Nature Climate Change 3:961–968
Eong OJ (1993) Mangroves—a carbon source and sink. Chemosphere 27:1097–1107
Folk RL (1980) Petrology of sedimentary rocks. Hemphill Publishing, Austin
FSI (2019) India State of forest report. 2019. Forest Survey of India, Ministry of Environment and Forests, Dehradun
Fujimoto K et al (1999) Belowground carbon storage of Micronesian mangrove forests. Ecological Research 14:409–413
Gilman EL, Ellison J, Duke NC, Field C (2008) Threats to mangroves from climate change and adaptation options. Aquatic Botany 89:237–250
Giri C et al (2011) Status and distribution of mangrove forests of the world using earth observation satellite data. Global Ecology and Biogeography 20:154–159
Hamilton S, Casey D (2016) Creation of high spatiotemporal resolution global database of continuous mangrove forest cover for the 21st century: a big-data fusion approach. Global Ecology and Biogeography 25:729–738
Harishma KM, Sandeep S, Sreekumar VB (2019) Biomass and carbon stocks in mangrove ecosystems of Kerala, southwest coast of India. Ecological Processes. https://doi.org/10.1186/s13717-020-00227-8
Hinrichs S, Nordhaus I, Geist SJ (2009) Status, diversity and distribution patterns of mangrove vegetation in the Segara Anakan lagoon, Java, Indonesia. Regional Environmental Change 9:275–289
Howard J, Sutton-Grier A, Herr D, Kleypas E, Mcleod J, Pidgeon E, Simpson S (2017) Clarifying the role of coastal and marine systems in climate mitigation. Frontiers in Ecology and the Environment. https://doi.org/10.1002/fee.1451
IPCC (2007) Climate Change 2007. In: Core Writing Team, Pachauri, RK, Reisinger A (eds) Synthesis report. Contribution of working groups I, II and III to the fourth assessment report of the intergovernmental panel on climate change. IPCC, Geneva, Switzerland, p 104
IPCC (2014) Summary for policymakers. In: Edenhofer O, Pichs-Madruga R, Sokona Y, Farahani E, Kadner S, Seyboth K, Adler A, Baum I, Brunner S, Eickemeier P, Kriemann B, Savolainen J, Schlömer S, von Stechow C, Zwickel T, Minx JC (eds) Climate change 2014: Mitigation of climate change. Contribution of working group III to the fifth assessment report of the intergovernmental panel on climate change.Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA
IPCC Climate Change (2007) The Physical Science Basis. In: Solomon S, Qin D, Manning M, Chen Z, Marquis M, Averyt KB, Tignor M, Miller HL (eds) Contribution of working group I to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA, p 996
Jagtap TG, Ansari ZA, Coutinho FB, Charulata S, Gaidhane DM (2004) Impact of climate change on mangrove forests along the south west coast: a case study from Kasargod, Kerala, India. In: Ravindranath, N.H., Sharma, S.K., Garg, A., Bhattacharya, S., Murthy, I.K. (Eds.) Proceedings of the workshop on vulnerability assessment and adaptation due to climate change on indian agriculture, forestry and natural ecosystem. Ministry of Environment and Forests, Government of India, pp 136–149
Jardine SL, Siikamäki JV (2014) A global predictive model of carbon in mangrove soils. Environmental Research Letters 9:104013
Jennerjahn TC, Ittekkot V (2002) Relevance of mangroves for the production and deposition of organic matter along tropical continental margins. The Science of Nature 89:23–30. https://doi.org/10.1007/s00114-001-0283-x
Kathiresan K, Rajendran N, Balakrishnan B, Thiruganasambandam R, Narayanasamy R (2021) Carbon sequestration and storage in planted mangrove stands of Avicennia marina. Regional Studies in Marine Science 43:101701
Kasawani I, Kamaruzaman J, Nurun-Nadhirah MI (2007) Biological diversity assessment of Tok Bali mangrove forest, Kelantan, Malaysia. WSEAS Transactions on Environment and Development 3(2):37–44
Kauffman JB, Bhomia RK (2017) Ecosystem carbon stocks of mangroves across Broad environmental gradients in West Central Africa: Global and regional comparisons. PLoS ONE 12:30187749
Kauffman JB, Donato D (2012) Protocols for the measurement, monitoring and reporting of structure, biomass and carbon stocks in mangrove forests. Center for International Forestry Research Center (CIFOR) Working paper 86
Kauffman JB, Arifanti VB, Bernardino AF, Ferreira TO, Murdiyarso D, Cifuentes MC, Norfolk J (2018) And details for land-use carbon footprints arise from quantitative and replicated studies. Frontiers in Ecology and the Environment 1749:12–13. https://doi.org/10.1002/fee.1749
Kauffman JB, Adame MF, Arifanti VB et al (2020) Total ecosystem carbon stocks of mangroves across broad global environmental and physical gradients. Ecological Monographs e01405. 10.1002/ecm.1405
Keith H, Mackey BG, Lindenmayer DB (2009) Re-evaluation of forest biomass carbon stocks and lessons from the world’s most carbon-dense forests. Proceedings of the National Academy of Sciences of the United States of America 106:11635–11640
Kennedy H, Beggins J, Duarte CM, Fourqurean JW, Holmer M, Marbà N, Middelburg JJ (2010) Seagrass sediments as a global carbon sink: isotopic constraints. Global Biogeochem Cycles 24(4). https://doi.org/10.1029/2010gb003848
Komiyama A, Poungparn S, Kato S (2005) Common allometric equations for estimating the tree weight of mangroves. Journal of Tropical Ecology 21:471–477. https://doi.org/10.1017/S0266467405002476
Komiyama A, Ong JE, Poungparn S (2008) Allometry, biomass, and productivity of mangrove forests. Aquatic Botany 89:128–137
Kristensen E, Bouillon S, Dittmar T, Marchand C (2008) Organic carbon dynamics in mangrove ecosystems. Aquatic Botany 89:201–219
Lamb AL, Wilson GP, Leng MJ (2006) A review of coastal paleoclimate and relative sea-level reconstructions using δ13C and C/N ratios in organic material. Earth-Science Reviews 75:29–57. https://doi.org/10.1016/j.earscirev.2005.10.003
Li SB, Chen PH, Huang JS, Hsueh ML, Hsieh LY, Lee CL, Lin HJ (2018) Factors regulating carbon sinks in mangrove ecosystems. Global Change Biology 24:4195–4210. https://doi.org/10.1111/gcb.14322
Lovelock CE, Cahoon DR, Friess DA, Guntenspergen GR, Krauss KW et al (2015) The vulnerability of Indo-Pacific mangrove forests to sea level rise. Nature 526:559–563. https://doi.org/10.1038/nature15538
Matsui N (1998) Estimated stocks of organic carbon in mangrove roots and sediments in Hinchinbrook Channel, Australia. Mangr. Salt Marsh. 2, 199–204
Mcleod E et al (2011) A blueprint for blue carbon: toward an improved understanding of the role of vegetated coastal habitats in sequestering CO2. Frontiers in Ecology and the Environment 9:552–560
Murdiyarso D et al (2015) The potential of Indonesian mangrove forests for global climate change mitigation. Nature Climate Change 5:8–11
Nair ASK, Sankar G (2002) Wetlands of Kerala. In: Jayakumar M (ed) Wetland conservation and management in Kerala. State Committee on Science, Technology and Environment, Kerala, India, pp 17–26
Pendleton L et al (2012) Estimating global ‘blue carbon’ emissions from conversion and degradation of vegetated coastal ecosystems. PLoS ONE 7:e43542
Polidoro BA et al (2010) The loss of species: Mangrove extinction risk and geographic areas of global concern. PLoS ONE 5:e10095
Rahman MM, Khan MNI, Hoque AKF, Ahmed I (2014) Carbon stock in the Sundarbans mangrove forest: spatial variations in vegetation types and salinity zones. Wetl Ecol Manag. https://doi.org/10.1007/s11273-014-9379-x
Rajasegar M, Sirnivasan M, Ajmal Khan S (2002) Distribution of sediment nutrients of Vellar estuary in relation to shrimp farming. Indian Journal of Geo-Marine Sciences 31(2):153–156
Ramachandran KK, Mohanan CN, Balasubrarnanian G, Kurian J, Thomas J (1986) The mangrove ecosystem of Kerala, its mapping inventory and some environmental aspects. State Committee on Science, Technology and Environment Project Report (1985-86), Thiruvananthapuram
Ranjan RK, Routh J, Ramanathan AL, Klump JV (2011) Elemental and stable isotope records of organic matter input and its fate in the Pichavaram mangrove–estuarine sediments (Tamil Nadu, India). Marine Chemistry 126:163–172. https://doi.org/10.1016/j.marchem.2011.05.005
Rani V, Bijoy Nandan S, Schwing PT (2021) Carbon source charecterisation and historical carbon burial in three mangrove ecosystems on the south west coast of India. Catena 197:104980. https://doi.org/10.1016/j.catena.2020.104980
Ray R, Chowdhury C, Majumder N, Dutta MK, Mukhopadhyay SK, Jana TK (2013) Improved model calculation of atmospheric CO2 increment in affecting carbon stock of tropical mangrove forest. Tellus B: Chemical and Physical Meteorology 65:18981
Ricke K, Drouet L, Caldeira K, Tavoni M (2018) Country-level social cost of carbon. Nature Climate Change 8:895–900. https://doi.org/10.1038/s41558-018-0282-y
Rovai AS, Twilley RR, Casta-neda-Moya E, Riul P, Cifuentes-Jara M, Manrow Villalobos M, Horta PA, Simonassi JC, Fonseca AL, Pagliosa PR (2018) Global controls on carbon storage in mangrove soils. Nature Climate Change 8:534–538. https://doi.org/10.1038/s41558-018-0162-5
Sahu SC, Kumar M, Ravindranath NH (2016) Carbon stocks in natural and planted mangrove forests of Mahanadi wetland, East coast of India. Current Science 110:2253–2260
Sahu G, Satpathy KK, Mohanty AK, Sarkar SK (2012) Variations in community structure of phytoplankton in relation to physicochemical properties of coastal waters, southeast coast of India. Indian Journal of Geo-Marine Sciences 41(3):223–241
Sanders CJ, Maher DT, Tait DR, Williams D, Holloway C, Sippo JZ, Santos IR (2016) Are global mangrove carbon stocks driven by rainfall? J Geophys Research: Biogeoscience 121:2600–2609
Satyanarayana B, Raman AV, Dehairs F, Kalavati C, Chandramohan P (2002) Mangrove floristics and zonation pattern of Coringa, Kakinada Bay, East coast of India. Wetlands Ecology and Management 10:25–39
ShyleshChandran MS, Ravi A, John SM, Sivan S, Asha MS, Mammen PC, Sajith Kumar KJ, Sruthi SN (2020) Ecosystem carbon stock of selected mangrove forests of Vypin-Cochin region, southwest coast of India. Wetlands 40:2263–2273. https://doi.org/10.1007/s13157-020-01365-7
Shunyang C, Bin C, Pramudji S, Eka DIW, Danyun O, Xijie Y, Weiwei Y, Guangcheng C (2018) Ecosystem carbon stock of a tropical mangrove forest in North Sulawesi, Indonesia. Acta Oceanologica Sinica 37:1285–1291. https://doi.org/10.1007/s13131-018-1290-5
Siikamäki J, Sanchirico JN, Jardine SL (2012) Global economic potential for reducing carbon dioxide emissions from mangrove loss. Proceedings of the National Academy of Sciences of the United States of America 109:14369–14374
Sitoe AA, Mandlate LJC, Guedes BS (2014) Biomass and carbon stocks of Sofala Bay mangrove forests. Forests 5:1967–1981. doi: https://doi.org/10.3390/f5081967
Sreelekshmi S, Preethy CM, Varghese R, Joseph P, Asha CV, Bijoy Nandan S, Radhakrishnan CK (2018) Diversity, stand structure, and zonation pattern of mangroves in southwest coast of India. Journal of Asia-Pacific Biodiversity 11:573–582. https://doi.org/10.1016/j.japb.2018.08.001
Sreelekshmi S, Joseph P, Varghese R, Preethy CM, Bijoy Nandan S (2020a) Structural variability and its relation to edaphic attributes of mangroves in the south-west coast of India. In: Lakes & Reservoirs: Science, Policy and Management for Sustainable Use. https://doi.org/10.1111/LRE.12314
Sreelekshmi S, Nandan SB, Kaimal SV, Radhakrishnan CK, Suresh VR (2020b) Mangrove species diversity, stand structure and zonation pattern in relation to environmental factors—A case study at Sundarban delta, east coast of India. Regional Studies in Marine Science 35:101111
Sreelekshmi S, Veetil BK, Bijoy Nandan S, Harikrishnan M (2021) Mangrove forests along the coastline of Kerala, southern India: Current status and future prospects. Regional Studies in Marine Science. https://doi.org/10.1016/j.rsma.2020.101573
Stringer CE, Trettin CC, Zarnoch SJ (2016) Soil properties of mangroves in contrasting geomorphic settings within the Zambezi River Delta, Mozambique. Wetlands Ecology and Management 24:139–152. https://doi.org/10.1007/s11273-015-9478-3
Suresh HS, Bhat DM, Ravindranath NH, Sukumar R (2017) Carbon stocks and sequestration potential of Indian Mangroves. Tropical Ecology 58(3):547–553
Trettin CC, Stringer CE, Zarnoch SJ (2016) Composition, biomass and structure of mangroves within the Zambezi River Delta. Wetlands Ecology and Management 24:173–186
Tue NT, Dung LV, Nhuan MT et al (2014) Carbon storage of a tropical mangrove forest in Mui Ca Mau National Park, Vietnam. CATENA 121:119–126. doi: https://doi.org/10.1016/j.catena.2014.05.008
Twilley RR, Chen RH, Hargis T (1992) Carbon sinks in mangroves and their implications to carbon budget of tropical coastal ecosystems. Water, Air, & Soil Pollution 64:265–288
van der Werf GR et al (2009) CO2 emissions from forest loss. Nature Geoscience 2:737–738
Vinod K, Koya AA, Kunhi Koya VA, Silpa PG, Asokan PK, Zacharia PU, Joshi KK (2018) Biomass and carbon stocks in mangrove stands of Kadalundi Estuarine Wetland, south-west coast of India. The Indian Journal of Fisheries 65(2):89–99. https://doi.org/10.21077/ijf.2018.65.2.72473-11
Xiong Y, Liao B, Wang F (2018) Mangrove vegetation enhances soil carbon storage primarily through in situ inputs rather than increasing allochthonous sediments. Marine Pollution Bulletin 131:378–385. https://doi.org/10.1016/j.marpolbul.2018.04.043
Zanne AE et al (2009) Global wood density database. Data from: ta worldwide wood economics spectrum Dryad Data Repository. PLoS ONE. https://doi.org/10.5061/dryad.234.
Zhu JK (2001) Plant salt tolerance. Trends in Plant Science 6(2):66–71
Acknowledgements
The first author is grateful to Kerala State Higher Education Council for the financial assistance in connection with the Kairali research Award-2020. Our sincere gratitude to the Director, School of Industrial Fisheries and the Head, Department of Marine Biology, Microbiology & Biochemistry, CUSAT for the research facilities and support.
Funding
SL received a research grant (No. A3/352) from Kerala State Higher Education Council in connection with the Kairali Research Award, 2019-20 instituted by the Kerala State Government.
Author information
Authors and Affiliations
Contributions
SL, BN & HK developed the research; SL, SK and RH performed the field work, collected and analysed the data, SL wrote the manuscript, SL, HK, BN, SK & RH reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Consent for Publication
Everybody entitled so gave their consent for this research being published.
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 34.9 KB)
Rights and permissions
About this article
Cite this article
Sreelekshmi, S., Harikrishnan, M., Nandan, S.B. et al. Ecosystem Carbon Stock and Stable Isotopic Signatures of Soil Organic Carbon Sources Across the Mangrove Ecosystems of Kerala, Southern India. Wetlands 42, 29 (2022). https://doi.org/10.1007/s13157-022-01540-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13157-022-01540-y