Abstract
Background
Indian population–based studies on the prevalence and risk factors for gastroesophageal reflux disease (GERD) are scanty, and a meta-analysis and a meta-regression of prevalence and risk factors based on the existing data have not yet been reported.
Methods
A systematic review of all the available publications from India reporting data regarding prevalence and risk factors of GERD was performed. Heterogeneity was assessed using I2 statistics. The meta-analysis was undertaken to measure the average proportion reported in the existing studies, and meta-regression models were used to explore the risk factors for it.
Results
The nine studies included 20,614 subjects; the prevalence of GERD ranged from 5% to 28.5%. The summary effect size (weighted average proportion) estimated by meta-analytic model was 0.1415 (95% confidence interval [CI] 0.099 to 0.197). The results for the test of heterogeneity that included tau2 (0.37, 95% CI 0.21 to 1.80), I2 (98.9%, 95% CI 98.01 to 99.77), and the Q-statistic (707.670; p < 0.0001) suggested high heterogeneity in the effect sizes. The pooled proportion of GERD (random-effects model) was 15.573 (95% CI 11.046 to 20.714). In the meta-regression model, sample size (p = 0.005) explained about 50% of the heterogeneity.
Conclusion
The pooled prevalence of GERD in the Indian population is 15.6 (95% CI 11.046 to 20.714). The risk factors were age, body mass index (BMI), non-vegetarian diet, tea/coffee intake, tobacco, and alcohol consumption. However, there was significant heterogeneity in the studies.

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gastroesophageal reflux disease (GERD) is associated with pathological reflux of acidic or non-acidic gastric contents into the esophagus and is characterized by heartburn, regurgitation, and chest pain [1,2,3,4]. Severe GERD may result in significant morbidity, impairment in quality of life, and work absenteeism, and, if left untreated, complications like peptic stricture, Barrett’s esophagus, and esophageal adenocarcinoma [5, 6]. However, the frequency, severity, and complications of GERD can vary according to the geographic and ethnic factors [7, 8]. Though GERD is a common problem in the Western population [5, 9,10,11], it may be less common in the Asian population in general and in India in particular [12,13,14].
Though multiple factors might contribute to the lower frequency and severity of GERD and Barrett’s esophagus in the Indian population, the dietary variation might be one of the most important contributors [3, 8, 15, 16]. A few earlier studies did show that non-vegetarian foods might contribute to the occurrence, symptom exacerbation, and severity of GERD [17,18,19]. Since vegetarianism is widespread in the Indian population, Indian epidemiological studies may uncover the dietary factors contributing to GERD that may have implications beyond India, as diet is a modifiable factor. However, Indian population–based data on prevalence and risk factors of GERD are scant. Moreover, easy over-the-counter availability of the proton pump inhibitors (PPI) and histamine 2 receptor antagonists (H2RA), high frequency of Helicobacter pylori (H. pylori) infection, which is known to reduce the severity of GERD, and cultural variation in the symptom reporting might also reduce the prevalence of GERD [20,21,22,23,24,25,26,27]. Since the studies on the prevalence and risk factors of GERD from India are scanty, we thought it worthwhile to undertake a meta-analysis and meta-regression on the existing population-based studies with aims (i) to evaluate prevalence of GERD in the Indian population, (ii) to evaluate its risk factors including the influence of dietary variation, and (iii) to critically analyze the lacunae in the current literature and directions for the future studies.
Methods
Search methodology, study selection, and data extraction
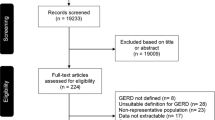
Two authors independently performed a literature search in PubMed (https://pubmed.ncbi.nlm.nih.gov/), Scopus, and Web of science (from January 2000 through May 2020) for abstracts matching one or more of the following keywords: “gastro-esophageal reflux,” “India,” “prevalence,” “risk factors,” and boolean operators AND/OR: (e.g. gastroesophageal reflux disease OR GERD) AND (India OR prevalence OR risk factors). This was based on the assumption that studies not including the above search terms in the abstract would not have reported the prevalence and risk factors for GERD in India. Two authors independently screened the titles and abstracts of the identified studies and further searched for the full texts of the potentially relevant publications. Studies in the English language were eligible for inclusion in the meta-analysis. The full text of the resulting papers were reviewed, and bibliographies of these were screened to look for other relevant studies. Eligibility criteria were pre-defined. Any discrepancy was resolved by consensus. After a thorough search, a total of 340 abstracts were found. After title and abstract reviews, 332 studies were excluded. The 8 studies that were selected were thoroughly reviewed, including bibliography. At this stage, one more population-based study from our group accepted for publication recently was also added [28]. Hence, nine studies were used in the final analysis (Fig. 1).
Definitions of significant GERD varied across the studies. Some studies utilized a pre-validated symptom score to diagnose and grade the severity of GERD. We recorded data for each study regarding the definition and severity criteria. Different risk factors for GERD have been assessed in various studies. All these data were extracted from relevant studies and appropriately inserted into a Microsoft Excel spreadsheet and subjected to meta-regression for the risk factors for GERD.
The sample size of the included studies ranged from 358 to 6174, with a combined total of 20,614 subjects [3, 7, 16, 20, 29,30,31,32,33] (Fig. 1). The following data were extracted from each paper: (1) author and publication year; (2) type of study (population-based or selected subjects); (3) sample size and GERD prevalence; (4) definition of GERD; (5) risk factors (age, body mass index [BMI], predominant diet, tea/coffee intake, tobacco smoking/chewing, alcohol intake) and predominant symptoms. The quality of the study was assessed by Jadad score (Table 1). The row of the table represented the data extracted from each study, and each column described the variables of interest, which were essential to compute effect sizes and plots. The proportions were transformed to make them follow a normal distribution to accurately estimate the summary proportion and increase the validity of the statistical analysis. The results were reported by converting the logits back into the proportions.
Data synthesis and analysis
MedCalc version 14 (Warandeberg 3, 1000 Brussels, Belgium) was used to generate forest plots of pooled prevalence with 95% confidence intervals (CIs). R::metafor package version 1.9-7 of R studio (R development core team, Vienna, Austria) was used for meta-regression. Data were analyzed using both fixed and random-effects models. Heterogeneity was assessed using I2 statistics. A fixed-effect model was used in the absence of heterogeneity (I2 < 50%), whereas if I2 was > 50%, a random-effects model was used.
A meta-analysis statistically synthesizes quantitative findings and measures the average proportion of multiple studies weighted by the inverse of sampling variances using the appropriate model. The forest plot was constructed to represent the degree of variation between effect sizes across studies, and it also helped to detect the outliers. The studies were identified as outliers whose 95% confidence intervals did not overlap with the overall summary effect; if the large studies were outliers, it might result in higher heterogeneity.
Meta-regression is a statistical technique used in meta-analysis to investigate the independent contribution of the changes in the main factors on the prevalence. A univariate model of meta-regression was evaluated before multivariate model to quantify the magnitude of each continuous moderator on the study effects. A restricted maximum likelihood (REML) estimation method with backward elimination regression procedure was used in meta-regression analysis. The scatter plots were constructed to indicate the association between the moderator and effect sizes through the slope of the regression line. The significantly positive or negative slope suggested whether the explanatory variable had a significant moderating effect or not. The I2 statistic quantified the overall heterogeneity of the model that could be accounted for by the real differences between studies; thus, subgroup analysis or meta-regression applied to identify potential moderating factors explained the inconsistencies between effect sizes across the studies.
Results
The search strategy initially revealed 340 studies, of which 324 were excluded after an initial review, and the remaining 16 were reviewed in detail for eligibility. Eight of these studies were excluded due to various reasons, and the remaining eight were eligible for the meta-analysis [3, 7, 16, 20, 29,30,31,32] (Fig. 1). Besides, an original population-based survey from the author’s group accepted for publication recently was also added [28]. Characteristics of all the studies included in the current meta-analysis, including the criteria to diagnose GERD, are presented in Table 1. The study cites are shown in Fig. 2. Four studies were population-based [7, 16, 30], and the other five were on selected populations such as medical students, patients’ attendants, doctors, nursing staff, and hospital employees [3, 20, 29,30,31](Table 1). The nine studies included 20,614 subjects, with the prevalence of GERD ranging from 5% to 28.5%. Different criteria were used for diagnosis of GERD in different studies, most of which are quite standard methods used to diagnose GERD in epidemiological surveys except for two studies, one by Bhalaghuru et al. and the other by Chowdhury et al. (Table 1) [7, 29]. The estimated prevalence of GERD in the Indian population was 14.15% (95% CI 9.9–19.7%) (Fig. 3). Analysis revealed high between-study variability (I2 98.9%). The model automatically selected the random-effects model in the presence of substantial heterogeneity. Thus, we have conducted a moderator analysis to examine how characteristics of studies were related to variation in the effect sizes across studies. Researchers have found different risk factors to be significant in GERD prevalence and severity. The risk factors found to be associated are depicted in Table 2. Briefly, three studies reported that smoking was not a risk factor; two showed that the younger population had GERD less often. A significant increase in GERD score was observed with higher BMI in five studies, non-vegetarian diet in three studies, with living in urban areas in two studies, with infrequent milk consumption in one study, with an increase in age in four studies, with inadequate sleep in one study, with excessive consumption of alcohol in two studies, with an increase in tea/coffee intake in four studies, and with an increase in intake of carbonated drinks in two studies.
a The forest plot shows the prevalence of gastroesophageal reflux disease (GERD) in India. The squares represent individual studies and the size of the square represents the effect size of each study included in the meta-analysis. The pink horizontal line with each square represents confidence limit of each study. Diamond represents pooled prevalence (fixed and random effect). b Funnel plot showing the publication bias of prevalence studies on GERD
The unadjusted meta-regression model was constructed for the effect of confounders. The estimated prevalence were 17.6 (95% CI = 10.07–29.04), 13.9 (95% CI = 8.60–21.89), and 15.4 (95% CI = 10.71, 21.66) for the subgroups (variable study type with categories; population-based/-selected population) and overall group of studies, respectively. The separate random-effects models were fitted for the subgroups of study population variable, and the difference between the two subgroup summary estimates was not significant (p = 0.522). Thus, the study population is not a significant moderating variable.
In the meta-regression model, sample size (p = 0.005) explained about 50% of the heterogeneity. A greater prevalence of GERD of 28.5% and 22.3% was noted in studies published in southern part of India [29, 30]. The slope of the estimated regression line of the scatter plot is neither completely horizontal nor very steep, suggesting a weak association with age, non-vegetarianism, and observed effects. By visual examination of scatter plots, we observed that nearly half of the studies fell outside of 95% CI bounds, which suggested that there might be more than one factor that could account for the heterogeneity in the effect sizes. The age (p = 0.53) and non-vegetarianism (p = 0.327) were found to be insignificant moderators. Similarly, the effects of other continuous moderators, BMI (kg/m2) (p = 0.771), publication year (p = 0.380), non-vegetarian diet (p = 0.327), tea/coffee intake (p = 0.935), tobacco use (p = 0.337), and alcohol consumption (p = 0.881), were evaluated through model and visually inspected through scatter plots, suggesting insignificant association of moderators with the observed effect sizes (proportion of GERD) (Fig. 4a–f).
Scatter plots of effect size (proportion of gastroesophageal reflux disease [GERD]) vs. a potential risk factor; a sample size, b publication year, c age (in years), d non-vegetarian diet, e tobacco consumption, and f alcohol consumption. The symbol for individual study is sized proportionally to the precision of the study (the larger the circle, the more precise is the estimated effect) and the dotted line represents the regression line for the analysis
Discussion
We conducted a meta-analysis and meta-regression assessing the prevalence and risk factors of GERD in the Indian population. The pooled prevalence of GERD in the Indian population was found to be 15.57, though there was significant heterogeneity in the studies. The risk factors for GERD included age, BMI, non-vegetarian diet, tea/coffee intake, tobacco, and alcohol consumption. To the best of our knowledge, this is perhaps the first Indian population–based meta-analysis and meta-regression on the prevalence and risk factors of GERD.
GERD is a common condition worldwide (Fig. 5) [34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57]. A systematic review and meta-analysis by Nirwan et al. demonstrated substantial variations in the pooled prevalence of GERD in different geographical regions and countries [58]. The prevalence of GERD varied from 8.7% to 33.1% in the Middle East, 11.6% in Australia, 23.0% in South America, 2.5% to 7.8% in East Asia, 8.8% to 25.9% in Europe, and 18.1% to 27.8% in North America (Fig. 5) [59]. The prevalence of GERD has been reported to be considerably higher in North America (19.5%) and Europe (25.9%). A high prevalence of GERD was reported in South America in the study by El-Serag et al., though another study by Nirwan et al. did report a lower pooled prevalence (12.8%) [59]. In a recent study, prevalence (age-standardized) was highest in the USA, Italy, Greece, New Zealand, north Africa and the Middle East, eastern Europe, Latin American, and the Caribbean countires [60]; in contrast, the prevalence was lowest in Asia Pacific, east Asia, Iceland, France, Denmark, and Switzerland [60]. Considering the variation in the population prevalence of several risk factors for GERD (including those revealed in this meta-analysis) in Western population as compared with the Indian population, difference in prevalence of GERD in different countries is not entirely unexpected [61]. These risk factors include intake of fatty and non-vegetarian foods, carbonated drinks, coffee/tea, high level of urbanization, and presence of obesity. In fact, several studies did support the fact that variation in the frequency of GERD in Asian and Western countries might be related to the frequency of different risk factors in different population [30, 5, 8, 18, 23, 38, 43, 54, 58, 59, 62]. Risk factors for GERD reported in the studies from the Western countries included age (35–59 years), use of non-steroidal anti-inflammatory drugs (NSAIDs), lower income, socioeconomic status, urbanization, genetic factors, family history of GERD, obesity, and consumption of carbonated drinks. Variation in prevalence in GERD has even been reported in different regions within the same country. For example, in the current systematic review, several studies from the southern part of India demonstrate a higher prevalence of GERD (22.2–28.5%) [29, 30] than those reported from the northern parts of India (11–18%; Fig. 2) [16, 20]. One of the reasons for such variation in prevalence of GERD in the southern and northern parts of India might be the differences in the diet in the two regions. Southern Indian diet is typically rice-based, with excess spice, tamarind, and non-vegetarian foods; in contrast, the northern Indian diet is wheat-based and less spicy and vegetarianism is common [63].
There are quite a few other factors that might explain regional differences in prevalence of GERD in different parts of the world. Differences in the definition/criteria used and ethnic and cultural variation on symptom reporting may affect the prevalence of GERD. Only a few studies used translated-validated questionnaire for assessing the symptoms [28]. House-to-house survey is known to show lower prevalence of functional GI disorders than internet survey [64]. H. pylori infection is known to reduce gastric acid secretion that may reduce the frequency and severity of GERD [13, 65]. Since the prevalence of H. pylori infection is high in India, this might contribute to the lower frequency of GERD in Indian population [13].
This is perhaps the most comprehensive meta-analysis and meta-regression study in Indian population on prevalence and risk factors of GERD. We pooled data from four population-based studies and five studies on selected subjects (medical students, patients’ attendants, doctors, nursing staff, and hospital employees) to analyze the prevalence of GERD [3, 7, 8, 16, 20, 29,30,31,32]. We have also identified that several earlier published studies used different survey methods and definitions and online instruments (questionnaires) to collect data from subjects, leading to a wide range in GERD prevalence. Most of the epidemiological studies on GERD in India are house-to-house surveys [64]. Furthermore, we have also investigated how risk factors affect the prevalence of GERD using meta-regression approach.
This meta-analysis, however, has a few limitations. Significant heterogeneity in the included studies is a limitation. The observed heterogeneity in the current study could be because of the different criteria used for diagnosis of GERD, selection criteria, and lack of controls. A number of studies are also small. In fact, meta-regression should ideally be conducted when the number of studies included in the meta-analysis is at least 10 [66]; there were 9 studies in the current meta-analysis. The diagnosis of GERD is based on symptoms alone. The gold standard for diagnosis of GERD is 24-h pH impedance monitoring rather than symptoms alone [13]. Several other conditions may manifest with symptoms of heartburn such as functional heartburn, esophageal hypersensitivity, and esophageal motor disorders [13]. All the reported studies are not population-based. In spite of all these limitations, we believe that the current meta-analysis does provide important information on epidemiology and risk of GERD in Indian population and suggests the future areas of research on this subject.
In conclusion, this meta-analysis showed the pooled prevalence of GERD in Indian population to be 15.57%, though there was significant heterogeneity in the studies included in this meta-analysis. On meta-regression, we found that age, BMI, non-vegetarian diet, tea/coffee intake, tobacco, and alcohol consumption are the risk factors for GERD in Indian population. These results may have several implications in clinical practice and research. The knowledge on risk factors may be used for public education to prevent GERD. Educating patients with GERD may help them to modify their lifestyle to reduce the severity and complications of GERD.
References
De Giorgi F, Palmiero M, Esposito I, Mosca F, Cuomo R. Pathophysiology of gastro-oesophageal reflux disease. Acta Otorhinolaryngol Ital. 2006;26:241–6.
Clarrett DM, Hachem C. Gastroesophageal reflux disease (GERD). Mo Med. 2018;115:214–8.
Bhatia SJ, Reddy DN, Ghoshal UC, et al. Epidemiology and symptom profile of gastroesophageal reflux in the Indian population: report of the Indian Society of Gastroenterology Task Force. Indian J Gastroenterol. 2011;30:118–27.
Savarino E, Marabotto E, Bodini G, et al. Epidemiology and natural history of gastroesophageal reflux disease. Minerva Gastroenterol Dietol. 2017;63:175–83.
Geeraerts A, Van Houtte B, Clevers E, et al. Gastroesophageal reflux disease-functional dyspepsia overlap: do birds of a feather flock together? Am J Gastroenterol. 2020;115(8):1167–82.
Spechler SJ. GERD and its complications. Mt Sinai J Med. 2000;67:106–11.
Chowdhury SD, George G, Ramakrishna K, et al. Prevalence and factors associated with gastroesophageal reflux disease in southern India: a community-based study. Indian J Gastroenterol. 2019;38:77–82.
Chandran S, Raman R, Kishor M, Nandeesh HP. The effectiveness of mindfulness meditation in relief of symptoms of depression and quality of life in patients with gastroesophageal reflux disease. Indian J Gastroenterol. 2019;38:29–38.
Qumseya BJ, Bukannan A, Gendy S, et al. Systematic review and meta-analysis of prevalence and risk factors for Barrett’s esophagus. Gastrointest Endosc. 2019;90:707–17. e1.
Nejat Pish-Kenari F, Qujeq D, Maghsoudi H. Some of the effective factors in the pathogenesis of gastro-oesophageal reflux disease. J Cell Mol Med. 2018;22:6401–4.
Yamasaki T, Hemond C, Eisa M, Ganocy S, Fass R. The changing epidemiology of gastroesophageal reflux disease: are patients getting younger? J Neurogastroenterol Motil. 2018;24:559–69.
Wang KY, Chen YW, Wang TN, et al. Predictor of slower gastric emptying in gastroesophageal reflux disease: survey of an Asian-Pacific cohort. J Gastroenterol Hepatol. 2019;34:837–42.
Bhatia SJ, Makharia GK, Abraham P, et al. Indian consensus on gastroesophageal reflux disease in adults: a position statement of the Indian Society of Gastroenterology. Indian J Gastroenterol. 2019;38:411–40.
Goh KL, Choi KD, Choi MG, et al. Factors influencing treatment outcome in patients with gastroesophageal reflux disease: outcome of a prospective pragmatic trial in Asian patients. BMC Gastroenterol. 2014;14:156.
Rogers BD, Gyawali CP. Why differences between New York and New Delhi matter in approach to gastroesophageal reflux disease. Indian J Gastroenterol. 2019;38:371–7.
Kumar S, Sharma S, Norboo T, et al. Population based study to assess prevalence and risk factors of gastroesophageal reflux disease in a high altitude area. Indian J Gastroenterol. 2011;30:135–43.
Jung JG, Kang HW, Hahn SJ, et al. Vegetarianism as a protective factor for reflux esophagitis: a retrospective, cross-sectional study between Buddhist priests and general population. Dig Dis Sci. 2013;58:2244–52.
Kubo A, Block G, Quesenberry CP Jr, Buffler P, Corley DA. Effects of dietary fiber, fats, and meat intakes on the risk of Barrett’s esophagus. Nutr Cancer. 2009;61:607–16.
Newberry C, Lynch K. The role of diet in the development and management of gastroesophageal reflux disease: why we feel the burn. J Thorac Dis. 2019;11:S1594–601.
Sharma PK, Ahuja V, Madan K, Gupta S, Raizada A, Sharma MP. Prevalence, severity, and risk factors of symptomatic gastroesophageal reflux disease among employees of a large hospital in northern India. Indian J Gastroenterol. 2011;30:128–34.
Baruah B, Kumar T, Das P, et al. Prevalence of eosinophilic esophagitis in patients with gastroesophageal reflux symptoms: a cross-sectional study from a tertiary care hospital in North India. Indian J Gastroenterol. 2017;36:353–60.
Zhao Y, Li Y, Hu J, et al. The effect of helicobacter pylori eradication in patients with gastroesophageal reflux disease: a meta-analysis of randomized controlled studies. Dig Dis. 2020;38:261–268.
Ghoshal UC, Singh R, Chang FY, et al. Epidemiology of uninvestigated and functional dyspepsia in Asia: facts and fiction. J Neurogastroenterol Motil. 2011;17:235–44.
Chourasia D, Misra A, Tripathi S, Krishnani N, Ghoshal UC. Patients with Helicobacter pylori infection have less severe gastroesophageal reflux disease: a study using endoscopy, 24-hour gastric and esophageal pH metry. Indian J Gastroenterol. 2011;30:12–21.
Ghoshal UC, Chourasia D. Gastroesophageal reflux disease and Helicobacter pylori: what may be the relationship? J Neurogastroenterol Motil. 2010;16:243–50.
Chourasia D, Ghoshal UC. Pathogenesis of gastro-oesophageal reflux disease: what role do Helicobacter pylori and host genetic factors play? Trop Gastroenterol. 2008;29:13–9.
Sandhu DS, Fass R. Current trends in the management of gastroesophageal reflux disease. Gut Liver. 2018;12:7–16.
Ghoshal UC, Singh R, Rai S. Prevalence and risk factors for gastroesophageal reflux disease in a rural Indian population. Indian J Gastroenterol. 2021;40 https://doi.org/10.1007/s12664-020-01135-7.
Bhalaghuru CM, Vijaya S, Jayanthi V. Symptomatic gastroesophageal reflux amongst hospital personnel in South India. Indian J Med Sci. 2011;65:355–9.
Wang HY, Leena KB, Plymoth A, et al. Prevalence of gastro-esophageal reflux disease and its risk factors in a community-based population in southern India. BMC Gastroenterol. 2016;16:36.
Sharma A, Sharma PK, Puri P. Prevalence and the risk factors of gastro-esophageal reflux disease in medical students. Med J Armed Forces India. 2018;74:250–4.
Arivan R, Deepanjali S. Prevalence and risk factors of gastro-esophageal reflux disease among undergraduate medical students from a southern Indian medical school: a cross-sectional study. BMC Res Notes. 2018;11:448.
Ghoshal UC, Singh R, Rai S, et al. Prevalence and risk factors for gastroesophageal reflux disease in a rural Indian population. Submitted to 6th Asian Postgraduate Course on Neurogastroenterology and Motility. 2020.
Jung HK, Halder S, McNally M, et al. Overlap of gastro-oesophageal reflux disease and irritable bowel syndrome: prevalence and risk factors in the general population. Aliment Pharmacol Ther. 2007;26:453–61.
Locke GR 3rd, Talley NJ, Fett SL, Zinsmeister AR, Melton LJ 3rd. Prevalence and clinical spectrum of gastroesophageal reflux: a population-based study in Olmsted County, Minnesota. Gastroenterology. 1997;112:1448–56.
Locke GR 3rd, Talley NJ, Fett SL, Zinsmeister AR, Melton LJ 3rd. Risk factors associated with symptoms of gastroesophageal reflux. Am J Med. 1999;106:642–9.
Talley NJ, Zinsmeister AR, Schleck CD, Melton LJ III. Dyspepsia and dyspepsia subgroups: a population-based study. Gastroenterology. 1992;102:1259–68.
El-Serag HB, Petersen NJ, Carter J, et al. Gastroesophageal reflux among different racial groups in the United States. Gastroenterology. 2004;126:1692–9.
Chiocca JC, Olmos JA, Salis GB, et al. Prevalence, clinical spectrum and atypical symptoms of gastro-oesophageal reflux in Argentina: a nationwide population-based study. Aliment Pharmacol Ther. 2005;22:331–42.
Isolauri J, Laippala P. Prevalence of symptoms suggestive of gastro-oesophageal reflux disease in an adult population. Ann Med. 1995;27:67–70.
Valle C, Broglia F, Pistorio A, Tinelli C, Perego M. Prevalence and impact of symptoms suggestive of gastroesophageal reflux disease. Dig Dis Sci. 1999;44:1848–52.
Diaz-Rubio M, Moreno-Elola-Olaso C, Rey E, Locke GR 3rd, Rodriguez-Artalejo F. Symptoms of gastro-oesophageal reflux: prevalence, severity, duration and associated factors in a Spanish population. Aliment Pharmacol Ther. 2004;19:95–105.
Lofdahl HE, Lane A, Lu Y, et al. Increased population prevalence of reflux and obesity in the United Kingdom compared with Sweden: a potential explanation for the difference in incidence of esophageal adenocarcinoma. Eur J Gastroenterol Hepatol. 2011;23:128–32.
Ronkainen J, Aro P, Storskrubb T, et al. High prevalence of gastroesophageal reflux symptoms and esophagitis with or without symptoms in the general adult Swedish population: a Kalixanda study report. Scand J Gastroenterol. 2005;40:275–85.
Terry P, Lagergren J, Wolk A, Nyrén O. Reflux-inducing dietary factors and risk of adenocarcinoma of the esophagus and gastric cardia. Nutr Cancer. 2000;38:186–91.
Mohammed I, Cherkas LF, Riley SA, Spector TD, Trudgill NJ. Genetic influences in gastro-oesophageal reflux disease: a twin study. Gut. 2003;52:1085–9.
Thompson WG, Heaton KW. Heartburn and globus in apparently healthy people. Can Med Assoc J. 1982;126:46–8.
Mostaghni A, Mehrabani D, Khademolhosseini F, et al. Prevalence and risk factors of gastroesophageal reflux disease in Qashqai migrating nomads, southern Iran. World J Gastroenterol. 2009;15:961–5.
Nasseri-Moghaddam S, Mofid A, Ghotbi MH, et al. Epidemiological study of gastro-oesophageal reflux disease: reflux in spouse as a risk factor. Aliment Pharmacol Ther. 2008;28:144–53.
Solhpour A, Pourhoseingholi MA, Soltani F, et al. Gastro-esophageal reflux symptoms and body mass index: no relation among the Iranian population. Indian J Gastroenterol. 2008;27:153–5.
Nouraie M, Radmard AR, Zaer-Rezaii H, Razjouyan H, Nasseri-Moghaddam S, Malekzadeh R. Hygiene could affect GERD prevalence independently: a population-based study in Tehran. Am J Gastroenterol. 2007;102:1353–60.
Nouraie M, Razjouyan H, Assady M, Malekzadeh R, Nasseri-Moghaddam S. Epidemiology of gastroesophageal reflux symptoms in Tehran, Iran: a population-based telephone survey. Arch Iran Med. 2007;10:289–94.
Cao H, Huang X, Zhi X, Han C, Li L, Li Y. Association between tea consumption and gastroesophageal reflux disease: a meta-analysis. Medicine (Baltimore). 2019;98:e14173.
Chen M, Xiong L, Chen H, Xu A, He L, Hu P. Prevalence, risk factors and impact of gastroesophageal reflux disease symptoms: a population-based study in South China. Scand J Gastroenterol. 2005;40:759–67.
Eslick GD, Talley NJ. Gastroesophageal reflux disease (GERD): risk factors, and impact on quality of life-a population-based study. J Clin Gastroenterol. 2009;43:111–7.
Kitapçioğlu G, Mandiracioğlu A, Caymaz Bor C, Bor S. Overlap of symptoms of dyspepsia and gastroesophageal reflux in the community. Turk J Gastroenterol. 2007;18:14–9.
Sperber AD, Halpern Z, Shvartzman P, et al. Prevalence of GERD symptoms in a representative Israeli adult population. J Clin Gastroenterol. 2007;41:457–61.
Nirwan JS, Hasan SS, Babar ZU, Conway BR, Ghori MU. Global prevalence and risk factors of gastro-oesophageal reflux disease (GORD): systematic review with meta-analysis. Sci Rep. 2020;10:5814.
El-Serag HB, Sweet S, Winchester CC, Dent J. Update on the epidemiology of gastro-oesophageal reflux disease: a systematic review. Gut. 2014;63:871–80.
Collaborators GBDG-oRD. The global, regional, and national burden of gastro-oesophageal reflux disease in 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol Hepatol. 2020;5:561–81.
Wong SW, Chan WK. Epidemiology of non-alcoholic fatty liver disease in Asia. Indian J Gastroenterol. 2020;39:1–8.
Ng M, Fleming T, Robinson M, et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2014;384:766–81.
Ghoshal UC, Singh R. Frequency and risk factors of functional gastro-intestinal disorders in a rural Indian population. J Gastroenterol Hepatol. 2017;32:378–87.
Sperber AD, Bangdiwala SI, Drossman DA, et al. Worldwide prevalence and burden of functional gastrointestinal disorders, results of Rome Foundation Global Study. Gastroenterology. 2020;S0016-5085(20)30487-X.
Chourasia D, Achyut BR, Tripathi S, Mittal B, Mittal RD, Ghoshal UC. Genotypic and functional roles of IL-1B and IL-1RN on the risk of gastroesophageal reflux disease: the presence of IL-1B-511*T/IL-1RN*1 (T1) haplotype may protect against the disease. Am J Gastroenterol. 2009;104:2704–13.
Shah A, Jones MP, Holtmann GJ. Basics of meta-analysis. Indian J Gastroenterol. 2020.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
SR, AK, and UCG declare that they have no conflict of interest.
Ethics statement
The study was performed conforming to the Helsinki declaration of 1975, as revised in 2000 and 2008 concerning human and animal rights, and the authors followed the policy concerning informed consent as shown on Springer.com.
Disclaimer
The authors are solely responsible for the data and the contents of the paper. In no way, the Honorary Editor-in-Chief, Editorial Board Members, the Indian Society of Gastroenterology, or the printer/publishers are responsible for the results/findings and content of this article.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rai, S., Kulkarni, A. & Ghoshal, U.C. Prevalence and risk factors for gastroesophageal reflux disease in the Indian population: A meta-analysis and meta-regression study. Indian J Gastroenterol 40, 209–219 (2021). https://doi.org/10.1007/s12664-020-01104-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12664-020-01104-0