Abstract
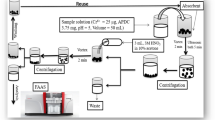
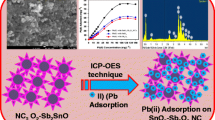
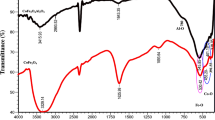
In this work, Co2SiO4 nanoparticles were facilely synthesized using the pechini sol gel method then modified by mordant red 3 as a novel composite for the separation and efficient preconcentration of palladium(II) ions from the aqueous chemical solutions prior to their determination by atomic absorption spectrometry. The synthesized products were characterized by X-ray diffraction (XRD), Fourier-transform infrared spectroscopy (FT-IR), transmission electron microscope (TEM), scanning electron microscope (SEM), and CHN elemental analysis. The successful modification of the Co2SiO4 nanoparticles by mordant red 3 was demonstrated by the appearance of a wide XRD peak centered at 2Ɵ=24o, SEM morphological change to flower-like shapes, the presence of carbon (C) and nitrogen (N), and the presence of characteristic organic functional groups using FT-IR. The maximum elimination capability of the synthesized nanocomposite towards palladium (II) ions is 166.39 mg/g. The adsorption of palladium (II) ions is spontaneous, chemical, and can be adequately described using the pseudo-second order kinetic model and Langmuir equilibrium isotherm. The nanocomposite was regenerated at least five times with a 0.5 M thiourea solution without a notable decrease in the % removal. The results confirmed that the optimized procedure is accurate (% recovery > 95%), reproducible (% RSD < 3.5%), and has a 0.5–450 µg/L dynamic linear range.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Data Availability
Data will be made available on request.
References
Ye FY, Hu M, Yu W, Zheng YS (2022) Highly selective detection of palladium ions by sulfur-containing tetraphenylethylene tetracycle helicate. Dye Pigment 208:110857. https://doi.org/10.1016/j.dyepig.2022.110857
Aruguete DM, Wallace A, Blakney T et al (2020) Palladium release from catalytic converter materials induced by road de-icer components chloride and ferrocyanide. Chemosphere 245:125578. https://doi.org/10.1016/j.chemosphere.2019.125578
Kielhorn J, Melber C, Keller D, Mangelsdorf I (2002) Palladium - A review of exposure and effects to human health. Int J Hyg Environ Health 205:417–432. https://doi.org/10.1078/1438-4639-00180
Zhang N, Ma Y, Shen Y, Gao X (2014) Determination of platinum, palladium, ruthenium, rhodium, and iridium in ultrabasic rock from the Great Dyke of Zimbabwe by inductively coupled plasma-optical emission spectrometry. Anal Lett 47:2072–2079. https://doi.org/10.1080/00032719.2014.893441
Müller M, Heumann KG (2000) Isotope dilution inductively coupled plasma quadrupole mass spectrometry in connection with a chromatographic separation for ultra trace determinations of platinum group elements (pt, pd, Ru, ir) in environmental samples. Fresenius J Anal Chem 368:109–115. https://doi.org/10.1007/s002160000494
Tokalioǧlu Ş, Oymak T, Kartal Ş (2004) Determination of palladium in various samples by atomic absorption spectrometry after preconcentration with dimethylglyoxime on silica gel. Anal Chim Acta 511:255–260. https://doi.org/10.1016/j.aca.2004.02.015
Mori I, Kawakatsu T, Fujita Y, Matsuo T (1999) Selective spectrophotometric determination of palladium(II) with 2(5- nitro-2-pyridylazo)-5-(N-propyl-N-3-sulfopropylamino)phenol(5-NO2.PAPS) and tartaric acid with 5-NO2.PAPS-niobium(V) complex. Talanta 48:1039–1044. https://doi.org/10.1016/S0039-9140(98)00309-9
Tu Z, Lu S, Chang X et al (2011) Selective solid-phase extraction and separation of trace gold, palladium and platinum using activated carbon modified with ethyl-3-(2-aminoethylamino)-2-chlorobut-2-enoate. Microchim Acta 173:231–239. https://doi.org/10.1007/s00604-011-0552-0
Mohammadi SZ, Afzali D, Taher MA, Baghelani YM (2010) Determination of trace amounts of palladium by flame atomic absorption spectrometry after ligandless-dispersive liquid-liquid microextraction. Microchim Acta 168:123–128. https://doi.org/10.1007/s00604-009-0267-7
Hassanien MM, Mortada WI, Kenawy IM (2015) Selective separation of palladium from synthetic highly active liquid waste by cloud point extraction using benzil mono-(2-pyridyl)hydrazone and Triton X-114. J Radioanal Nucl Chem 303:261–269. https://doi.org/10.1007/s10967-014-3430-5
Hu Q, Yang X, Huang Z et al (2005) Simultaneous determination of palladium, platinum, rhodium and gold by on-line solid phase extraction and high performance liquid chromatography with 5-(2-hydroxy-5-nitrophenylazo)thiorhodanine as pre-column derivatization regents. J Chromatogr A 1094:77–82. https://doi.org/10.1016/j.chroma.2005.07.090
Al-Wasidi AS, Naglah AM, Saad FA, Abdelrahman EA (2022) Modification of sodium aluminum silicate hydrate by thioglycolic acid as a new composite capable of removing and preconcentrating pb(II), Cu(II), and zn(II) ions from food and water samples. Arab J Chem 15:104178. https://doi.org/10.1016/j.arabjc.2022.104178
Al-Wasidi AS, AlSalem HS, Alshalawi AF et al (2022) Facile synthesis of a novel nanocomposite for determination of mercury and copper ions in food and water samples. Arab J Chem 15:104113. https://doi.org/10.1016/j.arabjc.2022.104113
Parastar M, Sheshmani S, Shokrollahzadeh S (2021) Cross-linked chitosan into graphene oxide-iron(III) oxide hydroxide as nano-biosorbent for pd(II) and cd(II) removal. Elsevier B.V, Amsterdam
Monier M, Abdel-Latif DA, Abou El-Reash YG (2016) Ion-imprinted modified chitosan resin for selective removal of pd(II) ions. J Colloid Interface Sci 469:344–354. https://doi.org/10.1016/j.jcis.2016.01.074
Kumar R, Sharma RK, Singh AP (2019) Grafting of cellulose with N-isopropylacrylamide and glycidyl methacrylate for efficient removal of Ni(II), Cu(II) and pd(II) ions from aqueous solution. Sep Purif Technol 219:249–259. https://doi.org/10.1016/j.seppur.2019.03.035
Naini N, Sid Kalal H, Almasian MR et al (2022) Phosphine-functionalized Fe3O4/SiO2/composites as efficient magnetic nanoadsorbents for the removal of palladium ions from aqueous solution: kinetic, thermodynamic and isotherm studies. Mater Chem Phys 287:126242. https://doi.org/10.1016/j.matchemphys.2022.126242
Gad HM, El Rayes SM, Abdelrahman EA (2022) Modification of silica nanoparticles by 2,4-dihydroxybenzaldehyde and 5-bromosalicylaldehyde as new nanocomposites for efficient removal and preconcentration of Cu(ii) and cd(ii) ions from water, blood, and fish muscles. RSC Adv 12:19209–19224. https://doi.org/10.1039/d2ra03177a
Khalifa ME, Abdelrahman EA, Hassanien MM, Ibrahim WA (2020) Application of mesoporous silica nanoparticles modified with dibenzoylmethane as a novel composite for efficient removal of cd(II), hg(II), and Cu(II) ions from aqueous media. J Inorg Organomet Polym Mater 30:2182–2196. https://doi.org/10.1007/s10904-019-01384-w
Bayat S, Sobhani A, Salavati-Niasari M (2018) Co2SiO4 nanostructures/nanocomposites: synthesis and investigations of optical, magnetic, photocatalytic, thermal stability and flame retardant properties. J Mater Sci Mater Electron 29:7077–7089. https://doi.org/10.1007/s10854-018-8695-y
Guo P, Wang C (2015) Synthesis and lithium storage performance of Co2SiO4 nanoparticles. RSC Adv 5:70661–70667. https://doi.org/10.1039/c5ra11327j
Vaselnia SY, Khajeh Aminian M, Dehghan Banadaki R (2022) Experimental and theoretical study on the structural, electronic, and optical properties within DFT + U, Fxc kernel for LRC model, and BSE approaches. Part II: CoSiO3 and Co2SiO4 pigments. Powder Technol 397:116999. https://doi.org/10.1016/j.powtec.2021.11.043
Mu Y, Zhang Y, Pei X et al (2022) Dispersed FeOx nanoparticles decorated with Co2SiO4 hollow spheres for enhanced oxygen evolution reaction. J Colloid Interface Sci 611:235–245. https://doi.org/10.1016/j.jcis.2021.12.099
Stoia M, Stefanescu M, Dippong T et al (2010) Low temperature synthesis of Co2SiO4/SiO2 nanocomposite using a modified sol-gel method. J Sol-Gel Sci Technol 54:49–56. https://doi.org/10.1007/s10971-010-2156-2
Shojaei B, Miri R, Bazyari A, Thompson LT (2022) Asphaltene adsorption on MgO, CaO, SiO2, and Al2O3 nanoparticles synthesized via the Pechini-type Sol – Gel method. Fuel 321:124136. https://doi.org/10.1016/j.fuel.2022.124136
Mirzaei A, Janghorban K, Hashemi B et al (2016) Highly stable and selective ethanol sensor based on α-Fe2O3 nanoparticles prepared by Pechini sol-gel method. Ceram Int 42:6136–6144. https://doi.org/10.1016/j.ceramint.2015.12.176
Chrunik M, Majchrowski A, Zasada D et al (2017) Modified Pechini synthesis of Bi2ZnB2O7 nanoparticles. J Alloys Compd 725:587–597. https://doi.org/10.1016/j.jallcom.2017.07.172
Zinatloo-Ajabshir S, Salavati-Niasari M (2019) Preparation of magnetically retrievable CoFe2O4@SiO2@Dy2Ce2O7 nanocomposites as novel photocatalyst for highly efficient degradation of organic contaminants. Compos Part B Eng 174:106930. https://doi.org/10.1016/j.compositesb.2019.106930
Boullosa-Eiras S, Vanhaecke E, Zhao T et al (2011) Raman spectroscopy and X-ray diffraction study of the phase transformation of ZrO2-Al2O3 and CeO2-Al 2O3 nanocomposites. Catal Today 166:10–17. https://doi.org/10.1016/j.cattod.2010.05.038
Youssef HM, Shah RK, Algethami FK et al (2021) Facile hydrothermal procedure for the synthesis of sodium aluminum silicate hydrate/analcime and analcime for effective removal of manganese(II) ions from aqueous solutions. J Inorg Organomet Polym Mater 31:1035–1046. https://doi.org/10.1007/s10904-020-01699-z
Almutairi MA, Algethami FK, Youssef HM (2020) Facile fabrication of novel analcime / sodium aluminum silicate hydrate and zeolite Y / faujasite mesoporous nanocomposites for efficient removal of Cu (II) and pb (II) ions from aqueous media. Integr Med Res 9:7900–7914. https://doi.org/10.1016/j.jmrt.2020.05.052
Behbahani M, Bagheri A, Gorji T et al (2013) Application of poly (N-phenylethanolamine) modified MWCNTs as a new sorbent for solid-phase extraction of trace palladium ions in soil and water samples. Sample Prep 1:10–17. https://doi.org/10.2478/sampre-2013-0002
Nagarjuna R, Sharma S, Rajesh N, Ganesan R (2017) Effective adsorption of precious metal palladium over polyethyleneimine-functionalized alumina nanopowder and its reusability as a catalyst for energy and environmental applications. ACS Omega 2:4494–4504. https://doi.org/10.1021/acsomega.7b00431
Behbahani M, Najafi F, Amini MM et al (2014) Solid phase extraction using nanoporous MCM-41 modified with 3,4-dihydroxybenzaldehyde for simultaneous preconcentration and removal of gold(III), palladium(II), copper(II) and silver(I). J Ind Eng Chem 20:2248–2255. https://doi.org/10.1016/j.jiec.2013.09.057
Farahani MD, Shemirani F, Gharehbaghi M (2013) Ferrofluid-based dispersive solid phase extraction of palladium. Talanta 109:121–127. https://doi.org/10.1016/j.talanta.2013.01.061
Ruhela R, Singh KK, Tomar BS et al (2012) Amberlite XAD-16 functionalized with 2-acetyl pyridine group for the solid phase extraction and recovery of palladium from high level waste solution. Sep Purif Technol 99:36–43. https://doi.org/10.1016/j.seppur.2012.08.018
Author information
Authors and Affiliations
Contributions
Faisal K Algethami conducted experimental work, organized the experimental results, wrote the manuscript, and corresponded the manuscript for publication.
Corresponding author
Ethics declarations
Ethics Approval
Author approves that the submitted work is original and has not been published elsewhere in any form or language (partially or in full).
Consent to Participate
Yes.
Consent for Publication
Yes.
Conflict of Interest
The author declares that he has no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Algethami, F.K. Efficient Removal and Preconcentration of Pd(II) Ions in Environmental Water Using Co2SiO4 Nanoparticles Modified by Mordant Red 3 as a Novel Adsorbent. Silicon 15, 5891–5909 (2023). https://doi.org/10.1007/s12633-023-02487-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-023-02487-3