Abstract
Hydroids are benthic cnidarians that have adapted to diverse marine environments through a wide variety of reproductive strategies and high phenotypic plasticity. This study aims at enhancing our understanding of the benthic hydroid diversity in underexplored areas of Brazil by analyzing hundreds of previously unexamined specimens. The study encompasses material collected from stations spanning the continental shelf and slope, ranging from the intertidal zone to a depth of 3,800 m across nine states in the North and Northeast regions of Brazil. This study represents the most extensive survey of benthic hydrozoans ever conducted in Brazil in terms of the sampled area, number of specimens collected, and bathymetric range. A total of 59 hydroid species, belonging to 32 genera and 14 families, were identified. In the states with the highest number of stations, we observed a greater number of specimens collected and, consequently, a greater richness, highlighting the importance of faunal surveys in neglected areas. The number of records and species richness declined with increasing depth, with 0–50 m exhibiting the highest number of records and species richness. Biogenic substrates, particularly sponges, hosted numerous epizoic hydroids. The significance of conducting new faunal surveys in underexplored areas of the Brazilian coastline is emphasized to address the knowledge gap regarding benthic hydrozoans in a vast area of the western South Atlantic.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hydroids are known from all marine habitats worldwide, spanning from shallow to abyssal waters, pole to pole (Calder 1998; Fernandez et al. 2020). Their wide distribution and relatively high abundance are supposed to be related to various factors, including asexual reproduction, a wide variety of life cycles, high phenotypic plasticity, and the diversity of biological traits (Cornelius 1995a, b; Cunha et al. 2016, 2022; Di Camillo et al. 2017; Fernandez et al. 2023). Some hydroid species form extensive three-dimensional “forests” for a wide range of organisms, from viruses to vertebrates (Gomes-Pereira and Tempera 2016; Di Camillo et al. 2017). However, the few diagnostic characters of hydroids have also led to taxonomic challenges, such as species with perplexing distribution records and puzzled phylogenetic affinities inconsistent with some presumed diagnostic characteristics (Maronna et al. 2016; Cunha et al. 2017).
Faunal knowledge of hydroids is highly heterogeneous across the world. Some coastal areas have been well sampled in important faunal studies (e.g., Millard 1975; Migotto 1996; Calder 1988, 1991, 1997, 1998; Schuchert 2003; Vervoort and Watson 2003; Gil and Ramil 2021), while others remain unexplored or uninvestigated. The South Atlantic is one such region (Fernandez et al. 2020), and several of these neglected areas occur along the extensive Brazilian coastline (Oliveira et al. 2016), with its wide variety of habitats and microclimates. This knowledge deficit makes it difficult to conserve, manage, and monitor the biodiversity of these areas (Amaral and Jablonski 2005). For example, only a small portion of the 342 hydroid species known from Brazil was recorded in the Northeast region (Oliveira et al. 2016; Mendonça et al. 2022), and this knowledge is derived from a few faunal studies (Maÿal 1983; Pires et al. 1992; Calder and Maÿal 1998; Kelmo and Santa-Isabel 1998; Shimabukuro et al. 2006; Mendonça et al. 2022). The situation is even worse in the North region, for which there are no published records of non-calcareous benthic hydroids on the Amazon continental shelf, with the northernmost record being in Ceará State (Oliveira et al. 2016). This knowledge gap contrasts with the great potential biodiversity for this region, given the geological and biological characteristics of the reefs along the North and Northeast coast of Brazil (Carneiro et al. 2022; Vale et al. 2022), which are expected to harbor a rich hydroid diversity.
This study aims to address this gap by accessing new collections of materials, shedding light on the diversity of hydroid communities along the North and Northeast coast of Brazil. This effort contributes to our knowledge of benthic hydrozoans in this extensive area of the western South American Atlantic.
Material and Methods
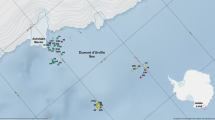
The study area comprised the Amazonia and Northeast Brazil marine ecoregions (Spalding et al. 2007), from Amapá (northernmost Brazilian state, 4.500000; -50.050000) to Alagoas State (-9.218975; -35.0564197). We studied material from 224 stations distributed along the continental shelf and the slope, from the intertidal zone to 3,800 m (Supplementary material). These stations were located within the sedimentary basins of Amazonas, Pará-Maranhão, Barreirinhas, Ceará, Potiguar, Paraíba, and Sergipe/Alagoas (Fig. 1).
Hydroid specimens were collected through various methods, including Van Veen sampling, dredging, or manually by SCUBA diving, as part of different projects aiming to characterize the benthic megafauna and abiotic parameters of Brazilian marine ecosystems over the last five decades. These collections were conducted without the presence of Hydrozoa specialists, potentially impacting the quality of collection, sorting, and preservation of some specimens. The 738 studied specimens are deposited in the Cnidarian collection at the Oceanography Museum Prof. Petrônio Alves Coelho at the Federal University of Pernambuco. We calculated the frequency of species occurrence across all stations as the percentage of each species’ records in relation to the totality of sampled states; and the frequency of occurrence within the sampled states as the percentage of states with records of each species among the nine states sampled. Canonical correspondence analysis using four metrics (Wilks 1935; Pillai 1956; Anderson 1984; Muller & Peterson 1984; Rao 2002) was performed to evaluate potential correlations between the independent variable, bathymetry, and the dependent variables (number of records and richness). We used the “igraph” package (Csardi and Nepusz 2006) to identify species groupings by state.
The specimens were preserved in 70% ethanol and were examined by using stereomicroscopes and compound microscopes. Materials were primarily identified based on the original species descriptions and relevant literature, with terminology following Millard (1975), Cornelius (1995a, b), and Bouillon et al. (2006). We have adopted the classification of Leptothecata suggested by Maronna et al. (2016), while for anthoathecates we have followed the classification available at WoRMS (2023).
Results
A total of 739 hydroid specimens were recorded along 3,382 km of coastline across nine states in the North and Northeast regions of Brazil, from Amapá to Alagoas. Of the 224 surveyed stations, approximately 86% (n = 193) were located on the continental shelf within depths of 0–100 m, with only 31 stations distributed on the slope.
We are aware of the caveat that the collection methods varied, and this likely influenced different richness results for each station, as well as the collection effort for each point. However, our intention was to compare to the best of our knowledge the hydroid communities in different geographic areas, and we do this conservatively, without assuming that these biases indicate significant absolute richness. The coast of Pernambuco state was the most thoroughly surveyed, with 48 stations (21%), followed by Pará (39 stations / 17%), and Maranhão (34 stations / 15%) (Table 1). These three states accounted for the highest number of collected hydroid specimens, with Pará having 191 records, Maranhão with 165 records, and Pernambuco with 93 records (Fig. 2). However, the highest species richness was found in Rio Grande do Norte, with 39 species (representing 66% of total richness), followed by Maranhão with 34 species (58% of richness), and Pará with 33 species (56% of richness) (Fig. 2). The reef habitats of the study area were found to provide favorable conditions for a diverse hydroid habitat. Some stations exhibited great richness, with up to 26 collected species. The results of the Canonical correspondence analysis showed no correlation between bathymetry and the number of species and records, with all metrics yielding similar significance value (p = 0.8164557).
Based on the grouping analysis within the study area, Rio Grande do Norte was characterized as having the highest number of exclusive species, followed by Paraíba, Maranhão, Pará, and Ceará, respectively. In contrast, Amapá, Piauí, Pernambuco and Alagoas did not exhibit species exclusive to these states. Furthermore, most of the species occurring in Amapá, Pará, Ceará, and Maranhão (states mainly located in the center of the diagram) were found in multiple states, illustrating species sharing between these regions (Fig. 3).
We found a total of 59 distinct hydroid species belonging to 32 genera and 15 families (Table 1 and Figs. 4 and 5). Of these species, only two are “anthoathecates”, while the rest belong to Leptothecata. Of these species, 25 are new records for the Northern coast, seven species for the North-eastern coast, two are new records for Brazil, eight for the South Atlantic, and two for the Atlantic Ocean (Table 1). However, eight morphotypes could not be identified to the species level due to damaged or fragmented colonies with limited distinguishing features and no reproductive structures. Out of the 59 morphotypes studied, 21 were fertile at the time of collection, and only four are known to produce free medusa or medusoid stages; the other 38 are species with fixed gonophores.
Regarding the distribution of the hydroids, 54% were circumglobal, while 29% were restricted to the Atlantic Ocean and adjacent seas, and 15% were recorded in both the Atlantic and another ocean (Table 1). It is worth noting that 80% (12) of the fifteen species of Sertulariidae recorded here have a circumglobal distribution. Conversely, among the species with distributions restricted to the Atlantic, we recorded all the three species of Thyroscyphidae and five out of the ten species of Aglaopheniidae.
The families Aglaopheniidae (192 colonies, comprising 26% of the total number of colonies), Thyroscyphidae (189, 25.5%, with Thyroscyphus ramosus, one of the three species of the family, being the most common), and Sertulariidae (147, or 20%) exhibited the highest number of recorded specimens (Fig. 4). Sertulariidae and Aglaopheniidae exhibited the highest richness (15 and 12 species, respectively). The most prevalent genera were Thyroscyphus (137 colonies) and Aglaophenia (131), primarily due to the presence of T. ramosus (122 colonies; frequency 16.2%) and Aglaophenia rhynchocarpa (67; frequency 9%), both of which were the most common species and the only species found in all states sampled from Amapá to Alagoas (Table 1). This was followed by Sertularelloides cylindritheca (52; frequency 7.0%), Hincksella formosa (51; frequency 6.7%), Aglaophenia latecarinata (46; frequency 6.2%), and Amphisbetia distans (42; frequency 5.6%), all of which exhibited a wide distribution in the study area, occurring respectively in 44.4%, 66.7% and 66.7% of the sampled states. Conversely, several species had isolated records, indicating a restricted distribution in the sampled area (Table 1).
Hydroids were collected from the intertidal zone to a depth of 3,888 m (Fig. 6). Among the observed diversity, 15 species had a bathymetric distribution range ≥ 300 m, with Eudendrium carneum, A. distans, H. formosa, S. cylindritheca, and T. ramosus recorded from 0 to 3,888 m (= eurybathic; see Table 1). On the other hand, species such as Parawrightia robusta and Gymnangium sinuosum had a limited bathymetric range, occurring at depths of only 39–43 m and 62–75 m, respectively (= stenobathic; see Table 1). Some hydroids were collected only once and, therefore, have a single depth record. The number of records and species richness declined with increasing depth. The range between 0–50 m had the highest number of records (387 records; 52% of the total number of records), as well as the greatest species richness (41 species; 70% of the total number of species), followed by the 51–100 m range with 239 records (32%) and 33 species (56%), and the 101–200 m range with 40 records (5%) and 29 species (49%) (Fig. 6). Only 16 species were collected in regions deeper than 201 m. Most species occurred only at the continental shelf (0–100 m) or on the upper slope (101–200 m). Conversely, nine species occurred exclusively on the slope at various depths (Table 1). Additionally, we extended the known bathymetry of 26 species in South America to deeper reefs in the mesophotic zone and the deep sea.
Thirty-six morphotypes of epizoic hydroids colonized biogenic substrates (Table 2), displaying a higher frequency in substrates such as sponges, other hydroids, and algae, but were also observed on mollusks, corals, bryozoans, and polychaete tubes. Sponges were the most frequent used basibiont, serving as substrates for 15 hydroid species. Oceanapia bartschi (Laubenfels, 1934) was the most frequently colonized sponge substrate, hosting 11 hydroid species. Nonetheless, the most frequent association was observed among hydroids themselves, with 19 species observed attached to other hydroids. Hydroids with larger and more robust colonies, such as T. ramosus and S. cylindritheca, were the most frequent substrates for other hydroids. Thyroscyphus ramosus was observed as a basibiont of 12 hydroid morphotypes. Another important cnidarian basibiont was the octocoral Nicella spicula, hosting eight different hydroid species (Table 2).
The majority of the epizoic hydroids presented small-sized colonies and were substrate-generalists, as the same species was observed on various distinct basibionts. For example, the hydroid H. formosa thrived on nine distinct substrates, while Aglaophenia species were also observed on a wide variety of substrates (Table 2). However, it is important to acknowledge that certain epizoic hydroids observed in this study were exclusively found on a single substrate. Nevertheless, we must refrain to conclusively state that they are substrate-specific, as this outcome could be influenced by sampling bias.
Discussion
This study represents the most comprehensive hydroid survey in Brazil to date in terms of the number of specimens studied, the length of the coastline covered, and bathymetric range covered (Oliveira et al. 2016). The richness of hydroids observed in this study enhances our understanding of the poorly studied benthic biodiversity in underexplored areas of Brazil (Aued et al. 2018). It particularly sheds light on the Northern and North-eastern coastlines, where research has been historically limited. Furthermore, it expands our knowledge of hydrozoans in the South Atlantic by documenting new occurrences and broadening the geographic and bathymetric range of most species (Table 3). The identification of 33 species along the Northern coast represents a substantial increase of 513% in the known hydroid species for this region, which previously only documented four recorded benthic calcified hydroid species. Similarly, among the 57 morphotypes identified in the Northeast, 20 are new records, contributing to a total of 144 known species in the area (Oliveira et al. 2016; Mendonça et al. 2022). It is worth noting that the Brazilian hydroid fauna exhibits greater species richness along the Southeastern and Southern coasts (216 species reported) due to greater sampling effort carried out in these areas (Oliveira et al. 2016).
In general, we observed that in states with the highest number of stations, there was a greater number of specimens collected and, consequently, greater richness. However, the highest richness was found in the Potiguar Basin area in Rio Grande do Norte State, which is characterized by rich hydroid and reef organism ecosystems, despite accounting for only the fifth-highest number of sampled stations. The benthos of the Potiguar Basin has recently become the focus of attention (Larrazábal-Filho et al. 2015, 2018; Cavalcanti et al. 2018; Dias et al. 2019) and represents one of the areas with the highest richness of deep-sea coral species in Brazil, offering substrates such as rocks, sponges and dead scleractinians (Cordeiro et al. 2020). These authors have observed the presence and abundance of several mesophotic scleractinians from depths from 102 to 108 m, often associated with sponges and gorgonian species of the genus Nicella, as well as other octocoral species. In our study, eight hydroid species were found to be epizoic on Nicella (Table 2). Black corals and octocorals form dense assemblages, creating complex habitats for associated species (Roberts et al. 2006). These dense assemblages, along with the proliferation of microhabitats, may explain the high number of hydroid species observed at some stations in the Potiguar Basin. For example, there was the occurrence of 26 hydroid species at a depth of 102 m during a single season, accompanied by the presence of 18 coral species at the same station (Cordeiro et al. 2020). This suggests favorable conditions for hydroid microhabitats and a rich diversity of other reef organisms.
The low number of anthoathecate hydroids is likely due to unfavorable conditions for collection, sorting, and poor specimen preservation. Anthoathecates are generally delicate animals due to the absence of perisarc protecting soft parts that are important diagnostic characters for the taxonomy of the group (Petersen 1990), and the absence of experts in the group during collection makes them more susceptible to improper processing and suboptimal preservation conditions, basically destroying or overlooking the materials. Additionally, the lack of important taxonomic characters poses challenges in identifying the specimens (Maggioni et al. 2018). Therefore, the richness of anthoathecate species must be considered underestimated at best.
The ability of hydroids to adapt to different conditions, including temperature and depth, as well as the organizational diversity of their colonies and life cycles, likely enables them to occur in all oceans (Leclère et al. 2007). In this study, slightly over half of the collected species exhibited a circumglobal distribution. This may support the great dispersal capacity of hydroids through rafting and advective life stages (Cornelius 1992). Interestingly, only four of the identified species in this study release medusae or medusoid stages (Table 1), indicating that life stages may not play a decisive role in wider distribution. Another explanation for wide distributions is an insufficient taxonomic knowledge at the species level (Cunha et al. 2020, 2022). This taxonomic uncertainty is also influenced by the lack or scarcity of diagnostic taxonomic characteristics and the reliance on plastic characteristics based on trophosomal and gonosomal morphology, as well as the limited understanding of many of their life cycles (Lindner et al. 2011). Indeed, some of these widely distributed species may represent species complexes, such as those occurring in the families Clytiidae and Obeliidae (Cunha et al. 2017) and some species of Sertulariidae (Moura et al. 2011).
The large number of Sertulariidae species collected in this study and their circumglobal distribution lend support to the wide distribution observed for most species of this family elsewhere (Bouillon et al. 2006). Conversely, 15 species found in this study are exclusively recorded in the Atlantic Ocean, including three Thyroscyphidae species and half of the Aglaopheniidae species. Assuming a general high dispersal capacity of hydroids and the plasticity of their life cycles may not be applicable in this case (Cornelius 1990). Interestingly, this distribution pattern of endemicity could be explained by the general system of ocean currents across the Atlantic (Miranda et al. 2015), which could play a role in the distribution of planktonic hydrozoans (Rodriguez et al. 2017), or by some historical factors influencing the distribution of certain biota (e.g., Miranda et al. 2021). Lastly, the distribution of the hydroid species could also be driven by trait-enviroment relationships, offering an ecological perspective to this conundrum (Fernandez et al. 2023).
The hydroid T. ramosus was the most common and widely distributed species in the study area, playing an important role in the reef ecosystems by providing substrate for the establishment of other hydrozoans’ larvae and forming associations with various reef organisms such as algae, sponges, mollusks, and polychaetes. This species has a broad distribution in Brazilian waters and is one of the most frequently observed and conspicuous hydroids in reef ecosystems, with robust colonies (Shimabukuro and Marques 2006), reaching heights exceeding 25 cm (this study). Due to their large and branched colonies, they provide substrate for the attachment and use of several organisms, particularly other hydroids (Migotto 1996). The fact that T. ramosus and A. rhynchocarpa were the two most frequent species at the sampled stations corroborates their wide distribution in the Western Atlantic, extending from Florida, USA (Calder 2013) to Santa Catarina, Brazil (Oliveira et al. 2016), including the Gulf of Mexico (Calder and Cairns 2009), the Caribbean (Galea 2008), and the West African coast (Vervoort 1959). Therefore, the records from the Amazon shelf, located in the northernmost part of the Brazilian coast, fill the knowledge gap regarding the distribution of these species in the Western Atlantic.
Richness of hydroid species and the total number of records decrease with increasing depth. The highest richness and number of records were found between 0 and 50 m (accounting for 69.5% of richness), consistent with previous studies (Calder 1998; Genzano et al. 2009; Ajala-Batista et al. 2020). This trend was followed by the 51–100 m range (representing 56% of richness), and the 101–200 m range (49% of richness). Although 86% of the sampled stations are located on the continental shelf at depths ranging from 0 to 100 m, the inclusion of stations on the slope during this study greatly expanded the bathymetric range for several species (Table 1). Knowledge of hydroid diversity in mesophotic and deep-sea reefs in Brazil is still incipient, and further studies in these areas are needed to fully understand their actual diversity, particularly considering that the range between 101 and 200 m is recognized as one of the richest in terms of hydroid fauna (Calder 1998). In Brazil, few records have been obtained for depths up to 375 m (Fernandez et al. 2020), and most existing records were collected at depths up to 90 m (Oliveira et al. 2016). These records were obtained during oceanographic expeditions such as HMS Challenger and Vega (Allman 1883, 58 m; Jäderholm 1903, 56 m, respectively) or through systematic studies on hydroid fauna in specific areas along the Brazilian coast (e.g., Maÿal 1983, up to 80 m; Migotto 1996, up to 25 m; Grohmann et al. 2003, up to 90 m).
Overall, approximately 1/4 of the hydroids in the study area displayed eurybathic characteristics, occurring from the intertidal zone up to 3,888 m, while 3/4 were stenobathic. These findings diverge from previous studies, which observed that 59% of Atlantic hydroid records occurring from 50 m were eurybathic (Fernandez and Marques 2018). Some species recorded up to 90 m on the continental shelf of southeastern Brazil have also been suggested as eurybathic (Grohmann et al. 2003), including species also found on the Amazon shelf, such as Plumularia margaretta, Lytocarpia tridentata, and Sertularelloides cylindritheca. The reduced number of eurybathic species in our study is likely due to sampling bias, as many species were only collected a few times, making an accurate analysis of their bathymetric range challenging. In the study area, hydroids such as E. carneum, A. rhynchocarpa, A. distans, H. formosa, S. cylindritheca, and T. ramosus are frequently found at different depths and occur from the intertidal zone to 3,888 m (Table 1). These species exhibit more restricted ranges in the western South Atlantic (Table 1) and, while common in shallow waters, they also have records in the deep North Atlantic (e.g., E. carneum up to 137 m, Fraser 1944; A. rhynchocarpa up to 365 m, Vervoort 1968; T. ramosus up to 457 m, Nutting 1915).
In addition to the 33 epibiotic hydroid species, a greater number may have been recorded if some hydroids from the analyzed collections had not detached from their original substrates during sample collection and sorting. Sponges and hydroids were the main substrates in the sampled reefs. Sponges possess complex canal systems that maintain continuous water flow, providing an ideal habitat for numerous hydroid species that can live on the surface or inside of sponge (Puce et al. 2005). Among the basibiont sponges on the Amazon shelf, the sponge O. bartschi, with its arborescent shape and prominent oscular tubes that facilitate continuous water flow, resulted as the most commonly inhabited by hydroids. Of the eleven hydroid species found on O. bartschi in this study, eight are also present in the Caribbean Sea, where this sponge also occurs (Rützler et al. 2014). This suggests that O. bartschi serves as an important substrate, promoting connectivity between the hydroid fauna of the Brazilian coast and that of the Caribbean Sea. The observed associations between hydroids and sponges were not species-specific, indicating that the selection of sponge substrates is also influenced by random larval settlement, as the hydroid hydrorhiza grew superficially on the sponges.
Conversely, hydroids serve as suitable substrates for smaller hydroid species, with epizoics benefiting from the protection offered by the host species' cnida against predators (Gili and Hughes 1995). In this study, nineteen out of the 33 epibiotic species occurred on other hydroids, a pattern well documented in the literature (e.g. Vervoort 1968; Calder 1991; Migotto 1996; Oliveira et al. 2016). Associations between hydroids are a successful strategy in areas characterized by high diversity and intense competition for space (Genzano and Rodriguez 1998). The erect, large, and sturdy colonies of hydroids form three-dimensional “forests” comparable to those created by gorgonians, fostering biotic interactions with various organisms, including other hydroid species (Di Camillo et al. 2017). This highlights the importance of hydroids as a key component of shallow and mesophotic areas. The majority of epibiont hydroids observed had small colonies measuring up to 3.0 mm (e.g., A. latecarinata, H. formosa, A. distans – Migotto 1996), suggesting that size is a limiting factor in the selection of basibionts by hydroid larvae. Some hydroids with larger colonies, such as Synthecium tubithecum, Sertularella diaphana, T. ramosus, and S. cylindritheca (Calder 1991; Migotto 1996), served as substrates for smaller bryozoans and other smaller hydroids, suggesting that the size of both the basibiont and epibiont colonies plays a role in substrate selection. Larger hydroid colonies facilitate the settlement of larvae of smaller species, as well as the settlement of non-larval propagules and overlapping vegetative growth. For instance, the hydroid T. ramosus was observed serving as a substrate for 12 different hydroid species, benefiting from settling larvae on stable substrates with ample space for colony development in a reef environment with strong hydrodynamics. Large colonies are common basibionts for many taxa because they provide additional surface area above the substrate, especially in the basal portions where organisms are particularly abundant (Hughes 1975; Garcia et al. 2009).
The results of our study suggest the larval settlement of hydroid larvae on the colonies of other hydroids occurs randomly, indicating that hydroids can be classified as substrate-generalists. Most species were observed on more than one basibiont, and records of these species on various other organisms exist in the literature (e.g., Vervoort 2006; Calder 2019). Aglaophenia species, in particular, demonstrate a generalist nature and grow on diverse facultative substrates (Calder 1997). In addition to being observed on five other hydroid species, two sponges, and a macroalgae in this study, Aglaophenia latecarinata has been previously observed as an epibiont on other substrates (e.g., Sargassum spp., Migotto 1996 and Calder 2013; sponge and bivalve shells, Galea 2010).
References
Ajala-Batista L, de Miranda LD, Haddad MA (2020) Diversity of estuarine and marine hydroids (Cnidaria, Hydrozoa) from subtropical ecosystems of Brazil. Mar Biodivers 50:97. https://doi.org/10.1007/s12526-020-01133-0
Allman GJ (1883) Report on the Hydroida dredged by H.M.S. Challenger during the years 1873–76. Part I. Plumulariidae. Report on the Scientific Results of the Voyage of H.M.S. Challenger 1873–76 Zoology 7(20):1–55. https://doi.org/10.5962/bhl.title.11299
Amaral A, Jablonski S (2005) Conservation of Marine and Coastal Biodiversity in Brazil. Conserv Biol 19:625–631. https://doi.org/10.1111/j.1523-1739.2005.00692.x
Anderson TW (1984) An introduction to multivariate statistical analysis, 2d edn. John Wiley, New York
Aued AW, Smith F, Quimbayo JP, Cândido DV, Longo GO et al (2018) Large-scale patterns of benthic marine communities in the Brazilian Province. PLoS ONE 13(6):e0198452. https://doi.org/10.1371/journal.pone.0198452
Bouillon J, Gravili C, Pagès F, Gili JM, Boero F (2006) An Introduction to Hydrozoa. Mémoires Du Muséum D’histoire Naturelle, Paris. https://doi.org/10.1086/586983
Calder DR (1988) Shallow-water hydroids of Bermuda: the Athecatae. Life Sci Contrib R Ont Mus 148:1–107. https://doi.org/10.5962/bhl.title.52225
Calder DR (1991) Shallow-water hydroids of Bermuda: the Thecatae, exclusive of Plumularioidea. Life Sci Contrib R Ont Mus 154:1–140. https://doi.org/10.2307/1352274
Calder DR (1997) Shallow-water hydroids of Bermuda: superfamily Plumularioidea. Life Sci Contrib R Ont Mus 161:1–85
Calder DR (1998) Hydroid diversity and species composition along a gradient from shallow waters to deep sea around Bermuda. Deep Sea Res Part I Oceanogr Res Pap 45:1843–1860. https://doi.org/10.1016/s0967-0637(98)00044-2
Calder DR (2013) Some shallow-water hydroids (Cnidaria: Hydrozoa) from the central east coast of Florida, USA. Zootaxa 3648(1):1–72. https://doi.org/10.11646/zootaxa.3648.1.1
Calder DR (2019) On a collection of hydroids (Cnidaria, Hydrozoa) from the southwest coast of Florida, USA. Zootaxa 4689:1–141. https://doi.org/10.11646/zootaxa.4689.1.1
Calder DR, Cairns SD (2009) Chapter 19. Hydroids (Cnidaria: Hydrozoa) of the Gulf of Mexico. In: Felder DL, Camp DK (eds) Gulf of Mexico. Origins, waters, and biota, Volume 1, Biodiversity. Texas A&M University Press, College Station, Texas, pp 381–394
Calder DR, Maÿal E (1998) Dry season distribution of hydroids in a small tropical estuary, Pernambuco, Brazil. Zool Verh 323:69–78
Di Camillo CG, Bavestrello G, Cerrano C, Gravili C, Piraino S, Puce S, Boero F (2017) Hydroids (Cnidaria, Hydrozoa): A neglected component of animal forests. In: Rossi S, Bramanti L, Gori A, Orejas C (eds) Marine animal forests, Springer, Cham, pp 397–427. https://doi.org/10.1007/978-3-319-21012-4_11
Carneiro PBM, Ximenes Neto AR, Jucá-Queiroz B et al (2022) Interconnected marine habitats form a single continental-scale reef system in South America. Sci Rep 12:17359. https://doi.org/10.1038/s41598-022-21341-x
Cavalcanti T, Santos GG, Pinheiro U (2018) Description of three species of Eurypon Gray, 1867 (Raspailiidae: Demospongiae: Porifera) from the Western Atlantic and a name to replace the the secondary homonym Eurypon topsenti. Zootaxa 4388(1):89–101. https://doi.org/10.11646/zootaxa.4388.1.6
Cordeiro RT, Neves BM, Kitahara MV, Arantes RCM, Pérez CD (2020) First assessment on Southwestern Atlantic equatorial deep-sea coral communities. Deep Sea Res Part I Oceanogr Res Pap 163:103344. https://doi.org/10.1016/j.dsr.2020.103344
Cornelius PFS (1990) Evolution in leptolid life-cycles (Cnidaria: Hydroida). J Nat Hist 24:579–594. https://doi.org/10.1080/00222939000770391
Cornelius PFS (1992) Medusa loss in leptolid Hydrozoan (Cnidaria) hydroid rafting, and abbreviated life-cycles among their remote-island faunae: an interim review. In: Bouillon J, Boero F, Cicogna F, Gili JM, Hughes RG (eds) Aspects of hydrozoan biology. Scientia Marina 56(2–3):245–261
Cornelius PFS (1995a) North-West European thecate hydroids and their medusae. Part 1. Introduction, Laodiceidae to Haleciidae. In: Barnes RSK, Crothers JH (eds) Synopses of the British Fauna. The Dorset Press, Dorchester, pp 1–347
Cornelius PFS (1995b) North-West European thecate hydroids and their medusae. Part 2. Sertulariidae to Campanulariidae. In: Barnes RSK, Crothers JH (eds) Synopses of the British Fauna 50. The Dorset Press, Dorchester, pp 1–386
Csardi G, Nepusz T (2006) The igraph software package for complex network research. Int J Complex Syst 1695(5):1–9
Cunha AF, Maronna MM, Marques AC (2016) Variability on microevolutionary and macroevolutionary scales: a review on patterns of morphological variation in Cnidaria Medusozoa. Org Divers Evol 16:431–442. https://doi.org/10.1007/s13127-016-0276-4
Cunha AF, Collins A, Marques AC (2017) Phylogenetic relationships of Proboscoida Broch, 1910 (Cnidaria, Hydrozoa): Are traditional morphological diagnostic characters relevant for the delimitation of lineages at the species, genus, and family levels? Mol Phylogenet Evol 106:118–135. https://doi.org/10.1016/j.ympev.2016.09.012
Cunha AF, Collins A, Marques AC (2020) When morphometry meets taxonomy: morphological variation and species boundaries in Proboscoida (Cnidaria: Hydrozoa). Zool J Linn Soc 190:417–447. https://doi.org/10.1093/zoolinnean/zlz166
Cunha AF, Carmelet-Rescan D, Marques AC, Morgan-Richards M (2022) Contrasting morphological and genetic patterns suggest cryptic speciation and phenotype–environment covariation within three benthic marine hydrozoans. Mar Biol 169:103. https://doi.org/10.1007/s00227-022-04088-x
Dias A, Santos GG, Pinheiro U (2019) A new species of Characella Sollas, 1886 (Tetractinellida; Demospongiae; Porifera) from deeper waters off the coast of Brazil. Zootaxa 4559(1):196–200. https://doi.org/10.11646/zootaxa.4559.1.13
Fernandez MO, Marques AC (2018) Combining bathymetry, latitude, and phylogeny to understand the distribution of deep Atlantic hydroids (Cnidaria). Deep Sea Res Part I Oceanogr Res Pap 133:39–48. https://doi.org/10.1016/j.dsr.2018.01.008
Fernandez MO, Collins A, Marques AC (2020) Gradual and rapid shifts in the composition of assemblages of hydroids (Cnidaria) along depth and latitude in the deep Atlantic Ocean. J Biogeogr 1:11. https://doi.org/10.1111/jbi.13853
Fernandez MO, Jaimes-Becerra A, Marques AC (2023) Trait-environment relationships of hydroids (Cnidaria: Hydrozoa) across large spatial and environmental gradients in the Atlantic Ocean. Glob Ecol Biogeogr 32:548–560. https://doi.org/10.1111/geb.13642
Fraser CM (1944) Hydroids of the Atlantic Coast of North America. University of Toronto Press, Toronto
Galea HR (2008) On a collection of shallow-water hydroids (Cnidaria: Hydrozoa) from Guadeloupe and Les Saintes, French Lesser Antilles. Zootaxa 1878:1–54. https://doi.org/10.11646/zootaxa.1878.1.1
Galea HR (2010) Additional shallow-water thecate hydroids (Cnidaria: Hydrozoa) from Guadeloupe and Les Saintes. French Lesser Antilles. Zootaxa 2570(1):1–40. https://doi.org/10.11646/zootaxa.2570.1.1
Garcia TM, Matthews-Cascon H, Franklin-Júnior W (2009) Millepora alcicornis (Cnidaria: Hydrozoa) as substrate for benthic fauna. Braz J Oceanogr 57(2):153–155. https://doi.org/10.1590/s1679-87592009000200009
Genzano GN, Rodriguez GM (1998) Association between hydroid species and their substrates from the intertidal zone of Mar del Plata (Argentine). Misc Zool 21(1):21–29
Gil M, Ramil F (2021) Hydroids (Cnidaria, Hydrozoa) from the Vema and Valdivia seamounts (SE Atlantic). Eur J Taxon 758:49–96. https://doi.org/10.5852/ejt.2021.758.1425
Gili JM, Hughes R (1995) The ecology of marine benthic hydroids. Oceanogr Mar Biol Ann Rev 33:351–426
Gomes-Pereira JN, Tempera F (2016) Hydroid gardens of Nemertesia ramosa (Lamarck, 1816) in the central North Atlantic. Mar Biodivers 46(1):85–94. https://doi.org/10.1007/s12526-015-0325-9
Grohmann PA, Nogueira CC, Vera MA (2003) Hydroids (Cnidaria, Hydrozoa) collected on the continental shelf of Brazil during the Geomar X Oceanographic Operation. Zootaxa 299:1–19. https://doi.org/10.11646/zootaxa.299.1.1
Hughes RG (1975) The distribution of epizoites on the hydroid Nemertesia antennina (L.). J Mar Biol Assoc UK 55:275–294. https://doi.org/10.1017/s0025315400015940
Jäderholm E (1903) Aussereuropäische Hydroiden im schwedischen Reichsmuseum. Arkiv for Zoologi 1:259–312
Kelmo F, de Santa-Isabel LM (1998) The athecatae hydroids (Cnidaria, Hydrozoa) from Northern Bahia. Brazil Rev Biol Trop 46(5):61–72
Larrazábal-Filho AL, Silva MC, Esteves AM (2015) Four new species of free-living marine nematodes of the family Desmodoridae (Nematoda: Desmodorida) and a redescription of Desmodora nini (Inglis, 1963) from the continental shelf off northeastern Brazil. Zootaxa 4021(1):63–92. https://doi.org/10.11646/zootaxa.4021.1.3
Larrazábal-Filho AL, Neres PF, Esteves AM (2018) The genus Bolbonema Cobb, 1920 (Nematoda: Desmodoridae): emended diagnosis, key to males, and description of three new species from the continental shelf off northeastern Brazil. Zootaxa 4420(4):551–570. https://doi.org/10.11646/zootaxa.4420.4.6
Leclère L, Schuchert P, Manuel M (2007) Phylogeny of the Plumularioidea (Hydrozoa, Leptothecata): evolution of colonial organisation and life cycle. Zool Scr 36:371–394. https://doi.org/10.1111/j.1463-6409.2007.00283.x
Lima MM, Cordeiro RTS, Pérez CD (2019) Black Corals (Anthozoa: Antipatharia) from the Southwestern Atlantic. Zootaxa 4692(1):1–67. https://doi.org/10.11646/zootaxa.4692.1.1
Lindner A, Govindarajan AF, Migotto AE (2011) Cryptic species, life cycles, and the phylogeny of Clytia (Cnidaria: Hydrozoa: Campanulariidae). Zootaxa 2980:23–36. https://doi.org/10.11646/zootaxa.2980.1.2
Maggioni D, Arrigoni R, Galli P, Berumen ML, Seveso D, Montano S (2018) Polyphyly of the genus Zanclea and family Zancleidae (Hydrozoa, Capitata) revealed by the integrative analysis of two bryozoan-associated species. Contrib Zool 87(2):87–104. https://doi.org/10.1163/18759866-08702003
Maronna MM, Miranda TP, Peña Cantero ÁL, Barbeitos MS, Marques AC (2016) Towards a phylogenetic classification of Leptothecata (Cnidaria, Hydrozoa). Sci Rep 6:18075. https://doi.org/10.1038/srep18075
Maÿal EM (1983) Distribuição de hidroides (Hydrozoa, Thecata) na costa do estado de Pernambuco. Brasil Bol Zool 6:1–13. https://doi.org/10.11606/issn.2526-3358.bolzoo.1983.121945
Mendonça LMC, Guimarães CRP, Haddad MA (2022) Taxonomy and diversity of hydroids (Cnidaria: Hydrozoa) of Sergipe. Northeast Brazil Zoologia 39:21032. https://doi.org/10.1590/s1984-4689.v39.e21032
Migotto AE (1996) Die Benthic shallow-water hydroids (Cnidaria, Hydrozoa) of the coast of São Sebastião, Brazil, including a checklist of Brazilian hydroids. Zool Verh 306:1–125
Millard NAH (1975) Monograph of the Hydroida of southern Africa. Ann S Afr Mus 68:1–513
Millard NAH, Bouillon J (1974) A collection of hydroids from Mozambique, East Africa. Ann S Afr Mus 65:1–40
Miranda TP, Genzano GN, Marques AC (2015) Areas of endemism in the Southwestern Atlantic Ocean based on the distribution of benthic hydroids (Cnidaria: Hydrozoa). Zootaxa 4033(4):484–506. https://doi.org/10.11646/zootaxa.4033.4.2
Miranda TP, Fernandez MO, Genzano G, Peña Cantero ÁL, Collins A, Marques AC (2021) Biodiversity and biogeography of hydroids across marine ecoregions and provinces of southern South America and Antarctica. Polar Biol 44:1669–1689. https://doi.org/10.1007/s00300-021-02909-1
Moura CJ, Cunha MR, Porteiro FM, Roges AD (2011) The use of the DNA barcode gene 16S mRNA for the clarification of taxonomic problems within the family Sertulariidae (Cnidaria, Hydrozoa). Zool Scr 40(5):520–537. https://doi.org/10.1111/j.1463-6409.2011.00489.x
Muller KE, Peterson BL (1984) Practical methods for computing power in testing the multivariate general linear hypothesis. Comput Stat Data Anal 2(2):143–158
Nutting CC (1915) American hydroids. Part III, the Campanularidae and the Bonneviellidae. Smithsonian Institution, US Nat Mus Spec Bull 4:1–126
Oliveira OM, Miranda TP, Araujo EM et al (2016) Census of Cnidaria (Medusozoa) and Ctenophora from south American marine waters. Zootaxa 4194(1):1–256. https://doi.org/10.11646/zootaxa.4194.1.1
Petersen KW (1990) Evolution and taxonomy in capitate hydroids and medusae (Cnidaria: Hydrozoa). Zool J Linn Soc 100:101–231. https://doi.org/10.1111/j.10963642.1990.tb01862.x
Pillai KS (1956) On the distribution of the largest or the smallest root of a matrix in multivariate analysis. Biometrika 43(1/2):122–127
Pires DO, Castro CB, Migotto AE, Marques AC (1992) Cnidários bentônicos do arquipélago de Fernando de Noronha, Brasil. Bol Mus Nac Zool 354:1–21
Puce S, Calcinai B, Bavestrello G, Cerrano C, Gravili C, Boero F (2005) Hydrozoa (Cnidaria) symbiotic with Porifera: a review. Mar Ecol 26:73–81. https://doi.org/10.1111/j.1439-0485.2005.00050.x
Rao CR (2002) Linear statistical inference and its applications, 2nd edn. John Wiley & Sons, New York
Roberts JM, Wheeler AJ, Freiwald A (2006) Reefs of the deep: the biology and geology of cold-water coral ecosystems. Science 312(5773):543–547. https://doi.org/10.1126/science.1119861
Rodriguez CS, Marques AC, Mianzan HW, Tronolone VB, Migotto AE, Genzano GN (2017) Environment and life cycles influence distribution patterns of hydromedusae in austral South America. Mar Biol Res 13(6):659–670. https://doi.org/10.1080/17451000.2017.1280170
Rützler K, Piantoni C, Van Soest R, Diaz M (2014) Diversity of sponges (Porifera) from cryptic habitats on the Belize barrier reef near Carrie Bow Cay. Zootaxa 3805:1–129. https://doi.org/10.11646/zootaxa.3805.1.1
Schuchert P (2003) Hydroids (Cnidaria, Hydrozoa) of the Danish expedition to the Kei Islands. Steenstrupia 27(2):137–256
Shimabukuro V, Marques AC (2006) Morphometrical analysis, histology, and taxonomy of Thyroscyphus ramosus (Cnidaria, Hydrozoa) from the coast of Brazil. Zootaxa 1184:29–42. https://doi.org/10.11646/zootaxa.1184.1.2
Shimabukuro V, Marques AC, Migotto AE (2006) Fauna de hidrozoários atecados (Hydrozoa, Anthoathecata) da costa do Estado do Ceará. Brasil Biota Neotrop 6(3):1–13. https://doi.org/10.1590/s1676-06032006000300009
Spalding MD, Fox HE, Allen GR et al (2007) Marine ecoregions of the world: a bioregionalization of coastal and shelf areas. BioScience 57(7):573–583. https://doi.org/10.1641/B570707
Vale NF, Braga JC, Moura RL et al (2022) Distribution, morphology and composition of mesophotic ‘reefs’ on the Amazon Continental Margin. Mar Geol 447:106779. https://doi.org/10.1016/j.margeo.2022.106779
Vervoort W (1959) The Hydroida of the tropical west coast of Africa. Atlantide Report. Scient Res Danish Exped Coasts Trop W Afr 1945-1946 5:211–325
Vervoort W (1968) Report on a collection of Hydroida from the Caribbean region, including an annotated checklist of Caribbean hydroids. Zool Verh 92(1):1–124
Vervoort W (2006) Leptolida (Cnidaria: Hydrozoa) collected during the CANCAP and Mauritania-II expeditions of the National Museum of Natural History, Leiden, the Netherlands (Anthoathecata, various families of Leptothecata and addenda) CANCAP-project. Contributions, no 128. Zool Meded 80(1):181–318
Vervoort W, Watson JE (2003) The marine fauna of New Zealand: Leptothecata (Cnidaria: Hydrozoa) (thecate hydroids). NIWA Biodivers Mem 119:5–538
Wilks SS (1935) On the independence of k sets of normally distributed statistical variables. Econometrica, J Econom Soc 3(3):309–326
WoRMS (2023). Hydrozoa. https://www.marinespecies.org/aphia.php?p=taxdetails&id=1337. Accessed 14 June 2023
Acknowledgements
The oceanographic campaigns were financed by Petrobras, the Brazilian company for exploration, processing, and transportation of oil and its derivatives; and by the Geomar Project, a Brazilian Navy program aimed at developing geological and geophysical knowledge for exploring the continental shelf of select states in the North and Northeast regions. The authors thank to these institutions for providing the collection data used in this study. We wish also to acknowledge the valuable contributions of numerous researchers who have assisted in collecting and preserving specimens over the years. We also feel grateful to the reviewers for their valuable criticisms and suggestions to the text.
Funding
This study received partial financial support from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (Grants-CNPq/MCTI/FACEPE/PROTAX No 001/2015, 440633/20150 CNPq); the Fundação de Amparo à Ciência e Tecnologia de Pernambuco (FACEPE/APQ-0913–2.04/17; APQ-1518-2.04/22); and Capes, Finance Code 001. AC Moura and UDR acknowledge FACEPE and FAPESB (Fundação de Amparo à Pesquisa do Estado da Bahia), respectively, for funding their post-graduate fellowship, and AC Marques was supported by CNPq (316095/2021–4). This is a partial contribution of NP-BioMar USP (AC Marques).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
No animal testing was performed during this study.
Sampling and field studies
All necessary permits for sampling and observational field studies have been obtained by the Brazilian Navy and Petrobras (Brazilian company for exploration of oil) from the competent authorities.
Data availability
All data generated during this study are included in this published article.
Author contributions
FFC, AC Marques and CDP designed the study. AC Moura and FFC performed laboratory work. CDP provided infrastructure and material. FFC and UDR analyzed the data and prepared the figures. The initial draft of the manuscript was written by FFC. All authors reviewed drafts and approved the final version of the manuscript.
Additional information
Communicated by D. Maggioni
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Moura, A.C., Campos, F.F., de Oliveira, U.D.R. et al. Hydroids (Cnidaria, Hydrozoa) from the Northern and North-eastern coast of Brazil: addressing knowledge gaps in neglected regions. Mar. Biodivers. 53, 81 (2023). https://doi.org/10.1007/s12526-023-01388-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12526-023-01388-3