Abstract
Soil is a suitable place for vegetation and plant growth. When this valuable resource is not preserved, shortage of food, erosion and damage of natural resources will be respected. Soil is a heterogeneous, diverse and dynamic system and investigation of its temporal and spatial changes is essential. In this paper spatial variability of some chemical and physical soil were investigated. Three hundred fifty eight soil samples were collected by systematic sampling strategy at 20 cm depth on a regular grid spacing of 500 × 500 m2 under different vegetation cover and processed for analysis in the laboratory. Soil chemical and physical parameters including pH, electrical conductivity, organic carbon, available phosphorus, available nitrogen, available potassium, sulphur, calcium, magnesium and sodium were measured. After data normalization, classical statistical analysis was used to describe soil properties and geo-statistical analysis was used to illustrate spatial correlation of soil characteristics. By using interpolating techniques, spatial distribution of these properties were prepared. Results indicated that calcium and phosphorus had strong and weak spatial dependence, respectively.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The spatial variability and heterogeneous geographical distribution of physical and chemical properties of cropland ecosystem soils are under the impact of physical and biological factors including topography, vegetation cover, soil microclimate, various grazing systems and rangeland management. Soil properties change in time and space continuously (Rogerio et al. 2006). Heterogeneity may occur at large scale (region) or small scale (community), even in the same type of soil or in the same community (Du Feng et al. 2008). Despite temporal and spatial changes in soil characteristics at small and large scales, awareness of these changes related to increasing profitability and sustainable agriculture management is necessary (Ayoubi and Khormali 2009). Distribution of vegetation is related to soil moisture and other soil properties such as soil aeration, soil texture, depth etc. Soil properties in relation to vegetative cover cause plant diversity and widespread geographical distribution of plants (Noy-Mire 1973; Burke 2001). Soil compaction following extensive cultivation and use of tractors cause homogenous spatial distribution of soil properties and increase vulnerability of soil and water loss, and consequently reduce available water for plants (Zhao et al. 2007). Cheng et al. (2007) reported that spatial variability of the above ground biomass in shrub lands is greater than grasslands. There is clear spatial relationship between plant and soil (Etema and Wardle 2002; Zhao et al. 2007; Covelo et al. 2008). Determining soil variability is important for ecological modeling, environmental predictions, precise agriculture and management of natural resources (Hangsheng et al. 2005; Wang et al. 2009). For a long time, spatial changes of soil characteristics have been attended by soil scientists. Precise and quantitative information about these changes is essential for environmental assessment of soil quality, risk of soil pollution and retro gradation of soil characteristics. Soil erosion studies as a part of environment and non-agricultural interpretations of soils has new challenges against soil scientists. Soil organic matter, nitrogen and phosphorus are the most important functions of ecosystems because they play a direct role in ecosystem processes such as plant growth and carbon cycle (Robertson et al. 1988). Organic matter is one of the most indexes of soil quality, thus investigation of changes and spatial distribution of organic carbon can be useful for evaluation of soil function and understanding of soil carbon decomposition processes and determination of soil quality trends (Venteris et al. 2004). Temporal and spatial investigation of data is essential for understanding soil spatial variability. Kresic (1997) revealed that geostatistics technique is the most confident, strongest and widest method for interpolation and has acknowledged that geostatistics is the strategy that considers spatial variance, location and distribution of samples. Geostatistics is a powerful tool for determining the spatial variability (Sauer et al. 2006). Geostatistical methods use mathematical and statistical functions for interpolation and their basis is statistical characteristics of data. This technique predicts unknown points based on autocorrelation and their spatial structure of measured points. Soil property maps show their spatial changes as well. Different methods exist for creating soil property maps, one of them is gathering samples from soil depths and analyzing the samples by using geostatistical technique (Hunter et al. 1982). Since the part of variations are caused by a number of randomly occurring events and geostatistics lead us to more accurate estimations with less error. In fact geostatistics investigate the variables that have spatial structure or continuous spatial distribution. Early principal of geostatistics is that the similarity between near samples decreases when the distance increases (Isaaks and Srivastava 1989; Goovaerts 1997). Many studies have shown the correlation between soil characteristics like organic matter and were illustrated in map (Zhang and McGrath 2004; Anderson et al. 2005; Jian-Bing et al. 2006). Zhao et al. (2007) reported that spatial variability of soil chemical and physical properties are affected by crop intensity and heavy cropping decreases soil water content (SWC) and soil organic carbon (SOC) but increases bulk density (BD) and shear strength (SS). Mohammadi and RaeisiGahrooee (2004) showed that spatial variation pattern of soil variables absolutely depends on cropland management history. Variogram of organic matter at some site has linear structure and does not access to threshold variance in regional scale. While the spatial pattern of this variable at enclosure site has strong structure and determine threshold variance. Fennessy and Mitsch (2001) evaluated spatial distribution of soil properties in 2 year period. They found that the spatial variability of organic matter and total nutrient of soil had decreased. Yong et al. (2006) investigated soil properties and their spatial pattern in a sandy grassland and reported that continuous grazing lead to decrease spatial dependence of soil organic carbon and total nitrogen. According to recent studies and confirmation of spatial relation between soil properties and plant at different ecosystems, knowledge of soil spatial variability for application purposes is necessary as well as model development (Sovik and Aagaard 2003). This research was done to investigate spatial variability of some chemical and physical soil properties in Bandipora agro-ecosystem of Lesser Himalayas.
Materials and Methods
Study Area
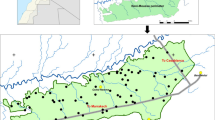
The study area is located in northern zone of Kashmir, India (74°27′08″E to 75°21′58″E and 34°10′53″N to 34°45′24″N). It has 1953 m mean altitude above sea surface and 298,300 ha area (Fig. 1). The land use of this area is forest, field crops, orchards and grasslands. The climate is temperate with mean annual precipitation of 990 mm, mostly falling in the winter, autumn and spring. Minimum and maximum monthly mean temperatures were −3.2 and 32.4 °C in January and July/August, respectively. The annual mean temperature is 18.2 °C.
Sampling
The sampling sites were selected in Bandipora district croplands. Soil samples were collected by a systematic sampling strategy on a regular grid spacing of 500 × 500 m2 from 20 cm depth. 325 points were selected and also 33 marginal points were added to increase the accuracy of research (358 soil samples in total). The UTM coordinates of soil samples were recorded for using in spatial analysis of soil characteristics.
Laboratory Analysis
The samples were air-dried and passed through a 2 mm sieve to prepare them for analysis. The methods applied were: Alkaline \( {\text{KMnO}}_{4}^{ - } \) method for nitrogen (Subbiah and Asija 1956), Walkley and Black (1934) wet oxidation procedure for organic carbon content and EDTA method for measuring calcium and magnesium (Lanyon and Heald 1982). Soil pH and electrical conductivity (EC) were also measured in the collected samples (McLean 1982). The amount of phosphorus was determined by Spectrophotometer (Olsen and Sommers 1982). Absorbable K and Na after extraction were measured using 1 N ammonium acetate (pH = 7) (Knudsen et al. 1982). Available sulphur was determined by following the turbidimetric method of Chesnin and Yien (1951). The methodology adopted is presented in the following flowchart

Spatial Analysis of Data
In order to know how data is distributed and accessing the statistical information summary, each soil characteristics were investigated using descriptive statistics. Geostatistics was used to investigate spatial variability of soil properties. In geostatistical studies, abnormal distribution of data have such effects that may lead to high fluctuations in variograms and reduces the reliability of analytical results, thus normalization of data is necessary. Normal distribution of data was estimated based on their skewness and the data within a range of −1 to +1 skewness were considered as normally distributed data (PazGonzales et al. 2000; Virgilio et al. 2007). This method is widely used in the analysis of soil ecological heterogeneity (Schlesinger et al. 1996). Since sulphur and calcium had skewness coefficient greater than −1, after elimination of imperfect data, Logarithmic conversion was chosen as the best method (Webster and Oliver 2001). For every variable before implementing geostatistical analysis, isotropy and anisotropy of each soil variable were controlled. Geostatistics is based on spatial correlation between samples and this correlation can be expressed with mathematical model called as “variogram”. In fact, variogram is defined as the functions which describe spatial variations of one variable and is defined by following formula:
N (h) is the number of sample pairs that are located by a particular distance (h) from each other. Z (xi) and Z (xi + h) are the values of regionalized variable at location xi and xi + h, respectively.
After calculating the variogram, fitting a theoretical model is necessary for generalization of deduction and estimation of variables from points which have not been sampled. For spatial interpolation and spatial mapping of soil characteristics, Kriging method was used. Overall Kriging method is a statistical estimator that gives statistical weight to each observation so their linear structure’s has been unbiased and has minimum estimation variance. This estimator has high application due to minimizing of error variance with unbiased estimation (Pohlmann 1993).
where, Z*(Xo) is, estimated variable at Xo location and Z*(Xo) is values of investigated variable at Xi location and λi is the statistical weight that is given to Z (Xi) sample located near Xo. N is the number of observations in the neighborhood of estimated point. Accuracy assessment of interpolation was done by using Cross-validation methods (Goovaerts 1997). The software package ARCGIS version 10.2 was used for geo-statistical analysis (ESRI 2014).
Results and Discussion
Sampling method was systematic with almost equal distances between soil samples in this study. Random sampling can generate points that are very close together so decreases accuracy of these studies (Weindorf and Zhu 2010). Davatgar (1998) reported whenever variables have been more randomly distributed and samples have been less continuous, nugget effect of variogram increases and precision of interpolation decreases. Also, McBratney and Webster (1983) and Wang and Qi (1998) expressed that a systematic sampling pattern provides more accurate results than random sampling pattern, and precision increased with addition sample size. Table 1 shows the summary statistics of soil characteristics. Coefficient of variation is used to show total changes. Among the investigated variables, EC had highest Coefficient of variation with 52.38 %. The result is in consistent with the research of Jafarian Jeloudar et al. (2009). pH had lowest coefficient variation with 7.25 %, which could be because of the uniform conditions in the region such as small changes in slope and its direction that led to uniformity of soil in this region. Cambardella et al. (1994) and Afshar et al. (2009) also found similar results.
Plotted variograms on different directions including 0°, 45°, 135° for all soil variables in this study showed that effective range and sill of variograms were uniform and there was no clear anisotropy, and soil properties were recognized isotropic. This shows the variability of variables is equal in different directions and changes depend on distance between samples (Mohammad Zamani et al. 2007). The ratio of nugget to sill (C0/C0 + C) reflects the spatial autocorrelation. If it is ≤25 %, spatial dependent of variable is strong, if the ratio is between 25 and 75 %, spatial dependent of variable is moderate and if it is >75 %, spatial dependent of variable is weak (Cambardella et al. 1994). Models presented in Table 2 were selected to soil characteristics because they had less residual sum of squares and better structure. Suitable model for soil characteristics was isotropic.
In the study area the spatial dependence of soil characteristics was different. Phosphorus and potassium had weak spatial dependence, because the fitted R2 was <0.50 (Emadi et al. 2008). pH, electrical conductivity, organic carbon, nitrogen, sulphur, magnesium and sodium had moderate, similar to what had been illustrated in research of Cambardella et al. (1994). Jafarian Jeloudar et al. (2009) also reported that organic matter had moderate spatial dependence according to the results of Yi-chang et al. (2009) and calcium had strong spatial dependence in the study area according to results of Cambardella et al. (1994), Lopez-granados et al. (2002) and Weindorf and Zhu (2010). Variables with strong spatial structure and very low nugget effect have high continuous distribution in this area. Strong spatial dependence can be controlled through the inherent variability of soil properties such as soil texture, mineralogy and less spatial dependence by non-intrinsic factors such as grazing (Cambardella et al. 1994).
Semivariograms and maps of soil characteristics are presented in Figs. 2 and 3. Semivariograms have different forms depending on the quality of data and the distance between samples (Davatgar et al. 2001). The results showed spatial distribution of sulphur content that can be described with spherical model according to results of Jian-Bing et al. (2008), Jafarian Jeloudar et al. (2009), Vasques et al. (2010) and Weindorf and Zhu (2010). Nitrogen can be described with exponential model according to results of Jian-Bing et al. (2006). Available phosphorus can be expressed with spherical method as had been showed in research of Mohammadi and RaeisiGahrooee (2004), Yi-chang et al. (2009). The value of nugget effect for EC and calcium is small which suggest that the random variance of variables is low in the study area. This means that near and away samples have similar and different values respectively. In other words, a small nugget effect and close to zero indicates a spatial continuity between the neighboring points. Results of Vieira and Paz Gonzalez (2003), Mohammad Zamani et al. (2007) showed that variogram of nitrogen had very small nugget effect equal to 0.006. Jian-Bing et al. (2008), Afshar et al. (2009) and Kamare (2010) reported that nugget effect of electrical conductivity was 0.0008. Assessment of fitted models showed that models of sulphur and calcium content had a higher regression coefficient and thus have more accuracy (Table 3).
Results showed that pH, EC, organic carbon, nitrogen, phosphorus, potassium, magnesium and sodium had highest effective range and sulphur and calcium had minimum effective range. The larger effective range has more widespread spatial structure and this expansion will increase the virtual range which can be used to estimate the amount of regional variable at unknown points. Effective range of some soil properties were higher than others which probably is due to same impact of intrinsic processes on these soil characteristics. Spatial structure of these parameters have been more widespread rather than others and also in sampling design, one can extend sampling interval up to effective range. The effective ranges were 378–82,660 meters in this study which represents an increase in soil heterogeneity or potential of retrospection processes. The results can be used to make recommendations of best management and modeling of soil and plant relationships in future studies.
Conclusions
Estimating the spatial variability of soil physical and chemical properties is a pre-requisite for soil and plant specific management. The resulted maps of soil properties along with their spatial structures has delineated the management zones to be attended first in future to improve the soil quality and can be used in making better future sampling designs to make efficient management decisions.
References
Afshar, H., Salehi, M. H., Mohammadi, J., & Mehnatkesh, A. (2009). Spatial variability of soil properties and irrigated wheat yield in quantitative suitability map, a case study: Share e Kian Area, Chaharmahaleva Bakhtiari province. Journal of Water and Soil, 23(1), 161–172.
Anderson, C. J., Mitsch, W. J., & Nairn, R. W. (2005). Temporal and spatial development of surface soil conditions at two created riverine marshes. Journal of Environmental Quality, 34, 2072–2081.
Ayoubi, S., & Khormali, F. (2009). Spatial variability of soil surface nutrients using principal component analysis and geostatistics: A case study of Appaipally village, Andhra Pradesh, India. Journal of Science and Technology of Agriculture and Natural Resources, Water and Soil Science, Isfahan University of Technology, 12(46), 609–622.
Burke, A. (2001). Classification and ordination of plant communities of the Nauklaft Mountain, Namibia. Journal of Vegetation Science, 12, 53–60.
Cambardella, C. A., Moorman, T. B., Parkin, T. B., Turco, D. L., Karlen, R. F., & Konopka, A. E. (1994). Field scale variability of soil properties in Central Iowa soils. Soil Science Society of America Journal, 58, 1501–1511.
Cheng, X., An, S., Chen, J., Li, B., Liu, Y., & Liu, S. (2007). Spatial relationships among species, above-ground biomass, N, and P in degraded grasslands in Ordos Plateau, northwestern China. Journal of Arid Environments, 68, 652–667.
Chesnin, L., & Yien, C. H. (1951). Turbidimetric determination of available sulphur. Proceedings of Soil Science Society of America, 15, 149–151.
Covelo, F., Rodríguez, A., & Gallardo, A. (2008). Spatial pattern and scale of leaf N and P resorption efficiency and proficiency in a Quercusrobur population. Plant and Soil, 311, 109–119.
Davatgar, N. (1998). Investigation spatial variability of some soil characteristics. M.Sc. Thesis, Faculty of agriculture, Tabriz University, pp. 108.
Davatgar, N., Neyshabouri, M. R., & Moghaddam, M. R. (2001). The analysis of information obtained from soil variables map by use of semivariogram models. Iranian Journal of Agricultural Sciences, 31(4), 725–735.
Du Feng, L. Z., XuXuexuan, Z. X., & Shan, L. (2008). Spatial heterogeneity of soil nutrients and aboveground biomass in abandoned old-fields of Loess Hilly region in Northern Shaanxi, China. Acta Ecologica Sinica, 28(1), 13–22.
Emadi, M., Baghernejad, M., Emadi, M., & Maftoun, M. (2008). Assessment of some soil properties by spatial variability in saline and sodic soils in Arsanjan plain, southern Iran. Pakistan Journal of Biological Sciences, 11(2), 238–243.
Etema, C., & Wardle, D. A. (2002). Spatial soil ecology. Trends in Ecology and Evolution, 17, 177–183.
Fennessy, M. S., & Mitsch, W. J. (2001). Effects of hydrology and spatial patterns of soil development in created riparian wetlands. Wetlands Ecology Management, 94, 103–120.
Goovaerts, P. (1997). Geostatistics for natural resources evaluation (p. 483). New York: Oxford University Press.
Hangsheng, L., Dan, W., Jay, B., & Larry, W. (2005). Assessment of soil spatial variability at multiple scales. Ecological Modelling, 182, 271–290.
Hunter, R. B., Romeny, E. M., & Wallace, A. (1982). Nitrate distribution in Majava Desert soils. Soil Science, 134, 22–30.
Isaaks, E. H., & Srivastava, R. M. (1989). An introduction to applied geostatistics (p. 561). New York: Oxford University Press.
Jafarian Jeloudar, Z., Arzani, H., Jafari, M., Kelarestaghi, A., Zahedi, G. H., & Azarnivand, H. (2009). Spatial distribution of soil properties using geostatistical methods in Rineh rangeland. Rangeland Journal, 3(1), 120–137.
Jian-Bing, W., Du-Ning, X., Hui, Z., & Yi-Kun, F. (2008). Spatial variability of soil properties in relation to land use and topography in a typical small watershed of the black soil region, northeastern China. Environmental Geology, 53, 1663–1672.
Jian-Bing, W., Du-Ning, X., Xing-Yi, Z., Xiu-Zhen, L., & Xiao-Yu, L. (2006). Spatial variability of soil organic carbon in relation to environmental factors of a typical small watershed in the black soil region, Northeast China. Environmental Monitoring and Assessment, 121, 597–613.
Kamare, R. (2010). Spatial variability of production, density and canopy cover percentage of Nitrariaschoberi L. in Meyghan Playa of Arak by using geostatistical methods. MSc Thesis, Tarbiat Modares University, pp. 76.
Knudsen, D., Peterson, G. A., & Pratt, P. F. (1982). Lithium, sodium, potassium. In A. L. Page (Ed.), Methods of soil analysis, part 2. Madison, WI: ASA-SSSA.
Kresic, N. (1997). Hydrogeology and groundwater modeling. Washington, D.C.: Lewis Publishers.
Lanyon, L. E., & Heald, W. R. (1982). Magnesium, calcium, strontium and barium. In A. L. Page, R. H. Miller, & D. R. Keeney (Eds.), Methods of soil analysis. Part 2 (2nd ed., pp. 247–262). Madison, WI: Agronomy No 9, American Society of Agronomy.
Lopez-Granados, F., Jurado-Exposito, M., Atenciano, S., Garcia-Ferrer, A., De la Orden, M. S., & Garcia-Torres, L. (2002). Spatial variability of agricultural soil parameters in southern Spain. Plant and Soil, 246, 97–105.
McBratney, A. B., & Webster, R. (1983). Optimal interpolation and isarithm mapping of soil properties. V. Co-regionalization and multiple sampling strategy. European Journal of Soil Science, 34, 137–162.
McLean, E. O. (1982). Soil pH and lime requirement. In A. L. Page (Ed.), Methods of soil analysis, part 2. Madison, WI: ASA-SSSA.
Mohammad Zamani, S., Auubi, S., & Khormali, F. (2007). Investigation of spatial variability soil properties and wheat production in some of farmland of sorkhkalateh of Golestan province. Journal of Science and Technical Agriculture and Natural Recourses, 11(40), 79–91.
Mohammadi, J., & RaeisiGahrooee, F. (2004). Fractal description of the impact of long-term grazing exclusion on spatial variability of some soil chemical properties. Journal of Science and Technology of Agriculture and Natural Resources, Water and Soil Science Isfahan University of Technology, 7(4), 25–37.
Noy-Mire, I. (1973). Multivariate analysis of the semi-arid vegetation of southern Australia. II. Vegetation catena and environmental gradients. Australian Journal of Botany, 22, 15–40.
Olsen, S. R., & Sommers, L. E. (1982). Phosphorus. In A. L. Page (Ed.), Methods of soil analysis, part 2: Chemical and microbiological properties (2nd ed., pp. 403–430). Madison, WI: Agron. No.9, American Society of Agronomy.
PazGonzales, A., Vieira, S. R., & Castro, T. (2000). The effect of cultivation on the spatial variability of selected properties of an umbric horizon. Geoderma, 97, 273–292.
Pohlmann, H. (1993). Geostatistical modeling of environment data. Catena, 20, 191–198.
Robertson, G. P., Huston, M. A., Evans, F. C., & Tiedje, J. M. (1988). Spatial variability in a successional plant community: patterns of nitrogen availability. Ecology, 69, 1517–1524.
Rogerio, C., Ana, L. B. H., & de Quirijn, J. L. (2006). Spatio- temporal variability of soil water tension in a tropical soil in Brazil. Geoderma, 133, 231–243.
Sauer, T. J., Cambardella, C. A., & Meek, D. W. (2006). Spatial variation of soil properties relating to vegetation changes. Plant and Soil, 280, 1–5.
Schlesinger, W. H., Raikes, J. A., Hartley, A. E., & Cross, A. F. (1996). On the spatial pattern of soil nutrients in desert ecosystems. Ecology, 77, 364–374.
Sovik, A. K., & Aagaard, P. (2003). Spatial variability of a solid porous framework with regard to chemical and physical properties. Geoderma, 113, 47–76.
Subbiah, B. V., & Asija, G. L. (1956). A rapid procedure for the estimation of available nitrogen in soils. Current Science, 25, 259–260.
Vasques, G. M., Grunwald, S., Comerford, N. B., & Sickman, J. O. (2010). Regional modeling of soil Carbon at multiple depth within a subtropical watershed. Geoderma, 156, 326–336.
Venteris, E. R., McCarty, G. W., Ritchie, J. C., & Gish, T. (2004). Influence of management history and landscape variables on soil organic carbon and soil redistribution. Soil Science, 169(11), 787–795.
Vieira, S. R., & Paz Gonzalez, A. (2003). Analysis of the spatial variability of crop yield and soil properties in small agricultural plots. Bragantia, Campina, 62, 127–138.
Virgilio, N. D., Monti, A., & Venturi, G. (2007). Spatial variability of switchgrass (Panicumvirgatum L.) yield as related to soil parameters in a small field. Field Crops Research, 101, 232–239.
Walkley, A. J., & Black, C. A. (1934). An estimation of the Degtjareff method for determining soil organic matter and a proposed modification of the chromic acid titration method. Soil Science, 37, 29–38.
Wang, X. J., & Qi, F. (1998). The effects of sampling design on spatial structure analysis of contaminated soil. The Science of the Total Environment, 224, 29–41.
Wang, Y., Zhang, X., & Huang, C. (2009). Spatial variability of soil total nitrogen and soil total phosphorus under different land uses in a small watershed on the Loess Plateau, China. Geoderma, 150, 141–149.
Webster, R., & Oliver, M. A. (2001). Geostatistics for environmental scientists. Brisbane: Wiley.
Weindorf, D. C., & Zhu, Y. (2010). Spatial variability of soil properties at Capulin volcano, New Mexico, USA: Implications for sampling strategy. Pedosphere, 20(2), 185–197.
Yi-chang, W., You-lu, B., Ji-yun, J., Fang, Z., Li-ping, Z., & Xiao-qiang, L. (2009). Spatial variability of soil chemical properties in the reclaiming marine foreland to yellow sea of China. Agricultural Sciences in China, 8(9), 1103–1111.
Yong, Z. S., Yul, L., & Halin, Z. (2006). Soil properties and their spatial pattern in a degraded sandy grassland under post- grazing restoration, Inner Mongolia, northern China. Biogeochemistry, 79, 297–314.
Zhang, C. S., & McGrath, D. (2004). Geostatistical and GIS analysis on soil organic carbon concentrations in grassland of southeastern Ireland from two different periods. Geoderma, 119, 261–270.
Zhao, Y., Peth, S., Krummelbein, J., Horn, R., Wang, Z., Steffens, M., et al. (2007). Spatial variability of soil properties affected by grazing intensity in Inner Mongolia grassland. Ecological Modeling, 205, 241–254.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Wani, M.A., Shaista, N. & Wani, Z.M. Spatial Variability of Some Chemical and Physical Soil Properties in Bandipora District of Lesser Himalayas. J Indian Soc Remote Sens 45, 611–620 (2017). https://doi.org/10.1007/s12524-016-0624-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12524-016-0624-z