Abstract
Synbiotic (probiotic bacteria and prebiotic) has beneficial effects on the gastrointestinal tract. This study was designed to investigate the effect of synbiotic supplementation on the growth of mild to moderate failure to thrive (FTT) children. A randomized, triple-blind, placebo-controlled trial was conducted involving 80 children aged 2–5 years with mild to moderate FTT, who were assigned at random to receive synbiotic supplementation (109 colony-forming units) or placebo for 30 days. The weights, height, and BMI were recorded in a structured diary, and the questionnaires were completed to monitor the numbers of infection episodes, gastrointestinal problems, admission to hospital, and appetite improvement during the study. Sixty-nine children completed the study. There were no differences in the demographic characteristic between the two groups. The mean weight was similar at baseline. After 30 days of intervention, the mean weight of the participants in the synbiotic group increased significantly than those in the placebo group (600 ± 37 vs. 74 ± 32 g/month P 0.000). BMI changes in synbiotic and placebo group were 0.44 and 0.07 kg/m2, and that the differences among the two groups were significant.(P 0.045) Furthermore, the height increment in synbiotic and placebo group was 0.41 and 0.37 cm respectively with no significant difference (P 0.761). Administration of 30-day synbiotic supplementation may significantly improve weight and BMI in Iranian children with mild to moderate FTT, but there is no effect on the height in this study. Further studies should be designed to found out the effect of synbiotic on growth parameters in undernourished and well-nourished children.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Malnutrition in children is still a major public health issue across the world, particularly in developing countries. According to the 2011 WHO Report, 27% of children under 5 years old are underweight [1].
In Iran as a developing country, 15.7% of children are underweight and 18.9% are stunted [2]. Under nutrition is a major risk factor of mortality and morbidity among the children under 5 years [3]. Failure to thrive (FTT) patients may deal with recurrent gastrointestinal and respiratory tract infections, cognitive defects, developmental delay, and emotional and socioeconomic problems [4]. So, it can cause a heavy economic burden on the health system [5].
Adequate nutrition is important for optimal growth and development [6]. Insufficient caloric intake and nutrient absorption are the common causes of FTT [7]. Macronutrient and micronutrients deficiencies through different ways such as poor nutrition and infectious disorders are the major cause of under nutrition [3, 8]. So prevention and treatment of this problem may be of value. Therefore, finding an effective, safe, and available treatment for weight gain in children with malnutrition is important.
Nowadays, there is a trend in consumption of probiotics for weight gain because of their beneficial effects on micronutrient and macronutrient absorption [9]. It is shown that widespread ingestion of probiotics may promote weight gain by changing the intestinal flora [10].
The other beneficial effects of probiotics is on infectious diarrhea and dysentery [11, 12], antibiotic associated diarrhea [13], upper respiratory infections [14, 15], necrotizing enterocolitis in very low birth weight infants [16,17,18], irritable bowel syndrome (IBS) [19], childhood atopy [20], colic [21], anemia [22] and children growth pattern [23].
Probiotics are live microorganisms that when administered in sufficient amounts may cause a health benefit on the host [24]. They work by regulating the immune system, inhibiting the pathogen adherent to gut epithelium, and improving nutrient absorption [9].
Prebiotics are nondigestible substances that provide a beneficial physiological effect for the host by selectively stimulating the favorable growth or activity of a limited number of indigenous bacteria. Prebiotics increase the absorption of minerals and trace elements [25].
The combination of probiotics and prebiotics named synbiotics exerts a beneficial effect beyond their own effect [26].
Because of the beneficial effects of synbiotics on nutrient absorption and immune system, different studies have evaluated the effects of synbiotic supplementation on growth parameters that the results were conflicting, so this study was established to evaluate the growth of undernourished children consuming a synbiotics or placebo sachet with weight, height, and BMI as the primary outcomes. As secondary outcomes, the study evaluated infectious episodes and gastrointestinal (GI) problems as well as the appetite improvement.
Materials and Methods
This study was a triple-blind, placebo-controlled, randomized nutritional intervention study conducted in Khatamolanbia Clinic, Shahid Sadoughi University of Medical Sciences, Yazd, Iran, from 2016 to 2017. Eighty children of age 24 to 59 months with mild to moderate undernourishment who were normal on the other parts of physical were recruited. The severity of malnutrition was determined according to the Gomez classification that mild, moderate, and severe status has been equivalent to 75–90%, 60–74%, and less than 60% of standard weight, respectively [27]. From a total of 80 children with mild to moderate FTT, 11 were excluded that the patients according to the exclusion criteria (6), loss of follow-up (3), and poor compliance (2).
Exclusion criteria were antibiotic consumption 2 weeks prior the study, diarrhea at the time of study, any congenital abnormality or chromosomal disorder, ICU admission more than 3 days, birth weight under 2500 g, and gestational age under 37 weeks.
The study protocol was approved by the research and ethical committee of Shahid Sadoughi University of Medical Sciences. Written informed consent was obtained from the patients’ parents or guardians.
The patients were randomly divided in two groups: synbiotic and control, using a random table numbers. The patients in the two groups were matched by age, sex, and weight. The case group was given a synbiotic sachet (Kidilact) which contained 109 colony-forming unit (CFU) of 7 effective strains including Lactobacillus acidophilus, Lactobacillus rhamnosus, Lactobacillus bulgaricus, Lactobacillus casei, Bifidobacterium infantis, Bifidobacterium breve, and Streptococcus thermophilus, as probiotics, and a prebiotic Fructooligosaccharides 1 g daily for 30 days. The children in the control group were given a placebo sachet daily which had a similar taste, smell, and appearance with synbiotic sachet and were supplied by the same company. The sachets were in the form of a powder reconstituted by parents with 5–10 ml of water and administered orally as a suspension. Products were manufactured according to current good manufacturing practices, and were packaged and coded at the Zist Takhmir Company. The pediatrician, parents, pharmacy, and statistical analyzer were not aware of the codes of synbiotic or placebo. The parents received the sachets at first visit by clinic pharmacy.
All patients were visited by a pediatrician at first. Physical examination and anthropometric measurements were recorded at first by the pediatrician as well as after 30 days of intervention by the same pediatrician. The children were weighed (measured with a Seca [Unicap, Australia] digital scale with an accuracy of 0.1 kg) and height measured (measured with a Somatometer [Stanley Mabo, Depose, France] with an accuracy of 0.1 cm) in light clothing without shoes using a digital standing scale by the same physician. Body mass index (BMI) was calculated as weight (kilograms) divided by height (meters) squared. In this study, we measured average of weight gain and other growth parameters in two groups of case and control to compare weight gain velocity (weight gain as gr/month), height increment velocity (height increment as cm/month), and BMI velocity (BMI difference as kg/m2/month) during the study.
Compliance of intake was assessed by pediatric assistant every week by telephone call. Furthermore, the parents were asked to return the empty boxes of sachets for assuring the compliance.
Moreover, numbers of upper respiratory disorders, gastrointestinal problems, antibiotic consumption, and appetite improvement and any adverse reaction to the medication were assessed weekly by telephone call.
Considering confidence level of 95% and a power of 80%, the sample size was estimated as minimum of 31 patients in each group. Results were reported as mean ± standard deviation (SD) for the quantitative variables. The groups were compared using the Student’s t test and the chi-square test (or Fisher’s exact test if required) for the categorical variables.
All the statistical analyses were performed using SPSS version 19.0 (SPSS Inc., Chicago, IL, USA). A P value < 0.05 was considered as statistically significant.
After finishing the statistical analysis, breaking the codes were done.
Results
Of 80 FTT children, 69 patients met the inclusion criteria and completed the study.
Forty one (59%) were females and 28 (41%) were males. Of these, 37 patients (24 girls, 13 boys) were in the synbiotic group and 32 patients (17 girls, 15 boys) were in the placebo group.
Age range was between 24 and 59 months with a median of 43 months old. Fifty six of 69 patients (81%) were mild under nutrition and 13 (19%) were moderate under nutrition.
The demographic characteristics of the 69 patients who completed the study are presented in Table 1. There was no significant difference in the weight, age, gender, and mode of delivery distribution among the two groups.
The mean initial children weight was 12.41 ± 1.59 kg (range 8.5–16 kg). While mean initial weight and follow-up weight, in the synbiotic group, were 12.05 ± 1.63 kg and 12.65 ± 1.66 kg, respectively. The mean weight of children in placebo group at the first and at the end of intervention was 12.53 ± 1.63 kg and 12.60 ± 1.51 kg respectively.
The average weight velocity after treatment was found to be significantly higher in the case group as compared to the control group (600 g vs. 74 g) (P 0.000) (Table 2). On further analysis, it was found that the effect of synbiotics on weight gain was not confined to a particular age group or malnutrition subgroups.
BMI in the case group at the first and at the end of this study was 13.68 ± .94 and 14.12 ± 1.11 respectively and in the control group was 13.82 ± .74 and 13.89 ± .63 respectively. BMI changes in the case group were statistically significant than the control group (P 0.045).
The mean height in the case group before and after treatment was 94.89 ± 5.95 and 95.30 ± 5.02 cm and in the control group was 95.28 ± 6.40 and 95.65 ± 5.97 respectively. The mean height increment in the synbiotic and control group was not statistically different (0.41 vs. 0.37 cm, P 0.761).
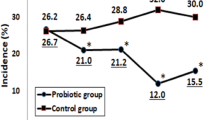
No difference on infectious disease episodes was seen in the two groups after 30 days of synbiotic intervention, whereas infection episodes in synbiotic and placebo group were 7 (20%) and 8 (27.58%) respectively (P 0.845).
GI problems occurred in the synbiotic and placebo group in 3 (9.37%) and 5 (13.51%) children respectively (P 0.575).
No statistical significance was observed between the synbiotic and placebo groups in antibiotic consumption (4 vs. 6 respectively) (P 0.52) (Table 3).
Sixteen patients (24.51%) reported appetite improvement, whereas 7 (20.91%) were in the synbiotic group and 9(28.12%) were in the control group. The differences among the two groups were not statistically significant (P 0.211) (Table 3).
Discussion
The present study evaluated the efficacy of 30-days administration of synbiotic supplementation on growth parameters in mild to moderate FTT children. We found that synbiotics can be beneficial in terms of weight and BMI gain, particularly in children who are under-nourished after 30 days of intervention, with a similar efficacy to other clinical studies. [15, 22, 28] However, synbiotic did not have a significant effect on frequency of infection and GI problems.
The product used in this study was Kidilact sachet which contained Lactobacillus acidophilus, Lactobacillus rhamnosus, Lactobacillus bulgaricus, Lactobacillus casei, Bifidobacterium infantis, Bifidobacterium breve, and Streptococcus thermophilus, as probiotics, and Fructooligosaccharides as a prebiotic. The reasons we chose this product were that it was well tolerated in children and also because of probiotic Bifidobacterium and Lactobacillus major benefits. As in a study by Million et al. [29], they concluded that different Lactobacillus species are associated with different effects on weight that are host-specific. They showed that Lactobacillus acidophilus administration causes a significant weight gain in human and in animal, while Lactobacillus gasseri was associated with weight loss both in obese humans and in animals. Also in our study, the synbiotic sachet consisted of Lactobacillus acidophilus but not Lactobacillus gasseri.
The effect of synbiotic supplementation on weight, height, and BMI that are important anthropometric parameters for child growth was reported in different studies. In various studies, it is shown that probiotics may promote weight gain [15, 28], while in the other studies, weight loss [30, 31] was observed as a result of synbiotic consumption. On the other hand, some other studies failed to show efficacy of probiotics and synbiotics on weight [38,39,40.] The results of these studies were different based on obesity or malnutrition of the cases. Hence, in a study by Ipar [32], they concluded that synbiotic can help obese children to lose weight. Also, age of the participants has an important role in the effectiveness of probiotics, while in a study by Dror et al. [23], synbiotics caused weight loss in adults and weight gain in children. The different effects of synbiotics in the studies may be due to variation in the dose and strains of probiotics, duration of intervention, characteristics of participants, statistical analyses, and different geographic regions.
It is proven that the gut senses alterations in nutrient availability and subsequently modulates the nutrient absorption. [33, 34] Also, the different effect of probiotics on obese and undernourished children may be due to gut microbiota differences between overweight and lean children [35]. Furthermore, the different gut microbiota may influence caloric intake, intestinal absorption, and energy balance [34].
In this study, we reported the positive effect of synbiotic on weight and BMI which was in accordance with the studies conducted by He [15], Famouri [28], Kerac [36], and Saran [37] on undernourished children and contrary to the studies by Gil-Compos [38], Gibson [39], and Scalabrin [40] that reported no effect of synbiotic consumption on weight gain in well-nourished children [41, 42].
It seems that different gut microbiota composition in undernourished and well-nourished children and also living in developed or developing countries may play an important role on the effects that synbiotic may have [43, 44].
In developed countries, synbiotic supplementation had no effect on weight gain [38,39,40,41,42, 45]. However, it had a significant effect on weight promotion in children living in developing countries [15, 22, 28, 37, 46].
Moreover, the composition of gut microbiota may vary due patient’s age [47], while in the studies that the subjects were infants or individuals younger than 7 months old, the effect of synbiotics on weight gain has not been established [38,39,40,41,42, 45]. But in the studies conducted on children with 1–5 years of age, the synbiotics supplementation had a significant effect on weight improvement [15, 22, 28, 37, 46].
Our study was done in Iran as a developing country and was conducted on undernourished children aged 2–5 years, so the results of our study were in accordance with the other studies that were done on undernourished children in developing countries.
In our study, we did not find any effect of synbiotics on height, while in studies by He [15], Saran [37], and Silva [48], they found a significant difference in height of the children. In He et al. [15], the children in the probiotic group had a change in height-for-age z score at 9 months of supplementation, and in Saran et al., the children in the probiotic group grew more in length compared to the control group, after 6 months. In Silva et al.’s study, height increment was shown after 101 days of probiotics consumption. In our study we just used 30 days of synbiotic, so short duration of synbiotic may not conclude the effectiveness of synbiotic on height.
Although it is described that lactobacillus/Bifidobacterium improved protection against rotavirus diarrhea and infection in pediatrics [49], in this study we have not seen any effect of synbiotic on prevention of infection episodes and GI problems.
The limitation of our study was exclusion of sever malnourished children, since they need additional treatments. The other limitation of this study was that we did not assess dietary intake of participants along the study and were therefore unable to control for this variable.
Conclusion
In conclusion, this study provides evidence that short course of synbiotic supplementation may cause weight and BMI gain in mild to moderate undernourished children.
Further studies are needed to better understand the mechanisms of synbiotics’ effects. It is recommended to evaluate gut microbiota and also the levels of micronutrient and macronutrient in plasma before and after the treatment.
References
on HIV/AIDS. JUNP, UNICEF (2010) Children and AIDS: Fifth Stocktaking Report, 2010. UNICEF
UNICEF (2008) The state of the world’s children 2009: maternal and newborn health. Unicef
Black RE, Allen LH, Bhutta ZA, Caulfield LE, de Onis M, Ezzati M, Mathers C, Rivera J (2008) Maternal and child undernutrition: global and regional exposures and health consequences. Lancet 371:243–260
Larson-Nath C, Biank VF (2016) Clinical review of failure to thrive in pediatric patients. Pediatr Ann 45:e46–e49
Gahagan S (2006) Failure to thrive: a consequence of undernutrition. Pediatr Rev 27:e1–e11
Matsuyama M, Harb T, David M, Davies PSW, Hill RJ (2017) Effect of fortified milk on growth and nutritional status in young children: a systematic review and meta-analysis. Public Health Nutr 20:1214–1225
Homan GJ (2016) Failure to thrive: a practical guide. Am Fam Physician 94:295–299
Victora CG, Adair L, Fall C, Hallal PC, Martorell R, Richter L, Sachdev HS, Maternal and Child Undernutrition Study Group (2008) Maternal and child undernutrition: consequences for adult health and human capital. Lancet 371:340–357
Scholz-Ahrens KE, Ade P, Marten B, Weber P, Timm W, Aςil Y, Glüer CC, Schrezenmeir J̈ (2007) Prebiotics, probiotics, and synbiotics affect mineral absorption, bone mineral content, and bone structure. J Nutr 137:838S–846S
Raoult D (2008) Human microbiome: take-home lesson on growth promoters? Nature 454:690–691
Sharif A, Kashani HH, Nasri E, Soleimani Z, Sharif MR (2017) The role of probiotics in the treatment of dysentery: a randomized double-blind clinical trial. Probiotics Antimicrob Proteins 9:380–385
Guarino A, Guandalini S, Lo Vecchio A (2015) Probiotics for prevention and treatment of diarrhea. J Clin Gastroenterol 49:S37–S45
Szajewska H, Kotowska M, Mrukowicz JZ, Arma′nska M, Mikolajczyk W (2001) Efficacy of lactobacillus GG in prevention of nosocomial diarrhea in infants. J Pediatr 138:361–365
Wolvers D, Antoine J-M, Myllyluoma E, Schrezenmeir J, Szajewska H, Rijkers GT (2010) Guidance for substantiating the evidence for beneficial effects of probiotics: prevention and management of infections by probiotics. J Nutr 140(3):698S–712S
He M, Yang YX, Han H, Men JH, Bian LH, Wang GD (2005) Effects of yogurt supplementation on the growth of preschool children in Beijing suburbs. Biomed Environ Sci: BES 18:192–197
Ganguli K, Walker WA (2011) Probiotics in the prevention of necrotizing enterocolitis. J Clin Gastroenterol 45:S133–S138
AlFaleh K, Anabrees J (2014) Probiotics for prevention of necrotizing enterocolitis in preterm infants. Evid Based Child Health: Cochrane Rev J 9:584–671
Barclay AR, Stenson B, Simpson JH, Weaver LT, Wilson DC (2007) Probiotics for necrotizing enterocolitis: a systematic review. J Pediatr Gastroenterol Nutr 45:569–576
Lee BJ, Bak Y-T (2011) Irritable bowel syndrome, gut microbiota and probiotics. J Neurogastroenterol Motil 17:252–266
Kalliomäki M, Salminen S, Poussa T, Isolauri E (2007) Probiotics during the first 7 years of life: a cumulative risk reduction of eczema in a randomized, placebo-controlled trial. J Allergy Clin Immunol 119:1019–1021
Ahanchian H, Javid A (2017) Probiotics and Synbiotics for Management of Infantile Colic. In: Nutritional modulators of pain in the aging population. Elsevier, pp 135–140
Sazawal S, Dhingra U, Hiremath G, Sarkar A, Dhingra P, Dutta A, Menon VP, Black RE (2010) Effects of Bifidobacterium lactis HN019 and prebiotic oligosaccharide added to milk on iron status, anemia, and growth among children 1 to 4 years old. J Pediatr Gastroenterol Nutr 51:341–346
Dror T, Dickstein Y, Dubourg G, Paul M (2017) Microbiota manipulation for weight change. Microb Pathog 106:146–161
Pineiro M, Stanton C (2007) Probiotic bacteria: legislative framework—requirements to evidence basis. J Nutr 137:850S–853S
Roberfroid M, Gibson GR, Hoyles L, McCartney AL, Rastall R, Rowland I, Wolvers D, Watzl B, Szajewska H, Stahl B, Guarner F, Respondek F, Whelan K, Coxam V, Davicco MJ, Léotoing L, Wittrant Y, Delzenne NM, Cani PD, Neyrinck AM, Meheust A (2010) Prebiotic effects: metabolic and health benefits. Br J Nutr 104:S1–S63
Schrezenmeir J, de Vrese M (2001) Probiotics, prebiotics, and synbiotics—approaching a definition. Am J Clin Nutr 73:361s–364s
Waterlow JC (1972) Classification and definition of protein-calorie malnutrition. Br Med J 3:566–569
Famouri F, Khoshdel A, Golshani A et al (2014) Effects of synbiotics on treatment of children with failure to thrive: a triple blind placebo-controlled trial. J Res Med Sci 19:1046
Million M, Angelakis E, Paul M, Armougom F, Leibovici L, Raoult D (2012) Comparative meta-analysis of the effect of Lactobacillus species on weight gain in humans and animals. Microb Pathog 53:100–108
Nagata S, Chiba Y, Wang C, Yamashiro Y (2017) The effects of the Lactobacillus casei strain on obesity in children: a pilot study. Benefic Microbes 8:535–543
Cakir M, Isbilen AA, Eyupoglu I et al (2017) Effects of long-term synbiotic supplementation in addition to lifestyle changes in children with obesity-related non-alcoholic fatty liver disease. Turk J Gastroenterol 28:377–384
Ipar N, Aydogdu SD, Yildirim GK, Inal M, Gies I, Vandenplas Y, Dinleyici EC (2015) Effects of synbiotic on anthropometry, lipid profile and oxidative stress in obese children. Benefic Microbes 6:775–781
Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI (2006) An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444:1027–1031
Jumpertz R, Le DS, Turnbaugh PJ et al (2011) Energy-balance studies reveal associations between gut microbes, caloric load, and nutrient absorption in humans. Am J Clin Nutr 94:58–65
Koleva P, Bridgman S, Kozyrskyj A (2015) The infant gut microbiome: evidence for obesity risk and dietary intervention. Nutrients 7:2237–2260
Kerac M, Bunn J, Seal A, Thindwa M, Tomkins A, Sadler K, Bahwere P, Collins S (2009) Probiotics and prebiotics for severe acute malnutrition (PRONUT study): a double-blind efficacy randomised controlled trial in Malawi. Lancet 374:136–144
Saran S, Gopalan S, Krishna TP (2002) Use of fermented foods to combat stunting and failure to thrive. Nutrition 18:393–396
Gil-Campos M, López MÁ, Rodriguez-Benítez MV et al (2012) Lactobacillus fermentum CECT 5716 is safe and well tolerated in infants of 1--6 months of age: a randomized controlled trial. Pharmacol Res 65:231–238
Gibson RA, Barclay D, Marshall H, Moulin J, Maire JC, Makrides M (2009) Safety of supplementing infant formula with long-chain polyunsaturated fatty acids and Bifidobacterium lactis in term infants: a randomised controlled trial. Br J Nutr 101:1706–1713
Scalabrin DM, Johnston WH, Hoffman DR, P’Pool VL, Harris CL, Mitmesser SH (2009) Growth and tolerance of healthy term infants receiving hydrolyzed infant formulas supplemented with Lactobacillus rhamnosus GG: randomized, double-blind, controlled trial. Clin Pediatr 48:734–744
Huet F, Lachambre E, Beck L, van Egroo LD, Sznajder M (2006) Evaluation of a formula with low protein content and supplemented with probiotic agents after breast milk weaning. Arch Pediatr 13:1309–1315
Puccio G, Cajozzo C, Meli F, Rochat F, Grathwohl D, Steenhout P (2007) Clinical evaluation of a new starter formula for infants containing live Bifidobacterium longum BL999 and prebiotics. Nutrition 23:1–8
De Filippo C, Cavalieri D, Di Paola M et al (2010) Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc Natl Acad Sci 107:14691–14696
Ley RE, Turnbaugh PJ, Klein S, Gordon JI (2006) Microbial ecology: human gut microbes associated with obesity. nature 444:1022–1023
Saavedra JM, Abi-Hanna A, Moore N, Yolken RH (2004) Long-term consumption of infant formulas containing live probiotic bacteria: tolerance and safety. Am J Clin Nutr 79:261–267
Surono IS, Koestomo FP, Novitasari N, Zakaria FR, Yulianasari, Koesnandar (2011) Novel probiotic Enterococcus faecium IS-27526 supplementation increased total salivary sIgA level and bodyweight of pre-school children: a pilot study. Anaerobe 17:496–500
Chandel DS, Perez-Munoz ME, Yu F, Boissy R, Satpathy R, Misra PR, Sharma N, Chaudhry R, Parida S, Peterson DA, Gewolb IH, Panigrahi P (2017) Changes in the gut microbiota after early administration of oral synbiotics to young infants in India. J Pediatr Gastroenterol Nutr 65:218–224
Silva MR, Dias G, Ferreira CLLF, Franceschini SCC, Costa NMB (2008) Growth of preschool children was improved when fed an iron-fortified fermented milk beverage supplemented with Lactobacillus acidophilus. Nutr Res 28:226–232
Vlasova AN, Kandasamy S, Chattha KS, Rajashekara G, Saif LJ (2016) Comparison of probiotic lactobacilli and bifidobacteria effects, immune responses and rotavirus vaccines and infection in different host species. Vet Immunol Immunopathol 172:72–84
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments. The study protocol was approved by the Research Ethical Committee of Shahid Sadoughi University of Medical Sciences. Written informed consent was obtained from the patients’ parents or guardians.
Consent for Publication
Not applicable.
Availability of Data and Material
The primary data for this study is available from the authors on direct request.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aflatoonian, M., Taghavi Ardakani, A., Modarresi, S.Z. et al. The Effect of Synbiotic Supplementation on Growth Parameters in Mild to Moderate FTT Children Aged 2–5 Years. Probiotics & Antimicro. Prot. 12, 119–124 (2020). https://doi.org/10.1007/s12602-018-9508-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12602-018-9508-6