Abstract
In 1998, a 6.1-ha wetland restoration project along the Rock River, IL, USA was designed to test five afforestation methods on former agricultural land, including planting bareroot trees, balled-and-burlapped trees, seedlings, acorns, or allowing natural regeneration. Fifteen years later, we assessed vegetation at the site to determine the effectiveness of alternative strategies and compare treatments to an adjacent floodplain forest. We also compared the cost of treatments to determine whether long-term restoration outcomes justified initial costs. After 15 years, lower cost treatments (acorn plantings and passive restoration) were dominated by dense reed canarygrass (Phalaris arundinacea) with sparse trees, whereas higher cost treatments (bareroot and balled-and-burlapped tree plantings) had developed closed tree canopies, and tended to have greater plant species richness, tree basal area, and density of stems >7.5-cm diameter. For every additional $10,000 per ha spent on restoration, predicted richness increased by 1 species per 250-m2 plot, predicted P. arundinacea biomass decreased by 61 g m−2, and predicted tree basal area increased by 3 m2 ha−1. Although some studies have indicated that passive regeneration alone is effective for afforestation, restoration of floodplains in the presence of P. arundinacea will require a more intensive approach.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Restoration practitioners are challenged to achieve desired ecological goals with minimal input of time and resources. In some situations, those goals can be achieved with little intervention, leading several authors to advocate spontaneous succession, sometimes referred to as “passive restoration,” as an efficient restoration strategy on disused lands (Mitsch et al. 1998; Halle 2007; Clewell and McDonald 2009; Rey Benayas et al. 2008; Birch et al. 2010). However, there are situations in which spontaneous succession is ineffective, for example, where limited propagule supply or non-native species invasion constrain recovery (Streever and Zedler 2000; Zahawi et al. 2014). In this study, we compare the effectiveness of alternative methods for restoring floodplain forest on former agricultural land in the presence of an invasive wetland grass. We contrast spontaneous succession with increasingly more expensive tree planting treatments.
An obvious advantage of spontaneous succession is low cost. Active intervention is not always necessary for ecosystem recovery. Costly hands-on intervention, such as planting late successional species, is inefficient if it is unnecessary for achieving restoration objectives; and efficiency is desirable because it liberates resources for land acquisition and restoration elsewhere (Higgs 1997; Holl and Aide 2011). Evidence from impaired ecosystems suggests that many recover just as quickly without direct intervention (Jírová et al. 2012; Jones et al. 2018). For example, spontaneous succession can lead to target vegetation in abandoned quarries (Řehounková and Prach 2008), and planting is not always necessary for restoring biotic structure and biogeochemical function in wetlands (Mitsch et al. 1998; Kamali and Hashim 2011; Moreno-Mateos et al. 2015). Thus, passive restoration strategies can enable otherwise cost-prohibitive regional- and landscape-scale restoration (Birch et al. 2010; Linhares de Rezende et al. 2015). In certain situations, active restoration can even be counter-productive. For example, planting may impose a community composition that is ill-suited to the local abiotic conditions (Mitsch et al. 1998). At worst, if planted species are inappropriately selected, they might inhibit natural colonization by more desirable species (Tropek et al. 2013; Bauman et al. 2015).
It is clear from the above examples that spontaneous succession can be both efficient and effective, but there are situations which preclude the use of spontaneous succession as the sole restoration tool. For example, active approaches to restoration, such as herbicide control and pre-emptive planting, may also be necessary where dominant undesirable species are likely to invade or reinvade a restoration site (Reinartz and Warne 1993; Boers et al. 2006; Reinecke et al. 2008; Middleton et al. 2010; Skinner et al. 2012). In addition, intensive planting may be necessary where desirable species are absent from the surroundings and cannot readily colonize (Castillo and Figueroa 2009; Gómez-Aparicio et al. 2009; De Steven et al. 2010; Klimkowska et al. 2010; Coiffait-Gombault et al. 2012; O'Connell et al. 2013). Prach and Hobbs (2008) proposed a conceptual model to describe the relative effectiveness of passive restoration vs. more expensive, hands-on technical approaches to restoration. At physically degraded sites, for example, toxic or extremely dry sites, hands-on technical approaches may be necessary to ameliorate stressful conditions (Prach and Pyšek 1994). At the other extreme, at productive, eutrophic sites, such as sites with high residual soil fertility from a history of intensive agriculture, it is probable that a few competitive, often non-native, plant species will dominate, and diversity will be low (Klimkowska et al. 2007; Cramer et al. 2008; Pywell et al. 2011; Jessop et al. 2015). At either extreme, Prach and Hobbs (2008) predict that the likelihood of attaining a targeted restoration outcome via spontaneous succession alone should decrease, and as a consequence, the monetary cost of attaining that targeted outcome should increase.
Afforestation of previously farmed floodplains has been studied extensively, particularly in the southeastern United States, and much of this research suggests that active tree planting may be necessary to reach restoration objectives. Several authors have noted a lack of natural regeneration of desired species, particularly heavy-seeded oaks [Quercus spp.] and hickories [Carya spp.], due to site or seed limitation (Kruse and Groninger 2003; Middleton 2003; Haynes 2004; Battaglia et al. 2008). Tree planting may be especially important when the restoration site is distant from potential seed sources (Allen 1997; Twedt 2004; De Steven et al. 2015). In addition, floodplains are stressful settings for tree seedlings (Battaglia et al. 1995; Middleton 2000; Kozlowski 2002), and it may be beneficial to plant more expensive, more mature trees, which are more flood-tolerant, rather than planting small seedlings or direct seeding (Dey et al. 2010). Larger individuals may also be more competitive with tall herbaceous plants that establish rapidly on the fertile soils of formerly agricultural floodplains (Stanturf et al. 2009).
Recently restored habitats, particularly in floodplain settings, may be especially vulnerable to invasion by non-native plant species due to low plant biomass, recently disturbed soils, and propagule delivery via hydrochory (Zedler and Kercher 2004; D’Antonio and Chambers 2006; Matthews et al. 2009). Once established, invasive species such as Phalaris arundinacea often persist in restored wetlands, or even increase in abundance through time, precluding the attainment of restoration goals for many years (Aronson and Galatowitsch 2008; Garbutt and Wolters 2008; Toth 2010; Matthews 2015).
Our objective in this study was to compare the relative effectiveness of passive restoration with the effectiveness of increasingly costly strategies for restoring floodplain forest on former agricultural land. We assessed vegetation in a reference forest and a restored forest 15 years after the imposition of five alternative tree planting treatments. Restoration goals were to establish a floodplain forest dominated by native plant species. We hypothesized that spontaneous succession would not achieve restoration goals in this situation for three reasons: (1) Previous authors have suggested that trees must be actively planted in floodplain afforestation projects to achieve desired stocking rates, especially when hard-mast species like oaks (Quercus spp.) are desired (Allen 1997; Kruse and Groninger 2003), (2) Floodplain wetlands are productive sites that are subject to non-native species invasion and dominance (Zedler and Kercher 2004), and (3) Our study site occurred within an agriculturally dominated, nutrient-enriched watershed (David et al. 2014).
Methods
Study Site Location and History
Our study site is a compensatory mitigation wetland restored by the Illinois Department of Transportation on 6.1 ha of former row crop agricultural field in the Rock River floodplain in northwest Illinois, USA (41.5542 N, −90.1835 W). The site was farmed for several years prior to restoration in 1997. Topography and hydrologic conditions are similar across the site. Between 1999 and 2003, the site was flooded by the Rock River an average of four times per year at an average maximum depth of 2.0 m (G.E. Pociask and J.W. Matthews, unpublished data). The site is underlain by Sawmill silty clay loam, a poorly drained, hydric soil (Elmer 2004). The restoration site is directly adjacent to a mature floodplain forest dominated by Acer saccharinum and Fraxinus pennsylvanica.
Restoration performance standards were established for the restored forest. Specifically, the site was required to establish jurisdictional wetland as defined by the U.S. Army Corps of Engineers (USACE 1987), 50% of the plant species present were required to be native, hydrophytic tree species were expected to establish naturally, and there were specific requirements (see below) for planted tree survival (Plocher et al. 2003). No hydrologic manipulation or earth-moving was performed prior to restoration. In 1997 the site was divided into 15 approximately 0.4-ha strips, each randomly assigned to one of 5 restoration treatments (Fig. 1, Fig. 2a-e). Four treatments involved introducing tree species on bare soil at various nursery stages: (1) 1.5–1.8 m tall bareroot trees at a density of 1,203 trees per ha; (2) larger, 5.0-cm stem diameter (~2.4–3.0 m tall) balled-and-burlapped trees at 272 trees per ha; (3) approximately 38 cm tall, 2-year-old seedlings planted at 2,474 per ha; and (4) acorns planted at 3,707 per ha. Acorns were collected from within 80 km of the project site. Required tree survival rates at the end of five years were 75% for bareroot trees, 90% for balled-and-burlapped trees, 33% for seedlings, and 20% for acorns. A fifth treatment with no tree planting was included to test the effectiveness of passive regeneration from the seedbank and adjacent natural forest. Number of replicates differed among treatment types (bareroot = 3, balled-and-burlapped = 2, seedlings = 5, acorns = 2, passive regeneration = 3; Fig. 1). We compared measures of vegetation structure, 15 years after planning, among six treatments: the five restoration treatments plus the adjacent reference forest.
Aerial photograph of study site along the Rock River, IL. Shaded overlays indicate restoration treatment strips, and rectangles indicate locations of the 50-m × 5-m sampling plots (n = 3 per treatment type). The reference floodplain forest, with three sampling plots, is northeast of the restoration site
Bareroot tree plantings included 1,460 Acer saccharinum, Betula nigra, Fraxinus pennsylvanica, Populus deltoides, Quercus bicolor, and Quercus palustris. Balled-and-burlapped plantings (220 trees) included these six species plus Platanus occidentalis, and seedling plantings (5,004 trees) included these six species plus P. occidentalis and Carya illinoiensis. Acorns (9,000) were Q. bicolor and Q. palustris. Trees were planted in rows to facilitate management, which included seeding of a temporary cover mix (Lolium perenne [55 kg ha−1] and Avena sativa [70 kg ha−1]), a one-time application of straw mulch, bi-annual mowing between rows, bi-annual application of glyphosate herbicide around the trees, and replanting in response to mortality (between 1999 and 2002, replanting included 26 balled-and-burlapped trees, 497 bareroot trees, and 6,500 seedlings). Control plots were not mowed or treated with herbicide, and acorn plots were mowed only. Vegetation monitoring and site management continued for 5 years.
We determined establishment costs for each treatment based on original contract documents. Costs are reported in $US as of 1997 – the start of restoration at the site. Site preparation was similar across all restoration treatments, including the passive treatment (approximately $2,613 per ha), and the costs reported here represent only the costs associated with the initial tree plantings and any replantings in response to mortality. We excluded costs of land acquisition, planning, permitting, and monitoring. Per tree costs varied, depending on species, from $210 to $260 for balled-and-burlapped trees, $55 to $65 for bareroot trees, and $11 to $12 for seedlings. Per hectare costs of tree planting were estimated at $95,679 for bareroot trees, $72,527 for balled-and-burlapped trees (which, although larger than bareroot trees, were planted at lower density), $65,848 for seedlings, $24,216 for acorns, and $0 for passive restoration. Adjusted for inflation, per hectare costs in 2019 would equate to $151,374 for bareroot trees, $114,745 balled-and-burlapped trees, $104,178 for seedlings, and $38,312 for acorns. We recognize that monetary cost is an imperfect proxy for restoration effort. For example, costs will vary depending on prices of source materials and labor. However, other proxies for restoration effort, such as total hours invested per treatment, were not recorded during restoration.
At the end of the 5-year monitoring period, three treatments were deemed to have acceptable tree survival relative to the number initially planted: balled-and-burlapped (97% survival), bareroot (100% survival), and seedlings (50% survival) (Plocher et al. 2003). However, germinating tree seedlings in acorn and passive restoration treatments had been overtopped by an invasive grass, reed canarygrass (Phalaris arundinacea) (Plocher et al. 2003), and so an additional 168 balled-and-burlapped oaks were planted on the acorn plots to meet regulatory requirements for compensatory mitigation credit.
Field Data Collection and Numerical Analyses
In 2013, 15 years after planting, we conducted vegetation surveys at the site to evaluate the long-term success of the site in the absence of active management. We compared characteristics of the planted forest plots to each other and to the adjacent mature floodplain forest community. We established three 50-m × 5-m vegetation plots per treatment type, plus three plots in the adjacent mature floodplain forest (18 plots total; Fig. 1). The long axis of the plots was arranged a direction perpendicular to the adjacent Rock River. Plot locations were selected randomly within each treatment strip. However, because two treatment types (balled-and-burlapped and acorn) were only replicated twice in the original experimental design, for these two treatments, we placed two plots in one treatment strip (Fig. 1).
In each 250-m2 plot, we measured the diameter-at-breast-height (DBH) of all woody stems larger than 5-cm DBH and calculated total basal area (m2 ha−1) for each plot. Stems of all woody species taller than 1 m, including stems <5-cm DBH, were tallied within each plot. We inventoried all vascular plant species within the plot to determine species richness and estimated the floristic quality of each plot by assigning each species a “Coefficient of Conservatism” (C) based on Taft et al. (1997) and averaging across all species in the plot to calculate the mean C. C-values range from zero to ten and provide a subjective rating of species fidelity to undegraded natural communities. Non-native species were assigned a C-value of zero. At 10-m intervals along a 50-m transect through the center of each plot, we measured herbaceous-layer species percent cover in a 0.25-m2 quadrat. All vascular plant species observed in each quadrat, as well as bare ground, were assigned a cover class (<1%, 1–5%, 6–25%, 26–50%, 51–75%, 76–95%, or 96–100%) to assess plant community composition at the plot-level. In addition, within each quadrat, we measured aboveground biomass of P. arundinacea in 30-cm × 30-cm subplots. Within each subplot, all live P. arundinacea was clipped to the soil surface, dried at 60 °C overnight, and dried biomass weighed. Biomass within the 5 subplots was averaged within each plot. We previously reported P. arundinacea biomass in Peralta et al. (2017).
We used Analyses of Variance (ANOVAs) to determine whether there were differences in each of six response variables (tree basal area, density of large woody stems [>7.5-cm DBH], density of small woody stems [<7.5-cm DBH, but taller than 1 m], plant species richness, mean C, or P. arundinacea biomass) among the six treatments (five restoration treatments plus the reference forest), with treatment as a fixed effect. Plots of residuals versus predicted values were inspected for normality, and Levene’s Test was used to determine if variances were homogenous. Residuals for density of small woody stems were not normally distributed, so a nonparametric Kruskal-Wallis test was used. Significant ANOVAs were followed by Tukey’s Honestly Significant Difference (HSD) test for post hoc comparisons among the six treatment types.
Results
Vegetation structure differed among some treatments after 15 years, and these differences were consistent with the 5-year results (Fig. 2). Strips that had been planted with bareroot or balled-and-burlapped trees had developed closed tree canopies with only sparse herbaceous plant cover, whereas acorn plantings and passive restoration plots had become dense stands of P. arundinacea. Average (±standard error) summed plant cover in the herbaceous layer was least (3.3 ± 2.1%) and bare soil cover greatest (95 ± 2.5%) in the bareroot plantings, and plant cover was greatest (86.5 ± 5.5%) and bare soil cover least (9.8 ± 5.1%) in the acorn plantings. Seedling plantings were spatially variable, with patches of trees and scattered open areas dominated by P. arundinacea. Herbaceous plant cover averaged 48.3 ± 17.5% in balled-and-burlapped plots, 34.5 ± 26.8% in seedling plots, and 82.3 ± 13.9% in passive regeneration plots. The reference forest plots were primarily composed of medium- to large-diameter (average DBH = 34 cm) Acer saccharinum (35%) and Fraxinus pennsylvanica (41%). Other woody species in the reference forests were Celtis occidentalis, Crataegus mollis, Gleditsia triacanthos, Morus alba, Ulmus americana, and the shrub Cephalanthus occidentalis. Herbaceous vegetation was sparse in reference plots (average cover 9.5 ± 3.2%); Symphyotrichum lanceolatum was the most abundant herbaceous species, and P. arundinacea comprised less than 1% of the herbaceous cover.
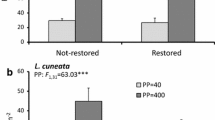
Tree basal area was small in acorn and passive restoration treatments, especially when compared to the densely planted bareroot plots and the reference plots (F5,12 = 4.26, p = 0.018; Fig. 3a). However, the only significant pairwise difference was between bareroot and acorn plantings. Overall stem density was similar among treatments (F5,12 = 2.60, p = 0.081). However, the balled-and-burlapped treatments and the reference plots contained fewer, but larger stems, whereas two of the passive restoration plots contained dense patches of small-diameter willows (Salix interior). When stem density was calculated separately for shrub- and sapling-layer stems, taller than 1 m, but smaller than 7.5-cm DBH, stem density did not differ significantly among treatments (Kruskal-Wallis Χ2, = 2.64, df = 5, p = 0.75; Fig. 3b). In contrast, density of larger (>7.5-cm DBH) stems differed among treatments (F5,12 = 10.95, p = 0.0004; Fig. 3c), and was greatest in the densely planted bareroot and seedling treatments. Relative to the more expensive, high density bareroot planting, the seedling and balled-and-burlapped treatments more closely matched reference forest plots in terms of total basal area and density of small and large stems. However, stem density in the seedling treatment varied within and among plots, with some large patches (up to approximately 25 m2) of P. arundinacea containing few or no trees. In addition to S. interior and the planted species listed in the Methods, woody species in the restored forest plots included Acer negundo, Celtis occidentalis, Gleditsia triacanthos, Juglans nigra, Morus alba, Salix nigra, and Ulmus americana.
Mean (±SE) tree basal area (a), density of small stems (b), density of large stems (c), Phalaris arundinacea biomass (d), plant species richness per plot (e), and mean Coefficient of Conservatism (f) by restoration treatment. Treatments are arranged, from left to right, in order of increasing initial cost. Different letters (a, b, c) above bars represent significant differences (Tukey’s HSD, p < 0.05) between treatments
As previously reported by Peralta et al. (2017), biomass of P. arundinacea differed significantly among treatments (F5,12 = 7.80, p = 0.002; Fig. 3d). P. arundinacea biomass was much greater in the acorn and passive restoration treatments than in the bareroot or reference treatments. Plant species richness also varied among treatment types (F5,12 = 4.77, p = 0.013; Fig. 3e) and was inversely related to P. arundinacea biomass (linear regression: r2 = 0.54, F1,16 = 18.95, p = 0.0004). Species richness was lower in the passive restoration plots than in the balled-and-burlapped or reference plots. Nevertheless, P. arundinacea was the most abundant herbaceous species in every restoration treatment type (followed by Leersia virginica and Symphyotrichum lanceolatum). Mean C values were similar among treatment types (F5,12 = 1.77, p = 0.194; Fig. 3f).
Assuming a linear relationship between cost and restoration outcomes—which may be unrealistic (Miller and Hobbs 2007), but is a close approximation over the range of our data—for each additional $10,000 ha−1 spent on restoration ($15,821 in 2019 dollars), P. arundinacea biomass decreased by 61 g m−2 (r2 = 0.64), tree basal area increased by 3 m2 ha−1 (r2 = 0.28), and plant species richness increased by one species per plot (r2 = 0.49).
Discussion
In a restored floodplain forest, alternative planting treatments that were imposed on the site at the outset of restoration have resulted in differences in vegetation structure after 15 years. Intensive and costly planting resulted in closed canopy forests, whereas more hands-off approaches to restoration resulted in species-poor wet meadows dominated by an invasive grass. Whether these differences are worth the additional investment in tree planting depends on the particular restoration goals. Our study site was restored as compensation for the destruction of naturally occurring wetlands. Spontaneous succession and low-cost acorn plantings resulted in failure to achieve legally required performance standards due to invasion by P. arundinacea and poor tree establishment. Thus, the cheapest approaches fell outside the realm of acceptability; the goal was to replace floodplain forest, and neither passive restoration nor acorn plantings achieved that goal. Our estimate of the cost of the acorn planting treatment did not include the costs associated with delayed balled-and-burlapped oak plantings into the acorn plots at the end of the five-year monitoring period to meet compliance requirements, which we estimated to be approximately $61,000 per ha. Direct seeding is initially cheaper than planting seedlings, and can be effective in some situations (Stanturf et al. 2009). Even with this delayed planting of balled-and-burlapped trees, however, the direct seeding treatment was ineffective and, ultimately, expensive.
A recent meta-analysis found that although revegetation accounts for a significant portion of the cost of wetland restoration, revegetation does not lead to improved biotic and functional outcomes (Moreno-Mateos et al. 2015). Our study is a reminder, however, that this finding is not universal. Similarly, other studies that have shown that increased investment in restoration plantings leads to improved restoration outcomes relative to plantations or spontaneous succession (Kanowski et al. 2003; Klimkowska et al. 2007; Munro et al. 2009; Middleton et al. 2010; Pywell et al. 2011). The important question is therefore not whether additional investment in active revegetation is worthwhile—in some situations it is not—but under which conditions is it worthwhile (Prach and Hobbs 2008; Holl and Aide 2011; Jones et al. 2018).
Intensive planting was necessary for ecosystem recovery in our study site, but we suspect that the observed lack of recovery in unplanted treatments was not due solely to slow colonization by woody species, but due to slow colonization relative to very high invasive grass productivity. A potential tree seed source was directly adjacent to our study site, and there is abundant evidence that proximity to remnant patches of target vegetation facilitates successful restoration (e.g., Hutchings and Booth 1996; Öster et al. 2009; Alsfeld et al. 2010; Helsen et al. 2013). Furthermore, the site was regularly flooded by the nearby river, and likely received propagules via hydrochory, which is known to be an important pathway for seed delivery to floodplain sites (e.g., Leyer 2006; Moggridge et al. 2009). Nevertheless, active and intensive tree planting was necessary. Soils at this site are particularly rich in ammonium, nitrate, and phosphate, even compared to other restored floodplains on former agricultural land in the region (Cohen 2018), and nutrient enrichment in wetlands is known to favor P. arundinacea dominance (Green and Galatowitsch 2002; Kercher and Zedler 2004; Perry et al. 2004). Thus, our study supports one end of the general model of Prach and Hobbs (2008), which suggests that more costly, active intervention is necessary for restoring eutrophic sites.
Unlike other studies which have shown a convergence of plant communities among experimentally imposed restoration treatments through time (e.g. Warren et al. 2002; Holl et al. 2014), the differences among initial treatments were long-lasting in our study site. Phalaris arundinacea, once established, was able to form a stable wet meadow community state for at least 15 years in the otherwise forested river floodplain. Zedler (2009) suggested that P. arundinacea is able to form an alternative stable state, driven by the production of a dense layer of thatch, which prevents germination of other species. Volunteer and planted tree seedlings do not establish well in existing stands of P. arundinacea (Hovick and Reinartz 2007; Adams et al. 2011; Thomsen et al. 2012). Thus, like some other invasive plants (e.g., Fike and Niering 1999; Rudgers et al. 2007), P. arundinacea seems to inhibit succession to forested communities. However, P. arundinacea is shade-intolerant and does not establish well under an existing plant canopy (Lindig-Cisneros and Zedler 2002; Adams et al. 2011). In our study site, treatment plots which were treated with glyphosate during the initial five years, and which rapidly developed closed tree canopies, effectively precluded P. arundinacea invasion. This led to persistent and abrupt ecological boundaries at the site, characteristic of alternative stable states (Wilson and Agnew 1992), and a reinforcement of initial differences in woody plant cover among restoration treatments. After 15 years, dense stands of willows had developed in two of the passive restoration plots, and although this may eventually result in a decrease in P. arundinacea abundance, it suggests a long temporal lag in the establishment of a woody canopy and a temporal loss of biodiversity support as a result of compensatory wetland mitigation (see Spyreas et al. 2010; Jessop et al. 2015).
Slow development of a closed canopy in low-cost planting treatments was exacerbated by seedling mortality. Tree mortality in response to flooding is a major constraint on floodplain forest restoration, and tree seedlings are particularly susceptible (King and Keeland 1999; Stanturf et al. 2004; Krzywicka et al. 2017; Matthews et al. in press). Restoration monitoring reports from our study site indicated poor survival of the smaller acorn and seedling plantings, and dominance by P. arundinacea by 2001, three years after restoration (Plocher et al. 2001). Tree seedlings in these treatments, especially in competition with invading P. arundinacea, may have been unable to grow large enough to escape flood risk. Furthermore, during our vegetation surveys, we noticed several planted trees that had been damaged or felled by beavers (Castor canadensis). In the openings created by beavers, P. arundinacea had established dense colonies.
Dominance by P. arundinacea has additional consequences for ecosystem structure and function. Invasion by P. arundinacea has been demonstrated to decrease biodiversity (Spyreas et al. 2010; Rojas and Zedler 2015) and lead to biological homogenization at a regional scale (Price et al. 2018). The high shoot density of P. arundinacea stands may promote sedimentation (Werner and Zedler 2002), which could have consequences for biogeochemical cycling. Indeed, Phalaris-dominated plots at our study sites had greater concentrations of soil nitrate and soil organic matter relative to forested plots, and initial restoration treatments influenced soil bacterial community composition after 15 years (Peralta et al. 2017).
Application of herbicide, when combined with other control strategies, and followed by planting of native vegetation, is recommended for eradicating P. arundinacea (Lavergne and Molofsky 2006). Initial control of P. arundinacea is important for establishing woody seedlings. Hovick and Reinartz (2007) and Thomsen et al. (2012) reported higher two-year survival rates for planted tree and shrub seedlings when planted into P. arundinacea stands after herbicide treatment relative to untreated controls. However, once established, P. arundinacea is difficult to control even with follow-up planting by native species, because it rapidly reinvades (Foster and Wetzel 2005; Adams and Galatowitsch 2006). Thus, rapid establishment of perennial vegetation cover is considered to be critical for preventing P. arundinacea reinvasion (Lindig-Cisneros and Zedler 2002; Iannone and Galatowitsch 2008). For example, Kim et al. (2006) found that after an initial herbicide application, dense planting of live willow stakes significantly decreased P. arundinacea biomass over two seasons. Our study indicates that a pre-emptive approach can be successful for controlling P. arundinacea in restorations where it is likely to invade – planting fast-growing trees which quickly form a closed canopy and shaded understory can prevent initial invasion. Similar to this study, Adams et al. (2011) found that acorn plantings grew too slowly to suppress P. arundinacea, and the authors recommend planting a mix of oaks and faster-growing trees which will create a shaded canopy. Added benefits of rapid canopy closure include facilitating the colonization and establishment of other forest species (McLeod et al. 2001; Stanturf et al. 2009; McClain et al. 2011; Reid et al. 2015; Wallace et al. 2017) and provision of vertical structure for wildlife habitat (Twedt et al. 2002; Dey et al. 2010).
Finally, this study illustrates the value of restoration as experimentation. Formal experimentation, a key component of adaptive restoration, is important for determining which restoration approaches are most successful in which contexts (Zedler et al. 2012). Conducting restoration as an experiment could help identify situations where passive restoration is and is not an effective strategy (Jones et al. 2018). As this study demonstrates, however, experimental learning often requires designing for uncertainty and accepting suboptimal outcomes as a part of the learning process (see Holling 1978). Such openness to enlightening failure may be particularly contentious in the case of compensatory wetland mitigation and other offset programs, for which success is legally mandated, and uncertainty is often unacknowledged.
References
Adams CR, Galatowitsch SM (2006) Increasing the effectiveness of reed canary grass (Phalaris arundinacea L.) control in wet meadow restorations. Restoration Ecology 14:441–451
Adams CR, Kauth PJ, Sorenson JW (2011) Assessing competition between reed canary grass (Phalaris arundinacea) and swamp white oak (Quercus bicolor). Ecological Restoration 29:332–338
Allen JA (1997) Reforestation of bottomland hardwoods and the issue of woody species diversity. Restoration Ecology 5:125–134
Alsfeld AJ, Bowman JL, Deller-Jacobs A (2010) The influence of landscape composition on the biotic community of constructed depressional wetlands. Restoration Ecology 18:370–378
Aronson MFJ, Galatowitsch S (2008) Long-term vegetation development of restored prairie pothole wetlands. Wetlands 28:883–895
Battaglia LL, Keough JR, Pritchett DW (1995) Early secondary succession in a southeastern U.S. alluvial floodplain. Journal of Vegetation Science 6:769–776
Battaglia LL, Pritchett DW, Minchin PR (2008) Evaluating dispersal limitation in passive bottomland forest restoration. Restoration Ecology 16:417–424
Bauman JM, Cochran C, Chapman J, Gilland K (2015) Plant community development following restoration treatments on a legacy reclaimed mine site. Ecological Engineering 83:521–528
Birch JC, Newton AC, Alvarez Aquino C, Cantarello E, Echeverria C, Kitzberger T, Schiappacasse I, Tejedor Garavito N (2010) Cost-effectiveness of dryland forest restoration evaluated by spatial analysis of ecosystem services. Proceedings of the National Academy of Sciences 107:21925–21930
Boers AM, Frieswyk CB, Verhoeven JTA, Zedler JB (2006) Contrasting approaches to the restoration of diverse vegetation in herbaceous wetlands. In: Bobbink R, Beltman B, Verhoeven JTA, Whigham DF (eds) Wetlands: functioning, biodiversity conservation, and restoration. Springer-Verlag, Berlin, pp 225–246
Castillo JM, Figueroa E (2009) Restoring salt marshes using small cordgrass, Spartina maritima. Restoration Ecology 17:324–326
Clewell A, McDonald T (2009) Relevance of natural recovery to ecological restoration. Ecological Restoration 27:122–124
Cohen DB (2018) Microbial community assembly and hydrologic variability as controls on potential denitrification rates in restored freshwater wetlands. University of Illinois at Urbana-Champaign, Dissertation
Coiffait-Gombault C, Buisson E, Dutoit T (2012) Using a two-phase sowing approach in restoration: sowing foundation species to restore, and subordinate species to evaluate restoration success. Applied Vegetation Science 15:277–289
Cramer VA, Hobbs RJ, Standish RJ (2008) What’s new about old fields? Land abandonment and ecosystem assembly. Trends in Ecology and Evolution 23:104–112
D’Antonio CM, Chambers JC (2006) Using ecological theory to manage or restore ecosystems affected by invasive plant species. In: Falk DA, Palmer MA, Zedler JB (eds) Foundations of restoration ecology. Island Press, Washington DC, pp 260–279
David MB, McIsaac GF, Schnitkey GD, Czapar GF, Mitchell CA (2014) Science assessment to support an Illinois nutrient loss reduction strategy. University of Illinois at Urbana-Champaign, College of Agricultural, Consumer and Environmental Sciences, Urbana IL
De Steven D, Sharitz RR, Barton CD (2010) Ecological outcomes and evaluation of success in passively restored southeastern depressional wetlands. Wetlands 30:1129–1140
De Steven D, Faulkner SP, Keeland BD, Baldwin MJ, McCoy JW, Hughes SC (2015) Understory vegetation as an indicator for floodplain forest restoration in the Mississippi River Alluvial Valley, U.S.a. Restoration Ecology 23:402–412
Dey DC, Gardiner ES, Kabrick JM, Stanturf JA, Jacobs DF (2010) Innovations in afforestation of agricultural bottomlands to restore native forests in the eastern USA. Scandanavian Journal of Forest Research 25(Suppl 8):31–42
Elmer SL (2004) Soil survey of Henry County, Illinois. United States Department of Agriculture, Natural Resources Conservation Service, in cooperation with Illinois agricultural Experiment Station. US government printing office, Washington DC
Fike J, Niering WA (1999) Four decades of old field vegetation development and the role of Celastrus orbiculatus in the northeastern United States. Journal of Vegetation Science 10:483–492
Foster RD, Wetzel PR (2005) Invading monotypic stands of Phalaris arundinacea: a test of fire, herbicide, and woody and herbaceous native plant groups. Restoration Ecology 13:318–324
Garbutt A, Wolters M (2008) The natural regeneration of salt marsh on formerly reclaimed land. Applied Vegetation Science 11:335–344
Gómez-Aparicio L, Zavala MA, Bonet FJ, Zamora R (2009) Are pine plantations valid tools for restoring Mediterranean forests? An assessment along abiotic and biotic gradients. Ecological Applications 19:2124–2141
Green EK, Galatowitsch SM (2002) Effects of Phalaris arundinacea and nitrate-N addition on the establishment of wetland plant communities. Journal of Applied Ecology 39:134–144
Halle S (2007) Science, art, or application—the “karma” of restoration ecology. Restoration Ecology 15:358–361
Haynes RJ (2004) The development of bottomland forest restoration in the lower Mississippi River Alluvial Valley. Ecological Restoration 22:170–182
Helsen K, Hermy M, Honnay O (2013) Spatial isolation slows down directional plant functional group assembly in restored semi-natural grasslands. Journal of Applied Ecology 50:404–413
Higgs ES (1997) What is good ecological restoration? Conservation Biology 11:338–348
Holl KD, Aide TM (2011) When and where to actively restore ecosystems? Forest Ecology and Management 261:1558–1563
Holl KD, Howard EA, Brown TM, Chan RG, de Silva TS, Mann ET, Russell JA, Spangler WH (2014) Efficacy of exotic control strategies for restoring coastal prairie grasses. Invasive Plant Science and Management 7:590–598
Holling CS (ed) (1978) Adaptive environmental assessment and management. John Wiley & Sons, Chichester
Hovick SM, Reinartz JA (2007) Restoring forest in wetlands dominated by reed canarygrass: the effects of pre-planting treatments on early survival of planted stock. Wetlands 27:24–39
Hutchings MJ, Booth KD (1996) Studies on the feasibility of re-creating chalk grassland vegetation on ex-arable land. I. The potential roles of the seed bank and the seed rain. Journal of Applied Ecology 33:1171–1181
Iannone BV III, Galatowitsch SM (2008) Altering light and soil N to limit Phalaris arundinacea reinvasion in sedge meadow restorations. Restoration Ecology 16:689–701
Jessop J, Spyreas G, Pociask GE, Benson TJ, Ward MP, Kent AD, Matthews JW (2015) Tradeoffs among ecosystem services in restored wetlands. Biological Conservation 191:341–348
Jírová A, Klaudisová A, Prach K (2012) Spontaneous restoration of target vegetation in old-fields in a central European landscape: a repeated analysis after three decades. Applied Vegetation Science 15:245–252
Jones HP, Jones PC, Barbier EB, Blackburn RC, Rey Benayas JM, Holl KD, McCrackin M, Meli P, Montoya D, Moreno Mateos D (2018) Restoration and repair of Earth's damaged ecosystems. Proceedings of the Royal Society of London B 285:20172577
Kamali B, Hashim R (2011) Mangrove restoration without planting. Ecological Engineering 37:387–391
Kanowski A, Catterall CP, Wardell-Johnson GW, Proctor H, Reis T (2003) Development of forest structure on cleared rainforest land in eastern Australia under different styles of reforestation. Forest Ecology and Management 183:265–280
Kercher SM, Zedler JB (2004) Multiple disturbances accelerate invasion of reed canary grass (Phalaris arundinacea L.) in a mesocosm study. Oecologia 138:455–464
Kim KD, Ewing K, Giblin DE (2006) Controlling Phalaris arundinacea (reed canarygrass) with live willow stakes: a density-dependent response. Ecological Engineering 27:219–227
King SL, Keeland BD (1999) Evaluation of reforestation in the lower Mississippi River Alluvial Valley. Restoration Ecology 7:348–359
Klimkowska A, Van Diggelen R, Bakker JP, Grootjans AP (2007) Wet meadow restoration in Western Europe: a quantitative assessment of the effectiveness of several techniques. Biological Conservation 140:318–328
Klimkowska A, Kotowski W, van Diggelen R, Grootjans AP, Dzierża P, Brzezińska K (2010) Vegetation re-development after fen meadow restoration by topsoil removal and hay transfer. Restoration Ecology 18:924–933
Kozlowski TT (2002) Physiological-ecological impacts of flooding on riparian forest ecosystems. Wetlands 22:550–561
Kruse BS, Groninger JW (2003) Vegetative characteristics of recently reforested bottomlands in the Lower Cache River Watershed, Illinois, U.S.A. Restoration Ecology 11:273–280
Krzywicka AE, Pociask GE, Grimley DA, Matthews JW (2017) Hydrology and soil magnetic susceptibility as predictors of planted tree survival in a restored floodplain forest. Ecological Engineering 103:275–287
Lavergne S, Molofsky J (2006) Control strategies for the invasive reed canarygrass (Phalaris arundinacea L.) in North American wetlands: the need for an integrated management plan. Natural Areas Journal 26:208–214
Leyer I (2006) Dispersal, diversity and distribution patterns in pioneer vegetation: the role of river-floodplain connectivity. Journal of Vegetation Science 17:407–416
Lindig-Cisneros R, Zedler JB (2002) Phalaris arundinacea seedling establishment: effects of canopy complexity in fen, mesocosm, and restoration experiments. Canadian Journal of Botany 80:617–624
Linhares de Rezende C, Uezu A, Scarano FR, Araujo DSD (2015) Atlantic Forest spontaneous regeneration at landscape scale. Biodiversity and Conservation 24:2255–2272
Matthews JW (2015) Group-based modeling of ecological trajectories in restored wetlands. Ecological Applications 25:481–491
Matthews JW, Peralta AL, Soni A, Baldwin P, Kent AD, Endress AG (2009) Local and landscape correlates of non-native species invasion in restored wetlands. Ecography 32:1031–1039
Matthews JW, Pociask GE, Price EPF, Krzywicka AE (in press) Flood exposure affects long-term tree survival in compensatory mitigation wetlands. Wetlands. https://doi.org/10.1007/s13157-019-01158-7
McClain CD, Holl KD, Wood DM (2011) Successional models as guides for restoration of riparian forest understory. Restoration Ecology 19:280–289
McLeod KW, Reed MR, Nelson EA (2001) Influence of a willow canopy on tree seedling establishment for wetland restoration. Wetlands 21:395–402
Middleton B (2000) Hydrochory, seed banks, and regeneration dynamics along the landscape boundaries of a forested wetland. Plant Ecology 146:169–184
Middleton BA (2003) Soil seed banks and the potential restoration of forested wetlands after farming. Journal of Applied Ecology 40:1025–1034
Middleton EL, Bever JD, Schultz PA (2010) The effect of restoration methods on the quality of the restoration and resistance to invasion by exotics. Restoration Ecology 18:181–187
Miller JR, Hobbs RJ (2007) Habitat restoration—do we know what we're doing? Restoration Ecology 15:382–390
Mitsch WJ, Wu X, Nairn RW, Weihe PE, Wang N, Deal R, Boucher CE (1998) Creating and restoring wetlands. BioScience 48:1019–1030
Moggridge HL, Gurnell AM, Mountford JO (2009) Propagule input, transport and deposition in riparian environments: the importance of connectivity for diversity. Journal of Vegetation Science 20:465–474
Moreno-Mateos D, Meli P, Vara-Rodríguez MI, Aronson J (2015) Ecosystem response to interventions: lessons from restored and created wetland ecosystems. Journal of Applied Ecology 52:1528–1537
Munro NT, Fischer J, Wood J, Lindenmayer DB (2009) Revegetation in agricultural areas: the development of structural complexity and floristic diversity. Ecological Applications 19:1197–1210
O'Connell JL, Johnson LA, Beas BJ, Smith LM, McMurry ST, Haukos DA (2013) Predicting dispersal-limitation in plants: optimizing planting decisions for isolated wetland restoration in agricultural landscapes. Biological Conservation 159:343–354
Öster M, Ask K, Cousins SAO, Eriksson O (2009) Dispersal and establishment limitation reduces the potential for successful restoration of semi-natural grassland communities on former arable fields. Journal of Applied Ecology 46:1266–1274
Peralta AL, Muscarella ME, Matthews JW (2017) Wetland management strategies lead to tradeoffs in ecological structure and function. Elementa: Science of the Anthropocene 5:74
Perry LG, Galatowitsch SM, Rosen CJ (2004) Competitive control of invasive vegetation: a native wetland sedge suppresses Phalaris arundinacea in carbon-enriched soil. Journal of Applied Ecology 41:151–162
Plocher AE, Larimore R, Keene D (2001) Wetland mitigation monitoring for FAP 585 (IL 92). Illinois Natural History Survey, Champaign
Plocher AE, Larimore R, Keene D (2003) Wetland mitigation monitoring for FAP 585 (IL 92). Illinois Natural History Survey, Champaign
Prach K, Hobbs RJ (2008) Spontaneous succession versus technical reclamation in the restoration of disturbed sites. Restoration Ecology 16:363–366
Prach K, Pyšek P (1994) Spontaneous establishment of woody plants in central European derelict sites and their potential for reclamation. Restoration Ecology 2:190–197
Price EPF, Spyreas G, Matthews JW (2018) Biotic homogenization of regional wetland plant communities within short time-scales in the presence of an aggressive invader. J Ecol 106:1180–1190
Pywell RF, Meek WR, Webb NR, Putwain PD, Bullock JM (2011) Long-term heathland restoration on former grassland: the results of a 17-year experiment. Biological Conservation 144:1602–1609
Řehounková K, Prach K (2008) Spontaneous vegetation succession in gravel-sand pits: a potential for restoration. Restoration Ecology 16:305–312
Reid JL, Holl KD, Zahawi RA (2015) Seed dispersal limitations shift over time in tropical forest restoration. Ecological Applications 25:1072–1082
Reinartz JA, Warne EL (1993) Development of vegetation in small created wetlands in southeastern Wisconsin. Wetlands 13:153–164
Reinecke MK, Pigot AL, King JM (2008) Spontaneous succession of riparian fynbos: is unassisted recovery a viable restoration strategy? South African Journal of Botany 74:412–420
Rey Benayas JM, Bullock JM, Newton AC (2008) Creating woodland islets to reconcile ecological restoration, conservation, and agricultural land use. Frontiers in Ecology and the Environment 6:329–336
Rojas IM, Zedler JB (2015) An invasive exotic grass reduced sedge meadow species richness by half. Wetlands Ecology and Management 23:649–663
Rudgers JA, Holah J, Orr SP, Clay K (2007) Forest succession suppressed by an introduced plant-fungal symbiosis. Ecology 88:18–25
Skinner RH, van der Grinten M, Gover AE (2012) Planting native species to control site reinfestation by Japanese knotweed (Fallopia japonica). Ecological Restoration 30:192–199
Spyreas G, Wilm BW, Plocher AE, Ketzner DM, Matthews JW, Ellis J, Heske EJ (2010) Biological consequences of invasion by reed canary grass (Phalaris arundinacea). Biological Invasions 12:1253–1267
Stanturf JA, Conner WH, Gardiner ES, Schweitzer CJ, Ezell AW (2004) Recognizing and overcoming difficult site conditions for afforestation of bottomland hardwoods. Ecological Restoration 22:183–193
Stanturf JA, Gardiner ES, Shepard JP, Schweitzer CJ, Portwood CJ, Dorris LC Jr (2009) Restoration of bottomland hardwood forests across a treatment intensity gradient. Forest Ecology and Management 257:1803–1814
Streever B, Zedler J (2000) To plant or not to plant. BioScience 50:188–189
Taft JB, Wilhelm GS, Ladd DM, Masters LA (1997) Floristic quality assessment for vegetation in Illinois, a method for assessing vegetation integrity. Erigenia 15:3–95
Thomsen M, Brownell K, Groshek M, Kirsch E (2012) Control of reed canarygrass promotes wetland herb and tree seedling establishment in an upper Mississippi River floodplain forest. Wetlands 32:543–555
Toth LA (2010) Restoration response of relict broadleaf marshes to increased water depths. Wetlands 30:263–274
Tropek R, Hejda M, Kadlec T, Spitzer L (2013) Local and landscape factors affecting communities of plants and diurnal Lepidoptera in black coal spoil heaps: implications for restoration management. Ecological Engineering 57:252–260
Twedt DJ (2004) Stand development on reforested bottomlands in the Mississippi Alluvial Valley. Plant Ecology 172:251–263
Twedt DJ, Wilson RR, Henne-Kerr JL, Grosshuesch DA (2002) Avian response to bottomland reforestation: the first 10 years. Restoration Ecology 10:645–655
USACE (US Army Corps of Engineers) (1987) Corps of Engineers wetlands delineation manual. Technical report Y-87-1. Environmental laboratory, US Army Corps of Engineers Waterways Experimental Station, Vicksburg, MS
Wallace KJ, Laughlin DC, Clarkson BD (2017) Exotic weeds and fluctuating microclimate can constrain native plant regeneration in urban forest restoration. Ecological Applications 27:1268–1279
Warren J, Christal A, Wilson F (2002) Effects of sowing and management on vegetation succession during grassland habitat restoration. Agriculture, Ecosystems and Environment 93:393–402
Werner KJ, Zedler JB (2002) How sedge meadow soils, microtopography, and vegetation respond to sedimentation. Wetlands 22:451–466
Wilson JB, Agnew ADQ (1992) Positive feedback switches in plant communities. Advances in Ecological Research 23:263–336
Zahawi RA, Reid JL, Holl KD (2014) Hidden costs of passive restoration. Restoration Ecology 22:284–287
Zedler JB (2009) Feedbacks that might sustain natural, invaded, and restored states in herbaceous wetlands. In: Hobbs RJ, Suding KN (eds) New models for ecosystem dynamics and restoration. Island Press, Washington, DC, pp 236–258
Zedler JB, Kercher S (2004) Causes and consequences of invasive plants in wetlands: opportunities, opportunists, and outcomes. Critical Reviews in Plant Sciences 23:431–452
Zedler JB, Doherty JM, Miller NA (2012) Shifting restoration policy to address landscape change, novel ecosystems, and monitoring. Ecology and Society 17:36
Acknowledgements
Illinois Department of Transportation provided funding for this project. George Geatz and Kyle Van den Bosch assisted with fieldwork. Dennis Skultety assisted with GPS/GIS.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Matthews, J.W., McIntyre, S., Peralta, A.L. et al. Long-Term Assessment of Alternative Strategies for the Restoration of Floodplain Forest in the Presence of an Invasive Grass, Phalaris arundinacea. Wetlands 40, 655–665 (2020). https://doi.org/10.1007/s13157-019-01204-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13157-019-01204-4