Abstract
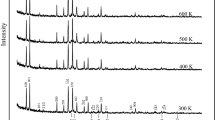
In-situ powder X-ray diffraction (XRD) and Fourier transform infrared (FTIR) spectra were measured on the natural crystals of calcite (Ca0.996Mg0.004CO3), dolomite (Ca0.497Mg0.454Fe0.046Mn0.003CO3) and magnesite (Mg0.988Ca0.010Fe0.002CO3), with a temperature up to 796 K. The thermal expansion coefficients were evaluated for these carbonate minerals, resulting in the values of 2.7×10-5, 3.3×10-5 and 3.5×10-5 K-1 for calcite, dolomite and magnesite, respectively. The magnitude of these coefficients is in the same order as those for the isothermal and elastic moduli of these carbonates (e.g., calcite<dolomite<magnesite). The IR-active internal modes of the CO3 group systematically shift to lower frequencies at elevated temperature, and the isobaric (γiP) and isothermal (γiT) Gruneisen parameters for the internal modes are generally smaller than 0.5. The corresponding anharmonic parameters (ai) are typically within the range of -1.5.+1×10-5 K-1, which are significantly smaller in magnitude than those for the external modes. We also calculate the thermodynamic properties (internal energy, heat capacities and entropy) at high temperatures for these carbonates, and the anharmonic contribution to thermodynamics shows an order of calcite>dolomite>magnesite. The Debye model (harmonic approximation) would be valid for magnesite to simulating the thermodynamic properties and isotope fractionation β-factor at high P-T condition.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References Cited
Adams, D. M., Williams, A. D., 1980. Vibrational Spectroscopy at very High Pressures. Part 26. An Infrared Study of the Metastable Phases of Ca[CO3]. Journal of the Chemical Society, Dalton Transactions, 8: 1482. https://doi.org/10.1039/dt9800001482
Andersen, F. A., Brečević, L., Beuter, G., et al., 1991. Infrared Spectra of Amorphous and Crystalline Calcium Carbonate. Acta Chemica Scandinavica, 45: 1018–1024. https://doi.org/10.3891/acta.chem.scand.45-1018
Antao, S. M., Hassan, I., Mulder, W. H., et al., 2009. In-situ Study of the R3c-R3m Orientational Disorder in Calcite. Physics and Chemistry of Minerals, 36(3): 159–169. https://doi.org/10.1007/s00269-008-0266-y
Antao, S. M., Mulder, W. H., Hassan, I., et al., 2004. Cation Disorder in Dolomite, CaMg(CO3)2, and Its Influence on the Aragonite+Magnesite. Dolomite Reaction Boundary. American Mineralogist, 89(7): 1142–1147. https://doi.org/10.2138/am-2004-0728
Biellmann, C., Gillet, P., 1992. High-Pressure and High-Temperature Behaviour of Calcite, Aragonite and Dolomite: A Raman Spectroscopic Study. European Journal of Mineralogy, 4(2): 389–394. https://doi.org/10.1127/ejm/4/2/0389
Biellmann, C., Gillet, P., Guyot, F., et al., 1993. Experimental Evidence for Carbonate Stability in the Earth’s Lower Mantle. Earth and Planetary Science Letters, 118(1/2/3/4): 31–41. https://doi.org/10.1016/0012-821x(93)90157-5
Bigeleisen, J., Mayer, M. G., 1947. Calculation of Equilibrium Constants for Isotopic Exchange Reactions. The Journal of Chemical Physics, 15(5): 261–267. https://doi.org/10.1063/1.1746492
Bottcher, M., Gehlken, P. L., Steele, D. F., 1997. Characterization of Inorganic and Biogenic Magnesian Calcites by Fourier Transform Infrared Spectroscopy. Solid State Ionics, 101–103: 1379–1385. https://doi.org/10.1016/s0167-2738(97)00235-x
Bottcher, M. E., Gehlken, P. L., Usdowski, E., 1992. Infrared Spectroscopic Investigations of the Calcite-Rhodochrosite and Parts of the Calcite-Magnesite Mineral Series. Contributions to Mineralogy and Petrology, 109(3): 304–306. https://doi.org/10.1007/bf00283320
Boulard, E., Gloter, A., Corgne, A., et al., 2011. New Host for Carbon in the Deep Earth. Proceedings of the National Academy of Sciences of the United States of America, 108(13): 5184–5187. https://doi.org/10.1073/pnas.1016934108
Boulard, E., Menguy, N., Auzende, A. L., et al., 2012. Experimental Investigation of the Stability of Fe-Rich Carbonates in the Lower Mantle. Journal of Geophysical Research: Solid Earth, 117(B2): B02208. https://doi.org/10.1029/2011jb008733
Brenker, F. E., Vollmer, C., Vincze, L., et al., 2007. Carbonates from the Lower Part of Transition Zone or Even the Lower Mantle. Earth and Planetary Science Letters, 260(1/2): 1–9. https://doi.org/10.1016/j.epsl.2007.02.038
Bromiley, F. A., Ballaran, T. B., Langenhorst, F., et al., 2007. Order and Miscibility in the Otavite-Magnesite Solid Solution. American Mineralogist, 92(5/6): 829–836. https://doi.org/10.2138/am.2007.2315
Catalli, K., Williams, Q., 2005. A High-Pressure Phase Transition of Calcite-III. American Mineralogist, 90(10): 1679–1682. https://doi.org/10.2138/am.2005.1954
Chang, L. L. Y., Howie, R. A., Zussman, J., 1996. Non-Silicates: Sulfates, Carbonates, Phosphates and Halides. The Geological Society, Longman, London. https://ci.nii.ac.jp/naid/10011703589/en/
Chen, P. F., Chiao, L. Y., Huang, P. H., et al., 2006. Elasticity of Magnesite and Dolomite from a Genetic Algorithm for Inverting Brillouin Spectroscopy Measurements. Physics of the Earth and Planetary Interiors, 155(1/2): 73–86. https://doi.org/10.1016/j.pepi.2005.10.004
Cynn, H., Hofmeister, A. M., Burnley, P. C., et al., 1996. Thermodynamic Properties and Hydrogen Speciation from Vibrational Spectra of Dense Hydrous Magnesium Silicates. Physics and Chemistry of Minerals, 23(6): 361–376. https://doi.org/10.1007/bf00199502
Dasgupta, R., Hirschmann, M. M., 2010. The Deep Carbon Cycle and Melting in Earth’s Interior. Earth and Planetary Science Letters, 298(1/2): 1–13. https://doi.org/10.1016/j.epsl.2010.06.039
Dorfman, S. M., Badro, J., Nabiei, F., et al., 2018. Carbonate Stability in the Reduced Lower Mantle. Earth and Planetary Science Letters, 489: 84–91. https://doi.org/10.1016/j.epsl.2018.02.035
Dorogokupets, P. I., 2007. Equation of State of Magnesite for the Conditions of the Earth’s Lower Mantle. Geochemistry International, 45(6): 561–568. https://doi.org/10.1134/s0016702907060043
Dorogokupets, P. T., Oganov, A. R., 2004. Intrinsic Anharmonicity in Equations of State of SOLIDS and Minerals. Doklady Earth Sciences, 395(2): 238–241
Dove, M. T., Powell, B. M., 1989. Neutron Diffraction Study of the Tricritical Orientational Order/disorder Phase Transition in Calcite at 1 260 K. Physics and Chemistry of Minerals, 16(5): 503–507. https://doi.org/10.1007/bf00197019
Dove, M. T., Swainson, I. P., Powell, B. M., et al., 2005. Neutron Powder Diffraction Study of the Orientational Order-Disorder Phase Transition in Calcite, CaCO3. Physics and Chemistry of Minerals, 32(7): 493–503. https://doi.org/10.1007/s00269-005-0026-1
Effenberger, H., Mereiter, K., Zemann, J., 1981. Crystal Structure Refinements of Magnesite, Calcite, Rhodochrosite, Siderite, Smithonite, and Dolomite, with Discussion of some Aspects of the Stereochemistry of Calcite Type Carbonates. Zeitschrift für Kristallographie-Crystalline Materials, 156(3/4): 233–243. https://doi.org/10.1524/zkri.1981.156.3-4.233
Fahad, M., Iqbal, Y., Riaz, M., et al., 2016. Metamorphic Temperature Investigation of Coexisting Calcite and Dolomite Marble-Examples from Nikani Ghar Marble and Nowshera Formation, Peshawar Basin, Pakistan. Journal of Earth Science, 27(6): 989–997. https://doi.org/10.1007/s12583-015-0643-7
Fahad, M., Saeed, S., 2018. Determination and Estimation of Magnesium Content in the Single Phase Magnesium-Calcite [Ca(1-x)MgxCO3(S)] Using Electron Probe Micro-Analysis (EPMA) and X-Ray Diffraction (XRD). Geosciences Journal, 22(2): 303–312. https://doi.org/10.1007/s12303-017-0059-8
Falini, G., Fermani, S., Gazzano, M., et al., 1998. Structure and Morphology of Synthetic Magnesium Calcite. Journal of Materials Chemistry, 8(4): 1061–1065. https://doi.org/10.1039/a707893e
Fei, Y., 1995. Thermal Expansion. In: Ahrens. T. J., ed., Mineral Physics & Crystallography: A Handbook of Physical Constants, Volume 2. American Geophysical Union, Washington, D.C. 29–44. https://doi.org/10.1029/rf002
Fiquet, G., Guyot, F., Itie, J. P., 1994. High-Pressure X-Ray Diffraction Study of Carbonates: MgCO3, CaMg(CO3)2, and CaCO3. American Mineralogist, 79(1–2): 15–23
Fiquet, G., Reynard, B., 1999. High-Pressure Equation of State of Magnesite; New Data and a Reappraisal. American Mineralogist, 84(5/6): 856–860. https://doi.org/10.2138/am-1999-5-619
Fiquet, G., Richet, P., Montagnac, G., 1999. High-Temperature Thermal Expansion of Lime, Periclase, Corundum and Spinel. Physics and Chemistry of Minerals, 27(2): 103–111. https://doi.org/10.1007/s002690050246
Fujimori, H., Komatsu, H., Ioku, K., et al., 2002. Anharmonic Lattice Mode of Ca2SiO4: Ultraviolet Laser Raman Spectroscopy at High Temperatures. Physical Review B, 66(6): 064306. https://doi.org/10.1103/physrevb.66.064306
Gong, Q., Deng, J., Wang, Q., et al., 2010. Experimental Determination of Calcite Dissolution Rates and Equilibrium Concentrations in Deionized Water Approaching Calcite Equilibrium. Journal of Earth Science, 21(4): 402–411. https://doi.org/10.1007/s12583-010-0103-3
Gillet, P., Guyot, F., Malezieux, J. M., 1989. High-Pressure, High-Temperature Raman Spectroscopy of Ca2GeO4 (Olivine Form): Some Insights on Anharmonicity. Physics of the Earth and Planetary Interiors, 58(2/3): 141–154. https://doi.org/10.1016/0031-9201(89)90050-2
Gillet, P., Biellmann, C., Reynard, B., et al., 1993. Raman Spectroscopic Studies of Carbonates Part I: High-Pressure and High-Temperature Behaviour of Calcite, Magnesite, Dolomite and Aragonite. Physics and Chemistry of Minerals, 20(1): 1–18. https://doi.org/10.1007/bf00202245
Gillet, P., McMillan, P., Schott, J., et al., 1996. Thermodynamic Properties and Isotopic Fractionation of Calcite from Vibrational Spectroscopy of 18O-Substituted Calcite. Geochimica et Cosmochimica Acta, 60(18): 3471–3485. https://doi.org/10.1016/0016-7037(96)00178-0
Grzechnik, A., Simon, P., Gillet, P., et al., 1999. An Infrared Study of MgCO3 at High Pressure. Physica B: Condensed Matter, 262(1/2): 67–73. https://doi.org/10.1016/s0921-4526(98)00437-2
Hazen, R. M., Downs, R. T., Jones, A. P., et al., 2013. Carbon Mineralogy and Crystal Chemistry. Reviews in Mineralogy and Geochemistry, 75(1): 7–46. https://doi.org/10.2138/rmg.2013.75.2
Holland, T. J. B., Redfern, S. A. T., 1997. Unit Cell Refinement from Powder Diffraction Data: The Use of Regression Diagnostics. Mineralogical Magazine, 61(404): 65–77. https://doi.org/10.1180/minmag.1997.061.404.07
Holland, T. J. B., Powell, R., 2004. An Internally Consistent Thermodynamic Data Set for Phases of Petrological Interest. Journal of Metamorphic Geology, 16(3): 309–343. https://doi.org/10.1111/j.1525-1314.1998.00140.x
Isshiki, M., Irifune, T., Hirose, K., et al., 2004. Stability of Magnesite and Its High-Pressure Form in the Lowermost Mantle. Nature, 427(6969): 60–63. https://doi.org/10.1038/nature02181
Koch-Müller, M., Jahn, S., Birkholz, N., et al., 2016. Phase Transitions in the System CaCO3 at High P and T Determined by in Situ Vibrational Spectroscopy in Diamond Anvil Cells and First-Principles Simulations. Physics and Chemistry of Minerals, 43(8): 545–561. https://doi.org/10.1007/s00269-016-0815-8
Lane, M. D., Christensen, P. R., 1997. Thermal Infrared Emission Spectroscopy of Anhydrous Carbonates. Journal of Geophysical Research: Planets, 102(E11): 25581–25592. https://doi.org/10.1029/97je02046
Lin, C. C., 2013. Elasticity of Calcite: Thermal Evolution. Physics and Chemistry of Minerals, 40(2): 157–166. https://doi.org/10.1007/s00269-012-0555-3
Litasov, K. D., Fei, Y. W., Ohtani, E., et al., 2008. Thermal Equation of State of Magnesite to 32 GPa and 2 073 K. Physics of the Earth and Planetary Interiors, 168(3/4): 191–203. https://doi.org/10.1016/j.pepi.2008.06.018
Liu, C. J., Zheng, H. F., Wang, D. J., 2017. Raman Spectroscopic Study of Calcite III to Aragonite Transformation under High Pressure and High Temperature. High Pressure Research, 37(4): 545–557. https://doi.org/10.1080/08957959.2017.1384824
Liu, J., Lin, J. F., Mao, Z., et al., 2014. Thermal Equation of State and Spin Transition of Magnesiosiderite at High Pressure and Temperature. American Mineralogist, 99(1): 84–93. https://doi.org/10.2138/am.2014.4553
Liu, L. G., Mernagh, T. P., 1990. Phase Transitions and Raman Spectra of Calcite at High Pressures and Room Temperature. American Mineralogist, 75(7-8): 801–806
Liu, Q., Tossell, J. A., Liu, Y., 2010. On the Proper Use of the Bigeleisen-Mayer Equation and Corrections to It in the Calculation of Isotopic Fractionation Equilibrium Constants. Geochimica et Cosmochimica Acta, 74(24): 6965–6983. https://doi.org/10.1016/j.gca.2010.09.014
Mao, Z., Armentrout, M., Rainey, E., et al., 2011. Dolomite III: A New Candidate Lower Mantle Carbonate. Geophysical Research Letters, 38(22): L22303. https://doi.org/10.1029/2011gl049519
Markgraf, S. A., Reeder, R. J., 1985. High-Temperature Structure Refinements of Calcite and Magnesite. American Mineralogist, 70(5–6): 590–600
Matas, J., Gillet, P., Ricard, Y., et al., 2000. Thermodynamic Properties of Carbonates at High Pressures from Vibrational Modelling. European Journal of Mineralogy, 12(4): 703–720. https://doi.org/10.1127/ejm/12/4/0703
Megaw, H. D., 1973. Crystal Structures: A Working Approach. Saunders, London. 563
Merlini, M., Sapelli, F., Fumagalli, P., et al., 2016. High-Temperature and High-Pressure Behavior of Carbonates in the Ternary Diagram CaCO3-MgCO3-FeCO3. American Mineralogist, 101(6): 1423–1430. https://doi.org/10.2138/am-2016-5458
Oganov, A. R., Dorogokupets, P. I., 2004. Intrinsic Anharmonicity in Equations of State and Thermodynamics of Solids. Journal of Physics: Condensed Matter, 16(8): 1351–1360. https://doi.org/10.1088/0953-8984/16/8/018
Oganov, A. R., Glass, C. W., Ono, S., 2006. High-Pressure Phases of CaCO3: Crystal Structure Prediction and Experiment. Earth and Planetary Science Letters, 241(1/2): 95–103. https://doi.org/10.1016/j.epsl.2005.10.014
Paquette, J., Reeder, R. J., 1990. Single-Crystal X-Ray Structure Refinements of Two Biogenic Magnesian Calcite Crystals. American Mineralogist, 75(9): 1151–1158
Pickard, C. J., Needs, R. J., 2015. Structures and Stability of Calcium and Magnesium Carbonates at Mantle Pressures. Physical Review B, 91(10): 104101
Polyakov, V. B., 1998. On Anharmonic and Pressure Corrections to the Equilibrium Isotopic Constants for Minerals. Geochimica et Cosmochimica Acta, 62(18): 3077–3085. https://doi.org/10.1016/s0016-7037(98)00220-8
Polyakov, V. B., Kharlashina, N. N., 1994. Effect of Pressure on Equilibrium Isotopic Fractionation. Geochimica et Cosmochimica Acta, 58(21): 4739–4750. https://doi.org/10.1016/0016-7037(94)90204-6
Redfern, S. A. T., Angel, R. J., 1999. High-Pressure Behaviour and Equation of State of Calcite, CaCO3. Contributions to Mineralogy and Petrology, 134(1): 102–106. https://doi.org/10.1007/s004100050471
Reynard, B., Caracas, R., 2009. D/H Isotopic Fractionation between Brucite Mg(OH)2 and Water from First-Principles Vibrational Modeling. Chemical Geology, 262(3/4): 159–168. https://doi.org/10.1016/j.chemgeo.2009.01.007
Reeder, R. J., 1983. Crystal Chemistry of the Rhombohedral Carbonates. Reviews in Mineralogy and Geochemistry, 11(1): 1–47
Reeder, R. J., Markgraf, S. A., 1986. High-Temperature Crystal Chemistry of Dolomite. American Mineralogist, 71(5–6): 795–804
Ross, N. L., 1997. The Equation of State and High-Pressure Behavior of Magnesite. American Mineralogist, 82(7/8): 682–688. https://doi.org/10.2138/am-1997-7-805
Ross, N. L., Reeder, R. J., 1992. High-Pressure Structural Study of Dolomite and Ankerite. American Mineralogist, 77(3–4): 412–421
Santillán, J., 2005. An Infrared Study of Carbon-Oxygen Bonding in Magnesite to 60 GPa. American Mineralogist, 90(10): 1669–1673. https://doi.org/10.2138/am.2005.1703
Santillán, J., Williams, Q., 2004. A High-Pressure Infrared and X-Ray Study of FeCO3 and MnCO3: Comparison with CaMg(CO3)2-Dolomite. Physics of the Earth and Planetary Interiors, 143–144: 291–304. https://doi.org/10.1016/j.pepi.2003.06.007
Santillán, J., Williams, Q., Knittle, E., 2003. Dolomite-II: A High-Pressure Polymorph of CaMg(CO3)2. Geophysical Research Letters, 30(2): 1054. https://doi.org/10.1029/2002gl016018
Stekiel, M., Nguyen-Thanh, T., Chariton, S., et al., 2017. High Pressure Elasticity of FeCO3-MgCO3 Carbonates. Physics of the Earth and Planetary Interiors, 271: 57–63. https://doi.org/10.1016/j.pepi.2017.08.004
Suito, K., Namba, J., Horikawa, T., et al., 2001. Phase Relations of CaCO3 at High Pressure and High Temperature. American Mineralogist, 86(9): 997–1002. https://doi.org/10.2138/am-2001-8-906
Thomson, A. R., Walter, M. J., Kohn, S. C., et al., 2016. Slab Melting as a Barrier to Deep Carbon Subduction. Nature, 529(7584): 76–79. https://doi.org/10.1038/nature16174
Titschack, J., Goetz-Neunhoeffer, F., Neubauer, J., 2011. Magnesium Quantification in Calcites [(Ca, Mg)CO3] by Rietveld-Based XRD Analysis: Revisiting a Well-Established Method. American Mineralogist, 96(7): 1028–1038. https://doi.org/10.2138/am.2011.3665
Urey, H. C., 1947. The Thermodynamic Properties of Isotopic Substances. Journal of the Chemical Society (Resumed), 562–581. https://doi.org/10.1039/jr9470000562
Valenzano, L., Noël, Y., Orlando, R., et al., 2007. Ab Initio Vibrational Spectra and Dielectric Properties of Carbonates: Magnesite, Calcite and Dolomite. Theoretical Chemistry Accounts, 117(5/6): 991–1000. https://doi.org/10.1007/s00214-006-0213-2
Wang, A. L., Pasteris, J. D., Meyer, H. O. A., et al., 1996. Magnesite-Bearing Inclusion Assemblage in Natural Diamond. Earth and Planetary Science Letters, 141(1/2/3/4): 293–306. https://doi.org/10.1016/0012-821x(96)00053-2
Wang, G., Wang, J., Wang, Z., et al., 2017. Carbon Isotope Gradient of the Ediacaran Cap Carbonate in the Shennongjia Area and Its Implications for Ocean Stratification and Palaeogeography. Journal of Earth Science. 28(2): 42–56. https://doi.org/10.1007/s12583-016-0923-x
Wang, M. L., Shi, G. H., Qin, J. Q., et al., 2018. Thermal Behaviour of Calcite-Structure Carbonates: A Powder X-Ray Diffraction Study between 83 and 618 K. European Journal of Mineralogy, 30(5): 939–949. https://doi.org/10.1127/ejm/2018/0030-2768
Wang, X., Ye, Y., Wu, X., et al., 2019. High-Temperature Raman and FTIR Study of Aragonite-Group Carbonates. Physics and Chemistry of Minerals, 46(1): 51–62. https://doi.org/10.1007/s00269-018-0986-6
Wei, S. H., Xu, X. X., 2018. Boosting Photocatalytic Water Oxidation Reactions over Strontium Tantalum Oxynitride by Structural Laminations. Applied Catalysis B: Environmental, 228: 10–18. https://doi.org/10.1016/j.apcatb.2018.01.071
Weir, C. E., Lippincott, E. R., van Valkenburg, A., et al., 1959. Infrared Studies in the 1- to 15-Micron Region to 30,000 Atmospheres. Journal of Research of the National Bureau of Standards Section A: Physics and Chemistry, 63A(1): 55–62. https://doi.org/10.6028/jres.063a.003
White, W. B., 1974. The Carbonate Minerals. In: Farmer, V. C., ed., The Infrared Spectra of Minerals. Mineralogical Society of Great Britain and Ireland, London. 227–284
Yang, J., Mao, Z., Lin, J.-F., et al., 2014. Single-Crystal Elasticity of the Deep-Mantle Magnesite at High Pressure and Temperature. Earth and Planetary Science Letters, 392: 292–299. https://doi.org/10.1016/j.epsl.2014.01.027
You, X. L., Jia, W. Q., Xu, F., et al., 2018. Mineralogical Characteristics of Ankerite and Mechanisms of Primary and Secondary Origins. Earth Science, 43(11): 4046–4055 (in Chinese with English Abstract). https://doi.org/10.3799/dqkx.2018.152
Zhang, J., Martinez, I., Guyot, F., et al., 1997. X-Ray Diffraction Study of Magnesite at High Pressure and High Temperature. Physics and Chemistry of Minerals, 24(2): 122–130. https://doi.org/10.1007/s002690050025s
Zhang, J., Reeder, R., 1999. Comparative Compressibilities of Calcite-Structure Carbonates: Deviations from Empirical Relations. American Mineralogist, 84(5–6): 861–870. https://doi.org/10.2138/am-1999-5-620
Zhang, X., Yang, S. Y., Zhao, H., et al., 2019. Effect of Beam Current and Diameter on Electron Probe Microanalysis of Carbonate Minerals. Journal of Earth Science, 30(4): 834–842. https://doi.org/10.1007/s12583-017-0939-x
Zheng, R., Pan, Y., Zhao, C., et al., 2013. Carbon and Oxygen Isotope Stratigraphy of the Oxfordian Carbonate Rocks in Amu Darya Basin. Journal of Earth Science, 24(1): 42–56. https://doi.org/10.1007/s12583-013-0315-4
Zhu, Y. F., Ogasawara, Y., 2002. Carbon Recycled into Deep Earth: Evidence from Dolomite Dissociation in Subduction-Zone Rocks. Geology, 30(10): 947–950. https://doi.org/10.1130/0091-7613(2002)030%3c0947:cridee%3e2.0.co;2
Acknowledgment
Many thanks to Profs. Kurt Leinenweber and Joseph Smyth for helpful and constructive discussion and revision on this manuscript. This work was supported by the National Key Research and Development Program of China (No. 2016YFC0600204), and the National Natural Science Foundation of China (Nos. 41590621, 41672041). EPMA and in-situ high-T FTIR experiments were carried out at the State Key Laboratory of Geological Processes and Mineral Resources, China University of Geosciences (Wuhan), while in-situ high-T powder XRD measurements were conducted at School of Chemical Science and Engineering, Tongji University. The final publication is available at Springer via https://doi.org/10.1007/s12583-019-1236-7.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Wang, X., Xu, X., Ye, Y. et al. In-situ High-Temperature XRD and FTIR for Calcite, Dolomite and Magnesite: Anharmonic Contribution to the Thermodynamic Properties. J. Earth Sci. 30, 964–976 (2019). https://doi.org/10.1007/s12583-019-1236-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12583-019-1236-7