Abstract
Anthopleura dalyae sp. nov, a new species of sea anemone (Actiniaria: Actiniidae) from the rocky intertidal zone of La Gallega reef, Veracruz, Mexico, is described and illustrated. This species is characterized by a column that is pale yellow proximally and dark-orange to bright-orange distally, longitudinal rows of verrucae from margin to limbus, and a series of marginal projections with acrorhagi. Anthopleura dalyae sp. nov. is distinguished from other species of Anthopleura from the Gulf of Mexico by several taxonomic characters, such as column coloration, number of tentacles, variable number of directive mesenteries and siphonoglyphs, as well as some differences on cnidae. Anthopleura dalyae sp. nov. is the fourth species of the genus occurring in shallow intertidal waters associated to coastal and coral reef environments of the region, and the third species of the genus Anthopleura recorded in the southern Gulf of Mexico.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The genus Anthopleura Duchassaing de Fonbressin & Michelotti, 1860 is one of the best known genera of sea anemones, and its members can be typically found inhabiting the intertidal and shallow subtidal ecosystems from both tropical and temperate regions (Daly et al. 2017; Spano and Häussermann 2017). This genus comprises about 45 valid species (Fautin 2016; Spano and Häussermann 2017), and can be differentiated from other members of Actiniidae Rafinesque, 1815 in having both acrorhagi and adhesive verrucae arranged in longitudinal endocoelic rows on the column (see Daly et al. 2017).
There are currently three known valid species of Anthopleura inhabiting coastal and coral reef environments of the Gulf of Mexico and the Caribbean Sea: Anthopleura krebsi Duchassaing de Fonbressin & Michelotti, 1860, Anthopleura pallida Duchassaing de Fonbressin & Michelotti, 1864, and Anthopleura texaensis (Carlgren & Hedgepeth, 1952) (Daly and den Hartog 2004).
In 1922, Watzl (1922) described the species Anthopleura varioarmata from specimens collected in the Bahamas, and Carlgren (1952) and Belém and Monteiro (1981) applied the name A. varioarmata to specimens from North Florida, USA, and Rio de Janeiro, Brazil, respectively. However, Cairns et al. (1986) and Daly and den Hartog (2004) recognized that the specimens of Watzl from the Bahamas actually belongs to A. krebsi. In addition, Daly and den Hartog (2004) provisionally refer the specimens identified by Carlgren (1952) and Belém and Monteiro (1981) to A. texaensis, but recognized that those specimens may belong to a different, as yet unnamed, species.
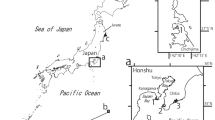
In the present study, we describe a new species of sea anemone, Anthopleura dalyae sp. nov., from specimens collected in the rocky intertidal zone of La Gallega reef, one of the 25 coral reef formations of the Veracruz Reef System, in Mexico (Tunnel 2007).
The taxonomic features of A. dalyae sp. nov. resembles, in many respects, to those of the specimens identified as A. varioarmata by Carlgren (1952) and Belém and Monteiro (1981). However, some differences are discussed, as well as the distinctions of A. dalyae sp. nov. from other species of Anthopleura from the region. Anthopleura dalyae sp. nov. is the fourth species of the genus reported in the Gulf of Mexico, and the third species of the genus reported in the coral reef environments of the southern Gulf of Mexico.
Materials and methods
Seven specimens were collected in the rocky intertidal zone of La Gallega reef (19°13′19.91″ N, 96°07′38.74″ W) (Fig. 1). Collections were made by hand, using a hammer and a chisel. Collected specimens were transferred to the laboratory and maintained in an aquarium to photograph their color while alive. Specimens were relaxed in 5% MgCl2 seawater solution, subsequently fixed in 10% seawater formalin, and stored in 70% ethanol. Squash preparations of small amounts of tissue (approximately 1 mm2) from tentacles, actinopharynx, filaments, column, and acrorhagi were made from five preserved specimens to study the cnidom. From each squash, the length and width of 40 undischarged capsules of each cnidae type present were haphazardly measured using a Nikon Labophot-2 light microscope (1000× oil immersion). Cnidae terminology follows Östman (2000). Measurements were obtained from living specimens. Longitudinal and cross-sectional serial sections 6–8 mm were made from two specimens dehydrated in ethanol and embedded in Paraplast (Fisher Scientific). Sections were stained with hematoxylin and eosin (Estrada-Flores et al. 1982). Type and voucher specimens were deposited in the Cnidarian Collection of the Gulf of Mexico and Mexican Caribbean Sea (registration code: YUC-CC-254-11) of Unidad Multidisciplinaria de Docencia e Investigación en Sisal (UMDI-Sisal) at the Universidad Nacional Autónoma de México (UNAM). Taxonomic classification follows Rodríguez et al. (2014).
Results
Suborder Enthemonae Rodríguez & Daly, 2014 in Rodríguez et al. (2014).
Superfamily Actinioidea Rafinesque, 1815.
Family Actiniidae Rafinesque, 1815.
Genus Anthopleura Duchassaing de Fonbressin & Michelotti, 1860.
Anthopleura dalyae sp. nov.
Anthopleura dalyae sp. nov. a Oral disk view. b Oral disk view: detail of the tentacle color variation. c Lateral view. d Pedal disk view. e Oral disk view: detail of the wide open mouth showing the siphonoglyphs. a–e Living specimen. f Preserved specimen: detail of marginal projections with acrorhagi. g Transversal section through distal column showing siphonoglyph. h Transversal section through proximal column showing mesenteric muscles. i Longitudinal section through distal column showing verrucae. j Longitudinal section through pedal disk showing basilar muscles. k Transversal section through tentacle showing longitudinal muscles from a tentacle. l Longitudinal section through margin showing marginal sphincter. Scale bars: a–e = 10 mm; f = 1 mm; g–l = 200 μm). Abbreviations: acr: acrorhagi, bm: basilar muscles, c: column, d: directive mesenteries, e: epidermis, f: fosse, g: gastrodermis, lm: longitudinal muscles, m: mesoglea, mp: marginal projection, od: oral disk, pd: pedal disk, pm: parietobasilar muscle, rm: retractor muscle, s: siphonoglyph, sc: spermatic cysts, sp: sphincter, t: tentacles, vr: verrucae
non Anthopleura varioarmata: Watzl, 1922: 33–36, 75.
? Anthopleura vario-armata [sic]: Carlgren, 1952: 373–374.
Anthopleura varioarmata: Belém and Monteiro, 1981: 193–203.
Anthopleura variarmata [sic]: Zamponi et al., 1998: 36, 38, 40.
Anthopleura texaensis: González-Muñoz, Tello-Musi and Simões, 2015: 101–118.
Type material: Holotype: one specimen (YUC-CC-254-11-000329), La Gallega reef, Veracruz, Mexico, (19°13′19.91″ N, 96°07′38.74″ W), 0.5 m depth, 6 January 2012, coll. R. González-Muñoz; paratypes: four specimens (YUC-CC-254-11-000317, 000330, 000331, 000332), with the same sampling data as the holotype; voucher of longitudinal and transversal histological sections from two more specimens (YUC-CC-254-11-000055, 000056), with the same sampling data as the holotype.
Diagnosis
Actiniidae with endocoelic adhesive verrucae from margin to just above the limbus. Column pale yellow proximally, dark-orange or bright-orange distally. Margin with endocoelic projections with acrorhagi (containing basitrichs and holotrichs) on the oral surface and settled in the fosse. Acrorhagi sometimes appear only on the marginal projections relative to the first and second cycles, but sometimes appear in those from almost all marginal projections.
Description
External anatomy
Fully expanded oral disk and tentacles 11–27 mm in diameter in living specimens (8–10 when preserved). Oral disk flat, smooth, 5–10 mm in diameter, olive-green to dark-green, sometimes with white spots marking the siphonoglyphs (Fig. 2a, b). Tentacles irregularly arranged in four to five cycles (between 64 and 82 in number in the specimens examined) (Fig. 2a, b, e). Tentacles short (3–8 mm in diameter in living specimens), conical, tapering distally, smooth, inner cycles longer than outer ones, contractile, pale-gray with orange flashes, sometimes with small white rounded spots (Fig. 2a, b). Fosse well marked. Column cylindrical, stout, 8–14 mm in diameter and 6–11 mm in height in living specimens (8–10 in diameter and 5–7 in height when preserved), pale yellow proximally and dark-orange or bright-orange distally in living specimens (Fig. 2c), beige proximally and gray distally when preserved. Column covered from margin to just above limbus with simple, endocoelic, adhesive verrucae arranged in 42–48 longitudinal series (Fig. 2c). Verrucae cup-shaped, more prominent distally than proximally, same color as column in live specimens (Fig. 2c, i). Margin with endocoelic marginal projections (29–41 in number in specimens examined); acrorhagi (containing basitrichs and holotrichs) attached to the oral face of the marginal projections and settled in the fosse; acrorhagi sometimes appear only on the marginal projections corresponding to the first and second cycles (Fig. 2f), but sometimes appear in those from almost all marginal projections. Pedal disk well-developed, 8–14 mm in diameter in living specimens (7–10 when preserved), slightly wider than column, pale yellow to beige (Fig. 2d). Acrorhagi pale orange while alive, milky-white in preservation.
Internal anatomy
Mesenteries irregularly arranged in four cycles (40–52 pairs in the specimens examined): mesenteries of the first, second, and some of the third cycles perfect, others imperfect; same number of mesenteries distally and proximally. Directive mesenteries in variable number (2–4 pairs in specimens examined), each attached to a well-developed siphonoglyph (Fig. 2e, g). Stronger mesenteries of the first three cycles fertile, except directives; gonochoric (?), only spermatic cysts were observed in specimens examined (Fig. 2h). Retractor muscles well-developed, restricted; parietobasilar muscles developed with short mesogleal pennon (Fig. 2h). Basilar muscles well-developed (Fig. 2j). Longitudinal muscles of tentacles and oral disk ectodermal (Fig. 2k). Marginal sphincter endodermal, strong, circumscribed, and palmate (Fig. 2l).
Cnidom
Basitrichs, spirocysts, holotrichs, microbasic b-mastigophores, microbasic p-mastigophores (Fig. 3). See Table 1 for size and cnidom distribution.
Natural history
Anthopleura dalyae sp. nov. lives attached to rocks, inside cracks and crevices, in the intertidal and subtidal zone. Azooxanthellate.
Known distribution
La Gallega reef, Veracruz Reef System, Gulf of Mexico; coast of Rio de Janeiro, Brazil (Belém and Monteiro 1981).
Etymology
Anthopleura dalyae sp. nov. is named after Dr. Marymegan Daly, in recognition of her valuable contributions to the taxonomic and phylogenetic study of cnidarians, particularly on sea anemones.
Differential diagnosis
Anthopleura dalyae sp. nov. is easily distinguished from other species of the genus Anthopleura from the Gulf of Mexico by its column coloration pattern. In addition, A. krebsi and A. pallida possess 24–48 tentacles and verrucae arranged in up to 24 longitudinal rows on the column, while A. dalyae sp. nov. and A. texaensis have a greater number of tentacles (about 64–82 in the former and up to 100 in the latter) and verrucae arranged in up to 48 longitudinal rows on the column (Daly and den Hartog 2004).
Aside from column coloration and number of tentacles, other differences between A. dalyae sp. nov. and A. texaensis lie on the type of the retractor muscle, being restricted in the former and diffuse in the latter (Table 2). Furthermore, two size categories of holotrichs from acrorhagi have been reported from A. krebsi, A. pallida, and A. texaensis (Daly and den Hartog 2004), while only one was observed from A. dalyae sp. nov., which is smaller, particularly compared with those of A. texaensis (Table 3).
In addition, the whole body size reported for the specimens of A. texaensis is somewhat greater than those reported from specimens of the other three species, and A. krebsi is the only of these four species reported as zooxanthellate (Daly and den Hartog 2004). Differences in anatomy and cnidae between these four species could also be observed in Tables 2 and 3, respectively.
Discussion
The anatomic features of the specimens examined by Carlgren (1952) and Belém and Monteiro (1981) fit well with those observed in A. dalyae sp. nov. Carlgren (1952) reported specimens with up to 100 tentacles, 2–4 siphonoglyphs, several pairs of directive mesenteries and mesenteries irregularly arranged, all strongest mesenteries fertile, band-like retractor muscles, and a marginal sphincter circumscribed. The specimens described by Carlgren (1952) only differ to those of A. dalyae sp. nov. in the column coloration, being reported as cream below and olive above, and in the number of tentacles (64–82 in A. dalyae sp. nov., while up to 100 are present in A. texaensis). In addition, Carlgren (1952) did not report basitrichs in the marginal projections, spirocysts in the tentacles, nor the microbasic b-mastigophores in the filaments, which were observed in A. dalyae sp. nov.
Specimens reported by Belém and Monteiro (1981) only differ from those of A. dalyae sp. nov. in some aspects of the cnidom. They did not report the microbasic b-mastigophores from the filaments that were found in A. dalyae sp. nov. Instead, they report a long curved type of basitrich, which was not observed in A. dalyae sp. nov.
Based on the descriptions by Carlgren (1952) and Belém and Monteiro (1981), it is most probable that those specimens are the same that here described as A. dalyae sp. nov. However, because the type specimen assigned to A. varioamata by Watzl (1922) was synonymized with A. krebsi (Daly and den Hartog 2004), according to the International Code of Zoological Nomenclature (Article 49, ICZN 1999), the name Anthopleura varioarmata becomes unavailable. The species A. dalyae sp. nov. is here described according to the standard morphological features currently used in actiniarian taxonomy. In addition, the specimens of Belém and Monteiro (1981) are here recognized as synonyms of A. dalyae sp. nov., but those specimens named by Carlgren (1952) as A. varioarmata need to be examined in order to corroborate its synonymy with A. dalyae sp. nov.
Due to the observation of an irregular number of directive mesenteries, siphonoglyphs (one specimen with two siphonoglyphs, five with three, and one with four), and tentacles, it maybe that A. dalyae sp. nov. undergoes asexual reproduction by longitudinal fission, which has also been reported for A. krebsi and A. texaensis (Belém and Pinto 1990; Daly and den Hartog 2004). Although the specimens of A. dalyae sp. nov. collected in La Gallega reef were previously reported as A. texaensis by González-Muñoz et al. (2015), the finding of a new species was apparent under a more thorough revision. The species A. krebsi has been previously reported in the Veracruz Reef System (González-Muñoz et al. 2015) and in the Mexican Caribbean (Instituto Nacional de Ecología, INE 1998), and A. pallida has been reported in Alacranes reef (González-Muñoz et al. 2013). Anthopleura dalyae sp. nov. is the third species of the genus reported from the coastal coral reef environments in the Mexican Atlantic.
References
Belém MJC, Monteiro DC (1981) Fauna de cnidários do Rio de Janeiro. III - Anthopleura varioarmata Watzl, 1922 (Actiniaria, Endomyaria), uma nova ocorrência de Actiniidae. Sem Biol Mar Acad Bras Ciên, pp 193–203
Belém MJC, Pinto SM (1990) Morphological and microanatomical study of Anthopleura krebsi Duchassaing & Michelotti, 1860 (Cnidaria, Anthozoa, Actiniidae), a new record in Brazil. An Acad Bras Cienc 62(2):183–192
Cairns S, den Hartog C, Arneson C (1986) Class Anthozoa (corals, anemones). In: Sterrer W, Schoepfer-Sterrer C (eds) Marine fauna and flora of Bermuda. A systematic guide to the identification of marine organisms. Wiley, New York, pp 159–194
Carlgren O (1952) Actiniaria from North America. Ark Zool 3(30):373–390
Carlgren O, Hedgepeth JW (1952) Actiniaria, Zoantharia and Ceriantharia from shallow water in the northwestern Gulf of Mexico. Publ Inst Mar Sci (Univ Texas) 2(2):141–172
Daly M, den Hartog JC (2004) Anthopleura (Cnidaria: Anthozoa: Actiniaria) from the Gulf of Mexico and Caribbean. Bull Mar Sci 74(2):401–421
Daly M, Crowley LM, Larson P, Rodríguez E, Heestand Saucier E, Fautin DG (2017) Anthopleura and the phylogeny of Actinioidea (Cnidaria: Anthozoa: Actiniaria). Org Divers Evol 17(3):545–564. https://doi.org/10.1007/s13127-017-0326-6
Duchassaing de Fonbressin P, Michelotti G (1860) Mémoire sur les coralliaires des Antilles. Imprimerie Royale, Turin
Duchassaing de Fonbressin P, Michelotti G (1864) Supplément au mémoire sur les coralliaires des Antilles. Imprimerie Royale, Turin
Estrada-Flores E, Peralta L, Rivas P (1982) Manual de técnicas histológicas. AGT Press, México
Fautin DG (2016) Catalog to families, genera, and species of orders Actiniaria and Corallimorpharia (Cnidaria: Anthozoa). Zootaxa 4145(1):1–449. https://doi.org/10.11646/zootaxa.4145.1.1
González-Muñoz R, Simões N, Tello-Musi JL, Rodríguez E (2013) Sea anemones (Cnidaria, Anthozoa, Actiniaria) from coral reefs in the southern gulf of Mexico. Zookeys 341:77–106
González-Muñoz R, Tello-Musi JL, Simões N (2015) Las anémonas del Sistema Arrecifal Veracruzano. In: Granados-Barba A, Ortíz-Lozano L, Salas-Monreal D, González-Gándara C (eds) Aportes al conocimiento del Sistema Arrecifal Veracruzano: hacia el Corredor Arrecifal del Suroeste del Golfo de México. Universidad Autónoma de Campeche, Campeche, pp 101–118
Instituto Nacional de Ecología (INE) (1998) Programa de Manejo del Parque Marino Nacional Costa Occidental de Isla Mujeres, Punta Cancún y Punta Nizuc. Instituto Nacional de Ecología, México
International Commission on Zoological Nomenclature (ICZN) (1999) International code of zoological nomenclature, 4th edn. International Trust for Zoological Nomenclature, London. https://doi.org/10.5962/bhl.title.50608
Östman C (2000) A guideline to nematocyst nomenclature and classification, and some notes on the systematic value of nematocysts. Sci Mar 64(S1):31–46
Rafinesque CS (1815) Analyse de la Nature ou Tableau de l’Univers et des Corps Organisés. CS Rafinesque, Palerme
Rodríguez E, Barbeitos MS, Brugler MR, Crowley LM, Grajales A, Gusmão L, Häussermann V, Reft A, Daly M (2014) Hidden among sea anemones: the first comprehensive phylogenetic reconstruction of the order Actiniaria (Cnidaria, Anthozoa, Hexacorallia) reveals a novel group of hexacorals. PLoS One 9(5):e96998. https://doi.org/10.1371/journal.pone.0096998
Spano C, Häussermann V (2017) Anthopleura radians, a new species of sea anemone (Cnidaria: Actiniaria: Actiniidae) from northern Chile, with comments on other species of the genus from the South Pacific ocean. Biodiv Nat Hist 3(1):1–11
Tunnel JW (2007) Reef distribution. In: Tunnell JW, Chávez EA, Withers K (eds) Coral reefs of the southern gulf of Mexico. Texas A&M University Press, Harte Research Institute, Texas, pp 14–22
Watzl O (1922) Die Actiniarien der Bahamainseln. Ark Zool 14(24):1–89
Zamponi MO, Belém MJC, Schlenz E, Acuña FH (1998) Distribution and some ecological aspects of Corallimorpharia and Actiniaria from shallow waters of the South American Atlantic coasts. Physis Secc A 55(128–129):31–45
Acknowledgements
The first author is grateful to the Programa de Becas Postdoctorales Latinoamericanas of the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET). This study was partially funded by the Universidad Nacional de Mar del Plata (project EXA747/16 to F.H.A.). We thank the Comisión Nacional de Áreas Naturales Protegidas (CONANP) and the staff at the Parque Nacional Sistema Arrecifal Veracruzano for their helpful assistance during field work. All specimens were collected under the consent of Mexican law, collecting permit approved by Comisión Nacional de Acuacultura y Pesca (number 07332.250810.4060). José Luis Tello-Musi and Nuno Simões from the Universidad Nacional Autónoma de México (UNAM) provided support for field work; Maribel Badillo-Alemán (UNAM) provided access and support to histological facilities; Gemma Martínez-Moreno (UNAM) and Patricia Guadarrama-Chávez (UNAM) helped with lab work and provided support in the microscopy lab. We thank the commentaries of the two anonymous reviewers which helped us to improve this manuscript.
Funding
This study was partially funded by the Universidad Nacional de Mar del Plata (project EXA747/16 to F.H.A.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed by the authors.
Sampling and field studies
All necessary permits for sampling and observational field studies have been obtained by the authors from the competent authorities and are mentioned in the acknowledgements, if applicable.
Additional information
Communicated by S. Piraino
This article is registered in ZooBank under urn:lsid:zoobank.org:pub:407AE066-7321-47D0-8EA3-E1525207B0EB.
Rights and permissions
About this article
Cite this article
González-Muñoz, R., Garese, A. & Acuña, F.H. Anthopleura dalyae sp. nov. (Cnidaria: Actiniaria), a new species of sea anemone from the southern Gulf of Mexico. Mar Biodiv 49, 683–690 (2019). https://doi.org/10.1007/s12526-018-0844-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12526-018-0844-2