Abstract
The hepatitis E virus (HEV) genotype 3 (GT3) is an emergent pathogen in industrialized countries. It is transmitted zoonotically and may lead to chronic hepatitis in immunocompromised individuals. We evaluated if the major antigen of HEV, the capsid protein, can be used in combination with immunobiotic bacterium-like particles (IBLP) for oral vaccination in a mouse model. We have cloned and expressed the RGS-His5-tagged HEV GT3 capsid protein (ORF2) in E. coli and purified it by NiNTA. IBLP were obtained from two immunobiotic Lactobacillus rhamnosus strains acid- and heat-treated. ORF2 and the IBLP were orally administered to Balb/c mice. After three oral immunizations (14-day intervals), blood, intestinal fluid, Peyer´s patches, and spleen samples were drawn. IgA- and IgG-specific antibodies were determined by ELISA. Mononuclear cell populations from Peyer’s patches and spleen were analyzed by flow cytometry, and the cytokine profiles were determined by ELISA to study cellular immunity. Orally administered recombinant ORF2 and IBLP from two L. rhamnosus strains (CRL1505 and IBL027) induced both antigen-specific humoral and cellular immune responses in mice. IBLP027 was more effective in inducing specific secretory IgA in the gut. IFN-γ, TNF-α, and IL-4 were produced by Peyer’s plaques lymphocytes stimulated with ORF2 ex vivo suggesting a mixed Th1/Th2-type adaptive immune response in immunized mice. Oral vaccines are not invasive, do not need to be administered by specialized personal, and elicit both systemic and local immune responses at the port of entry. Here, we present an experimental oral vaccine for HEV GT3, which could be further developed for human and/or veterinary use.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The hepatitis E virus is an emergent hepatotropic virus, which causes hepatitis outbreaks in developing countries and has extraordinarily high mortality in pregnant women (ca. 30%) [1, 2]. Four genotypes cause disease in humans: genotype 1 (GT1), which is responsible of epidemics in developing countries in Asia and Africa [3, 4]; GT2, which was found in Mexico; and GT4, which was exclusively isolated in Asia [5]. In addition, GT3, which has a zoonotic origin, has been associated with cases worldwide [5] and to the development of chronic hepatitis in immunocompromised individuals [6, 7]. Recently, other genotypes found in monkey, rabbit, camel, and rat were also reported to cause human infections underlining the strong zoonotic potential of this virus [8,9,10,11]. HEV is a small naked virus with a simple-stranded RNA (ssRNA) genome of approximately 7.2 kb that encodes for three open reading frames (ORF). The ORF1 encodes a poly-protein involved in genomic replication, ORF3 a phosphoprotein with putative regulation functions, and ORF2 the single capsid protein, the main immunogenic antigen. The lack of an efficient cell culture system for HEV and the absence of a well-characterized small animal model are drawbacks in HEV research. Despite that, researches have advanced in the knowledge of infection biology of HEV that has allowed the generation of a vaccine. An intramuscular subunit vaccine consisting of a fragment of ORF2 from HEV GT1 (Heicolin®) has been licensed in China for vaccination of healthy adults. This vaccine elicits protective IgG antibodies, which reduced the incidence of the disease but did not affect the rate of infection because the virus is neutralized only when it reaches the bloodstream after replication.
Mucosal vaccines offer several advantages in comparison to systemic vaccines. These include the induction of selective mucosal immunity that protects the host at the infection port of entry, and no need of needle or specialized personnel for its administration and therefore mass administration is possible. However, subunit vaccines to be administered at mucosal sites need to be administered with adjuvants to ensure immunity, as mucosal sites, especially the intestinal mucosa, tend to be tolerant.
Several studies have demonstrated that Gram-positive enhancer matrix [12] particles also known as bacterium-like particles (BLP) stimulate potent immune responses when administered together with microbial antigens [12, 13]. Most studies evaluating the ability of BLP to improve immune responses to vaccine antigens have been performed with BLP obtained from Lactococcus lactis [14]. BLP were used as a non-genetically modified delivery system for pathogen’s epitopes by anchoring antigens on their surface via peptidoglycan binding protein domains fused to the antigen of interest [15,16,17]. These complexes delivered orally or nasally induced mucosal and systemic protective immunity to different bacterial and viral pathogens including Streptococcus pneumoniae [18], influenza virus [19,20,21], and respiratory syncytial virus [22]. The adjuvant properties of BLP have also been used for improving the efficacy of existing vaccines [13, 23]. This strategy was tested for influenza vaccination using a seasonal influenza vaccine mixed with L. lactis derived BLP. It was reported that the use of BLP reduced the antigen doses needed to obtain a protective response, elicited both local and systemic immune responses, and shifted the immune response to a Th1-type [13, 23].
The immunomodulatory ability of lactic acid bacteria (LAB) is a strain-specific property. The distinct ability of LAB strains to modulate the immune system has been attributed to the different microbial-associated molecular patterns (MAMPs) that interact with distinct pattern recognition receptors (PRRs) expressed in immune and non-immune cells [24,25,26,27,28]. Therefore, it is possible to speculate that BLP obtained from different LAB strains would have distinct adjuvant capacities. As our starting material is an immunomodulatory strain, the obtained BLP conserve the immunomodulatory properties and we refer to them as immunobiotic bacterium-like particles (IBLP).
In this study, we evaluated the adjuvant activity of IBLP produced from immunomodulatory L. rhamnosus strains when mixed with purified HEV ORF2 from GT3 and administered orally to mice.
Material and Methods
Microorganisms
L. rhamnosus CRL1505 and L. rhamnosus IBL027 [29] were obtained from the CERELA culture collection and the IBL culture collection, respectively. They were kept freeze in 50% glycerol. For experiments, bacteria were activated and finally cultured over night at 37 °C (final log phase) in Man–Rogosa–Sharpe broth (MRS, Oxoid).
Production of Immunobiotic Bacterium-Like Particles
Fresh overnight cultures (100 ml) of L. rhamnosus IBL027 and L. rhamnosus CRL1505 were collected by centrifugation (10 min, 13,000×g) and washed once with sterile distilled water. Afterwards, the pellets were suspended in 20 ml of 0.1 M HCl and treated at 99 °C in a water bath for 45 min. Next, the cells were washed three times in 50 ml sterile phosphate buffer saline (PBS), pH 7.4, with vigorous vortexing. After the last washing step, cells were resuspended in 10 ml PBS and stored at − 20 °C. The number of IBLP particles per milliliter was adjusted to the CFU/ml determined in the starting culture. IBLP obtained from L. rhamnosus IBL027 and L. rhamnosus CRL1505 were designated as IBLP1505 and IBLP027, respectively.
Recombinant Viral Proteins
The HEV ORF2 protein is a 66-kDa recombinant polypeptide corresponding to amino acid residues 112-608 of the capsid protein of HEV GT3. HEV ORF2 genotype 3 (GT3) contained in a pMK plasmid flanked with attB sites was obtained by synthesis at GeneArt Gene Synthesis (TermoFisher Scientific) and further subcloned into pETG-N-RGS-His-[rfB] vector [30] by Gateway® recombinatorial cloning, and transformed into E. coli Rossetta (DE3). Recombinant protein expression was induced with IPTG (1 mM) for 3 h. ORF2 protein was purified under native and denaturing condition using NiNTA chromatography (ThermoFisher Scientific) following the manufacturer’s instructions. The purified protein was examined by SDS-PAGE using 10 % separating gels and quantified by Bradford (Bio-Rad).
Animals and Treatments
Animals were housed individually and fed a conventional balanced diet ad libitum during the experiments. All efforts were made to minimize the number of animals and their suffering.
Six-week-old BALB/c male mice were obtained from a closed colony kept at CERELA. Mice were orally immunized three times every 14 days with purified HEV ORF2 protein obtained under native (N) or denaturing conditions (D). For immunization, 75 μg of N protein or 7.5 μg of D protein were administered alone or combined with IBLP1505 or IBLP027 as adjuvants at a final dose of 108 cells/mouse/day. Therefore, seven groups of 5 mice each were used including controls, mice receiving purified HEV ORF2 (ORF2N and ORF2D groups), HEV ORF2 plus IBLP1505 (ORF2N+IBLP1505 and ORF2D+IBLP1505 groups) or HEV ORF2 plus IBLP027 (ORF2N+IBLP027 and ORF2D+IBLP027 groups). Humoral- and cellular-specific immune responses were evaluated at the end of the last immunization (day 42) (Fig. 1).
Immunization scheme. Balb/c mice were grouped in seven groups with five mice each and orally immunized three times every 14 days. HEV ORF2 protein obtained under native (ORF2N) or denaturing conditions (ORF2D) was given alone or adjuvanted with IBLP [immunobiotic bacterium-like particles (IBLP), from L. rhamnosus CRL1505 (IBLP1505) or L. rhamnosus IBL027 (IBLP027)]. Seven groups of five mice each were used including controls, mice receiving purified HEV ORF2 (ORF2N and ORF2D groups), HEV ORF2 plus IBLP1505 (ORF2N+IBLP1505 and ORF2D+IBLP1505 groups), or HEV ORF2 plus IBLP027 (ORF2N+IBLP027 and ORF2D+IBLP027 groups). Intestinal fluid, blood, spleen, and Peyer’s patches were drawn 42 days after the 1st oral immunization
Tissue and Fluid Sampling
Blood samples were obtained through cardiac puncture and intestinal fluid samples (IF) were obtained as described previously [31]. Briefly, the small intestine was exposed, and two sequential lavages were performed in each mouse by injecting sterile PBS. The recovered fluid was centrifuged for 10 min at 10,000 rpm. The supernatant fluids were kept frozen at − 70 °C for subsequent analyses. In addition, Peyer’s plaques and spleen were removed aseptically and treated for immune cell isolation as described below.
Transaminases Determination
To evaluate hepatic toxicity, the levels of ALT and AST in serum samples were determined using the automatized COBAS 4000 c311 for blood chemistry (Roche).
Flow Cytometry Studies
Peyer’s patches and spleen were teased gently to release cells, which were filtered through a cell strainer (Becton, Dickinson, San Jose, CA, USA) to remove debris. After washing the cells twice in PBS supplemented with 2% fetal bovine serum, they were counted using Trypan Blue. Suspensions were adjusted to 5 × 106 cells/ml for flow cytometry studies. Cell suspensions were pre-incubated with anti-mouse CD32/CD16 monoclonal antibody (Fc block) for 15 min at 4 °C. Primary antibodies (anti-mouse CD45-PE, anti-mouse CD3-FITC, anti-mouse CD4-biotin, anti-mouse CD8-biotin, anti-mouse CD24-FITC, and anti-mouse B220-PE, BD PharMingen) were incubated with cells for 30 min at 4 °C and washed with FACS buffer. Streptavidin-PerCP was used as a second-step reagent. Cells were acquired on a BD FACSCaliburTM flow cytometer (BD Biosciences) and data were analyzed with FlowJo software (TreeStar).
Cytokine Determination
Tumor necrosis factor (TNF)-α, interferon (IFN)-γ, and interleukin (IL)-4 were measured in supernatants of stimulated mononuclear cells isolated from Peyer’s patches or spleen with commercially available enzyme-linked immunosorbent assay (ELISA) kits following the manufacturer’s recommendations (R&D Systems, MN, USA).
Anti-HEV ORF2-Specific antibodies
ELISA was performed to determine HEV ORF2-specific antibodies in sera and intestinal fluids. Purified HEV GT3 ORF2 was coated (1 μg/ml) onto 96-well high-binding microtiter plates, blocked with 1% gelatin and incubated with diluted mouse sera or diluted mouse intestinal fluid for 1 h. Bound mouse antibodies were detected using HRP-conjugated antibodies directed to mouse IgG-Fc or IgA (MP Biomedicals, Inc.). Endpoint enzymatic activity was detected with TMB as substrate. The reaction was stopped with 0.1 M H3PO4 and measured at 495 nm in a microplate reader.
Statistical Analysis
Experiments were performed in triplicate and results were expressed as mean ± standard error (SE). After verification of the normal distribution of data, two-way ANOVA was used. Tukey’s test (for pairwise comparisons of the means) was used to test for differences between the groups. Differences were considered significant at P < 0.05.
Results
Development of HEV ORF2-IBLP Experimental Vaccines
We first aimed to produce and purify recombinant HEV ORF2 from GT3. In our hands, HEV ORF2 was mainly expressed in soluble form and to a lesser extent in inclusion bodies as it was reported for ORF2 from GT1 [32]. Under native conditions, ORF2 was present in monomeric and trimeric forms, whereas under denaturing conditions, as expected, only the monomeric form was detected (data not shown).
On the other hand, we obtained IBLP derived from the immunomodulatory strains L. rhamnosus IBL027 and L. rhamnosus CRL1505. IBLPs were assessed by electronic microscopy after treating bacteria with acid and heat. The elliptical shape of lactobacilli is conserved as well as the cell wall, whereas cytoplasmic membrane is disrupted and there is loss of intracellular content (data not shown). There were no morphological differences between the strains.
Experimental vaccines were developed with native and denatured proteins to reach two different concentrations related to the maximal yield obtained under native and denaturing conditions.
Humoral Immune Response Induced by HEV ORF2-IBLP Vaccines
We performed immunization experiments comparing the immune responses to the HEV ORF2 antigen under native and denaturing conditions with and without IBLP supplementation. The experiments included seven groups of mice: controls, ORF2N, ORF2D, ORF2N+IBLP1505, ORF2D+IBLP1505, ORF2N+IBLP027, and ORF2D+IBLP027 as described in the “Materials and Methods” section and Fig. 1. The different formulations were administered orally on day 0, and mice were boosted with the respective formulations on days 14 and 28 after the priming. Animals were euthanized on day 42 after priming for the evaluation of specific humoral and cellular immune responses. Mice were monitored daily to evaluate potential adverse effects of experimental vaccines. No signs of illness or discomfort were observed in any of the experimental groups. No changes in body weight gain were observed with respect to control animals (data not shown). In addition, the study of liver enzymes did not reveal alterations of liver function. All experimental groups showed ranges between 27.5 and 33 ± 7.14 IU/L for AST and 21 and 26.5 ± 6.3 IU/L for ALT, which correspond to reference values published elsewhere [33].
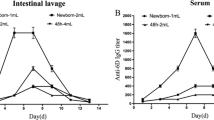
The determination of specific anti-HEV ORF2 antibodies showed that the proteins alone did not induce production of serum IgG or intestinal IgA (Fig. 2). However, when IBLP were used as adjuvants, IgG and IgA production increased significantly compared to non-vaccinated mice. IBLP1505 were more effective in inducing specific IgG than IBLP027. The latter only enhanced IgG production with the higher dose of the native protein HEV ORF2 (Fig. 2). In contrast, IBLP027 were more potent in inducing secretory IgA in the intestinal lumen.
Effect of oral immunization with HEV capsid protein (ORF2) administered with IBLP on adaptive humoral immune response. Serum (a) and intestinal levels (b) of specific anti-HEV ORF2 antibodies in serum or intestinal fluid, respectively. Antibodies were determined by ELISA. Fold increase was determined as the relation between respective treatment groups and the control groups and are expressed in arbitrary units (A.U.). Data was obtained as triplicate determinations of pooled samples from each mouse group. HEV ORF2 protein obtained under native (ORF2N) or denaturing conditions (ORF2D) was given alone or adjuvanted with IBLP [immunobiotic bacterium-like particles (IBLP), from L. rhamnosus CRL1505 (IBLP1505) or L. rhamnosus IBL027 (IBLP027)]. Seven groups of five mice each were used including controls, mice receiving purified HEV ORF2 (ORF2N and ORF2D groups), HEV ORF2 plus IBLP1505 (ORF2N+IBLP1505 and ORF2D+IBLP1505 groups), or HEV ORF2 plus IBLP027 (ORF2N+IBLP027 and ORF2D+IBLP027 groups). ANOVA followed by Dunnet’s test was conducted. *P < 0.05 was considered significant
Cellular Immune Response Induced by HEV ORF2-IBLP Vaccines
For the evaluation of the local cellular-specific immune response, lymphocytes from Peyer’s patches were isolated and evaluated by flow cytometry. As shown in Fig. 3, the frequency of CD45+CD3+CD4+ T cells was similar in mice immunized with ORF2N with or without the addition of IBLP. On the contrary, CD45+CD3+CD4+ T cells in ORF2D+IBLP027 and ORF2D+IBLP1505 immunized mice were higher than in the ORF2D group. Frequencies in CD45+CD3+CD8+ cytotoxic T cells were not significantly different among the different treated groups and the control group in Peyer’s patches (Fig. 3). When the B cell population was analyzed, it was found that Peyer’s patches CD24+B220+ cells were significantly increased in ORF2N and ORF2D when compared to control mice. In addition, administration of IBLPs improved the frequency of CD24+B220+ B cells when compared to their respective controls (Fig. 3). In order to further characterize the cellular immune response in the intestinal mucosa, mononuclear cells were isolated from Peyer’s patches and cultured. Then, cells were stimulated with the ORF2 protein and the levels of IFN-γ, TNF-α, and IL-4 were determined by ELISA on the supernatants. Immune cells from ORF2N and ORF2D groups produced IFN-γ and IL-4 in response to ORF2 stimulation in higher concentrations than those observed in control cells while only ORF2N group-derived cells produced higher levels of TNF-α than controls (Fig. 4). Both IBLP1505 and IBLP027 improved the levels of IFN-γ and TNF-α, when compared to ORF2N and ORF2D groups while the concentration of IL-4 was similar in all the immunized groups (Fig. 4).
Effect of oral immunization with HEV capsid protein (ORF2) administered with IBLP on lymphocytes from Peyer’s patches. Cell populations from Peyer’s plaques of mice immunized with HEV ORF2 were analyzed by flow cytometry. HEV ORF2 protein obtained under native (ORF2N) or denaturing conditions (ORF2D) was given alone or adjuvanted with IBLP [immunobiotic bacterium-like particles (IBLP), from L. rhamnosus CRL1505 (IBLP1505) or L. rhamnosus IBL027 (IBLP027)]. Seven groups of five mice each were used including controls, mice receiving purified HEV ORF2 (ORF2N and ORF2D groups), HEV ORF2 plus IBLP1505 (ORF2N+IBLP1505 and ORF2D+IBLP1505 groups), or HEV ORF2 plus IBLP027 (ORF2N+IBLP027 and ORF2D+IBLP027 groups). Different letters above bars indicate significant differences between groups. P < 0.05 was considered significant
Effect of oral immunization with HEV capsid protein (ORF2) administered with IBLP on cytokine production of mononuclear cells isolated from Peyer’s patches stimulated ex vivo with ORF2. HEV ORF2 protein obtained under native (ORF2N) or denaturing conditions (ORF2D) was given alone or adjuvanted with IBLP [immunobiotic bacterium-like particles (IBLP), from L. rhamnosus CRL1505 (IBLP1505) or L. rhamnosus IBL027 (IBLP027)]. Seven groups of five mice each were used including controls, mice receiving purified HEV ORF2 (ORF2N and ORF2D groups), HEV ORF2 plus IBLP1505 (ORF2N+IBLP1505 and ORF2D+IBLP1505 groups), or HEV ORF2 plus IBLP027 (ORF2N+IBLP027 and ORF2D+IBLP027 groups). Different letters above bars indicate significant differences between groups. P < 0.05 was considered significant
We also aimed to study the systemic specific immune response by evaluating the same parameters described above in mononuclear cells from spleens of immunized mice. As shown in Fig. 5, the frequency of CD45+CD3+CD4+ and CD45+CD3+CD8+ T cells was similar in all the experimental groups. On the other hand, the frequency of CD24+B220+ B cells in ORF2N and ORF2D cells was higher than in controls. It was also observed that spleen B cells were similar between ORF2D, ORF2D+IBLP1505, and ORF2D+IBLP027 while were higher in ORF2N+IBLP1505 and ORF2N+IBLP027 when compared to their respective control (Fig. 5). Spleen immune cells from ORF2N and ORF2D groups produced IFN-γ, TNF-α, and IL-4 in response to ORF2 stimulation in higher concentrations than those observed in control cells (Fig. 6). Both IBLP1505 and IBLP027 improved the levels of IFN-γ and TNF-α, when compared to ORF2N and ORF2D groups while the concentration of IL-4 was enhanced only in the ORF2N+IBLP027 and ORF2N+IBLP1505 groups (Fig. 6).
Effect of oral immunization with HEV capsid protein (ORF2) administered with IBLP on lymphocyte populations in spleen. Cell populations from spleen of mice immunized with HEV ORF2 were analyzed by flow cytometry. HEV ORF2 protein obtained under native (ORF2N) or denaturing conditions (ORF2D) was given alone or adjuvanted with IBLP [immunobiotic bacterium-like particles (IBLP), from L. rhamnosus CRL1505 (IBLP1505) or L. rhamnosus IBL027 (IBLP027)]. Seven groups of five mice each were used including controls, mice receiving purified HEV ORF2 (ORF2N and ORF2D groups), HEV ORF2 plus IBLP1505 (ORF2N+IBLP1505 and ORF2D+IBLP1505 groups), or HEV ORF2 plus IBLP027 (ORF2N+IBLP027 and ORF2D+IBLP027 groups). Different letters above bars indicate significant differences between groups. P < 0.05 was considered significant
Effect of oral immunization with HEV capsid protein (ORF2) administered with IBLP on cytokine production of mononuclear cells isolated from spleen stimulated ex vivo with ORF2. Mononuclear cells from spleen of mice immunized with HEV ORF2 were isolated and stimulated with ORF2 and cytokines were determined in cell culture supernatants by ELISA. HEV ORF2 protein obtained under native (ORF2N) or denaturing conditions (ORF2D) was given alone or adjuvanted with IBLP [immunobiotic bacterium-like particles (IBLP), from L. rhamnosus CRL1505 (IBLP1505) or L. rhamnosus IBL027 (IBLP027)]. Seven groups of five mice each were used including controls, mice receiving purified HEV ORF2 (ORF2N and ORF2D groups), HEV ORF2 plus IBLP1505 (ORF2N+IBLP1505 and ORF2D+IBLP1505 groups), or HEV ORF2 plus IBLP027 (ORF2N+IBLP027 and ORF2D+IBLP027 groups). Different letters above bars indicate significant differences between groups. P < 0.05 was considered significant
Discussion
The ORF2 protein is the only component of HEV capsid and both conformational and linear epitopes have been identified in its C-terminal domain [34,35,36]. There is a commercially available subunit vaccine for hepatitis E in China, formulated with protein ORF2 from HEV genotype 1, which is applied intramuscularly and elicits good protection at systemic level after three doses. It should be noted that although ORF2 is conserved across genotypes, there are different immunoreactivities between proteins derived from different genotypes [37]. In addition, this vaccine is not indicated for risk populations such as pregnant women and children [38] and does not confer mucosal immunity. Naturally acquired immunity to HEV seems to wane over time; thus, re-infections are possible, and the cross-sectional epidemiological studies conducted globally may underestimate the prevalence of HEV infection because of the natural extinction of anti-HEV IgG [39]. Therefore, HEV-specific immunity elicited by the vaccine probably does not protect life-long and needs to be boosted. The only available data indicate that 87% of the vaccinated population with three doses had detectable specific antibodies up to 5 years post-vaccination [40].
Taking into consideration the disadvantages of systemic vaccines, some researchers are focused on the development of safe and efficient mucosal vaccines. The advantages of oral vaccines rely on the higher acceptance due to the convenience of applying the vaccine without needles, which saves time and work, and because mucosal vaccines stimulate adaptive immunity at the port of entry of the pathogen preventing pathogen dissemination. In this regard, we investigated in this work whether a subunit oral vaccine for HEV consisting of the purified HEV capsid protein ORF2 administered with IBLPs derived from two different immunomodulatory strains of L. rhamnosus was able to elicit specific humoral and cellular immunity in mice.
LAB are non-pathogenic and generally regarded as safe microorganisms according to the US Food and Drug Administration [41, 42]. Apart from being used for the conservation of food, they have multiple applications and some strains can selectively modulate the immune system [43]. Due to their natural immunoadjuvant activities, in the past 20 years, some LAB, especially Lactococcus lactis and L. plantarum, were used as mucosal delivery vehicles of heterologous antigens and therapeutic molecules [44,45,46]. Recently, the immunogenicity of a recombinant L. lactis expressing the HEV ORF2 protein on its surface was tested in mice. The oral immunization with this recombinant strain resulted in the induction of specific humoral and cellular responses against the capsid antigen [47]. These results encourage the use of LAB as mucosal adjuvants for the development of HEV vaccines. However, genetically modified organisms pose a challenge to human use because of the needed strict control of their attenuation and/or absence of reversion to virulence, their genetic stability and antibiotic resistance [48].
An easier and safer approach of the use of LAB as mucosal adjuvants and delivery vectors was explored in earlier studies by Saluja et al. [23], who demonstrated that the influenza subunit vaccine orally administered with BLP from L. lactis (known as GEM, Gram-positive enhanced matrix) generated an antigen-specific immune response [23]. BLP conserve their cell wall structure, and the peptidoglycan is even more exposed because of the acid-heat depletion of surface proteins and teichoic acids. Peptidoglycan is recognized by the Toll-like receptor 2 (TLR2) and in this way, it could serve as a mucosal adjuvant. In previous studies, we have demonstrated that L. rhamnosus CRL1505 alive or heat-killed is able to increase immunity against infections [26, 49]. L. rhamnosus CRL1505 can modulate immunity by triggering TLR2 signaling in intestinal epithelial cells and antigen-presenting cells [24], and it was demonstrated that the main structural component responsible for the immunomodulatory activity of the CRL1505 is its peptidoglycan [50, 51]. In addition, the CRL1505 strain, when orally administered to mice, is able to improve the production of TNF-α, IL-6, and IFN-γ in the intestine and serum without inducing any detrimental inflammatory effect [26, 49, 52]. This change induced by L. rhamnosus CRL1505 on cytokine’s profiles could support an efficient antigen presentation. Interestingly, the immunomodulatory capacity of L. rhamnosus CRL1505 is not fully shared by other strains of the same or related species or their peptidoglycans [50], indicating that LAB strains have to be carefully selected in order to use those with the highest adjuvant activities as carriers and adjuvants for vaccines.
In this study, we show that orally administered ORF2 protein produced in E. coli under native conditions combined with IBLP obtained either from L. rhamnosus CRL1505 or IBL027 induced humoral antigen-specific immune responses in mice. IBLP1505 elicited a slightly stronger anti-HEV ORF2 IgG response in serum than IBLP027, but the latter also induced specific secretory IgA in the gut lumen, key for mucosal defenses. Considering that the port of entry of HEV is the intestinal epithelial cell, it is desirable that a mucosal vaccination induces protection at this site, apart from generating systemic anti-HEV IgG. Of interest, ORF2+IBLPs experimental vaccines also stimulated cellular immune responses. We found that Peyer’s patches T and B cell populations increased in number, regardless of the IBLPs used, whereas in the spleen, only the B cell population augmented when IBLPs were used as adjuvants. We also evaluated the specific production of IFN-γ, TNF-α, and IL-4 upon stimulation of mononuclear cells isolated from Peyer’s patches and spleen, reflecting mucosal and systemic immune activation, respectively. Elevated levels of IFN-γ, TNF-α, and IL-4 suggested that ORF2 combined with IBLPs induced a mixed Th1/Th2-type adaptive immune response. Both types of IBLPs were able to increase the responsiveness of mononuclear cells to the specific viral antigen (ORF2).
The delivery of mucosal vaccines is more accepted in humans and is more convenient than intramuscular vaccination because it saves time and qualified personal involved in vaccination is not necessary. In addition, the use of mucosal vaccination is especially attractive for animal vaccination, especially for zoonotic infections such as hepatitis E. This study presents evidence on the possible use of recombinant proteins adjuvanted with IBLPs from immunomodulatory lactobacilli for oral vaccination.
References
Nimgaonkar I, Ding Q, Schwartz RE, Ploss A (2018) Hepatitis E virus: advances and challenges. Nat Rev Gastroenterol Hepatol 15:96–110. https://doi.org/10.1038/nrgastro.2017.150
Teshale EH, Hu DJ (2011) Hepatitis E: epidemiology and prevention. World J Hepatol 3:285–291. https://doi.org/10.4254/wjh.v3.i12.285
Kmush B, Wierzba T, Krain L, Nelson K, Labrique AB (2013) Epidemiology of hepatitis E in low- and middle-income countries of Asia and Africa. Semin Liver Dis 33:15–29. https://doi.org/10.1055/s-0033-1338111
Nelson KE, Labrique AB, Kmush BL (2018) Epidemiology of genotype 1 and 2 hepatitis E virus infections. Cold Spring Harb Perspect Med. https://doi.org/10.1101/cshperspect.a031732
Lu L, Li C, Hagedorn CH (2006) Phylogenetic analysis of global hepatitis E virus sequences: genetic diversity, subtypes and zoonosis. Rev Med Virol 16:5–36. https://doi.org/10.1002/rmv.482
Shrestha A, Adhikari A, Bhattarai M, Rauniyar R, Debes JD, Boonstra A, Lama TK, Al Mahtab M, Butt AS, Akbar SMF, Aryal N, Karn S, Manandhar KD, Gupta BP (2017) Prevalence and risk of hepatitis E virus infection in the HIV population of Nepal. Virol J 14:228–227. https://doi.org/10.1186/s12985-017-0899-x
Riveiro-Barciela M, Buti M, Homs M, Campos-Varela I, Cantarell C, Crespo M, Castells L, Tabernero D, Quer J, Esteban R, Rodriguez-Frias F (2014) Cirrhosis, liver transplantation and HIV infection are risk factors associated with hepatitis E virus infection. PLoS One 9:e103028. https://doi.org/10.1371/journal.pone.0103028
Geng Y, Zhao C, Geng K, Wang C, Wang X, Liu H, Wang Y (2019) High seroprevalence of hepatitis E virus in rabbit slaughterhouse workers. Transbound Emerg Dis 66:1085–1089. https://doi.org/10.1111/tbed.13130
Sahli R, Fraga M, Semela D, Moradpour D, Gouttenoire J (2019) Rabbit HEV in immunosuppressed patients with hepatitis E acquired in Switzerland. J Hepatol 70:1023–1025. https://doi.org/10.1016/j.jhep.2019.01.025
Lee GH, Tan BH, Teo EC, Lim SG, Dan YY, Wee A, Aw PP, Zhu Y, Hibberd ML, Tan CK, Purdy MA, Teo CG (2016) Chronic infection with camelid hepatitis E virus in a liver transplant recipient who regularly consumes camel meat and milk. Gastroenterology 150:355–357. https://doi.org/10.1053/j.gastro.2015.10.048
Shimizu K, Hamaguchi S, Ngo CC, Li TC, Ando S, Yoshimatsu K, Yasuda SP, Koma T, Isozumi R, Tsuda Y, Fujita H, Pham TT, Le MQ, Dang AD, Nguyen TQ, Yoshida LM, Ariyoshi K, Arikawa J (2016) Serological evidence of infection with rodent-borne hepatitis E virus HEV-C1 or antigenically related virus in humans. J Vet Med Sci 78:1677–1681. https://doi.org/10.1292/jvms.16-0200
Nganou-Makamdop K, van Roosmalen ML, Audouy SA, van Gemert GJ, Leenhouts K, Hermsen CC, Sauerwein RW (2012) Bacterium-like particles as multi-epitope delivery platform for Plasmodium berghei circumsporozoite protein induce complete protection against malaria in mice. Malar J 11:50. https://doi.org/10.1186/1475-2875-11-50
Van Braeckel-Budimir N, Haijema BJ, Leenhouts K (2013) Bacterium-like particles for efficient immune stimulation of existing vaccines and new subunit vaccines in mucosal applications. Front Immunol 4:282. https://doi.org/10.3389/fimmu.2013.00282
Heine SJ, Franco-Mahecha OL, Chen X, Choudhari S, Blackwelder WC, van Roosmalen ML, Leenhouts K, Picking WL, Pasetti MF (2015) Shigella IpaB and IpaD displayed on L. lactis bacterium-like particles induce protective immunity in adult and infant mice. Immunol Cell Biol 93:641–652. https://doi.org/10.1038/icb.2015.24
Audouy SA, van Roosmalen ML, Neef J, Kanninga R, Post E, van Deemter M, Metselaar H, van Selm S, Robillard GT, Leenhouts KJ, Hermans PW (2006) Lactococcus lactis GEM particles displaying pneumococcal antigens induce local and systemic immune responses following intranasal immunization. Vaccine 24:5434–5441. https://doi.org/10.1016/j.vaccine.2006.03.054
Audouy SA, van Selm S, van Roosmalen ML, Post E, Kanninga R, Neef J, Estevao S, Nieuwenhuis EE, Adrian PV, Leenhouts K, Hermans PW (2007) Development of lactococcal GEM-based pneumococcal vaccines. Vaccine 25:2497–2506. https://doi.org/10.1016/j.vaccine.2006.09.026
van Roosmalen ML, Kanninga R, El Khattabi M, Neef J, Audouy S, Bosma T, Kuipers A, Post E, Steen A, Kok J, Buist G, Kuipers OP, Robillard G, Leenhouts K (2006) Mucosal vaccine delivery of antigens tightly bound to an adjuvant particle made from food-grade bacteria. Methods 38:144–149. https://doi.org/10.1016/j.ymeth.2005.09.015
Zhu B, Liu J, Fu Y, Zhang B, Mao Y (2018) Spatio-temporal epidemiology of viral hepatitis in China (2003-2015): implications for prevention and control policies. Int J Environ Res Public Health. https://doi.org/10.3390/ijerph15040661
Saluja V, Amorij JP, van Roosmalen ML, Leenhouts K, Huckriede A, Hinrichs WL, Frijlink HW (2010) Intranasal delivery of influenza subunit vaccine formulated with GEM particles as an adjuvant. AAPS J 12:109–116. https://doi.org/10.1208/s12248-009-9168-2
de Haan A, Haijema BJ, Voorn P, Meijerhof T, van Roosmalen ML, Leenhouts K (2012) Bacterium-like particles supplemented with inactivated influenza antigen induce cross-protective influenza-specific antibody responses through intranasal administration. Vaccine 30:4884–4891. https://doi.org/10.1016/j.vaccine.2012.04.032
Keijzer C, Haijema BJ, Meijerhof T, Voorn P, de Haan A, Leenhouts K, van Roosmalen ML, van Eden W, Broere F (2014) Inactivated influenza vaccine adjuvanted with bacterium-like particles induce systemic and mucosal influenza A virus specific T-cell and B-cell responses after nasal administration in a TLR2 dependent fashion. Vaccine 32:2904–2910. https://doi.org/10.1016/j.vaccine.2014.02.019
Rigter A, Widjaja I, Versantvoort H, Coenjaerts FE, van Roosmalen M, Leenhouts K, Rottier PJ, Haijema BJ, de Haan CA (2013) A protective and safe intranasal RSV vaccine based on a recombinant prefusion-like form of the F protein bound to bacterium-like particles. PLoS One:8. https://doi.org/10.1371/journal.pone.0071072
Saluja V, Visser MR, van Roosmalen ML, Leenhouts K, Huckriede A, Hinrichs WL, Frijlink HW (2010) Gastro-intestinal delivery of influenza subunit vaccine formulation adjuvanted with Gram-positive enhancer matrix (GEM) particles. Eur J Pharm Biopharm 76:470–474. https://doi.org/10.1016/j.ejpb.2010.08.003
Villena J, Chiba E, Vizoso-Pinto MG, Tomosada Y, Takahashi T, Ishizuka T, Aso H, Salva S, Alvarez S, Kitazawa H (2014) Immunobiotic Lactobacillus rhamnosus strains differentially modulate antiviral immune response in porcine intestinal epithelial and antigen presenting cells. BMC Microbiol 14:126–114. https://doi.org/10.1186/1471-2180-14-126
Laino J, Villena J, Kanmani P, Kitazawa H (2016) Immunoregulatory effects triggered by lactic acid bacteria exopolysaccharides: new insights into molecular interactions with host cells. Microorganisms. https://doi.org/10.3390/microorganisms4030027
Villena J, Vizoso-Pinto MG, Kitazawa H (2016) Intestinal innate antiviral immunity and immunobiotics: beneficial effects against Rotavirus infection. Front Immunol 7:563. https://doi.org/10.3389/fimmu.2016.00563
Barbieri N, Herrera M, Salva S, Villena J, Alvarez S (2017) Lactobacillus rhamnosus CRL1505 nasal administration improves recovery of T-cell mediated immunity against pneumococcal infection in malnourished mice. Benefic Microbes 8:393–405. https://doi.org/10.3920/BM2016.0152
Garcia-Castillo V, Zelaya H, Ilabaca A, Espinoza-Monje M, Komatsu R, Albarracin L, Kitazawa H, Garcia-Cancino A, Villena J (2018) Lactobacillus fermentum UCO-979C beneficially modulates the innate immune response triggered by Helicobacter pylori infection in vitro. Benefic Microbes 9:829–841. https://doi.org/10.3920/BM2018.0019
Vizoso-Pinto MG, Saavedra L, Hebert EM, Raya Tonetti F, Albarracin L, Alvarez S, Kitazawa H, Villena J (2017) Draft genome sequence of immunobiotic Lactobacillus rhamnosus strain IBL027, a potential adjuvant for mucosal vaccine development. Genome Announc 5:e01268–e01217. https://doi.org/10.1128/genomeA.01268-17
Vizoso Pinto MG, Pfrepper KI, Janke T, Noelting C, Sander M, Lueking A, Haas J, Nitschko H, Jaeger G, Baiker A (2010) A systematic approach for the identification of novel, serologically reactive recombinant Varicella-Zoster Virus (VZV) antigens. Virol J 7:165. https://doi.org/10.1186/1743-422X-7-165
Salva S, Villena J, Alvarez S (2010) Immunomodulatory activity of Lactobacillus rhamnosus strains isolated from goat milk: impact on intestinal and respiratory infections. Int J Food Microbiol 141:82–89. https://doi.org/10.1016/j.ijfoodmicro.2010.03.013
Osterman A, Vizoso Pinto MG, Haase R, Nitschko H, Jager S, Sander M, Motz M, Mohn U, Baiker A (2012) Systematic screening for novel, serologically reactive hepatitis E virus epitopes. Virol J 9:28. https://doi.org/10.1186/1743-422X-9-28
Schnell MA, Hardy C, Hawley M, Propert KJ, Wilson JM (2002) Effect of blood collection technique in mice on clinical pathology parameters. Hum Gene Ther 13:155–161. https://doi.org/10.1089/10430340152712700
Aggarwal R, Shukla R, Jameel S, Agrawal S, Puri P, Gupta VK, Patil AP, Naik S (2007) T-cell epitope mapping of ORF2 and ORF3 proteins of human hepatitis E virus. J Viral Hepat 14:283–292. https://doi.org/10.1111/j.1365-2893.2006.00796.x
Wang H, Zhang W, Gu H, Chen W, Zeng M, Ji C, Song R, Zhang G (2017) Identification and characterization of two linear epitope motifs in hepatitis E virus ORF2 protein. PLoS One. https://doi.org/10.1371/journal.pone.0184947
Zhang H, Dai X, Shan X, Meng J (2008) The Leu477 and Leu613 of ORF2-encoded protein are critical in forming neutralization antigenic epitope of hepatitis E virus genotype 4. Cell Mol Immunol 5:447–456. https://doi.org/10.1038/cmi.2008.56
Behloul N, Wen J, Dai X, Dong C, Meng J (2015) Antigenic composition and immunoreactivity differences between HEV recombinant capsid proteins generated from different genotypes. Infect Genet Evol 34:211–220. https://doi.org/10.1016/j.meegid.2015.06.026
Zhao Q, Zhang J, Wu T, Li SW, Ng MH, Xia NS, Shih JW (2013) Antigenic determinants of hepatitis E virus and vaccine-induced immunogenicity and efficacy. J Gastroenterol 48:159–168. https://doi.org/10.1007/s00535-012-0701-1
Gu G, Huang H, Zhang L, Bi Y, Hu Y, Zhou YH (2015) Hepatitis E virus seroprevalence in pregnant women in Jiangsu, China, and postpartum evolution during six years. BMC Infect Dis 15:560. https://doi.org/10.1186/s12879-015-1308-y
Zhang J, Zhang XF, Huang SJ, Wu T, Hu YM, Wang ZZ, Wang H, Jiang HM, Wang YJ, Yan Q, Guo M, Liu XH, Li JX, Yang CL, Tang Q, Jiang RJ, Pan HR, Li YM, Shih JW, Ng MH, Zhu FC, Xia NS (2015) Long-term efficacy of a hepatitis E vaccine. N Engl J Med 372:914–922. https://doi.org/10.1056/NEJMoa1406011
Salminen S, von Wright A, Morelli L, Marteau P, Brassart D, de Vos WM, Fonden R, Saxelin M, Collins K, Mogensen G, Birkeland SE, Mattila-Sandholm T (1998) Demonstration of safety of probiotics -- a review. Int J Food Microbiol 44:93–106. https://doi.org/10.1016/s0168-1605(98)00128-7
Fontana L, Bermudez-Brito M, Plaza-Diaz J, Munoz-Quezada S, Gil A (2013) Sources, isolation, characterisation and evaluation of probiotics. Br J Nutr 109(Suppl 2):35–50. https://doi.org/10.1017/S0007114512004011
Zago M, Scaltriti E, Bonvini B, Fornasari ME, Penna G, Massimiliano L, Carminati D, Rescigno M, Giraffa G (2017) Genomic diversity and immunomodulatory activity of Lactobacillus plantarum isolated from dairy products. Benefic Microbes 8:597–604. https://doi.org/10.3920/BM2016.0223
Cortes-Perez NG, Kharrat P, Langella P, Bermudez-Humaran LG (2009) Heterologous production of human papillomavirus type-16 L1 protein by a lactic acid bacterium. BMC Res Notes 2:167. https://doi.org/10.1186/1756-0500-2-167
Leblanc JG, Aubry C, Cortes-Perez NG, de Moreno de Leblanc A, Vergnolle N, Langella P, Azevedo V, Chatel JM, Miyoshi A, Bermudez-Humaran LG (2013) Mucosal targeting of therapeutic molecules using genetically modified lactic acid bacteria: an update. FEMS Microbiol Lett. https://doi.org/10.1111/1574-6968.12159
Marelli B, Perez AR, Banchio C, de Mendoza D, Magni C (2011) Oral immunization with live Lactococcus lactis expressing rotavirus VP8 subunit induces specific immune response in mice. J Virol Methods 175:28–37. https://doi.org/10.1016/j.jviromet.2011.04.011
Gao S, Li D, Liu Y, Zha E, Zhou T, Yue X (2015) Oral immunization with recombinant hepatitis E virus antigen displayed on the Lactococcus lactis surface enhances ORF2-specific mucosal and systemic immune responses in mice. Int Immunopharmacol 24:140–145. https://doi.org/10.1016/j.intimp.2014.10.032
Leunda A, Baldo A, Goossens M, Huygen K, Herman P, Romano M (2014) Novel GMO-based vaccines against tuberculosis: state of the art and biosafety considerations. Vaccines (Basel) 2:463–499. https://doi.org/10.3390/vaccines2020463
Kolling Y, Salva S, Villena J, Marranzino G, Alvarez S (2015) Non-viable immunobiotic Lactobacillus rhamnosus CRL1505 and its peptidoglycan improve systemic and respiratory innate immune response during recovery of immunocompromised-malnourished mice. Int Immunopharmacol 25:474–484. https://doi.org/10.1016/j.intimp.2015.02.006
Kolling Y, Salva S, Villena J, Alvarez S (2018) Are the immunomodulatory properties of Lactobacillus rhamnosus CRL1505 peptidoglycan common for all lactobacilli during respiratory infection in malnourished mice? PLoS One. https://doi.org/10.1371/journal.pone.0194034
Clua P, Kanmani P, Zelaya H, Tada A, Kober A, Salva S, Alvarez S, Kitazawa H, Villena J (2017) Peptidoglycan from immunobiotic Lactobacillus rhamnosus improves resistance of infant mice to respiratory syncytial viral infection and secondary pneumococcal pneumonia. Front Immunol 8:948. https://doi.org/10.3389/fimmu.2017.00948
Villena J, Salva S, Núñez M, Corzo J, Tolaba R, Faedda J, Font G, Alvarez S (2012) Probiotics for everyone! The novel immunobiotic Lactobacillus rhamnosus CRL1505 and the beginning of social probiotic programs in Argentina. doi:https://doi.org/10.6000/1927-3037/2012.01.03.05
Acknowledgments
We thank Dr. Roque Carrero Valenzuela for sharing his facilities with us.
Funding
This study received financial support from the Agencia Nacional de Ciencia y Técnica (PICT-2016-0853 and PICT-2016-0410), CONICET (PIP057), and Fundación Allende.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Statement on the Welfare of Animals
This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the Guidelines for Animal Experimentation of the Reference Centre for Lactobacilli (CERELA-CONICET, Tucuman, Argentina). The CERELA Institutional Animal Care and Use Committee prospectively approved this research under the protocol BIOT-CRL-17.
Conflict of Interest
The authors declare that they have no conflict of interest. LA, FRT, and MM hold CONICET scholarships. SGA, JV, and MGVP are members of CONICET.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Arce, L.P., Raya Tonetti, M.F., Raimondo, M.P. et al. Oral Vaccination with Hepatitis E Virus Capsid Protein and Immunobiotic Bacterium-Like Particles Induce Intestinal and Systemic Immunity in Mice. Probiotics & Antimicro. Prot. 12, 961–972 (2020). https://doi.org/10.1007/s12602-019-09598-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12602-019-09598-7