Abstract
Chitosan, an important biopolymer with many uses, is made by deacetylation of chitin. Classical hydrolysis at high temperatures (above 100 °C) in 50 % concentrated sodium hydroxide during several hours produces chitosan, a poly-beta-glucosamine. This chemical deacetylation step is often performed under pressure in boiling ethyleneglycol and tends to degrade polymer chain length, thus the interesting polymer properties. It is convenient to search for a more economical production pathway in order to make chitosan available for environmental applications, such as use in water suspended matter flocculation. Chitin was extracted from the exoskeletons of shrimps and its deacetylation kinetics was followed for 35 days at 20 and 35 °C. The results obtained using different techniques (conductivity, viscometry, 13C NMR and thermogravimetry) showed that the deacetylation degree of chitin increased with the incubation time reaching 99 and 88 % at 35 and 20 °C, respectively, after 35 days. Moreover, during the incubation time, the molecular weight of chitosan showed moderate decrease compared to deacetylated chitin at high temperature. Clarification or urban waste water with chitosan biopolymer demonstrated good results at 1.5 mg/L concentrations.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Chitosan is an expensive biopolymer (more than one thousand euros per kilogram at Aldrich, for example), despite being sources from abundant marine wastes, including crab, shrimp or lobster shells. The main source of chitosan remains chitin which is extracted from crustacean exoskeletons and chemically treated by basic hydrolysis (deacetylation). There are two reasons why chitosan is costly: there exist high value applications for chitosan, particularly in medical and antibiotic fields, and the preparation methods involve many steps. Starting from chitin, the process includes demineralization, deproteinization, deacetylation of chitin and chitosan isolation. For example, sodium hydroxide and hydrochloric acid solutions were used for deproteination and demineralisation, respectively, while acetone was used for decolourisation [1]. The amount of chitin and subsequently chitosan obtained was ~35 and 25 % respectively of the dry weight of the shells. The chitin was deacetylated using sodium hydroxide at 100 °C. In another example Yong describes “Chitosan production involves four major steps, viz., deproteination, demineralization, bleaching and deacetylation. These four processes require excessive usage of strong alkali at different stages, and drives chitosan’s production cost up” [2]. Alternative biological methods to the chemical treatments have been developed, as, for example, the lactic acid fermentation pathway in a packed bed column reactor where the maximal percentages of demineralization and deproteinization after 96 h reached 92 and 94 %, respectively [3]. However, even submerged fermentation reactions can use complex methods, such as steam explosion, so that biologically derived chitosan is not necessarily less costly than chemically obtained chitosan [4]. Engineering optimization was performed with Taguchi design to evaluate chitosan production starting from jumbo squid pen by Jung [5], who concluded that optimal treatment conditions for making chitosan were: 40 % NaOH at 90 °C for 6 h with three separate steps (2 + 2 + 2 h) or 50 % NaOH at 90 °C for 6 h with one step, or 50 % KOH at 90 °C for 4 h with three steps (1 + 1 + 2 h) or 6 h with one step. Another study, using factorial design by Younes et al. [6], found that nitrogen atmosphere had a protective effect against chitosan degradation during deacetylation.
Typically, the deacetylation of chitin is performed with very high concentrations of NaOH and heating under pressure at high temperatures, ranging from 100 to 140 °C [7]. Under these conditions, the molecular weight of chitosan is reduced by polymer chain breaking that leads to the decrease of the flocculent properties. Furthermore, the deacetylation process is not cost effective due to heating [8, 9]. Therefore, to promote environmental use of chitosan [10], the transformation process of crustacean exoskeletons should be simplified to lessen production costs.
The propreties of chitin and chitosan have been revieved [11] and the use of chitosan for coagulation/flocculation processes has been detailed [12], but without consideration for the method of deacetylation of chitin. In this report, our aim is to show that chitosan can be derived from chitin with as economical approach at room temperature and pressure in simple plastic vessels and that the resulting polymer works well in clarifying urban waste waters.
Materials and Methods
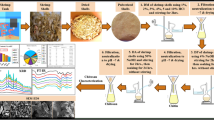
Exoskeletons (shells) of fresh shrimps were used in this study to extract chitosan. The deacetylation kinetics of chitin was followed in order to improve the conditions for obtaining a less expensive product with good flocculent properties (Fig. 1). One gram of chitin was suspended in a 100 mL vial containing 50 mL of 40 % NaOH. The resulting product was then filtered, washed with water until neutral pH and then oven dried at 50 °C for 24 h. The experiment was repeated for reaction times ranging from 3 to 35 days at 20 and 35 °C.
Conductimetric titrations were performed by dissolving 150 mg of each chitin product in 10 ml of 0.1 N HCl, adjusting the volume to 200 ml with deionized (DI) water and titration with a solution of 0.1 N NaOH. The degree of deacetylation (DDA) was calculated according to Eq. 1 below [13]:
where V 1 and V 2 are the equivalent volumes of NaOH as shown in Fig. 2, m is the mass of the sample (g), 203 is the molecular weight of the acetylated monomer (g/mol) and 42 is the difference between the molecular weight of the acetylated monomer and that of the deacetylated one (g/mol). N (Normality): equivalent/liter).
The molecular weight of each sample of deacetylated chitin was calculated from the intrinsic viscosity denoted [η] using the following Mark-Houwink equation [14]:
where k and a are constants and M is the molecular weight (Dalton). The intrinsic viscosity of each sample was determined experimentally using a capillary tube viscometer. This was performed by measuring the time taken for the solvent, consisting of 1 % acetic acid, to flow through the capillary tube. Time measurements were made in 10 replicates for better accuracy. The intrinsic viscosity can be determined either from the reduced or inherent viscosity. The reduced viscosity η red is given by the equation:
where C i is the concentration of chitosan and η sp (specificity viscosity) is given by:
where t and t 0 are the flow times of the chitosan solution and the solvent, respectively.
The inherent viscosity η inh is defined by the following equation:
where η rel (relative viscosity) is given by:
Different values of reduced an inherent viscosities were obtained for each sample of different concentrations (0.01, 0.025, 0.05, 0.075 and 0.1 g/dl). The plot of reduced or inherent viscosity as a function of the concentration is a straight line whose intercept is equal to the intrinsic viscosity.
The determination of molecular weight of the chitosan from its intrinsic viscosity using the Mark-Houwink equation requires knowledge of constants a and K that depend essentially on the temperature and the used solvent. Based on Rao [15] who used the same solvent at 30 °C, the following values of 0.72 and 4.74 × 10−5 dl/g were assigned to a and K, respectively.
Qualitative analyses using 13C NMR and thermogravimetry were performed on the different samples in order to confirm the results obtained by the conductivity method.
Results and Discussion
Chitosan was prepare directly by deacetylation of crude chitin in this work. The multiple chitin preparation steps prior to deacetylation were omitted because the impurities can be removed during or after the deacetylation procedure: the minerals are not soluble in the final dilute acetic acid dissolution step, the proteins remain soluble and do not precipitate from the deacetylated solutions when the pH is raised during the rinsing step. Therefore, the impurities present in chitin, including proteins, fats, minerals and other contaminants [16], need not be pre-removed for environmental applications of chitosan.
The change in conductivity during the titration with sodium hydroxide is represented by Figs. 2 and 3 at 35 and 20 °C, respectively. These titration curves exhibit two inflection points. The difference in volume of NaOH (V 2 − V 1) between these two points (Fig. 2) corresponds to the amount of HCl required to protonate the amino groups of the deacetylated chitin [17]. The results of DDA range from 50 to 99 %. The titration of chitin is described by the following reaction.

.
In Fig. 2, the conductivity decreases while the excess HCl is neutralized, followed by a slope change when the protonated amines are neutralized by base, and finally the conductivity increases when the pH rises with excess base. The volume of base comprised between points V1 and V2 correspond to the number of moles of amino groups titrated in the chitosan. Similar curves are observed for chitin extracted at 20 °C, as illustrated in Fig. 3. From these results, the degrees of deacetylation can be derived, and are illustrated in Fig. 4. It can be observed that the chitin reacts faster at 35 °C than at 20 °C. A high degree of deacetylation occurs before 10 days time, sufficient to make a chitosan solution in 1 % acetic acid. On the other hand, the viscosities of the solutions tend to decrease faster too and were investigated. The evolution of the reduced viscosities η red and inherent viscosities η inh as a function of the concentration of deacetylated chitin (chit D35-T20) is shown by Fig. 5. Similar calculations gave the viscosities of the products obtained at 35 °C. From these results, the plot of molecular weight as a function of extraction times was established for both temperatures and is illustrated in Fig. 6. Clearly, the small increase in hydrolysis temperature results in a significant decrease in molecular weight of the chitosan. The value of molecular weight (MW) of the chitosan depends intimately on the preparation process as it can induce depolymerization of macromolecular chains and/or degradation during its production [18]. Macromolecular chains of chitosan generally have a molecular weight of about 100–1500 kDa [12]. This molecular weight determines the desired application for the chitosan. The chitin products obtained in this investigation have relatively high molecular weights, compared with other samples mentioned in the literature [19]. This is probably due to the treatment at low temperature (20 and 35 °C) during the deacetylation of chitin, as temperature has a strong effect on the molecular weight of chitosan. These high molecular weights would provide a better behavior to chitosan during flocculation. Molecular weights typically encountered in this type of chitosan have values between 100 and 500 kDa [11].
The results of 13C NMR analysis shown in Fig. 7 clearly indicate the presence of peaks of high intensity corresponding to the acetyl group (CH3 and C=O) in the spectrum of undeacetylated chitin. These peaks appeared at very low intensities in the case of deacetylated chitin (chitD7-T35, chitD35-T20) and were almost absent in the spectrum of the sample chitD35-T35.
The solid state 13C spectrum is ideal for identifying the presence or absence of methyl groups present in the acetylated form. The spectrum allows identification of the other carbons labeled as depicted in Fig. 1. The attributions are from Ref. [20–22]. It can be observed that the acetyl group gradually decreases in intensity as the deacetylation progresses. The other peaks shift during the reaction except for the C3 and C5 peaks. The absence of extraneous peaks shows the chitosan is pure and free of other protein or impurities [22].
A typical thermogravimetric curve is illustrated in Fig. 8a. Thermogravimetry also shows differences according to deacetylation degree: there are two weight losses the first (M1) corresponding to the acetyl group portion and the second (M2) to the cyclic part of the polymer. The M1/M2 ratio gradually decreases from 1.3 to 0.8 during deacetylation (see Fig. 8b).
Clarification of urban waters usually involves treatment with coagulation and flocculation additives. Biodegradable or biocompatible additives are sought to promote ecofriendly processes and renewable biopolymers would be favored over synthetic ones. The chitosan biopolymers obtained in our work were conveniently derived from marine wastes, at different temperatures including room temperature and atmospheric pressure. Advanced deacetylation occurs after 15 days in base without stirring and simply macerating in a plastic flask. Deacetylation is faster when heating at 120 °C for a few hours, but requires investing in equipment. To compare the results given by high or low temperature deacetylation, the variation in turbidity of waste water was examined following the action of each type of chitin. The results plotted in Fig. 9, and summarized in the table, show a significant reduction in turbidity together with reduced COD, reduced suspended particles and volatile matters. The best results were derived with the use of chitD15-T20 at 1.5 mg/l concentrations. At higher concentrations of chitosan, self-aggregation disfavored the flocculation and the results were not as good as at lower concentrations.
At the optimum dosage of 1.5 mg/l of chitosan, the results presented in Table 1 show that contaminants were reduced to levels acceptable according to local regulations.
Conclusion
The kinetics of deacetylation of the chitin, incubated at 20 and 35 °C for 35 days showed that the degree of deacetylation (DDA) increased with time of incubation reaching 88 and 99 % at 20 and 35 °C, respectively. A moderate decrease in molecular weight of the chitin which did not exceeded 170 and 226 kDa for the samples incubated at 35 and 20 °C, respectively, was observed. This would provide for the chitin a better behavior in flocculation. Qualitative analysis of different samples of deacetylated chitin by 13C NMR and thermogravimetry were in good agreement with the results obtained by the conductivity method. This low cost method for obtaining chitosan should help recovery of chitin in developing countries lacking industrial facilities to transform shrimp waste into a valuable polymer. Such easily obtained biopolymers can be an economical and eco-friendly polymer for use in treating waste waters.
References
Mohammed, M.M., Williams, P.A., Tveresovskaya, O.: Extraction of chitin from prawn shells and conversion to low molecular mass chitosan. Food Hydrocoll. 31(2), 166–171 (2013)
Yong, S.K., Shrivastava, M., Srivastava, P., Kunhikrishnan, A., Bolan, N.: Environmental applications of chitosan. Rev. Environ. Contam. Toxicol. 233, 1–43 (2015)
Pacheco, N., Garnica-Gonzalez, M., Gimeno, M., Bárzana, E., Trombotto, S., David, L., Shirai, K.: Structural characterization of chitin and chitosan obtained by biological and chemical methods. Biomacromolecules 12(9), 3285–3290 (2011)
Villa-Lerma, G., González-Márquez, H., Gimeno, M., López-Luna, A., Bárzana, E., Shirai, K.: Ultrasonication and steam-explosion as chitin pretreatments for chitin oligosaccharide production by chitinases of Lecanicillium lecanii. Bioresour. Technol. 146, 794–798 (2013)
Jung, J., Zhao, Y.: Characteristics of deacetylation and depolymerization of (sup)-chitin from jumbo squid (Dosidicus gigas) pens. Carbohydr. Res. 346(13), 1876–1884 (2011)
Younes, I., Ghorbel-Bellaaj, O., Chaabouni, M., Rinaudo, M., Souard, F., Vanhaverbeke, C., Jellouli, K., Nasri, M.: Use of a fractional factorial design to study the effects of experimental factors on the chitin deacetylation. Int. J. Biol. Macromol. 70, 385–390 (2014)
No, H.K., Meyers, S.P.: Preparation and characterization of chitin and chitosan. A review. J. Aquat. Food Prod. Technol. 4(2), 27–52 (1995)
de Moura, C.M., de Moura, J.M., Soares, N.M., de Almeida Pinto, L.A.: Evaluation of molar weight and deacetylation degree of chitosan during chitin deacetylation reaction: used to produce biofilm. Chem. Eng. Process. 50(4), 351–355 (2011)
Tolaimate, A., Desbrieres, J., Rhazi, M., Alagui, A.: Contribution to the preparation of chitins and chitosans with controlled physico-chemical properties. Polymer 44(26), 7939–7952 (2003)
Zeng, D., Wu, J., Kennedy, J.F.: Application of a chitosan flocculant to water treatment. Carbohydr. Polym. 71(1), 135–139 (2008)
Rinaudo, M.: Chitin and chitosan: properties and applications. Prog. Polym. Sci. 31(7), 603–632 (2006)
Crini, G., Badot, P.M., Guibal É.: Chitine et chitosane: du biopolymère à l’application. Presses universitaires de Franche-Comté (2009)
Yu, G.E., Morin, F.G., Nobes, G.A.R., Marchessault, R.H.: Degree of acetylation of chitin and extent of grafting PHB on chitosan determined by solid state N-15 MMR. Macromolecules 32(2), 518–520 (1999)
Kasaai, M.R.: Calculation of Mark-Houwink-Sakurada (MHS) equation viscometric constants for chitosan in any solvent-temperature system using experimental reported viscometric constants data. Carbohydr. Polym. 68(3), 477–488 (2007)
Rao, D.G.: Studies on viscosity-molecular weight relationship of chitosan solutions. J. Food Sci. Technol. (India) 30(1), 66–67 (1993)
Muzzareli, R.A.A.: Chitins and chitosans as immunoadjuvants and non-allergenic drug carriers. Mar. Drugs 8(2), 292–312 (2010)
de Alvarenga, E.S., de Oliveira, C.P., Bellato, C.R.: An approach to understanding the deacetylation degree of chitosan. Carbohydr. Polym. 80(4), 1155–1160 (2010)
Jia, Z., Shen, D.: Effect of reaction temperature and reaction time on the preparation of low-molecular-weight chitosan using phosphoric acid. Carbohydr. Polym. 49(4), 393–396 (2002)
Gupta, K.C., Jabrail, F.H.: Effects of degree of deacetylation and cross-linking on physical characteristics, swelling and release behavior of chitosan microspheres. Carbohydr. Polym. 66(1), 43–54 (2006)
Zhang, M., Hagga, A., Sekiguchi, H., Hirano, S.: Chitin isolated from beetle larva cuticle and silkworm (Bombix mori) pupaexuvia. Int. J. Biol. Macromol. 27, 99–105 (2000)
Lavertu, M., Xia, Z., Serregi, A.N., Berrada, M., Rodrigues, A., Wang, D., Buschmann, M.D., Gupta, A.: A validated 1H NMR method for the determination of the degree of deacetylation of chitosan. J. Pharm. Biomed. Anal. 32, 1149–1158 (2003)
Paulino, A.T., Simionato, J., Garcia, J.C., Nozaki, J.: Characterization of chitosan and chitin produced from silkworm chrysalides. Carbohydr. Polym. 64, 98–103 (2006)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kadouche, S., Farhat, M., Lounici, H. et al. Low Cost Chitosan Biopolymer for Environmental Use Made from Abundant Shrimp Wastes. Waste Biomass Valor 8, 401–406 (2017). https://doi.org/10.1007/s12649-016-9593-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-016-9593-2