Abstract
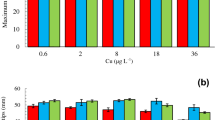
Seawater salinity is greatly influenced by tide, evaporation and rain falls. In this study, we investigated the growth and photosynthetic responses of zygote-derived Ulva fasciata Delile germlings to short-term (minutes) and prolonged (days) exposure to different salinity gradients, to evaluate the effect of salinity variation on the early stage of life history in this seaweed. The results showed that, the maximum net photosynthetic rates (NPRm) of U. fasciata germlings was observably decreased in desalted (25 and 15) and high (45) salinity seawater in short-term exposure tests (in minutes). However, after 30 min, the photosynthesis activity in medium salinity (25) was maintained at a relative high level (above 70%). After 8 d prolonged culture, the photosynthesis and mean relative growth rate (RGR) of germlings were all markedly lowered, whereas the malondialdehyde (MDA) contents increased as the salinity desalted from 34 to 15. The salinity decrease from 34 to 25 had no significant effect on the RGR, but obviously influenced the morphology of the germlings. High salinity level (45) significantly depressed the RGR and photosynthesis of U. fasciata germlings, while it notably increased the MDA contents. The results showed that the salinity elevation had more detrimental effects on Ulva fasciata germlings than salinity decrease did. The germlings grown at the salinity seawater levels from 25 to 34, performed preferable photosynthetic acclimation both in temporary and prolonged culture. Broad salinity tolerance from 25 to 34 in U. fasciata germlings may have partly evolved as a response to regular diurnal tides.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Ahmad I, Hellebust J A. 1988. The relationship between inorganic nitrogen metabolism and proline accumulation in osmoregulatory responses of two euryhaline microalgae. Plant Physiology, 88(2): 348–354
Bisson M A, Kirst G O. 1979. Osmotic adaption in the marine alga Griffithsia monilis (Rhodophyceae): the role of ions and organic compounds. Australian Journal of Plant Physiology, 6(4): 523–538
Bowler C, van Montagu M, Inze D. 1992. Superoxide dismutase and stress tolerance. Annual Review of Plant Physiology and Plant Molecular Biology, 43: 83–116
Chang W-C, Chen M-H, Lee T-M. 1999. 2, 3, 5 triphenyltetrazolium chloride reduction in the viability assay of Ulva fasciata (Chlorophyta) in response to salinity stress. Botanical Bulletin Academia Sinica, 40: 207–212
Corney H J, Sasse J M, Ades P K. 2003. Assessment of salt tolerance in eucalypts using chlorophyll fluorescence attributes. New Forests, 26(3): 233–246
Davison I R, Pearson G A. 1996. Stress tolerance in intertidal seaweeds. Journal of Phycology, 32(2): 197–211
Dawes C J. 1998. Marine Botany. 2nd ed. New York: John Wiley & Sons, Inc, 82–85
Dickson D M, Wyn Jones R G, Davenport J. 1980. Steady state osmotic adaptation in Ulva lactuca. Planta, 150(2): 158–165
Durack P J, Wijffels S E, Matear R J. 2012. Ocean salinities reveal strong global water cycle intensification during 1950 to 2000. Science, 336(6080): 455–458
Florides G A, Christodoulides P. 2009. Global warming and carbon dioxide through sciences. Environment International, 35(2): 390–401
Gessner E, Schramm W. 1971. Salinity: plants. In: Kinne O, ed. Marine Ecology. Volume 1. London: Wiley-Interscience, 705–720
González-Moreno S, Gómez-Barrera J, Perales H, et al. 1997. Multiple effects of salinity on photosynthesis of the protist Euglena gracilis. Physiologia Plantarum, 101(4): 777–786
Heath R L, Packer L. 1968. Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Archives of Biochemistry and Biophysics, 125(1): 189–198
Hellebust J A. 1976. Effect of salinity on photosynthesis and mannitol synthesis in the green flagellate Platymonas suecica. Canadian Journal of Botany, 54(15): 1735–1741
Henley W J. 1993. Measurement and interpretation of photosynthetic light-response curves in algae in the context of photoinhibition and diel changes. Journal of Phycology, 29(6): 729–739
Hiraoka M, Oka N. 2008. Tank cultivation of Ulva prolifera in deep seawater using a new “germling cluster” method. Journal of Applied Phycology, 20(1): 97–102
Huang Qing, Shen Hanming. 2009. To die or to live: the dual role of poly (ADP-ribose) polymerase-1 in autophagy and necrosis under oxidative stress and DNA damage. Autophagy, 5(2): 273–276
IPCC (Intergovernmental Panel on Climate Change). 2007. Climate Change 2007: Synthesis Report. Cambridge, UK: Cambridge University Press
Lartigue J, Neill A, Hayden B L, et al. 2003. The impact of salinity fluctuations on net oxygen production and inorganic nitrogen uptake by Ulva lactuca (Chlorophyceae). Aquatic Botany, 75(4): 339–350
Lee Y-H, Kim D-J, Kim H-K. 2003. Characteristics of the seawater quality variation on the South Coastal Area of Korea. KSCE Journal of Civil Engineering, 7(2): 123–130
Liu Jingwen, Dong Shuangxiu, Ma Shen. 2001. Effects of temperature and salinity on growth of G. tenuistipitata var. liui, U. pertusa, G. filicina and NH4-N uptake of G. tenuistipitata var. liui. Haiyang Xuebao (in Chinese), 23(2): 109–116
Lu Congming, Vonshak A. 2002. Effects of salinity stress on photosystem II function in cyanobacterial Spirulina platensis cells. Physiologia Plantarum, 114(3): 405–413
Mantri V A, Singh R P, Bijo A J, et al. 2011. Differential response of varying salinity and temperature on zoospore induction, regeneration and daily growth rate in Ulva fasciata (Chlorophyta, Ulvales). Journal of Applied Phycology, 23(2): 243–250
Ohno M. 2006. Recent developments in the seaweed cultivation and industry in Japan. In: Phang S M, Critchley A T, Ang P O, et al., eds. Advances in Seaweed Cultivation and Utilisation in Asia. Kuala Lumpur: University of Malaya Maritime Research Center, 1–20
Richter M, Rühle W, Wild A. 1990. Studies on the mechanism of photosystem II photoinhibition I. A two-step degradation of D1-protein. Photosynthesis Research, 24(3): 229–235
Satoh K, Smith C M, Fork D C. 1983. Effects of salinity on primary processes of photosynthesis in the red alga Porphyra perforata. Plant Physiology, 73(3): 643–647
Schmitt R W. 1996. If rain falls on the ocean-does it make a sound? Fresh water's effect on ocean phenomena. Oceanus, 39(2): 4–8
Steen H. 2004. Effects of reduced salinity on reproduction and germling development in Sargassum muticum (Phaeophyceae, Fucales). European Journal of Phycology, 39(3): 293–299
Wong C-L, Phang S-M. 2004. Biomass production of two Sargassum species at Cape Rachado, Malaysia. Hydrobiologia, 512(1–3): 79–88
Xia Jianrong, Li Yongjun, Zou Dinghui. 2004. Effects of salinity stress on PSII in Ulva lactuca as probed by chlorophyll fluorescence measurements. Aquatic Botany, 80(2): 129–137
Yamochi S. 2013. Effects of desiccation and salinity on the outbreak of a green tide of Ulva pertusa in a created salt marsh along the coast of Osaka Bay, Japan. Estuarine, Coastal and Shelf Science, 116: 21–28
Ying Weihai, Alano C C, Garnier P, et al. 2005. NAD+ as a metabolic link between DNA damage and cell death. Journal of Neuroscience Research, 79(1–2): 216–223
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation item: The National Natural Science Foundation of China under contract Nos 41276148 and 41076094.
Rights and permissions
About this article
Cite this article
Chen, B., Zou, D. Altered seawater salinity levels affected growth and photosynthesis of Ulva fasciata (Ulvales, Chlorophyta) germlings. Acta Oceanol. Sin. 34, 108–113 (2015). https://doi.org/10.1007/s13131-015-0654-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13131-015-0654-3