Abstract
During the induction of Japanese eel maturation, administering maturation-inducing steroids (MIS) or their precursors at an inappropriate maturational status is a major cause of poor egg quality. In this study, we investigated the feasibility of controlling oocyte maturation progress by rearing in cold and warm water to manipulate the timing of MIS administration. Mature females with oocytes at the migratory nucleus stage were reared for two terms (3 days to 1 day and 1 day to 0 days before MIS administration) at 20/20 °C, 20/15 °C, 15/20 °C, or 15/15 °C, and the maturational status was monitored based on their lipid droplet morphology and oocyte diameter. Oocytes matured faster at 20 °C than at 15 °C in either term. Next, the mature females were reared at 15 or 20 °C depending on the maturational status of each female 3 days and 1 day before MIS administration; the immature females were reared at 20 °C to accelerate their maturation. Consequently, the maturational status of most females was similar at MIS administration. After improvement, this method would lead most females to the optimum maturational status at MIS administration by properly rearing in cold and warm water.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Japanese eel, Anguilla japonica, is a representative freshwater aquaculture species in Japan (Tanaka 2015). The glass eels used for aquaculture are all caught in the wild. The catch of glass eels for cultivation has been declining significantly for decades (Arai 2014). In 2014, the International Union for Conservation of Nature (IUCN) classified the Japanese eel as “Endangered” on the IUCN Red List of Threatened Species (Kaifu and Yokouchi 2019). The increased prices of glass eels has led to business crises in aquaculture farms and their related industries, such as eel restaurants (Tanaka et al. 2006; Tanaka 2015). Therefore, developing a technique to produce glass eels artificially is highly desirable.
Since male and female eels do not mature in captivity, artificial maturation induction using hormonal treatment is crucial for producing glass eels (Kagawa et al. 2005). Studies on maturation induction in Japanese eels started around 1960 (Tanaka 2015). Artificial fertilization, using eggs obtained from females injected with salmon pituitary extracts (SPE) and sperm from males injected with human chorionic gonadotropin (hCG), proved successful in 1973 (Yamamoto and Yamauchi 1974). In the early 1990s, oral estradiol-17β administration facilitated the stable production of feminized eels to address the problem that the sex ratio of cultured eels is skewed toward males (Tachiki et al. 1997). In the late 1990s, improved methods were developed for maturation induction in both females and males, and obtaining sufficient gametes consistently became possible (Ohta and Izawa 1996; Ohta et al. 1997; Kagawa et al. 1997, 1998). Furthermore, leptocephali larvae were successfully produced (Tanaka et al. 2001) and subsequently metamorphosed into glass eels (Tanaka et al. 2003) using a novel eel larval feed containing dogfish shark eggs (Squalus sp.). Finally, the full life cycle of this species was cultured in 2010 (Masuda et al. 2012).
Despite these efforts, glass eels have not been produced commercially to date (Tanaka 2015), as the survival rates from eggs to the leptocephali and glass eel stages are very low. One of the impediments for the mass production of glass eels is the unstable quality of eggs obtained from females induced to maturation by hormonal treatments (Kagawa et al. 2013; Kazeto et al. 2019). Hatchability highly varies among batches of eggs from different females (Unuma et al. 2004). In the current protocols for obtaining eggs, the female eels are injected with SPE, weekly until the oocytes develop to the migratory nucleus stage (Kazeto et al. 2019). The mature females are successively injected with SPE, for priming, and a maturation-inducing steroid (MIS; 17α, 20β-dihydroxy-4-pregene-3-one) or its precursor (17α-hydroxyprogesterone; 17α-OHP) to induce final oocyte maturation and ovulation. During maturation induction, oogenesis progress and consequent egg quality are influenced by environmental water temperature (Sato et al. 2006; Dou et al. 2008; Horie et al. 2008; Unuma et al. 2012; Yoshikawa 2012). In the current protocols, vitellogenesis is induced at 15–20 °C (primarily 15 °C), and final oocyte maturation and ovulation at 20–23 °C (primarily 20 °C) (Unuma et al. 2012). Thus, the seed production facilities usually rear eels at 15–20 °C during repeated SPE injections and at 20–23 °C from priming with SPE to egg collection.
The timing of MIS injection strongly affects the quality of eggs. We classified the fusion of lipid droplets in Japanese eel oocyte into 10 stages for use as a biomarker for inducing final oocyte maturation and ovulation, and found that stage 6 (average diameter of the five largest droplets; 90–110 µm) was the best for MIS administration (Unuma et al. 2011). If all the females are injected with MIS at stage 6, quality deterioration due to MIS injection at inappropriate timing could be alleviated. However, we cannot predict when each female reaches the optimum stage, owing to individual differences in oocyte maturation progress. Therefore, to administer MIS to all females at the best time, researchers would have to monitor them 24 h every day during the maturation-induction period. Such a method is unrealistic in seed production studies. It is more practical to inject MIS on scheduled days and times, as has been conducted to date in the current protocols.
Our final goal is to develop a method to control oocyte maturation progress of each female in order to administer MIS to all the females at the optimum period (stage 6) at a scheduled day and time based on the current protocol for inducing Japanese eel maturation. As an initial step, in the present study, we investigated the effects of rearing water temperature manipulation for 3 days before MIS injection, on oocyte maturation progress. As mentioned above, rearing at low (15–20 °C) and high (20–23 °C) temperatures usually helps maturation induction in female eels. It is convenient to use these two types of rearing water to control oocyte maturation progress. Therefore, we reared the eels at 15 and 20 °C to accelerate and decelerate oocyte maturation.
Materials and methods
Broodstock
Glass eels, purchased from a commercial dealer, were feminized by feeding an estradiol-17β-supplemented commercial eel feed (10 mg/kg feed) for 6 months (Tachiki et al. 1997) and were reared on normal commercial feed under routine culture conditions in freshwater ponds for 1–4 years at the Hamanako Branch, Shizuoka Prefectural Research Institute of Fishery, Shizuoka, Japan. Some glass eels were reared without feeding estradiol-17β to obtain the male broodstock. The female and male broodstock were acclimated to seawater and used for maturation induction, after which they were not fed.
Maturation induction
The female and male eels were induced to maturation according to the methods reported by Kagawa et al. (1997) and Ohta et al. (1997), respectively, with modifications.
Briefly, 84 females (1082 ± 261 g, mean ± standard deviation [SD]) were reared in three 2000 L tanks in Experiment 1, and 36 females (527 ± 128 g) were reared in the same tank in Experiment 2. The tanks were supplied with sand-filtered seawater at 15 °C (flow-through system). The female eels were intraperitoneally injected with SPE (20 mg/kg body weight) every Monday for 10–17 weeks. The body weight was measured before each injection.
The male eels were intraperitoneally injected with human chorionic gonadotropin (2000 IU/kg body weight) every Monday for up to 20 weeks. Sperm were repeatedly obtained by stripping the spermiating males, diluted 100 times with artificial seminal plasma (Ohta and Izawa 1996), and used for artificial fertilization within 3 weeks. Sperm motility was assessed under a microscope prior to use.
The female and male eels were anesthetized with 2-phenoxyethanol dissolved in seawater (2 mL/L) for injecting hormones or stripping for gamete collection. Every effort was made to minimize animal suffering.
Experiment 1
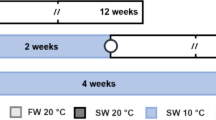
Experiment 1 (Fig. 1a) was conducted to investigate whether oocyte maturation could be accelerated or decelerated by manipulating the rearing temperature for 3 days before MIS injection (from Monday evening to Thursday evening). Female eels with > 5% increased body weight were used. These females were randomly divided into the 20/20 °C, 20/15 °C, 15/20 °C, and 15/15 °C groups (14 individuals/group). On Monday, females in the 20/20 °C and 20/15 °C groups were reared at 20 °C and those in the 15/20 °C and 15/15 °C groups were reared at 15 °C. On Wednesday, females in the 20/20 °C and 15/20 °C groups were reared at 20 °C and those in the 20/15 °C and 15/15 °C groups were reared at 15 °C. To assess maturation progress, the oocytes were collected five times (Monday, Tuesday, Wednesday evenings, Thursday morning, and Thursday evening) in a noninvasive manner from the gonopore by aspiration with a soft plastic cannula, and the lipid droplet stage and oocyte diameter were determined using 10 oocytes from each female, as described later. To facilitate comparison of the 3-day progress in the lipid droplet stage and oocyte diameter in the different groups, only females with stage 2 lipid droplets and 750–850 µm oocyte diameter on Monday evening were used for data analyses. The females were intraperitoneally injected with SPE (20 mg/kg body weight) on Wednesday evening for priming, and with 17α-OHP (2 mg/kg body weight), a precursor of eel MIS, on Thursday evening to induce final oocyte maturation and ovulation. After 17α-OHP injection, all females were reared at 20 °C, which is the optimum temperature for inducing final oocyte maturation and ovulation (Unuma et al. 2012). The eggs were gently stripped from ovulated females 15–19 h after 17α-OHP injection and artificially fertilized immediately for the hatchability test.
a, b Experimental procedures for maturation induction in Japanese eel Anguilla japonica. Female eels were injected with salmon pituitary extract (SPE) every Monday, and reared at specific temperatures when the body weight index increased by > 5% (a, Experiment 1) or the lipid droplet stage reached stage 2 and above (b, Experiment 2). Such females were reared at 15 and/or 20 °C for two terms (Monday to Wednesday and Wednesday to Thursday). Thus, the females were reared at 20/20 °C, 20/15 °C, 15/20 °C, or 15/15 °C for 3 days from Monday to Thursday. In Experiment 1, the females were randomly allocated to the four temperature groups on Monday. In Experiment 2, the temperature was chosen for each female based on its lipid droplet stage on Monday and Wednesday. The oocytes were collected from the gonopore with a cannula five (Experiment 1) or three (Experiment 2) times from Monday to Thursday, for analyzing the lipid droplet stage and oocyte diameter. The females were injected with SPE on Wednesday evening for priming and with 17α-hydroxyprogesterone (17α-OHP) on Thursday evening for final oocyte maturation and ovulation. After 17α-OHP injection, all females were reared at 20 °C. The eggs were stripped from ovulated females 15–19 h after 17α-OHP injection and used for artificial fertilization
Experiment 2
Experiment 2 (Fig. 1b) was conducted to investigate whether the maturational status of oocytes could be adjusted to the optimum stage for MIS injection (stage 6) (Unuma et al. 2011) by manipulating temperature adaptively for each female for 3 days before MIS injection. The female eels with stage 2 and above lipid droplets on Monday were used. These females were reared at 15 or 20 °C based on their individual lipid droplet stage. On Monday, the females at stage 2 (27 individuals) were reared at 20 °C to accelerate oocyte maturation, and those at stage 3 (6 individuals) were reared at 15 °C to decelerate oocyte maturation. On Wednesday, the females at stages 3–4 (24 reared at 20 °C tanks and 2 at 15 °C) were reared at 20 °C to accelerate oocyte maturation, and those at stage 5 (3 reared at 20 °C and 4 at 15 °C) were reared at 15 °C to decelerate oocyte maturation. Thus, 24, 3, 2, and 4 individuals were assigned to the 20/20 °C, 20/15 °C, 15/20 °C, and 15/15 °C groups, respectively. To assess maturation progress, the oocytes were collected with a cannula three times (Monday, Wednesday, and Thursday evenings), and the lipid droplet stage and oocyte diameter were determined using 15 oocytes from each female, as described later. The other procedures were the same as those described in Experiment 1.
Determination of lipid droplet stage and oocyte diameter
To assess the maturational status of each female, the oocytes collected with cannulas were immersed in 0.9% NaCl and photographed under a stereoscopic microscope. A total of 10 (Experiment 1) or 15 (Experiment 2) oocytes were randomly chosen among the most advanced mode of developing oocytes and used for determining both lipid droplet stage and oocyte diameter. The morphology of lipid droplets in each oocyte was classified into stages 1 to 10 as reported by Unuma et al. (2011), and the median value of 10 (Experiment 1) or 15 (Experiment 2) oocytes was defined as the stage for the individual. The long and short diameters of each oocyte were measured with digital calipers and averaged. The oocyte diameter increase rates from Monday to Wednesday and from Wednesday to Thursday were calculated for each female using the following formula:
Hatchability test
The eggs (2 g) were inseminated with 1 mL pre-diluted milt (sperm motility > 50%) and dispersed in 100 mL seawater. Then, 3 mL of this seawater, containing approximately 120 eggs, was used to assess hatchability by employing a conventional method using Petri dishes in Experiment 1 (Unuma et al. 2005) or using an individual rearing method using 48-well tissue culture microplates in Experiment 2 (Unuma et al. 2004). The Petri dishes and microplates, containing inseminated eggs, were incubated at 25 °C. The hatched larvae were counted 3 days after insemination. The hatching rate was calculated for each female using the following formula:
Statistical analysis
Data are expressed as mean ± SD unless otherwise specified. The Kruskal–Wallis test or the Mann–Whitney U test were performed to examine the differences in oocyte diameter increase rates and hatching rates among groups using BellCurve for Excel version 2.0 (Social Survey Research Information Co., Ltd; Tokyo, Japan). Statistical significance was set at P < 0.05.
Results
Experiment 1
Figure 2 shows oocyte maturation progress for 3 days before 17α-OHP injection in the female eels randomly reared at 20/20 °C, 20/15 °C, 15/20 °C, and 15/15 °C.
a–d Progress of oocyte maturation in Japanese eel Anguilla japonica reared at 15 and/or 20 °C for 3 days before 17α-hydroxyprogesterone (17α-OHP) injection (Experiment 1). The females with > 5% increase in body weight were randomly reared at 20/20 °C (n = 3), 20/15 °C (n = 5), 15/20 °C (n = 6), and 15/15 °C (n = 7) from Monday to Wednesday and from Wednesday to Thursday. The oocytes were collected from the gonopore with a cannula on Monday, Tuesday, and Wednesday evenings, and Thursday morning and evening. The lipid droplet stage and oocyte diameter were determined using 10 oocytes from each female at each cannulation. a Frequency distribution of the lipid droplet stage from Monday to Thursday. b Changes in the lipid droplet stage. Values are the median of those for 3–7 females. c Changes in the oocyte diameter. Upper panel shows 20/20 °C and 20/15 °C groups, and lower panel 15/20 °C and 15/15 °C groups. Values are the mean ± standard deviation of those for 3–7 females. d Rate of increase in oocyte diameter. Values are the mean ± standard deviation of those for 3–7 females
The frequency distribution of the lipid droplet stage from Monday to Thursday evening is shown in Fig. 2a. From Monday to Wednesday evening, some females reared at 20 °C (20/20 °C and 20/15 °C groups) proceeded to stage 5, while those reared at 15 °C (15/20 °C and 15/15 °C groups) proceeded to stage 4 at most. From Wednesday to Thursday evening, the females reared consecutively at 20 °C (20/20 °C group) proceeded to stage 6, while those reared alternatively at 15 °C (20/15 °C group) proceeded to stage 5 at most. However, the females reared alternatively at 20 °C (15/20 °C group) proceeded to stages 4–6, while those reared consecutively at 15 °C (15/15 °C group) proceeded to stages 3–5.
Changes in the median lipid droplet stage from Monday to Thursday evening are shown in Fig. 2b. On Monday evening, the medians in all groups were stage 2. The median in the 20/20 °C group proceeded to stage 3 on Tuesday evening, stage 4 on Wednesday evening, stage 6 on Thursday morning, and remained at stage 6 until Thursday evening. The median in the 20/15 °C group was similar to that in the 20/20 °C group until Wednesday evening; however, after transfer to 15 °C, it remained at stage 4 until Thursday morning and proceeded to stage 5 on Thursday evening. The median in the 15/20 °C group remained at stage 2 until Tuesday evening, proceeded to stage 3.25 on Wednesday evening, stage 5 on Thursday morning, and remained at stage 5 until Thursday evening. The median in the 15/15 °C group was similar to that in the 15/20 °C group until Wednesday evening, but remained at stage 3 until Thursday morning and proceeded to stage 4 on Thursday evening.
Changes in the mean oocyte diameters from Monday to Thursday evening are shown in Fig. 2c. The mean diameters in the 20/20 °C, 20/15 °C, 15/20 °C and 15/15 °C groups increased from 765.3 ± 6.4, 781.4 ± 33.7, 780.8 ± 22.5, and 784.0 ± 13.9 μm, respectively, on Monday evening to 862.9 ± 20.9, 885.9 ± 31.9, 856.0 ± 22.0, and 867.1 ± 25.0 μm, respectively, on Wednesday evening, and 949.4 ± 8.7, 959.3 ± 17.2, 963.2 ± 28.7, and 947.7 ± 19.2 μm, respectively, on Thursday evening.
The rate of increase in oocyte diameter from Monday to Thursday evening is shown in Fig. 2d. The increase rate from Monday to Wednesday was higher in the order of 20/15 °C (13.5 ± 4.2%), 20/20 °C (12.8 ± 3.7%), 15/15 °C (10.6 ± 3.2%), and 15/20 °C (9.7 ± 3.4%) groups. The increase in the females reared at 20 °C (20/15 °C and 20/20 °C groups) was higher than that in those reared at 15 °C (15/15 °C and 15/20 °C groups), although the differences were not significant between groups. The rates of increase in oocyte diameter from Wednesday to Thursday was higher in the order of 15/20 °C (12.6 ± 3.6%), 20/20 °C (10.1 ± 2.4%), 15/15 °C (9.3 ± 2.2%), and 20/15 °C (8.4 ± 4.1%) groups. The increase rate in the females reared at 20 °C (15/20 °C and 20/20 °C groups) was higher than that in those reared at 15 °C (15/15 °C and 20/15 °C groups), although the differences were not significant between groups.
These changes in lipid droplet stage and oocyte diameter indicated that oocyte maturation progressed faster at 20 °C than at 15 °C despite changing the rearing temperature.
Figure 3 shows the hatchability of the eggs obtained from female eels reared at 15 °C and/or 20 °C for 3 days before 17α-OHP injection. Hatching rates were higher in the order of 20/20 °C (35.9 ± 46.6%), 20/15 °C (21.5 ± 31.3%), 15/20 °C (10.7 ± 24.2%), and 15/15 °C (0.8 ± 2.0%) groups (Fig. 3a). Hatchability was lower in the groups where females were reared at 20 °C for fewer days, although the differences were not significant between groups. Relationship between the lipid droplet stage at 17α-OHP injection and hatching rates is shown in Fig. 3b. Fewer days of rearing at 20 °C resulted in a higher number of stages 3–4 females and lower hatching rates (< 20%).
a, b Hatchability of eggs obtained from Japanese eel Anguilla japonica reared at 15 and/or 20 °C for 3 days before 17α-hydroxyprogesterone (17α-OHP) injection (Experiment 1). The females with > 5% increase in body weight were randomly reared at 20/20 °C (n = 3), 20/15 °C (n = 5), 15/20 °C (n = 6), and 15/15 °C (n = 7) from Monday to Wednesday and from Wednesday to Thursday. The females were injected with 17α-OHP on Thursday evening for final oocyte maturation and ovulation. The eggs were stripped from ovulated females and used for artificial fertilization. a Hatching rates in the females reared at the four temperature conditions. Values are the mean ± standard deviation of those for 3–7 females. b The relationship between lipid droplet stage at 17α-OHP injection and hatching rate. To avoid overlap, the plots are slightly shifted along the x-axis
Experiment 2
Figure 4 shows oocyte maturation progress for 3 days before 17α-OHP injection in the female eels adaptively reared at 15 and 20 °C, according to their individual lipid droplet stage on Monday and Wednesday evenings.
a–g Progress of oocyte maturation in Japanese eel Anguilla japonica reared at 15 and/or 20 °C for 3 days before 17α-hydroxyprogesterone (17α-OHP) injection (Experiment 2). On Monday, stage 2 and stage 3 females were reared at 20 and 15 °C, respectively. On Wednesday, stage 3–4 and stage 5 females were reared at 20 and 15 °C, respectively. Thus, the females were reared at 20/20 °C (n = 24), 20/15 °C (n = 3), 15/20 °C (n = 2), and 15/15 °C (n = 4) from Monday to Wednesday and from Wednesday to Thursday. The oocytes were collected from the gonopore with a cannula on Monday, Wednesday, and Thursday evenings. The lipid droplet stage and oocyte diameter were determined using 15 oocytes from each female at each cannulation. a Frequency distribution of the lipid droplet stage from Monday to Thursday. b Changes in the lipid droplet stage from Monday to Wednesday. Values are the median of those for 27 (20 °C) or 6 (15 °C) females. c Changes in the oocyte diameter from Monday to Wednesday. d Rate of increase in oocyte diameter from Monday to Wednesday. In (c) and (d), values are the mean ± standard deviation of those for 27 (20 °C) or 6 (15 °C) females. e Changes in the lipid droplet stage from Wednesday to Thursday. Values are the median of those for 2 to 24 females. f Changes in the oocyte diameter from Wednesday to Thursday. Upper panel shows 20/20 °C and 20/15 °C groups, and lower panel 15/20 °C and 15/15 °C groups. g Rate of increase in oocyte diameter from Wednesday to Thursday. In (f) and (g), values are the mean ± standard deviation of those for 2–24 females
The frequency distribution of the lipid droplet stage from Monday to Thursday evening is shown in Fig. 4a. From Monday to Wednesday evening, the females reared at both 20 and 15 °C proceeded to stages 3–5. From Wednesday to Thursday evening, 24 females reared consecutively at 20 °C (20/20 °C group) proceeded to stages 4–8, among which stage 5 was the most common. Moreover, all three females reared alternatively at 15 °C (20/15 °C group), two females reared alternatively at 20 °C (15/20 °C group), and four females reared consecutively at 15 °C (15/15 °C group) proceeded to stage 5.
Changes in median lipid droplet stage, mean oocyte diameter, and rate of increase in oocyte diameter from Monday to Wednesday are shown in Fig. 4b–d, respectively. The median stage proceeded from stage 2 to 3 at 20 °C and from stage 3 to 5 at 15 °C. The mean oocyte diameter increased from 827.1 ± 35.4 to 883.1 ± 22.9 μm at 20 °C and from 869.5 ± 21.6 to 913.3 ± 18.3 μm at 15 °C. The rate of increase in oocyte diameter at 20 °C (6.9 ± 2.5%) was higher than that at 15 °C (5.1 ± 2.4%), although the difference was not significant between the temperature.
Changes in median lipid droplet stage, mean oocyte diameter, and rate of increase in oocyte diameter from Wednesday to Thursday are shown in Fig. 4e–g, respectively. The median stage proceeded from stage 3 to 5 in both 20/20 °C and 15/20 °C groups, while it remained at stage 5 in both 20/15 °C and 15/15 °C groups. Consequently, the median in all groups was stage 5 on Thursday evening. The mean oocyte diameter increased from 882.5 ± 22.0 to 939.3 ± 24.1 μm in the 20/20 °C group, from 887.3 ± 35.0 to 934.3 ± 29.8 μm in the 20/15 °C group, from 899.0 ± 12.7 to 958 ± 56.6 μm in the 15/20 °C group, and from 920.5 ± 17.4 to 954.5 ± 14.3 in the 15/15 °C group. The rate of increase in oocyte diameter was higher in the order of 15/20 °C (6.5 ± 4.8%), 20/20 °C (6.5 ± 2.4%), 20/15 °C (5.3 ± 1.0%), and 15/15 °C (3.7 ± 0.5%) groups. The increase rate in the females reared at 20 °C (15/20 °C and 20/20 °C groups) was higher than that in those reared at 15 °C (20/15 °C and 15/15 °C groups), although the difference was not significant between groups.
These changes in lipid droplet stage and oocyte diameter indicated that oocyte maturation accelerated and decelerated depending on the rearing temperature (20 or 15 °C) for each female; as a result, their maturational status (lipid droplet stage) was around stage 5 at 17α-OHP injection (Thursday evening).
Figure 5 shows the hatchability of the eggs obtained from the female eels reared at 15 and/or 20 °C for 3 days before 17α-OHP injection. The hatching rates in the 20/20 °C, 20/15 °C, 15/20 °C, and 15/15 °C groups were 43.4 ± 27.6%, 79.9 ± 8.0%, 28.6 ± 20.9%, and 67.2 ± 32.3%, respectively (Fig. 5a). Significant difference was not detected between groups. Thus, the lower hatchability in females reared at 20 °C for fewer days, as observed in Experiment 1, was ameliorated. The relationship between lipid droplet stage at 17α-OHP injection and hatching rate is shown in Fig. 5b. The hatching rates were not low even in females reared at 20 °C for fewer days.
a, b Hatchability of eggs obtained from Japanese eel Anguilla japonica reared at 15 and/or 20 °C for 3 days before 17α-hydroxyprogesterone (17α-OHP) injection (Experiment 2). On Monday, stage 2 and stage 3 females were reared at 20 and 15 °C, respectively. On Wednesday, stages 3–4 and stage 5 females were reared at 20 and 15 °C, respectively. Thus, the females were reared at 20/20 °C (n = 24), 20/15 °C (n = 3), 15/20 °C (n = 2), and 15/15 °C (n = 4) from Monday to Wednesday and from Wednesday to Thursday. The females were injected with 17α-OHP on Thursday evening for final oocyte maturation and ovulation. The eggs were stripped from ovulated females and used for artificial fertilization. a Hatching rates in the females reared at the four temperature conditions. Values are the mean ± standard deviation of those for 2–24 females. b The relationship between lipid droplet stage at 17α-OHP injection and hatching rate. To avoid overlap, the plots are slightly shifted along the x-axis
Discussion
In the current protocol for inducing final oocyte maturation and ovulation in Japanese eels, mature females with oocytes at the migratory nucleus stage are successively injected with SPE (for priming) and MIS (or its precursor) based on the weekly scheduled program, for example, SPE injection for priming on Wednesday evening, MIS injection on Thursday evening, and egg collection on Friday morning (Unuma et al. 2012; Kagawa et al. 2013; Kazeto et al. 2019). Such a weekly scheduled method was established because it is convenient for researchers to prepare for the operations on a fixed day and time. However, oocyte maturation does not always proceed as expected. Thus, the females are often administered MIS at inappropriate times, which is one of the causes of poor egg quality (Unuma et al. 2011). Therefore, in this study, we investigated the feasibility of optimizing the maturational status of female eels on a fixed day and time, by rearing at high and low temperatures. The results demonstrated that oocyte maturation progress in each female can be accelerated or decelerated by proper rearing at 15 and 20 °C.
In the first experiment, females with stage 2 lipid droplets were reared at 15 or 20 °C, as determined beforehand. Oocytes matured faster at 20 °C than at 15 °C from Monday to Wednesday and from Wednesday to Thursday (Fig. 2). As a result, the females briefly reared in warm water were at stages lower than stage 6, the optimum stage for MIS injection, and had poor hatching rates (Fig. 3).
In the second experiment, females with stage 2 and 3 lipid droplets were reared at 15 or 20 °C based on their maturational status. On Monday and Wednesday, the females with oocytes of higher stages were reared at 15 °C. Oocytes matured faster at 20 °C than at 15 °C from Monday to Wednesday and from Wednesday to Thursday (Fig. 4), as in Experiment 1. Consequently, the maturational status of most females was leveled at stage 5 at MIS precursor administration. Unfortunately, the number of females injected with the MIS precursor at stage 6 (optimum stage) was limited. This indicates that the division points for cold and warm rearing water (stages 2 and 3 on Monday evening and stages 4 and 5 on Wednesday evening) were not appropriate.
To develop a method for controlling oocyte maturation progress in each female for administering MIS to all females at the optimum stage, an appropriate way for rearing in cold and warm water should be determined in future studies. If the division points for rearing at cold and warm temperatures are modified, it may lead most females to the optimum stage (stage 6) for MIS (or its precursor) injection on the scheduled day and time. However, this method uses SPE to induce vitellogenesis and prime for final oocyte maturation. Recombinant gonadotropic hormones for anguillid species have been synthesized using various methods (Kamei et al. 2003; Kazeto et al. 2008; Kobayashi et al. 2010; Peñaranda et al. 2018). Recently, recombinant follicle-stimulating hormone and luteinizing hormone of Japanese eel have been commercially produced using Chinese hamster ovary cell lines (Kazeto and Tanaka 2020), which are more effective than conventional SPE or hCG injections for inducing oogenesis and spermatogenesis (Ohta et al. 2017; Kazeto and Tanaka 2020). When SPE is replaced with recombinant gonadotropins in future maturation induction protocols, the influence of temperature on oocyte maturation progress should be re-examined to modify the division points for rearing at cold and warm temperatures.
Egg quality in eels, as well as in other fish, is dependent on various factors (Kjørsvik et al. 1990; Brooks et al. 1997). For instance, the broodstock type (silver, feminized, or cultivated) and environmental conditions before maturation induction affect the responsiveness of females to repeated SPE injections and, as a result, egg quality (Ijiri et al. 1998; Okamura et al. 2008; Chai et al. 2010). Nutritional conditions during broodstock rearing influence the biochemical composition of eggs, such as vitamins and fatty acids, which alters hatchability and larval survival (Furuita et al. 2003, 2006, 2007, 2009; Yamamoto et al. 2011). Furthermore, the period from ovulation to insemination strongly affects fertility and hatchability because eel eggs rapidly over-ripen after ovulation (Ohta et al. 1996; Unuma et al. 2012). Therefore, egg quality does not depend entirely on the timing of final oocyte maturation and ovulation induction. As shown in Figs. 3b and 5b, hatchability was poorer in Experiment 1 than in Experiment 2, even in the females injected with MIS precursor at the same stage. The egg quality in Experiment 1 may have been reduced for some of the abovementioned reasons, such as environmental conditions before maturation induction. Moreover, some eggs obtained from the females injected with MIS precursor at stage 6 showed poor hatchability in both experiments, which may also have been caused by some of the abovementioned factors.
The poor quality of eggs obtained from anguillid species in captivity is an urgent matter to be addressed for developing eel propagation practices worldwide (Damsteegt et al. 2020; Kottmann et al. 2020). For decades, several studies on maturation induction have intensively been conducted in various eel species such as European eel A. anguilla (Müller et al. 2003; Pedersen 2004; Di Biase et al. 2017), New Zealand eel A. australis (Lokman and Young 2000; Lokman et al. 2015), and American eel A. rostrata (Oliveira and Hable 2010). To induce maturation in females, similar protocols as those used for Japanese eel are used, and unstable egg quality is a common problem to be addressed. Among these species, the influence of the timing of MIS injection on egg quality has been examined in A. anguilla, using the lipid droplet-based oocyte maturation scale (Palstra et al. 2005; da Silva et al. 2018). da Silva et al. (2018) reported that the range of lipid droplet diameters in oocytes obtained from European female eels producing high-quality eggs was 117–160 µm upon MIS administration, which is larger than that at stage 6 (90–110 µm) in Japanese eels. This means that the optimum maturational status for inducing final oocyte maturation and ovulation is different among species. If the optimum timing for MIS administration as well as the influence of temperature on oocyte maturation progress are clarified in each species, this method, using rearing at high and low temperatures to level the uneven oocyte maturation progress, may also be used in other anguillid species.
In summary, oocyte maturation progress can be accelerated or decelerated by rearing each female at 15 and/or 20 °C to correct the uneven oocyte maturation progress. It is not necessary to additionally rear them at a specific temperature if they are reared at low (around 15 °C) and high (around 20 °C) temperatures necessary for weekly SPE injections and ovulation induction, respectively. It would be possible to lead most females to the optimum maturation stage for MIS administration (stage 6) after determining the appropriate division points for rearing at cold and warm temperatures in future studies.
Change history
13 August 2021
A Correction to this paper has been published: https://doi.org/10.1007/s12562-021-01537-2
References
Arai T (2014) Do we protect freshwater eels or do we drive them to extinction? Springerplus 3:534
Brooks S, Tyler CR, Sumpter JP (1997) Egg quality in fish: what makes a good egg? Rev Fish Biol Fish 7:387–416
Chai Y, Tosaka R, Abe T, Sago K, Sago Y, Hatanaka E, Ijiri S, Adachi S (2010) The relationship between the developmental stage of oocytes in various seasons and the quality of the egg obtained by artificial maturation in the feminized Japanese eel Anguilla japonica. Aquacult Sci 58:269–278
da Silva FFG, Jacobsen C, Kjørsvik E, Støttrup JG, Tomkiewicz J (2018) Oocyte and egg quality indicators in European eel: Lipid droplet coalescence and fatty acid composition. Aquaculture 496:30–38
Damsteegt EL, Thomson-Laing G, Wylie MJ, Lokman PM (2020) Effects of estradiol and 11-ketotestosterone pre-treatment on artificial induction of maturation in silver female shortfinned eels (Anguilla australis). PLoS One 15:e0229391
Di Biase A, Lokman PM, Govoni N, Casalini A, Emmanuele P, Parmeggiani A, Mordenti O (2017) Co-treatment with androgens during artificial induction of maturation in female eel, Anguilla anguilla: effects on egg production and early development. Aquaculture 479:508–515
Dou S-Z, Yamada Y, Okamura A, Shinoda A, Tanaka S, Tsukamoto K (2008) Temperature influence on the spawning performance of artificially-matured Japanese eel, Anguilla japonica, in captivity. Environ Biol Fishes 82:151–164
Furuita H, Ohta H, Unuma T, Tanaka H, Kagawa H, Suzuki N, Yamamoto T (2003) Biochemical composition of eggs in relation to egg quality in the Japanese eel, Anguilla japonica. Fish Physiol Biochem 29:37–46
Furuita H, Unuma T, Nomura K, Tanaka H, Okuzawa K, Sugita T, Yamamoto T (2006) Lipid and fatty acid composition of eggs producing larvae with high survival rate in the Japanese eel. J Fish Biol 69:1178–1189
Furuita H, Hori K, Suzuki T, Sugita T, Yamamoto T (2007) Effect of n-3 and n-6 fatty acids in broodstock diet on reproduction and fatty acid composition of broodstock and eggs in the Japanese eel Anguilla japonica. Aquaculture 267:55–61
Furuita H, Unuma T, Nomura K, Tanaka H, Sugita T, Yamamoto T (2009) Vitamin contents of eggs that produce larvae showing a high survival rate in the Japanese eel Anguilla japonica. Aquacul Res 40:1270–1278
Horie N, Utoh T, Mikawa N, Yamada Y, Okamura A, Tanaka S, Tsukamoto K (2008) Influence of artificial fertilization methods of the hormone-treated Japanese eel Anguilla japonica upon the quality of eggs and larvae (comparison between stripping-insemination and spontaneous spawning methods). Nippon Suisan Gakkaishi 74:26–35 (in Japanese with English abstract)
Ijiri S, Kayaba T, Takeda N, Tachiki H, Adachi S, Yamauchi K (1998) Pretreatment reproductive stage and oocyte development induced by salmon pituitary homogenate in the Japanese eel Anguilla japonica. Fish Sci 64:531–537
Kagawa H, Tanaka H, Ohta H, Okuzawa K, Iinuma N (1997) Induced ovulation by injection of 17,20β-dihydroxy-4-pregnen-3-one in the artificially matured Japanese eel, with special reference to ovulation time. Fish Sci 63:365–367
Kagawa H, Iinuma N, Tanaka H, Ohta H, Okuzawa K (1998) Effects of rearing period in seawater on induced maturation in female Japanese eel Anguilla japonica. Fish Sci 64:77–82
Kagawa H, Tanaka H, Ohta H, Unuma T, Nomura K (2005) The first success of glass eel production in the world: basic biology on fish reproduction advances new applied technology in aquaculture. Fish Physiol Biochem 31:193–199
Kagawa H, Sakurai Y, Horiuchi R, Kazeto Y, Gen K, Imaizumi H, Masuda Y (2013) Mechanism of oocyte maturation and ovulation, and its application to seed production in the Japanese eel. Fish Physiol Biochem 39:13–17
Kaifu K, Yokouchi K (2019) Increasing or decreasing?–Current status of the Japanese eel stock. Fish Res 1:220–105348
Kamei H, Ohira T, Yoshiura Y, Uchida N, Nagasawa H, Aida K (2003) Expression of a biologically active recombinant follicle stimulating hormone of Japanese eel Anguilla japonica using methylotropic yeast. Gen Comp Endocrinol 134:244–254
Kazeto Y, Tanaka T (2020) Study on artificial induction of maturation and ovulation in Japanese eel using the recombinant gonadotropins: application to the seed production. Nippon Suisan Gakkaishi 86:364–366 (in Japanese)
Kazeto Y, Kohara M, Miura T, Miura C, Yamaguchi S, Trant JM, Adachi S, Yamauchi K (2008) Japanese eel follicle-stimulating hormone (Fsh) and luteinizing hormone (Lh): production of biologically active recombinant Fsh and Lh by Drosophila S2 cells and their differential actions on the reproductive biology. Biol Reprod 79:938–946
Kazeto Y, Tanaka T, Suzuki H, Ozaki Y, Fukada H, Gen K (2019) Development and validation of enzyme-linked immunosorbent assays specific for follicle-stimulating hormone and luteinizing hormone in Japanese eel. Fish Sci 85:829–837
Kjørsvik E, Mangor-Jensen A, Holmefjord I (1990) Egg quality in fishes. Adv Mar Biol 26:71–113
Kobayashi M, Hayakawa Y, Park W, Banba A, Yoshizaki G, Kumamaru K, Kagawa H, Kaki H, Nagaya H, Sohn YC (2010) Production of recombinant Japanese eel gonadotropins by baculovirus in silkworm larvae. Gen Comp Endocrinol 167:379–386
Kottmann JS, Jørgensen MGP, Bertolini F, Loh A, Tomkiewicz J (2020) Differential impacts of carp and salmon pituitary extracts on induced oogenesis, egg quality, molecular ontogeny and embryonic developmental competence in European eel. PLoS One 15:e0235617
Lokman PM, Young G (2000) Induced spawning and early ontogeny of New Zealand freshwater eels (Anguilla dieffenbachii and A. australis). NZJ Mar Freshwat Res 34:135–145
Lokman PM, Wylie MJ, Downes M, Di Biase A, Damsteegt EL (2015) Artificial induction of maturation in female silver eels, Anguilla australis: The benefits of androgen pre-treatment. Aquaculture 437:111–119
Masuda Y, Imaizumi H, Oda K, Hashimoto H, Usuki H, Teruya K (2012) Artificial completion of the Japanese eel, Anguilla japonica, life cycle: challenge to mass production. Bull Fish Res Agency 35:111–117
Müller T, Váradi B, Horn P, Bercsényi M (2003) Effects of various hormones on the sexual maturity of European eel (Anguilla anguilla L.) females from farm and lakes. Acta Biol Hung 54:313–322
Ohta H, Izawa T (1996) Diluent for cool storage of the Japanese eel (Anguilla japonica) spermatozoa. Aquaculture 142:107–118
Ohta H, Kagawa H, Tanaka H, Okuzawa K, Hirose K (1996) Changes in fertilization and hatching rates with time after ovulation induced by 1720β-dihydroxy-4-pregnen-3- one in the Japanese eel, Anguilla japonica. Aquaculture 139:291–301
Ohta H, Kagawa H, Tanaka H, Okuzawa K, Iinuma N, Hirose K (1997) Artificial induction of maturation and fertilization in the Japanese eel, Anguilla japonica. Fish Physiol Biochem 17:163–169
Ohta H, Sato Y, Imaizumi H, Kazeto Y (2017) Changes in milt volume and sperm quality with time after an injection of recombinant Japanese eel luteinizing hormone in male Japanese eels. Aquaculture 479:150–154
Okamura A, Yamada Y, Horie N, Utoh T, Mikawa N, Tanaka S, Tsukamoto K (2008) Effects of silvering state on induced maturation and spawning in wild female Japanese eel Anguilla japonica. Fish Sci 74:642–648
Oliveira K, Hable WE (2010) Artificial maturation, fertilization, and early development of the American eel (Anguilla rostrata). Can J Zool 88:1121–1128
Palstra AP, Cohen EGH, Niemantsverdriet PRW, van Ginneken VJT, van den Thillart GEEJM (2005) Artificial maturation and reproduction of European silver eel: development of oocytes during final maturation. Aquaculture 249:533–547
Pedersen BH (2004) Fertilisation of eggs, rate of embryonic development and hatching following induced maturation of the European eel Anguilla anguilla. Aquaculture 237:461–473
Peñaranda DS, Gallego V, Rozenfeld C, Herranz-Jusdado JG, Pérez L, Gómez A, Giménez I, Asturiano JF (2018) Using specific recombinant gonadotropins to induce spermatogenesis and spermiation in the European eel (Anguilla anguilla). Theriogenology 107:6–20
Sato N, Kawazoe I, Suzuki Y, Aida K (2006) Effects of temperature on vitellogenesis in Japanese eel Anguilla japonica. Fish Sci 72:961–966
Tachiki H, Nakagawa T, Tamura K, Hirose K (1997) Effects of oral administration of estradiol-17β to young on gonadal sex and growth of Japanese eel Anguilla japonica. Suisanzoshoku 45:61–66 (in Japanese with English abstract)
Tanaka H (2015) Progression in artificial seedling production of Japanese eel Anguilla japonica. Fish Sci 81:11–19
Tanaka H, Kagawa H, Ohta H (2001) Production of leptocephali of Japanese eel (Anguilla japonica) in captivity. Aquaculture 201:51–60
Tanaka H, Kagawa H, Ohta H, Unuma T, Nomura K (2003) The first production of glass eel in captivity: fish reproductive physiology facilitates great progress in aquaculture. Fish Physiol Biochem 28:493–497
Tanaka H, Nomura K, Yamamoto T, Oku H (2006) Development of artificial diets and rearing systems for eel larvae - the first success of production of glass eel in captivity—. Bull Fish Res Agen Suppl 5:63–69 (in Japanese with English abstract)
Unuma T, Kondo S, Tanaka H, Kagawa K, Nomura K, Ohta H (2004) Determination of the rates of fertilization, hatching and larval survival in the Japanese eel, Anguilla japonica, using tissue culture microplates. Aquaculture 241:345–356
Unuma T, Kondo S, Tanaka H, Kagawa K, Nomura K, Ohta H (2005) Relationship between egg specific gravity and egg quality in the Japanese eel, Anguilla japonica. Aquaculture 246:493–500
Unuma T, Hasegawa N, Sawaguchi S, Tanaka T, Matsubara T, Nomura K, Tanaka H (2011) Fusion of lipid droplets in Japanese eel oocytes: stage classification and its use as a biomarker for induction of final oocyte maturation and ovulation. Aquaculture 322–323:142–148
Unuma T, Sawaguchi S, Hasegawa N, Tsuda N, Tanaka T, Nomura K, Tanaka H (2012) Optimum temperature of rearing water during artificial induction of ovulation in Japanese eel. Aquaculture 358–359:216–223
Yamamoto K, Yamauchi K (1974) Sexual maturation of Japanese eel and production of eel larvae in the aquarium. Nature 251:220–222
Yamamoto T, Furuita H, Matsunari H, Sugita T, Mineshima F, Nakagawa T, Ishida T (2011) Effects of fortification of nutrients to female broodstock by diet alone and by both diet and direct injection on the egg quality of Japanese eel Anguilla japonica. Aquacult Sci 59:123–130
Yoshikawa M (2012) Improvement in hatching rates in the Japanese eel Anguilla japonica by the control of rearing temperatures in the late stage of maturation in the female parents. Aquaculture 338–341:223–227
Acknowledgements
We are grateful to the staff of the Shizuoka Prefectural Research Institute of Fishery and Ocean for technical assistance. We also thank Drs. Sayumi Sawaguchi, and Natsuki Hasegawa at the Japan Fisheries Research and Education Agency for helpful discussion. This study was supported by a grant-in-aid (Development of seed production of the eel) from the Ministry of Agriculture, Forestry and Fisheries, Government of Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original version of this article was revised: In this article the black down arrows in Fig. 2a. that should indicate white letters ‘20°C’ inside the arrows have disappeared. It has now appeared correctly in this article.
Rights and permissions
About this article
Cite this article
Tanaka, T., Adachi, S., Nomura, K. et al. Effects of rearing temperature manipulation on oocyte maturation progress in Japanese eel. Fish Sci 87, 681–691 (2021). https://doi.org/10.1007/s12562-021-01531-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12562-021-01531-8