Abstract
We investigated the inter-annual and seasonal variation of the aboveground net primary productivity (ANPP), and leaf nitrogen and phosphorus concentrations across two different types of wet meadows (spring-fed and riparian) in Tierra del Fuego Island, Southern Patagonia. In addition, we estimated grazing effects on ANPP, the nature of nutrient limitation, and the resorption efficiencies for both nutrients. ANPP showed a high inter-annual variation (CV = 49–57%), typical seasonal dynamics with a summer peak, and higher annual means at the spring-fed meadows (532 g∙m−2∙yr.−1) compared to the riparian meadows (310 g∙m−2∙yr.−1) with a higher contribution of graminoids (50–65%). Sheep grazing reduced a 66% the annual mean of ANPP (108 g∙m−2∙yr.−1) in riparian meadows. Leaf nitrogen concentrations were similar to reported values for graminoids from temperate and cold regions (1.5–2.5%), while leaf phosphorus concentrations were markedly higher (0.2–0.3%), indicating a nitrogen limitation. Resorption efficiency was 54.2% for nitrogen and 48.2% for phosphorus, exhibiting incomplete resorption proficiency for both nutrients. Our results represent valuable information to design sustainable management strategies for wetlands according to their spatial and temporal variation, and to define a baseline of vegetation functioning to assess the impact of climate change in high-latitude southern ecosystems.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Wetlands occupy an extent of 7 to 10 million km2 representing about 5 to 8% of the Earth’s land surface (Mitsch and Gosselink 2000). They spread over a wide latitudinal and ecosystem range across cold, temperate and tropical climates and varying from coastal mangroves and marshlands to inland grasslands (Whigham et al. 1993; Lehner and Döll 2004). Despite this relative low percentage, wetlands are main providers of important ecosystem services at a global scale, such as water and oxygen supply and balance, natural water filtering, primary productivity, nutrient cycling, habitat and biodiversity conservation, basin water balance, among others (Newman et al. 2006; Mitsch and Gosselink 2000). These ecosystems embedded in arid or semiarid regions are particularly important because they constitute an oasis for agriculture within generally unfavorable local conditions (Hollis 1990; Belsky et al. 1999).
Wetlands are currently threatened by vegetation and soil degradation processes due to unsustainable grazing (Davis and Froend 1999; Taboada et al. 1999), biological invasions (Zedler and Kercher 2004; Levin et al. 2006), drainage for urbanization or real estate purposes, and unpredictable effects of global warming (Erwin 2009), particularly in high-latitudes (Pauchard et al. 2016). Hence, the study of spatial and temporal variation of above-ground net primary productivity (hereafter ANPP) of wetland meadows and the related environmental drivers is fundamental for conservation and sustainable grazing management. In addition, the study of environmental drivers linked to the spatial and temporal variation of ANPP allows us to design grazing strategies to adapt or mitigate to unfavorable environmental conditions (Wang et al. 2012). However, this kind of studies is strongly limited by the poor availability of field data on ANPP and environmental drivers with extended spatial and temporal coverage (Knapp and Smith 2001; Jobbágy et al. 2002).
At a regional scale, climate variability (i.e. mainly annual and seasonal rainfall), is the main environmental driver of ANPP inter-annual variation (Sala et al. 1988; Knapp and Smith 2001). In sub humid ecosystems the relationship between ANPP and annual rainfall at a regional scale is strong (Knapp and Smith 2001; Jobbágy and Sala 2000), but that correlation seems to vanish at high mean annual rainfall (Oesterheld et al. 1999; Huxman et al. 2004). However, at local scales ANPP temporal variation is less explained by annual rainfall (Paruelo et al. 1999; Knapp and Smith 2001), and local drivers such as soil biogeochemistry, hydrological regime, grazing, fire and “memory effects” gain importance (Burke et al. 1997; Oesterheld et al. 2001).
Wetland ANPP is also frequently limited by nitrogen or phosphorus (Verhoeven et al. 1996; Güsewell 2004, 2005; Elser et al. 2007), which has also an effect on the floristic composition and succession dynamics of plant communities (Chapin et al. 1986). Moreover, global warming is expected to provoke a significant impact on nutrients cycling in nutrient-limited ecosystems from high-latitude regions (Van Heerwaarden 2003). Hence, fertilization experiments and studies on nutrient concentrations in biomass are important to reveal possible nutrient limitations and to determine which nutrients are deficient (Venterink et al. 2001; Wassen et al. 2002; Niinements and Kull 2005). Information on N:P ratios can give a broad idea about the limiting nutrient for plants (Koerselman and Meuleman 1996; Güsewell 2004).
In continental Patagonia, southern South America, wetland meadows occupy a 2–8% of the total area of the region (780,000 km2) representing a minor extent in comparison to more xerophytic communities like grass or shrub steppes (Iriondo 1989; Bran et al. 2004). However, in Tierra del Fuego Island this percentage is higher and varies depending on the zone (ca. 8–25%; Anchorena et al. 2016). Despite their minor extent, wetlands have high productivity and offer forage that have allowed continuous sheep grazing on these rangelands during the last century (Golluscio et al. 1998; Anchorena et al. 2001). Wetland meadows vary across Patagonia due to differences in lithology, location within particular watershed basin, latitude, topography, hydrology, soil type and land use (Bran et al. 2004; Collantes et al. 2013). They are small patches scattered within the landscape, primarily composed of grasses, sedges and forbs, mostly associated to permanent or transient shallow water streams with soils rich in organic matter and nutrients (Mazzoni and Vázquez 2004; Perotti et al. 2005). Nowadays, unsustainable grazing has led to wetlands severe degradation, disturbances in water dynamics and physical-chemical soil properties, decreases in vegetation cover, soil erosion, and a decrease in soil fertility (del Valle 1993; Utrilla et al. 2005, 2006; Gaitán et al. 2011). In addition, human activities result in the introduction of exotic species, threatening meadows biodiversity (Iglesias and Pérez 1998; Perotti et al. 2005). Moreover, predicted increases in temperature and decreases in precipitation and river water flows in the region for next decades may accelerate the current degradation processes (Vera et al. 2006; Kitoh et al. 2011).
In this study, we address different questions related to the functioning and degradation of wetlands, particularly: 1) Which are the main differences in the ANPP between riparian- and spring-fed meadows in different functional groups? 2) Which nutrient (N or P) is limiting the growth of the meadows? 3) Which are the grazing effects on the ANPP? and 4) In which extent do rainfall and temperature explain the inter-annual variation of ANPP and leaf nutrient concentrations? To answer these questions we analyze long-term data on ANPP and nutrients from different types of wetland meadows taking into account different co-dominant plant functional groups. We hope our work will contribute to sustainable management of these ecosystems in cold sub-humid and high-latitude regions of South Patagonia, Argentina.
Materials & Methods
Study Area
The steppe region of Tierra del Fuego is part of the Magellanic steppe, a humid grassy vegetation type extending south of the Patagonian semidesert on the continent, at approximately 51°25′ S, and reaching the Nothofagus forests at approximately 54° S in the Tierra del Fuego Island (León et al. 1998; Collantes et al. 1999). In the Fuegian steppe, daily and seasonal variations of temperature are small (from a monthly mean of 0 °C in July to 10 °C in January; Estadísticas Meteorológicas 1986), cloudiness is high, winds are strong, and the Falklands ocean stream determines a colder climate than expected at its latitudinal location (Collantes et al. 1999). Spatially, the main climatic gradient has a southwest-northeast direction, due to the rain shadow produced by the Andes Mountains on the predominant westerlies. Within 150 km, annual rainfall decreases from 450 mm in the southwest of the area to ca. 280 mm in the northeastern extreme. The relief of the steppe region is flat to undulating, with some low to medium-height hills (Codignotto and Malumián 1992). The areas covered during Pleistocene by continental glaciation are dominated by till and outwash deposits of coarse texture. In contrast, large areas not covered formerly by glaciers or meltwater deposits are built of old Tertiary rocks of medium to fine texture.
At a regional scale, highland vegetation is dominated by tussock grasslands of Festuca gracillima Hooker f. and its variants are mostly controlled by climate and lithology (Collantes et al. 1999), while lowlands are covered by wetland communities dominated by hygrophilous vegetation (Collantes et al. 2009). Wetland meadows of the Magellanic region are affected by two main environmental gradients: a regional latitudinal-climatic gradient of increasing rainfall and decreasing temperature towards the southwest, and a local-topographic gradient that controls the water table depth (Collantes et al. 2009); resulting in a main floristic gradient of wetlands that differentiate into dry and wet meadows (Collantes et al. 2013). Wet meadows have organic or mineral soils, and a water table between 0 and 40 cm below the surface in summer. They occupy depressed areas of valleys and are characterized by dwarf sedges (Carex vallis-pulchrae Phil., C. macrosolen), rushes (Juncus scheuchzerioides Gaudich.), and soft grasses (Hordeum lechleri [Steud.] Schenck, Deschampsia antarctica Desv., and P. pratensis, among others). Generally, the vegetation and soil type of wet meadows are closely related to the wetland hydrology determining two different types of wet meadows: spring-fed or riparian (Collantes et al. 2009). In spring-fed meadows groundwater flow is dispersed and not concentrated to a defined stream network as is the case in riparian meadows. Hence, soil types associated to Fuegian spring-fed meadows are Histosols or Fluvisols, while in riparian meadows the dominant soil type is Gleysols (Collantes et al. 2013). Generally, spring-fed meadows are associated to neutral soils (pH 6–7), with high CEC and carbon, nitrogen, and cations content (Ca, Mg, Na and K), and more abundance of sedges and rushes compared to riparian meadows (Collantes et al. 2009, 2013). On the other hand, riparian meadows are associated to more acidic soils (pH 5.5–6) with low CEC, carbon, nitrogen, and cations content, and to plant communities richer in grass species.
Field Surveys
In order to characterize the inter-annual and seasonal variation of ANPP and nutrients (i.e. leaf nitrogen and phosphorus) we analyzed data from different field studies (Fig. 1). To describe the inter-annual variation in ANPP we evaluated two meadows representative for the main types of wet meadows described by Collantes et al. (2013) (i.e. spring-fed and riparian) during 6–9 growing seasons. ANPP studies on both meadow types were located in paddocks (i.e. large fenced areas, ~2.000–2500 ha) managed under long-term continuous grazing with an average stocking of ca. 1 sheep·ha−1·yr.−1. However, punctual grazing pressure on meadows (about 15% of paddocks area, with the remaining 85% being upland communities) varied between a stocking rate of 3.2 and 10 sheep·ha−1·yr.−1 (Anchorena et al. 2001). ANPP was estimated with the method based on the peak of biomass recorded at the end of growing season (January–February). This procedure also known as peak standing crop or peak live biomass is a simple and most common method which uses the maximum amount of live plus recent dead material (current growing season) as estimate of ANPP (Sala et al. 2000; Scurlock et al. 2002; Ruppert and Linstädter 2014). In the wet spring-fed meadow (ca. 30 ha) we cut all vegetation at the ground level (1 cm) from 10 sampled quadrats (0.1 m2) randomly set during nine consecutive growing seasons (1987/88–1995/96) from a grazing exclosure (800 m2) set in 1986. In the wet riparian meadow (ca. 15 ha) we followed the same protocol to characterize the ANPP inter-annual variation, but we followed six consecutive growing seasons (1996/97–2001/02) from a grazing exclosure (800 m2) set in 1996. To analyze grazing effects, we used ten mobile cages (1 m2) to exclude sheep grazing near to the exclosure area in the wet riparian meadow. Floristic similarities between exclosure and grazed area (Sørensen index, Mueller-Dombois and Ellenberg 1974) ranged from 68% to 80% when the sampling started. This is about the maximum similarity that can be expected for replicated stands (Mueller-Dombois and Ellenberg 1974). Cages were located at the end of the winter in the grazed area, and biomass was cut at the beginning (i.e. outside the caged site to estimate biomass availability) and at the end of the growing season. During the next winter, all cages were randomly re-located in the meadow avoiding previously cut areas to estimate biomass availability, ANPP and animal intake for the next growing season following the same protocol. Harvested biomass samples were conserved in freezers, processed in the laboratory and separated in three main fractions (i.e. green graminoids, green forbs, and standing dead biomass), oven-dried at 65 °C during 72 h, and then weighed. To track inter-annual changes in leaf nutrient status we measured the nitrogen and phosphorus concentration for each main fraction. The same biomass samples used for ANPP were taken into account in the riparian meadow under grazing during five consecutive growing seasons (1996/97–2000/01).
Study area and location of surveyed wet meadows: Isla Grande de Tierra del Fuego (dashed square) at the southern extreme of South America (a); study area (dashed rectangle) in the Isla Grande de Tierra del Fuego (Arg.) (b); and specific locations for the surveyed wet meadows in the Río Grande basin (c). In panel “c” ring markers indicated riparian meadows, while star markers indicated spring-fed meadows
To estimate the seasonal variation of ANPP we studied five wetland meadows during three growing seasons (1999/00–2001/02), where we estimated ANPP for each different season (3-months) by multiple successive cuts in specific months (February, May, August and November) in 10 mobile cages (1 m2) per meadow located in grazed paddocks (Fig. 1). The harvested biomass samples were processed as previously mentioned to estimate the ANPP for each season. By subtracting the final recorded biomass inside cages (BiomassIC tf in Eq. 1) and the available biomass outside cages at the beginning of each growing period (BiomassOC t0 in Eq. 1), we estimated the seasonal ANPP. In all cases, ANPP was expressed as grams of dry matter per square meter and year.
In order to track the seasonal changes in the nutrient status we measured the leaf nitrogen and phosphorus concentration for each main fraction from ten biomass samples used to estimate seasonal ANPP, but only during two consecutive growing seasons (2000/01 and 2001/02). Nitrogen concentration was determined by MicroKjeldahl method on a hot plate with sulphuric acid (Page et al. 1982). Phosphorus concentration was determined by digesting biomass samples on a hot plate with nitric and perchloric acid. Then, phosphorus concentration was measured by spectrophotometry and colorimetric technique with vanadate-molybdate reagent (Jackson 1964).
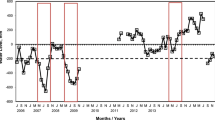
Weather data were obtained from an autonomous data logger station (LICOR LI-1200) installed in the study area with respective sensors to record daily: rainfall, aerial temperature (mean, maximum and minimum), soil temperature (mean) and radiation. In addition, we complemented this dataset with meteorological records taken in the headquarters of a ranch located close to the studied area (14 km) and the recorder station from the National Meteorological Service located in the Río Grande airport (27 km). The average total annual precipitation is 361 mm, falling as rain and snow, and having a slight seasonal peak in December–January (Fig. B1 in Appendix B). During the periods of the study, mean annual rainfall was lower than the long-term average, with several years below 220 mm during a drought cycle between 1995 and 2004 (Fig. B1).
Data Analysis
To detect differences in ANPP throughout years we used linear models and conducted ANOVAs separated by main fractions (i.e. total, graminoids, and forbs) and within each type of meadow (spring-fed and riparian). Similarly, to detect differences in leaf nutrient concentration throughout years we used linear models and conducted ANOVAs separated by main fractions (i.e. graminoids, forbs, and standing dead biomass) and within each nutrient (i.e. nitrogen and phosphorus). In order to detect differences in ANPP across seasons we used mixed linear models and conducted ANOVAs separated by main fractions (i.e. total, graminoids, and forbs), where season was the fixed effect and meadow was the block random effect with the ANPP measures repeated across different years. Likewise, to detect differences in leaf nutrient concentration across seasons we used mixed linear models and conducted ANOVAs separated by each main fraction and nutrient with the same fixed and random effects as ANPP analysis.
To detect relationships between ANPP and nutrients concentration with climatic drivers, we conducted Pearson correlation analyses among them. Specifically, we correlated the ANPP for different fractions, leaf N and P concentration with monthly data on mean temperature and rainfall of the respective growing season (i.e. September to February). In addition, we conducted correlation analysis between ANPP and precedent standing dead biomass to detect possible negative feedbacks on productivity.
To evaluate grazing effects, we estimated the animal intake and the harvest index in grazed areas near to grazing exclosures of wet riparian meadow during four growing seasons. Animal intake was calculated as the difference between the ANPP estimates and the final recorded biomass outside cages for a particular growing season (Eq. 2a).
Then, we related the animal intake with the respective ANPP for the same growing season to obtain the harvest index expressed as a proportion of ANPP (Eq. 2b).
Finally, we estimated the nutrient resorption efficiency based on the nitrogen and phosphorus concentration in green and standing dead biomass according to the eq. 3, and the nutrient proficiency as the N or P concentration in standing dead biomass.
Results
Inter-Annual Variation of ANPP
The spring-fed meadow approximately doubled the total ANPP of the riparian meadow at ungrazed conditions. In the spring-fed meadow, the total ANPP varied between 200 and 1000 g∙m−2∙yr.−1 with a mean ± SE value ca. 532 ± 60.8 g∙m−2∙yr.−1 (Fig. 2a), whereas in the riparian meadow the total ANPP varied between 100 and 500 g∙m−2∙yr.−1 with a mean ± SE value ca. 310 ± 32.7 g∙m−2∙yr.−1 (Fig. 3a). However, the analysis of 6–9 years of ANPP data yielded that both wetland meadows exhibited a high and similar relative inter-annual variation (ca. 5 times between the minimum and maximum ANPP), mostly explained by the graminoids fraction (50–65%; Figs. 2 and 3b) and in a lesser extent by the forbs (5–25%) (Figs. 2c and 3c; Tables A1-A3).
Aboveground net primary productivity (ANPP) across growing seasons for total (a), graminoids (b) and forbs (c) in ungrazed conditions from a wet spring-fed meadow. Different letters indicate statistical differences (p < 0.05) across growing seasons. Dashed lines indicate the temporal mean for each panel
Aboveground net primary productivity (ANPP) across growing seasons for total (a), graminoids (b) and forbs (c) under two grazing conditions from a wet riparian meadow. Different lower-case letters indicate statistical differences (p < 0.05) across growing seasons in ungrazed condition, while different upper-case letters indicate statistical differences in grazed condition. Dashed lines indicate the temporal mean for ungrazed condition and continuous lines indicate the temporal mean for grazed condition
The riparian meadow had a lower total ANPP at grazing conditions, ranging between 50 and 200 g⋅m−2⋅yr−1, with a mean ± SE value ca. 108 ± 9.4 g⋅m−2⋅yr−1 (Fig. 3a), and a higher contribution of graminoids (Fig. 3b, c). Based on the results of ANPP at grazed conditions and available biomass during four growing seasons, we estimated a mean animal intake of 45±29 g⋅m−2⋅yr−1 and a mean harvest index of 43±17%. Also, we found a negative significant correlation between the current green ANPP and the standing dead biomass during the previous year (r = −0.593, p = 0.033).
Seasonal Variation of ANPP
The ANPP at grazed conditions showed a high seasonal variation, especially between cold and warm seasons, with differences across fractions (Fig. 4; Table A4). The mean total ANPP during the cold season (i.e. fall and winter) was not different from zero, indicating a higher senescence than production during this season (Fig. 4a), whereas in the warm season, the mean total ANPP was about 70 and 150 g∙m−2∙yr.−1 in spring and summer, respectively (Fig. 4a). Total ANPP variation across seasons was mostly explained by the graminoids contribution (50–70%; Fig. 4b), and in a lesser extent by the forbs (5–20%; Fig. 4c). In addition, senescence was higher during the fall compared to the winter for both fractions (i.e. graminoids and forbs), but we did not detect differences in the productivity between the spring and the summer (Fig. 4b, c).
Inter-Annual Variation of Leaf Nutrient Concentrations
Overall analysis of the 5-years dataset showed that leaf nutrient concentrations varied inter-annually in a lesser extent than ANPP, and depended on biomass fractions, (graminoids and forbs > standing dead) and nutrient type (nitrogen or phosphorus) (Fig. 5; Tables A5-A6). In graminoids, leaf nitrogen concentration ranged between 1.4 and 2% (Fig. 5a), while in forbs it ranged between 1.3 and 1.9% and had a remarkable decrease in the 1998/99 and 1999/00 growing seasons (Fig. 5b). Standing dead was the biomass fraction with the lowest nitrogen concentration (0.9–1.25%) and variability compared to the measured concentrations in graminoids or forbs (Fig. 5c). Leaf phosphorus concentration in graminoids ranged between 0.18 and 0.25%, and exhibited higher concentrations in the last growing season (Fig. 5d). In forbs, leaf phosphorus concentration ranged between 0.18 and 0.28%, and presented a similar pattern as nitrogen dynamics (Fig. 5e). Also, standing dead biomass was the fraction exhibiting lower phosphorus concentrations (0.11–0.16%) and variability compared to the graminoids or forbs (Fig. 5f).
Seasonal Variation of Leaf Nutrient Concentrations
Leaf nutrient concentration dynamics across seasons showed differences among biomass fractions (graminoids, forbs and standing dead) and nutrient type (nitrogen or phosphorus) (Fig. 6; Tables A7-A8). In the graminoids, leaf nitrogen concentration ranged between 1.6 and 2.5%, having the lowest concentration in summer (Fig. 6a), while in the forbs it ranged between 1.5 and 2.1% and the lowest concentrations were measured in winter and summer (Fig. 6b). Nitrogen concentration across seasons in standing dead biomass showed no differences, with the lowest global mean around 1.3% (Fig. 6c). Leaf phosphorus concentration ranged between 0.2 and 0.26% in the graminoids, with the lowest concentration in the fall (Fig. 6d); whereas in the forbs, it ranged between 0.16 and 0.29%, and the lowest concentrations were measured in winter and summer (Fig. 6e). No differences were detected in the phosphorus concentration across seasons in standing dead biomass, with the lowest global mean around 0.14% (Fig. 6f).
Leaf nitrogen (a-c) and phosphorous (d-f) concentration across seasons for graminoids (a, d), forbs (b, e), and standing dead (c, f) biomass fractions in grazed condition from different meadows (5) and years or growing seasons (2). Different letters indicate statistical differences (p < 0.05) across seasons within each panel
Based on previous results of nutrients concentrations across different meadows and growing seasons, the estimation of N:P ratio of green biomass in the warm season was 7.4±2.5 (mean±SD), while the same ratio in the cold season reached to 11.3±4.7. Based on the difference in nutrients concentrations between green and senescent biomass in consecutive seasons we estimated the resorption efficiency of nutrients, that was 54.2±6.9% for nitrogen and 48.2±6.7% for phosphorus. In addition, the resorption proficiency was incomplete for both nutrients (i.e. N > 0.7% and P > 0.05% in senescent leaves).
Climatic Controls of ANPP and Nutrients
ANPP was positively correlated with the monthly rainfall at the beginning of the growing season (October; Table 1), and negatively correlated with the mean monthly temperature of October, especially for the total and graminoids fractions (Table 1).
Nutrients have shown different correlative responses to rainfall and temperature, and in different months within the growing season. In the graminoids, the nitrogen concentration was negatively correlated with the monthly rainfall and positively with the mean monthly temperature at the beginning of the growing season (Oct-Nov; Table 2); while phosphorus concentrations correlated negatively with October rainfall and with the mean temperature in January (Table 2). In the forbs, nitrogen concentration was negatively correlated with October rainfall, while phosphorus concentration correlated positively with September rainfall, and negatively with December mean temperature (Table 2).
Discussion
Inter-Annual and Seasonal Variation of ANPP
Mean total ANPP of both types of wetland meadows (300 and 550 g∙m−2∙yr.−1 for riparian and spring-fed meadows, respectively) was higher than the ANPP reported for other cold regions, such as Asian meadows (Guo et al. 2006; Ganjurjav et al. 2015), European alpine meadows (Ren et al. 2010), fen meadows in Northern Europe (Peregon et al. 2005, 2008), and America (Van der Valk and Bliss 1971; Weltzin et al. 2000; Chapin et al. 2004). The oceanic influence on the climate of Tierra del Fuego Island may determine more humid and mild winters and a longer growing season compared with more continental areas of similar latitude (Koremblit and Forte Lay 1991). However, our results indicate a higher inter-annual variation in ANPP for both types of wet meadows (CV = 49 and 57% for spring-fed and riparian meadows, respectively), reaching to 5 times between the min and max values recorded for studied periods (6–9 years). This inter-annual variation of ANPP is higher than that reported from colder ecosystems like alpine or northern meadows and boreal fen peatlands (Thormann and Bayley 1997; Peregon et al. 2008; Ren et al. 2010), where mostly the max estimates of ANPP have doubled the minimums. Nevertheless, the inter-annual variation of ANPP is similar to that reported from temperate wetlands and marshlands dominated by graminoids (Altesor et al. 2005; Ewe et al. 2006). Probably, the inter-annual variation in mean temperature at the beginning of the growing season (Sept-Oct) regulates the length of growing season favoring the alternation of years or short cycles with more harsh or mild winters according to the functioning of oceanic streams. On the other hand, methodological differences can explain the higher inter-annual variation of ANPP here encountered. Particularly, the time frame covered by our ANPP measurements (e.g. > 6-yr) or decisions about the inclusion of dead matter to estimate ANPP can influence the inter-annual variation recorded (Scurlock et al. 2002).
Our estimates on ANPP from meadows in Tierra del Fuego Island are relatively similar to previous reports from northern Patagonian locations, especially from the riparian meadow type and estimates based on the central zone of meadows (Utrilla et al. 2005, 2006; Buono et al. 2010), but lower than the ANPP estimates from Irisarri et al. (2012). Differences may be due to the different locations, i.e. higher latitude and colder sites of this study, and degradation conditions. In addition, the reported spatial variation of 2–3 times in the mean ANPP among different meadows was generally higher than our estimates. The high spatial variation reported at a regional scale may be associated to larger differences in the main environmental controls of ANPP such as radiation, rainfall, temperature, salinity, water table depth and pH (Gaitán et al. 2011; Crego et al. 2014a) that were not embraced in our study. On the other hand, the seasonal variation of ANPP was typical of the region, with a major peak at the end of growing season, a minor peak during spring, and a higher senescence rate during the cold season.
Effects of Grazing on ANPP
Sheep grazing resulted in a 66% decrease of the mean total ANPP, and in an increase of the inter-annual variation (CV = 67%) in riparian meadows of the Fuegian steppe with respect to the adjacent ungrazed enclosures. Grazing effects were significant, despite the fact that field measurements begun only one year after the set of exclosures and we would expect changes to be more gradual in vegetation composition. Our results agree with most results in sub-humid and semi-arid grasslands, where domestic grazing generally reduced the ANPP (Milchunas and Lauenroth 1993; Irisarri et al. 2016). However, the relative reduction of ANPP by grazing (66%) was quite high compared with other grasslands (10–30%), probably environmental and evolutionary factors conditioned this response (Milchunas and Lauenroth 1993). On the other hand, the intake estimates and harvest index of sheep are within the ranges reported for Patagonian steppes in accordance to the respective ANPP values here reported (Golluscio et al. 1998). Grazing history by sheep during the last 120 years in the Fuegian steppe has caused large changes in the floristic composition of rangelands (Cingolani et al. 2005), particularly in the meadows (Collantes et al. 2013). Generally, tall and soft graminoids are replaced by cushion shrubs, short graminoids and forbs (e.g. Deyeuxia poaeoides, Hordeum pubiflorum, Koeleria fueguina and Juncus scheuchzerioides are replaced by Caltha sagittata, Azorella trifurcata and Poa spiciformis). This replacement of plant species explained the remarkable decrease of the ANPP, and turning the ANPP also more sensible to the weather conditions in each growing season, and consequently increasing the inter-annual variability.
Inter-Annual and Seasonal Variation of Leaf Nutrient Concentrations
The analysis of inter-annual and seasonal variation of leaf nutrient concentrations in the green biomass revealed that seasonal variation of nitrogen and phosphorus concentration was slightly higher than inter-annual variation, particularly in forbs. Previous works have also reported subtle differences across seasons in leaf N and P concentrations, particularly in forbs, compared to the main sources of variation such as soil nutrient availability and across plant species or functional types (Chapin et al. 1980; Güsewel and Koerselman 2002; Wang and Moore 2014). Compared to other wetlands around the World, leaf nitrogen concentrations in green biomass were similar (1.5–2.5%), but leaf phosphorus concentrations were quite higher (0.2–0.3%). Specifically, our estimates are higher than the 75th percentile for phosphorus concentrations in grasslands reported by Chapin et al. (1986), probably explained by the role of waterlogged conditions in wet meadows during the growing season (Rubio and Lavado 1999). Alternatively, this differential accumulation of phosphorus in plant tissues can be also related to luxury consumption (Oyarzábal and Oesterheld 2009) or possible mycorrhizal symbiosis (Marschner and Dell 1994) that in both cases deserve more studies to disentangle their role in Fuegian meadows.
Although the N:P ratio in plant tissues (Koerselman and Meuleman 1996; Güsewell 2004, 2005) was subject of much debate, most authors seem to agree that this ratio can be used to determine the type of growth limitation (N or P) (Verhoeven et al. 1996; Tessier and Raynal 2003; Güsewell and Koerselman 2002). Güsewell (2004) proposed N:P ratios <10 and >20 as critical thresholds in terrestrial ecosystems to determine N or P limitation, respectively. According to Güsewell (2004), our results point to a nitrogen limitation in vegetation of wet meadows in Tierra del Fuego during the growing season. Indeed, our results on N:P ratio agree with the high biomass increases (3–4 times compared to the unfertilized treatment) in response to the N fertilization (360 kg·ha−1 ammonium nitrate) reported from field experiments conducted in meadows on organic soils of Tierra del Fuego (San Martino 2003). However, the use of other indicators of N and P limitation, as resorption proficiency, are also recommended because N:P ratio is a relative measure and can not represent low absolute concentrations simultaneously in both nutrients (Killingbeck 1996).
Resorption proficiency is defined as the ability of a species to reduce N and P concentrations in senescent leaves below established benchmark levels, either by resorption before senescence (high resorption efficiency) or by having low nutrient requirements, i.e. < 0.7% N for all species, < 0.04% P in evergreens, and <0.05% P in deciduous species (Killingbeck 1996). Resorption of nutrients from senescing leaves is of great ecological significance, because it is a major strategy for conserving plant nutrients for growth, nutrient absorption and competition (Aerts and Chapin 2000). We find that nutrient concentration in standing dead biomass was relatively high and stable throughout years and across seasons. This result indicate an incomplete resorption proficiency in both nutrients, but remarkable higher and thus less complete in phosphorus. These results are in accordance with our previous conclusion indicating a higher demand for nitrogen, but do not support the notion that resorption is a key process to recycle nutrients at plant level in these wetlands. Probably, soil nutrients availability related to plant requirements (Ven Heerwaarden et al. 2003; Rejmánková 2005), and the low internal remobilization besides decomposition rates due to the cold weather explain this result (Escartín et al. 2010).
Effects of Rainfall and Temperature on ANPP and Leaf Nutrient Concentrations
Overall results showed that ANPP and nutrient dynamics were associated to rainfall, as expected for a water-limited ecosystem, i.e. annual precipitation ranges between 280 and 450 mm·year−1. In fact, all fractions of ANPP (i.e. total, graminoids, and forbs) were positively associated with rainfall, and negatively with temperature. Even though wet meadows have more water availability than upland plant communities (e.g. fescue steppe or shrublands), the role of rainfall at the beginning of the growing season seems to be also important to determine the ANPP in these ecosystems.
The negative correlation of ANPP with temperature may be caused by a length reduction of the growing season due to the balance between water losses by evapotranspiration and the soil water holding capacity. This shortening in the length of growing season promotes in turn early and longer drought periods as it was reported in other sub-humid grasslands (Epstein et al. 1997). Furthermore, the occurrence of a significant correlation between the ANPP and standing dead biomass of the preceding growing season indicates the existence of “memory” effects or short-term cycles of productivity mainly controlled by the accumulation of litter on aboveground (Oesterheld et al. 2001; Wiegand et al. 2004).
Leaf nutrient concentrations were also correlated to rainfall and temperature, depending on nutrient type (N or P) and plant functional group (i.e. graminoids or forbs). Leaf nitrogen concentration in graminoids and forbs was negatively associated with rainfall, probably due to an increased biomass dilution effect (Lü et al. 2012). It is also possible that in wetter years the waterlogged conditions throughout the growing season slowed down decomposition processes in soil and thus reduced availability of nitrogen for plants. By contrast, leaf phosphorus concentration was negatively associated with rainfall in graminoids, but positively in forbs. These differential responses of leaf nutrient concentration and resorption efficiency to environmental factors among different plant functional types or plant species were previously reported, where the functional group or species identity is more important than climate factors or soil conditions to determine plant nutrient responses (Kao et al. 2003; Ven Heerwaarden et al. 2003; Güsewell 2005; He et al. 2008).
The sub-artic and sub-antarctic wetlands play a significant role in the global carbon cycle. The increase of atmospheric CO2 and the global warming can affect the ANPP and the carbon pools stored in terrestrial ecosystems, particularly at higher latitudes (Pauchard et al. 2016). Based on climate scenarios projections and species distribution models, Crego et al. (2014b) reveal a reduction ca. 8% of the suitable area for meadows in Tierra del Fuego Island by 2050. In this context, our results are relevant because they define an ANPP and nutrients baseline of the ecosystem functioning from the high productive ecosystems in Southern Patagonia, useful to estimate and mitigate global warming effects in the future. To conclude, our results bring valuable information to design conservation practices and grazing sustainable managements according to the spatial and temporal variation of these important environments and main providers of multiple ecosystem services in Patagonian rangelands.
References
Aerts R, Chapin FS (2000) The mineral nutrition of wild plants revisited: a re-evaluation of process and patterns. Advances in Ecological Research 30:1–67
Altesor A, Oesterheld M, Leoni E, Lezama F, Rodríguez C (2005) Effects of grazing on community structure and productivity of a Uruguayan grassland. Plant Ecology 179:83–91
Anchorena J, Cingolani AM, Livraghi E, Collantes MB, Stoffella S (2001) Manejo del pastoreo de ovejas en Tierra del Fuego. Edipubli, Buenos Aires
Anchorena J, Dieguez H, Collantes MB, Cingolani A (2016) A vegetation map for the land use planning of the southernmost rangelands of the world: the steppes of Tierra del Fuego. Proceedings of the 10th International Rangeland Congress. Saskatoon, Canada
Belsky AJ, Matzke A, Uselman S (1999) Survey of livestock influences on stream and riparian ecosystems in the western United States. Journal of Soil and Water Conservation 54:419–431
Bran D, Gaitan J, Ayesa J, López C (2004) La vegetación de los mallines del NO de la Patagonia. Los mallines en la Patagonia Argentina. Esquel, Taller
Buono G, Oesterheld M, Nakamatsu V, Paruelo JM (2010) Spatial and temporal variation of primary production of Patagonian wet meadows. Journal of Arid Environments 74:1257–1261
Burke IC, Lauenroth WK, Parton WJ (1997) Regional and temporal variation in net primary production and nitrogen mineralization in grasslands. Ecology 78:1330–1340
Chapin FS III, Johnson DA, McKendrick JD (1980) Seasonal movement of nutrients in plants of differing growth from in an Alaskan tundra ecosystem: Implications for herbivory. Journal of Ecology 68:189–209
Chapin FS III, Vitousek PM, Van Cleve K (1986) The nature of nutrient limitation in plant communities. The American Naturalist 127:48–58
Chapin CT, Bridgham SD, Pastor J (2004) pH and nutrient effects on above-ground net primary production in a Minnesota, USA bog and fen. Wetlands 24:186–201
Cingolani AM, Noy-Meir I, Díaz S (2005) Grazing effects on rangeland diversity: A synthesis of contemporary models. Ecological Applications 15:757–773
Codignotto JO, Malumián N (1992) Geología de la región al norte del paralelo de 54° S de la Isla Grande de Tierra del Fuego. Revista de la Asociación Geológica Argentina 36:44–88
Collantes MB, Anchorena J, Stoffella S, Escartín C, Rauber R (2009) Wetlands of the Magellanic Steppe (Tierra del Fuego, Argentina). Folia Geobotanica 44:227–245
Collantes MB, Anchorena J, Cingolani AM (1999) The steppes of Tierra del Fuego: floristic and growthform patterns controlled by soil fertility and moisture. Plant Ecology 140:61–75
Collantes MB, Escartín C, Braun K, Cingolani A, Anchorena J (2013) Grazing and grazing exclusion along a resource gradient in Magellanic meadows of Tierra del Fuego. Rangeland Ecology and Management 66:688–699
Crego RD, Didier KA, Nielsen CK (2014a) Modeling meadow distribution for conservation action in arid and semi-arid Patagonia, Argentina. Journal of Arid Environments 102:68–75
Crego RD, Nielsen CK, Didier KA (2014b) Climate change and conservation implications for wet meadows in dry Patagonia. Environmental Conservation 41:122–131
Davis JA, Froend R (1999) Loss and degradation of wetlands in southwestern Australia: underlying causes, consequences and solutions. Wetlands Ecology and Management 7:13–23
del Valle HF (1993) Mallines del ambiente árido, pradera salina y estepa arbustivo-graminosa en el NO del Chubut. In: Paruelo, JM, MB Bertiller, TM Schlichter and FR Coronato (eds) Secuencias de deterioro en distintos ambientes Patagónicos. Su Caracterización mediante el Modelo de Estados y Transiciones: 31-39. Convenio Argentino-Alemán de Cooperación Técnica Instituto Nacional deTecnología Agropecuaria (INTA) and Deutsche Gesellschaftfür Technische Zusammenarbeit (INTA-GTZ) (Ludepa-SMR), Buenos Aires
Elser JJ, Bracken MES, Cleland EE, Gruner DS, Harpole WS, Hillebrand H, Ngai JT, Seabloom EW, Shurin JB, Smith JE (2007) Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecology Letters 10:1135–1142
Epstein HE, Lauenroth WK, Burke IC (1997) Effects of temperature and soil texture on ANPP in the U.S. Great Plains. Ecology 78:2628–2631
Erwin KL (2009) Wetlands and global climate change: the role of wetland restoration in a changing world. Wetlands Ecological Management 17:71–84
Escartín C, Collantes M, Braun K (2010) Descomposición de la broza de las formas de vida dominantes en las comunidades de vegas de Tierra del Fuego. IV Reunión Binacional de Ecología. Buenos Aires, Argentina
Estadísticas Meteorológicas (1986) Fuerza Aérea Argentina, Buenos Aires
Ewe SML, Gaiser EE, Childers DL, Iwaniec D, Rivera-Monroy VH, Twilley RR (2006) Spatial and temporal patterns of aboveground net primary productivity (ANPP) along two freshwater-estuarine transects in the Florida Coastal Everglades. Hydrobiologia 569:459–474
Gaitán JJ, Ayesa JA, Umaña F, Raffo F, Bran DB (2011) Cartografía del área afectada por cenizas volcánicas en las provincias de Río Negro y Neuquén. Informe Técnico. Instituto Nacional de Tecnología Agropecuaria (INTA), Estación Experimental S.C. de Bariloche
Ganjurjav H, Gao Q, Zhang W, Liang Y, Li Y, Cao X, Wan Y, Li Y, Danjiu L (2015) Effects of warming on CO2 fluxes in an alpine meadow ecosystem on the central Qinghai–Tibetan Plateau. PLoS One 10:e0132044
Golluscio RA, Deregibus VA, Paruelo JM (1998) Sustainability and range management in the Patagonian steppes. Ecología Austral 8:265–284
Guo R, Wang XK, Ouyang ZY, Li YN (2006) Spatial and temporal relationships between precipitation and ANPP of four types of grasslands in northern China. Journal of Environmental Sciences 18:1024–1030
Güsewell S (2004) N:P ratios in terrestrial plants: Variation and functional significance. The New Phytologist 164:243–266
Güsewell S, Koerselman W (2002) Variation in nitrogen and phosphorus concentrations of wetland plants. Perspectives in Plant Ecology, Evolution and Systematics 5:37–61
He JS, Wang L, Flynn DFB, Wang X, Ma W, Fang J (2008) Leaf nitrogen:phosphorus stoichiometry across Chinese grassland biomes. Oecologia 155:301–310
Hollis GE (1990) Environmental impacts of development on wetlands in arid and semi-arid lands. Hydrological Sciences Journal 35:411–428
Huxman TE, Snyder KA, Tissue D, Leffler AJ, Ogle K, Pockman WT, Sandquist DR, Potts DL, Schwinning S (2004) Precipitation pulses and carbon fluxes in semiarid and arid ecosystems. Oecologia 141:254–268
Iglesias GJ, Pérez AA (1998) Patagonia. In: Canevari P, Blanco DE, Bucher E, Castro G, Davidson I (eds) Los humedales de la Argentina. Clasificación, situación actual, conservación y legislación. Wetlands International 46, Secretaria de Recursos Naturales y Desarrollo Sustentable, Buenos Aires, pp 116–135
Iriondo M (1989) Quaternary lakes of Argentina. Palaeogeography, Palaeoclimatology and Palaeoecology 70:81–88
Irisarri JGN, Oesterheld M, Paruelo JM, Texeira MA (2012) Patterns and controls of above-ground net primary production in meadows of Patagonia. A remote sensing approach. Journal of Vegetation Science 23:114–126
Irisarri JGN, Derner JD, Porensky LM, Augustine DJ, Reeves JL, Mueller KE (2016) Grazing intensity differentially regulates ANPP response to precipitation in North American semiarid grasslands. Ecological Applications 26:1370–1380
Jackson M (1964) Soil chemical analysis. Englewood Cliffs, Prentice Hall Inc, pp 153–154
Jobbágy EG, Sala OE (2000) Controls of grass and shrub aboveground production in the Patagonian steppe. Ecological Applications 10:541–549
Jobbágy EG, Sala OE, Paruelo JM (2002) Patterns and control of primary production in the Patagonian Steppe: a remote sensing approach. Ecology 83:307–319
Kao JT, Titus JE, Zhu WX (2003) Differential nitrogen and phosphorus retention by five wetland plant species. Wetlands 23:979–987
Killingbeck KT (1996) Nutrients in senesced leaves: keys to the search for potential resorption and resorption proficiency. Ecology 77:1716–1727
Kitoh A, Kusunoki S, Nakaegawa T (2011) Climate change projections over South America in the late 21st century with the 20 and 60 km mesh Meteorological Research Institute atmospheric general circulation model (MRI-AGCM). Journal of Geophysical Research 116:D06105. https://doi.org/10.1029/2010JD014920
Knapp AK, Smith MD (2001) Variation among biomes in temporal dynamics of aboveground primary production. Science 291:481–484
Koerselman W, Meuleman AFM (1996) The vegetation N:P ratio: a new tool to detect the nature of nutrient limitation. Journal of Applied Ecology 33:1441–1450
Koremblit G, Forte Lay JA (1991) Contribución al estudio agroclimático del norte de Tierra del Fuego (Argentina). Anales del Instituto de la Patagonia Serie Cs Naturales 20:125–133
Lehner B, Döll P (2004) Development and validation of a global database of lakes, reservoirs and wetlands. Journal of Hydrology 296:1–22
León RJC, Bran DE, Collantes M, Paruelo JM, Soriano A (1998) Grandes unidades de vegetación de la Patagonia extra andina. Ecología Austral 8:125–144
Levin LA, Neira C, Grosholz ED (2006) Invasive cordgrass modifies wetland trophic function. Ecology 87:419–432
Lü X-T, Kong D-L, Pan Q-M, Simmons ME, Han X-G (2012) Nitrogen and water availability interact to affect leaf stoichiometry in a semi-arid grassland. Oecologia 168:301–310
Marschner H, Dell B (1994) Nutrient uptake in mycorrhizal symbiosis. Plant and Soil 159:89–102
Mazzoni E, Vázquez M (2004). Ecosistemas de mallines y paisajes de la Patagonia Austral (Provincia de Santa Cruz). Informe Técnico Instituto Nacional De Tecnología Agropecuaria. EEA INTA. Santa Cruz. pp 31
Milchunas DG, Lauenroth WK (1993) Quantitative Effects of Grazing on Vegetation and Soils over a Global Range of Environments. Ecological Monographs 63:327–366
Mitsch WM, Gosselink JG (2000) Wetlands, 5rd edn. John Wiley and Sons, Inc., New York
Mueller-Dombois D, Ellenberg H (1974) Aims and Methods of Vegetation Ecology. John Wiley & Sons, New York, 547 pp
Newman BD, Wilcox BP, Archer SR, Breshears DD, Dahm CN, Duffy CJ, McDowell NG, Phillips FM, Scanlon BR, Vivoni ER (2006) Ecohydrology of water-limited environments—a scientific vision. Water Resources Research 42:W06302. https://doi.org/10.1029/2005WR004141
Niinements Ü, Kull K (2005) Co-limitation of plant primary productivity by nitrogen and phosphorus in a species-rich wooded meadow on calcareous soils. Acta Oecologica 28:345–356
Oesterheld M, Loreti J, Semmartin M, Paruelo JM (1999) Grazing, fire, and climate effects on primary productivity of grasslands and savannas. In: Walker LR (ed) Ecosystems of the world. Ecosystems of disturbed ground. Elsevier, Amsterdam, pp 287–306
Oesterheld M, Loreti J, Semmartin M, Sala OE (2001) Inter-annual variation in primary production of a semi-arid grassland related to previous-year production. Journal of Vegetation Science 12:137–142
O’Neill RV, DeAngelis DL (1981) Comparative productivity and biomass relations of forest ecosystems. In: Reichle D (ed) Dynamic properties of forest ecosystems. Cambridge University Press, Cambridge, pp 411–449
Oyarzábal M, Oesterheld M (2009) Phosphorus reserves increase regrowth after defoliation. Oecologia 159:717–724
Page AL, Miller RH, Keeney DR (1982) Methods of Soil Analysis. Part 2 – Chemical and Microbiological Properties, 2nd edn. American Society of Agronomy and Soil Science Society of America, Madison, pp 1153
Paruelo JM, Beltrán A, Jobbagy EG, Sala OE, Golluscio RA (1998) The climate of Patagonia: general patterns and controls on biotic processes. Ecología Austral 8:85–101
Paruelo JM, Lauenroth WK, Burke IC, Sala OE (1999) Grassland precipitation use efficiency varies across a resource gradient. Ecosystems 2:64–68
Pauchard A, Milbau A, Albihn A, Alexander J, Burgess T, Daehler C et al (2016) Non-native and native organisms moving into high elevation and high latitude ecosystems in an era of climate change: new challenges for ecology and conservation. Biological Invasions 18:345–353
Peregon A, Maksyutov S, Kosykh NP, Mironycheva-Tokareva NP (2008) Map-based inventory of wetland biomass and net primary production in western Siberia. Journal of Geophysical Research G: Biogeosciences 113:G01007
Peregon A, Maksyutov S, Kosykh N, Mironycheva-Tokareva N, Tamura M, Inoue G (2005) Application of the multiscale remote sensing and GIS to mapping net primary production in west Siberian wetlands. Phyton 4:543–550
Perotti MG, Diéguez MC, Jara FG (2005) State of the knowledge of moist soils of the Patagonic north (Argentina): excellent aspects and importance for the conservation of the regional biodiversity. Revista Chilena de Historia Natural 78:723–737
Rejmánková E (2005) Nutrient resorption in wetland macrophytes: comparison across several regions of different nutrient status. The New Phytologist 167:471–482
Ren Z, Li Q, Chu C, Zhao L, Zhang J, Ai D, Yang Y, Wang G (2010) Effects of resource additions on species richness and ANPP in an alpine meadow community. Journal of Plant Ecology 3:25–31
Rubio G, Lavado R (1999) Acquisition and allocation of resources in two waterlogging-tolerant grasses. The New Phytologist 143:539–546
Ruppert JC, Linstädter A (2014) Convergence between ANPP estimation methods in grasslands – A practical solution to the comparability dilemma. Ecological Indicators 36:524–531
Sala OE, Chapin IFS, Armesto JJ, Berlow E, Bloomfield J, Dirzo R, Huber Sanwald E, Huenneke LF, Jackson RB, Kinzig A, Leemans R, Lodge DH, Mooney HA, Oesterheld M, Leroy Poff N, Sykes MT, Walker BH, Walker M, Wall DH (2000) Global biodiversity scenarios for the year 2100. Science 287:1770–1774
Sala OE, Parson WJ, Joyce LA, Lauenroth WK (1988) Primary production of the central grassland region of the United States: spatial pattern and major controls. Ecology 69:40-45
San Martino L (2003) Fertilización de mallines en la Patagonia. Ediciones INTA, Santa Cruz, p 44
Scurlock JMO, Johnson K, Olson RJ (2002) Estimating net primary productivity from grassland biomass dynamics measurements. Global Change Biology 8:736–753
Taboada MA, Lavado RS, Svartz H, Segat AMI (1999) Structural stability changes in a grazed grassland Natraquoll of the flooding Pampa, Argentina. Wetlands 19:50–55
Tessier JT, Raynal DJ (2003) Use of nitrogen to phosphorus ratios in plant tissue as an indicator of nutrient limitation and nitrogen saturation. Journal of Applied Ecology 40:523–534
Thormann MN, Bayley SE (1997) Aboveground plant production and nutrient content of the vegetation in six peatlands in Alberta, Canada. Plant Ecology 131:1–16
Utrilla V, Brizuela M, Cibils A (2005) Riparian habitats (mallines) of Patagonia—a key grazing resource for sustainable sheep-farming operations. Outlook on Agriculture 34:55–59
Utrilla V, Brizuela M, Cibils A (2006) Structural and nutritional heterogeneity of riparian vegetation in Patagonia (Argentina) in relation to seasonal grazing by sheep. Journal of Arid Environment 67:661–670
Van der Valk AG, Bliss LC (1971) Hydrarch sussession and net primary production of oxbow lakes in central Alberta. Canadian Journal of Botany 49:1177–1199
Ven Heerwaarden LM, Toet S, Aerts R (2003) Nitrogen and phosphorus resorption efficiency and proficiency in six sub-artic bog species after 4 years of nitrogen fertilization. Journal of Ecology 91:1060–1070
Venterink HO, van der Vliet RE, Wassen MJ (2001) Nutrient limitation along a productivity gradient in wet Meadows. Plant and Soil 234:171–179
Vera C, Silvestri G, Liebmann B, Gonzalez P (2006) Climate change scenarios for seasonal precipitation in South America. Geophysical Research Letters 33. https://doi.org/10.1029/2006GL025759
Verhoeven JTA, Koerselman W, Meuleman AFM (1996) Nitrogen- or phosphorus-limited growth in herbaceous, wet vegetation: relations with atmospheric inputs and management regimes. Trends in Ecology and Evolution 11:494–497
Wang M, Moore TR (2014) Carbon, nitrogen, phosphorus, and potassium stoichiometry in an ombrotrophic peatland reflects plant functional type. Ecosystems 17:673–684
Wang S, Duan J, Xu G, Wang Y, Zhang Z, Rui Y, Luo C, Xu B, Zhu X, Chang X (2012) Effects of warming and grazing on soil N availability, species composition, and ANPP in an alpine meadow. Ecology 93:2365–2376
Wassen MJ, Peeters WHM, Venterink HO (2002) Patterns in vegetation, hydrology, and nutrient availability in an undisturbed river floodplain in Poland. Plant Ecology 165:27–43
Weltzin JF, Pastor J, Harth C, Bridgham SD, Updegraff K, Chapin CT (2000) Response of bog and fen plant communities to warming and water-table manipulations. Ecology 81:3464–3478
Whigham DF, Dykyjova D, Hejny S (1993) Wetlands of the World I: Inventory, Ecology and Management. Kluwer Academic Publishers, Dordrecht
Wiegand T, Snyman HA, Kellner K, Paruelo JM (2004) Do grasslands have a memory: modeling phytomass production of a semiarid South African grassland. Ecosystems 7:243–258
Zedler JB, Kercher S (2004) Causes and consequences of invasive plants in wetlands: opportunities, opportunists, and outcomes. Critical Reviews in Plant Sciences 23:431–452
Acknowledgements
We thank to the owners of María Behety Ranch to permit conduct our experiments in their property. We also thank to Mr. José Bonilla for their field assistance and two anonymous reviewers for their constructive comments, which greatly improved the original manuscript. The studies reported in this manuscript comply with the current laws of the Republic of Argentina. This work was supported by grants from the Agencia Nacional de Promoción de Científica y Tecnológica (PICT 1998-3458 and 141-1998/03), and the Consejo Nacional de Investigaciones Científicas y Técnicas (PMT-SID 0403 and PIP-0265).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOC 845 kb)
Rights and permissions
About this article
Cite this article
Cipriotti, P.A., Collantes, M.B., Rauber, R.B. et al. Inter-Annual and Seasonal Variation of ANPP and Leaf Nutrient Concentration in Cold-Temperate Wetlands of Tierra del Fuego. Wetlands 38, 37–49 (2018). https://doi.org/10.1007/s13157-017-0955-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13157-017-0955-8