Abstract
A study of phytoplankton diversity and its seasonal variation was conducted by sampling water from Boukourdane Lake between February 2013 and January 2015. Fifteen physical and chemical variables were recorded, and their relationship with the density of 162 phytoplankton species was established using Canonical Correspondence Analysis, which showed that the density of phytoplankton was higher when minerals and nutrients increased. An anthropogenic impact was also most evident in structuring phytoplankton assemblages at sites close to the lake where agriculture was concentrated. The results obtained from the Margalef, Shannon–Wiener, and Equitability diversity indices indicated mediocre water quality. Determinations of the trophic state were made by examining several diverse criteria, such as transparency and the concentrations of chlorophyll a and phosphorus. Our study area was characterized as mesotrophic according to the Organisation for Economic Co-operation and Development classification.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Wetlands are one of the most productive ecosystems on Earth (Ghermandi et al. 2008) and are known to sustain a varied diversity of both flora and fauna. Diversity, distribution, and variation in the biotic parameters provide a good indication of energy turnover in aquatic environments (Forsberg 1982). Within these environments, phytoplankton is located at the base level and is represented as a major source of organic carbon (Gaikwad et al. 2004). Species diversity and dynamics respond to changes in environmental gradients and may characterize many interactions that can establish the intricate pattern of community structure. However, data on the phytoplankton and the physicochemical features of Boukourdane Lake are completely lacking.
Lakes have been classified according to their trophic state since the early part of the twentieth century. A eutrophic lake is characterized by high nutrients and high plant growth, unlike an oligotrophic lake, whereas mesotrophic lakes fall somewhere in between eutrophic and oligotrophic lakes. Although lakes may be lumped into a few trophic classes, each lake has a unique constellation of attributes that contribute to its trophic status (LakeAccess 2006). Three main factors determine the trophic state of a lake (transparency, the concentration of phosphorus ions, and the concentration of Chlorophyll a). The determination of trophic status is a useful means of classifying lakes and describing the productivity of the system. The infertile soils in basins release relatively low concentrations of nitrogen and phosphorus, which leads to less productive systems classified as oligotrophic or mesotrophic. Catchment basins or agricultural regions rich in organic matter and fertilizers produce higher nutrient levels, which induce more productive systems and eutrophic lakes (LakeAccess 2006).
Moreover, recognizing and understanding the relative influence of natural- and human-induced processes on hydrological and biochemical functioning and phytoplankton abundance are prerequisites for improved water resources management.
To determine some physicochemical features for water as well as the temporal distribution patterns of the phytoplankton community, 162 freshwater phytoplankton species were recorded during a survey of the catchment area of the Boukourdane dam in northern Algeria. Seasonal fluctuations in density, diversity, and composition of seven classes of phytoplankton identified in Boukourdane Lake in relation to environmental factors are described in the present study. This relationship was examined using canonical correspondence analysis (CCA) to ascertain the interdependence between physicochemical descriptors and phytoplankton assemblages in 192 samples collected over a period of 2 years across four sampling stations.
The aims of this paper were (a) to examine the phytoplankton composition, diversity, and dynamics under a changing nutrient rate and (b) to assess the Boukourdane dam water quality and trophic state according to SEQ-EAU and The Organisation for Economic Co-operation and Development (OCDE) guidelines.
Materials and methods
Study area
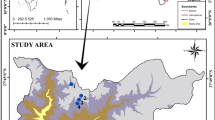
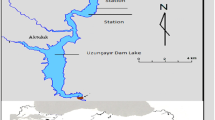
Boukourdane dam is located on the bed of Wadi El Hachem, at the juncture of two principal intermittent wadis, Menacer and Fedjana (Fig. 1). The study area is approximately 1.3 km south of the Village of Sidi Amar (Wilaya of Tipasa), located at a latitude of 35° 32′ N and a longitude of 2° 18′ E and an altitude of 119.5 m. The climate is typically Mediterranean, with a dry and hot period of 5 months stretching from mid-May to mid-October. The site is also 11 km from the Mediterranean Sea in a catchment area of over 177 km2. The average annual contribution of the Boukourdane dam reservoir is 2 million m3, presenting a full capacity of 101.5 million m3 with 700 mm annual rainfall. The reservoir has a maximum depth of 34 m and a total surface area of more than 536 ha.
Boukourdane dam is intended to supply the cities of Cherchell, Tipasa, and Nador with drinking water and enable irrigation of the valley of Wadi El Hachem and the areas of Hadjout and the Sahel.
Field sampling and lab analysis
We selected four stations distributed on the lake of the dam for monitoring (Fig. 1): S1 (in wadi Menacer), S2 (at the juncture of the two wadis Menacer and Fedjana), S3 (in wadi Fedjana), and S4 (near the dyke).
The sampling stations were selected based on the hydrographic system and potential sources of pollution. Sampling, transport, and conservation of the water samples were conducted based on the protocols described by Rodier et al. (2009). Samples were collected monthly over a period of 2 years, from January 2013 to January 2015, to obtain a representative image of water quality and its seasonal and annual evolution.
Five parameters were measured in situ; water temperature, pH, conductivity, and dissolved oxygen were determined using a Multi 340i/SET WTW analyzer, and a Secchi disk was used for transparency. Additionally, nine physicochemical parameters (nitrate, nitrite, ammonium, phosphorus, sulfate, calcium, magnesium, chloride, and suspended matter) were analyzed at the laboratory. Sulfate was measured using the gravimetric method, and molar titration was used to evaluate chloride, calcium, and magnesium. Filtration was used to measure the suspended matter (Rodier et al. 2009), and the colorimetric method with a continuous flow on an automated chain (SKALAR) was used to determine the nitrate, nitrite, ammonium, and phosphorus levels. Chlorophyll (a) was measured using the fluorometric method. Care was taken to not contaminate or modify the samples, which were transported to the laboratory in a cooler at 4 °C. Upon arrival in the laboratory, samples were analyzed according to the protocol described by Rodier et al. (2009), as described below.
A 50-μm mesh net was used for phytoplankton sampling. Samples were preserved in a 5% (final concentration) neutralized formaldehyde solution. The cells were counted on a ZEISS-WINKEL inverted microscope using the Utermöhl (1958) method.
The determinations of the trophic state were made by examining several diverse criteria, such as transparency, concentrations of chlorophyll a and phosphorus, and changes from oligotrophy to eutrophy; the changes did not occur at specific places, nor did they all occur at the same place or at the same rate. Some lakes may be considered oligotrophic by one criterion and eutrophic by another, which sometimes leads to the classification of lakes that show characteristics of both oligotrophy and eutrophy as mesotrophic (Carlson 1977). We characterized our study area according to the OCDE classification.
Statistical analysis
Dominance (Pi), diversity index Shannon–Wiener (H′), equitability index (E) (Shannon and Weaver 1949), and richness index Margalef (Dmg) (Margalef 1958) were used to calculate community metrics of phytoplankton.
where ni is the individual number of species i, Pi is the ratio of the individual number of the species i and total individual number N, and S is the total number of species in 1 month.
The differences in the physicochemical parameters were evaluated by the Kruskal–Wallis test. The overall dataset was analyzed by canonical correspondence analysis (CCA) to measure the contribution of the environmental parameters to the phytoplankton density. All analyses were conducted using the Ade4 package of the R programme (R Development Core Team 2014).
Results
Characterization of environmental conditions
The water temperature fluctuated with seasonal variations and climatic conditions, with the highest temperature recorded in the summer of 2014 (28.70 °C) and the lowest in winter (11.3 °C) (Table 1). Water in the reservoir tended to be alkaline, with pH values between 7.11 and 9.95. The highest pH was recorded in the spring of 2013. The dissolved oxygen levels varied between 1.40 and 11.61 mg L−1, with the highest value recorded in the winter of 2014. The nitrate and ammonium concentrations were low; the maximum nitrate value was recorded in the spring of 2014, whereas the minimum value was recorded in the summer of 2013 and autumn of 2014. Overall, the nitrate values ranged from 0.002 to 2.93 mg L−1. The maximum ammonium level was recorded in the spring of 2014 (1.27 mg L−1). The sulfate values fluctuated between 47.79 and 5768 mg L−1, with the highest concentrations recorded during the summer of 2014. The phosphorus ion levels ranged from 0.001 to 1.802 mg L−1, with the maximum value recorded in the spring of 2013.
Characterization of the phytoplankton community
A total of 162 phytoplankton species were recorded from 192 samples within the study period. The taxa belonging to Chlorophyceae exhibited the highest number of species (55), followed by Bacillariophyceae (diatoms) (47), Cyanophyceae (17), Euglenophyceae (15) and the less diverse Dinophyceae (10), Zygnematophyceae (10), Chrysophyceae (6), Xanthophyceae (1), and Coccolithophyceae (1).
We observed the proliferation of diatoms during almost the entire study period (Fig. 2); the highest dominance was marked during the winter season by the species Nitzschia acicularis, for which we obtained a maximum of 94.49%. Chlorophyceae mostly proliferated in the spring of 2013, with the maximum dominance of 29.78% by the species Pediastrum duplex. The maximum dominance of Euglenophyceae was registered during the spring of 2013 by the Phacus sp species, with a rate of 2.12%.
For Cyanophyceae, the highest dominance was recorded during the winter of 2013 (30.64%) by the species Microcystis aeroginosa, and a peak of Merismopedia sp. was also noted in the same season.
Zygnematophyceae were dominant in the spring of 2013, with 6.38%, and the dominate species was Closterium acutum var acutum.
Dinophyceae recorded their high dominance during the summer season, mainly in the summer of 2013, with a dominance of 15.76%, and in the spring of 2013, Peridinium cinctum was dominant (12.76%).
Xanthophyceae were recorded only during the spring of 2013, with a low rate (0.03%), by the species Ophiocytium capitatum.
Coccolithophyceae were recorded during the spring of 2013, with a dominance of 19.14%. They were represented only by the species Hymenomonas roseola. Chrysophyceae remained as a co-existing species in all periods; they were mainly represented by the species Dinobryon pediforme in the spring of 2014 (96.98%). These two groups fell sharply in winter.
Bioindices and water quality
The determination of the Schannon-Weiner index is important to the study of the structure of phytoplankton; it is the simplest index in its category and is thus the most widely used. The Shannon–Wiener index values varied between 0.36 and 3.49 (bits/ind). The values of the richness index ranged from 5.49 to 17.39 (Fig. 3).
The equitability results (Fig. 4) are similar and are superimposed on those of the Shannon–Wiener index. All values obtained were less than 0.50.
Discussion
The temperatures recorded during the study period indicate that the water of the Boukourdane dam reservoir is favorable to aquatic life, with mediocre quality according to the SEQ-EAU (2014) guidelines. Significant differences in temperatures were found during the study period (p < 0.001). The temporal evolution of the temperature for all of the stations indicates that the water temperature followed that of the air (Arab and Arab 2017a, b). The data recorded at the four stations did not vary greatly (p > 0.05), which is similar to the results reported by Ouhmidou et al. (2015). The pH values were slightly alkaline because of the limestone substrate of the geological formations in the area catchment, which is rich in carbonates, making it possible to buffer the water. Based on the pH, the water in the reservoir is of good quality according to SEQ-EAU (2014). Additionally, the pH did not significantly differ among stations (p > 0.05). Overall, the pH of all stations decreased during the summer, which was likely because of increasing water temperature (p < 0.001). These findings agree with those reported by El Ghachtoul et al. (2005) and Ouhmidou et al. (2015). The dissolved oxygen rates showed the water in the Boukourdane reservoir is well oxygenated (SEQ-EAU. 2014), with significantly lower values in summer than in winter (p < 0.001) because reduction of the solubility of this element when the temperature increased (Ouhmidou et al. 2015). The observed low nitrate concentrations indicate that the water is of good quality according to SEQ-EAU (2014), with a significant difference (p < 0.001). Its temporal evolution was also observed in the studies of Ouhmidou et al. (2015) and Martinelli et al. (1999). The presence of nitrates in surface water can be linked either to the contributions from agricultural processes (e.g., leaching of fertilizers containing nitrate) or to the transformation into nitrate of ammonium coming from domestic or animal discharges (Djabri 1996; El Ouali Lalami et al. 2011; Errochdi et al. 2012; Arab and Arab 2017a, b). The maximum sulfate concentrations were recorded during the dry season.
In North Algeria, most of the problems associated with the direct discharge of domestic wastewater have been successfully attenuated. The current Algerian regulation focus is on the much more difficult problem of controlling non-point sources (NPS) of nutrient pollution such as agricultural drainage, stormwater runoff, and inadequate on-site septic systems. NPS pollution is particularly difficult to control because it is diffuse, not attributable to a small number of polluters, and associated with fundamental changes in the landscape, such as agriculture, urbanization, and shoreline development (LakeAccess 2006). Boukourdane Lake was classified as mesotrophic based upon the OCDE fixed boundary system (Forsberg 1982) (Table 2). Depending on Secchi depth, it was classified, as eutrophic and mesotrophic for the other parameters (chlorophyll a, phosphorus). The work of Hamaidi (2012) and El Houati (2015) on the same lake reservoir indicated trophic conditions comparable to those obtained in this study.
The study of phytoplankton diversity and its seasonal variation according to the physicochemical parameters was established using Canonical Correspondence Analysis (CCA). The projection of the variables and the species was carried out on two axes and represent 83.15% of the total inertia (Fig. 5); this showed that the density of phytoplankton was higher when minerals and nutrients increased. Where, Nitzschia acicularis, was the most abundant species in the phytoplankton community (Arab and Arab 2017b), the community consisted of assemblages of species with morphological and physiological characteristics with different ecological organizations. Boukourdane Lake was dominated by Chlorophyceae compared with Diatoms for small and disturbed sites; the same results were found by Moshood (2009).
Phosphorus, carbonate, and conductivity ions are physicochemical parameters that significantly influence diatoms (Soininen and Kononen 2004; Cvetkoska et al. 2018), which is consistent with our results where we recorded these parameters with Cyclotella sp1, Cyclotella menegheniana, Nitzschia acicularis, and Synedra acus. We also found that different species of Cyclotella and Nitzschia were strongly related to conductivity and phosphorus ions, the same results reported by El Haouati et al. (2015).
Abubacker et al. (1996) reported that species such as Cyclotella sp., Gomphonema sp., and Nitzschia sp. were tolerant to eutrophic conditions. Similar studies by Bhatt et al. (2001), Jindal and Vatsal (2005), and Ghavzan et al. (2006) reported that forms such as Nitzschia palea and Synedra ulna were pollution-resistant and showed an elevation of their dominance values during the spring period over 2 years of study, which indicated a high pollution load.
Among the species that dominated the summer season were Nitzschia acicularis and Gyrosigma acuminatum (Arab and Arab 2017b); a similar trend was observed by Rolland (2009). These two species were the main diatoms. Whereas the PEG-model generalized this point on diatoms that often develop in early spring (Sommer et al. 1986), we noticed that the genus Cymateupleura was found in the autumn of 2014 (C. elliptica) and in the winter of 2015 (C. solea), unlike El Haouati et al. (2015), who found C. solea during the spring season, for the same environmental conditions where we recorded a re-increase in the mineralization of water.
Nitzschia palea is a common species in Algerian surface waters (Baudrimont 1973; Chaïb et al. 2011) and is known for its resistance to organic pollution (Lange-Bertalot 1979) and relatively high concentrations of H2S (Baudrimont 1973). We recorded its highest concentration in the spring of 2013 when we noticed a considerable increase in sulfate ions (1255.024 mg L−1); this is not consistent with the results of El Haouati et al. (2015), in which observations were taking during the fall season. The calcium ion is one of the parameters that significantly influence the diatoms (Soininen and Kononen 2004), this finding is consistent with a study conducted by El Haouati et al. (2015), although we recorded the species Achnanthes minutissima.
We have recorded the maximum dominance of the centric diatom Cyclotella ocellata during the fall season, which confirms Berrada et al. (1999).
Most of the initial detritus from domestic waste is incompletely mineralized. Therefore, such detritus sinks and accumulates on the lake bottom, which results in the extension of areas of anoxia in the water column throughout the entire ecosystem. Release into the ecosystem of large amounts of untreated domestic sewage leads to the growth of a taxonomy dominated by Chlorophytaceae, the Cyanophyceae and Euglenophyceae. Those genera are characteristic of polluted and organic substances-rich environments (Akpan and Offem 2004), including the genera Scenedesmus and Microcystis, which are known for their preference for eutrophic environments (Reynolds et al. 2000).
Chlorophyceae (Tetraspora gelatinosa, Pediastrum duplex, Pediastrum simplex, Chlorella sp) and zygnematophyceae (Staurastrum gracile) seem to be regulated by mineralization elements, such as magnesium, calcium, and chloride, and are related to the pH. Additionally, species (Scenedesmus bijugatus, Tetraedron minimum) were found to be related to chlorophyll a; these species were previously recorded by Atanle et al. (2013) in the same environment.
Atici and Alas (2012) studied the trophic status of Mamasin Dam Lake (Aksaray-Turkey) and found Chlorella and Scenedesmus sp. as pollution indicators. Most of the Scenedesmus sp. were present in Boukourdane Lake, with Scenedesmus ecornis having the highest abundance among all species.
Vyas and Kumar (1968) reported the presence of euglenophyceae during the rainy season. In contrast, these were present mostly throughout the year during our study and were found in low loads during summer.
Some cyanobacteria species, such as Anabaena sp., Cylindrspermopsis raciborskii, and Microcystis aeroginosa, appeared in the autumn of 2013 and 2014 (Arab and Arab 2017b). These results corroborate those of Padisák (1997), Groga (2012), and Radji et al. (2013), who have shown that the modifications in conjunction with the lowering of the lake level have a direct impact on the phytoplankton community for some major genera of cyanobacteria (Cylindrospermopsis, Microcystis, Anabaena). These environmental conditions are related to physical factors and are very favorable to the growth of these cyanobacteria as shown by various works of Wetzel (2001), which coincides with the high temperatures recorded in this period (Mowe et al. 2015). Cyanophyceae have optimum growth at high temperatures (28–32 °C), although they are able to tolerate and survive at lower temperatures (Beaver et al. 2018; Ribeiro et al. 2018). Therefore, they support abundant organic matter and low levels of dissolved oxygen. In September, phytoplankton individuals in the reservoir were in a mixed water column and were less affected by dilution. Moreover, the preceding phytoplankton increase and cyanobacterial dominance were observed when the water volume decreased to the lowest level, which occurred after a period without flushing in the reservoir, indicating the impact of flushing on phytoplankton dynamics; the same results were found by Katsiapi et al. (2011). While pointing out the presence of certain species that we found in our surveys as potentially toxic, such as Anabaena sp. Merismopedia sp., Microcystis aeruginosa, and Oscillatoria sp., our results confirm those of Schembri et al. (2001), Loez and Salibian (2008), and Radji et al. (2013).
Atanle et al. (2013) found a strong correlation between dinophyceae and mineralized water, which corroborates our result of the Dinobryon pediform species in water rich in carbonates and with a high conductivity; he also found that Peridinium cinctum and Peridinium aciculiferum were related to sulphate, phosphates, and ammonium ions.
For phytoplankton communities, Cottingharn (1999) suggests that anthropogenic stressors (like nutrient input) affect lake phytoplankton; however, predicting phytoplankton dynamics is not an easy task (Kagalou et al. 2003).
Diversity indices can serve as a good indicator of the overall quality of water. Non-polluted waters are often characterized by high diversity, with a large number of species and no single species dominating in numbers over others. However, with pollution causing stress, sensitive species are eliminated and tolerant species increase in numbers to become dominant. Bioindices of species diversity can be derived from species counts and we used two categories: species richness (Margalef index), a combination of richness, and dominance (Shannon–Wiener index). These diversity indices were developed by considering the number of species and their relative abundances, which means that the higher the values of these diversity indices, the more the oligotrophic state of water bodies (Margalef 1958). The fall in the value of the Margalef index shows the rise in the level of pollution (Arab and Arab 2017b). When calculating the Shannon–Wiener index, a value of this index above 3 indicates clean water, whereas values lower than this would indicate pollution. Comparing the data on the species diversity indices of Boukourdane Lake, it has been observed in 2 years that the values of the Margalef richness index were 5.49–17.39 and those of the Shannon–Wiener index were 0.36–3.49. The higher values indicate the healthy status of the lake, and the lowest values show the unhealthy and polluted status of the lake. The Shannon–Wiener index of species diversity for the entire planktonic community has been found to decrease with the increase in eutrophication (Kajak 1983; Rogozin 2000; Paturej 2006). The equitability index values (0.06–0.61) confirm those of Shannon–Wiener. The common characteristic of eutrophic ecosystems is known to be the presence of a few dominant species with high density (Green 1993).
The species diversity of organisms determined at various sites revealed that minimum species diversity was in winter and summer because of the elimination of sensitive forms and the flourishing of tolerant forms, which indicate a higher pollution load.
Conclusions
This work is based on a recent field study and includes various data available for Lake Boukourdane. The physical and chemical analysis of water and the study of the phytoplankton community based on sampling performed during an annual cycle were used to determine the trophic status of Boukourdane Lake and also the effects of pressure and subsequent impacts in the study area. This Lake is under a disturbance of its ecosystem in terms of physical chemistry and phytoplankton diversity levels due to the anthropological influence in its proximity; it is engaged in a eutrophication process. The imbalance of the ecosystem from continuous nutrient inputs and domestic untreated discharges from extensive agricultural areas around the lake is causing the instability of phytoplankton communities. These observations are signs of advanced functional degradation of the ecosystem. Therefore, the results of this study could help in pollution management and projects for the preservation of this Wetland. The rapid respond and evolution of phytoplankton communities related to the environmental changes increase interest in their ecological follow-up.
References
Abubacker MN, Kannan V, Sridharan VT, Chandramohan M, Rajavelu S (1996) Physico chemical and biological studies on Uyyakondan Canal water of river Cauvery. Pollut Res 15(3):257–259
Akpan ER, Offem JO (2004) Seasonal variation in water quality of the Cross river, Nigeria. Rev d’Hydrobiol Trop 26:95–103
Arab S, Arab A (2017a) Effect of the physico-chemical parameters on the distribution of the fecal flora in a dam reservoir (Algeria). Rev Ecol (Terre Vie) 72(3):269–280 http://hdl.handle.net/2042/62314
Arab S, Arab A (2017b) Bioévaluation de la qualité des eaux d’un écosystème lacustre le cas du barrage de Boukourdane wilaya de TIPASA. Dissertation, University of science and technologie Houari Boumedien
Atanle K, Bawa LM, Kokou K, Djaneye-boundjou G, Edorh MT (2013) Distribution saisonnière du phytoplancton en fonction des caractéristiques physico-chimiques du lac de Zowla. J Appl Biosci 64:4847–4857. https://doi.org/10.4314/jab.v64i1.88474
Atici T, Alas A (2012) A study on the trophic status and phytoplanktonic algae of Mamasin Dam Lake (Aksara Turkey). Turk J Fish Aquat Sci 12:595–601. https://doi.org/10.4194/1303-2712-v12_3_06
Baudrimont R (1973) Recherche sur les diatomées des eaux continentales de l’Algérie, écologie et paléoécologie. Dissertation, University of Bordeaux.
Beaver JR, Kirsch JE, Tausz CE, Samples EE, Renicker TR, Scotese KC, McMaster HA, Blasius-Wert BJ, Zimba PV, Casamatta DA (2018) Long-term trends in seasonal plankton dynamics in Lake Mead (Nevada-Arizona, USA) and implications for climate change. Hydrobiologia 822:85–109. https://doi.org/10.1007/s10750-018-3638-4
Berrada DF, Berrada R, Benzekri A (1999) Dynamique du phytoplancton en relation avec certains paramètres physico-chimiques dans la retenue El Kansera (Maroc). Ann Limnol Int J Lim 35(3):155–166. https://doi.org/10.1051/limn/1999022
Bhatt JP, Jain A, Bhaskar A, Pandit MK (2001) Pre-impoundment study of biotic communities of Kistobazar Nala in Purulia (West Bengal). Curr Sci 81:10
Carlson RE (1977) A trophic state index for lakes. Limnol Oceanogr 22(2):361–369. https://doi.org/10.4319/lo.1977.22.2.0361
Chaïb N, Alfarhan AH, Al-rasheid KAS, Samraoui B (2011) Environmental determinants of diatoms assemblages along a North African wadi, the Kebir-East, North-East Algeria. Int J Limnol 70:33–40. https://doi.org/10.4081/jlimnol.2011.33
Cottingharn KL (1999) Nutrients and zooplankton as multiple stressors of phytoplankton communities: evidence from size structure. Limnol Oceanogr 44(8):10–827
Cvetkoska A, Pavlov A, Jovanovska E, Tofilovska S, Blanco S, Ector L, Wagner-Cremer F, Levkov Z (2018) Spatial patterns of diatom diversity and community structure in ancient Lake Ohrid. Hydrobiologia 819:197–215. https://doi.org/10.1007/s10750-018-3637-5
Djabri L (1996) Mécanismes de la pollution et vulnérabilité des eaux de la Seybouse. Origines géologiques, industrielles, agricoles et urbaines. Dissertation, University of Annaba
El Ghachtoul Y, Alaoui mhamidi M, Gabi H (2005) Eutrophisation des eaux des retenues des barrages Smir et Sehla (maroc): causes, conséquences et consignes de gestion. Rev Sci Eau 18:75–89. https://doi.org/10.7202/705577ar
El Haouati H, Arab A, Tudesque L, Lek S, Samraoui B (2015) Study of the diatoms of Reghaia lake, northern Algeria. Rev Ecol (Terre Vie) 70:44–75 http://hdl.handle.net/2042/56309
El Houati H (2015) Adaptation d’un indice phytoplanctonique pour l’évaluation de la qualité des eaux des écosystèmes lacustres Algériens. Dissertation, University of science and technologie Houari Boumedien
El Ouali Lalami A, Merzouki M, El Hillali O, Maniar S, Ibnsouda koraichi S (2011) Pollution des eaux de surface de la ville de Fès au Maroc : typologie, origine et conséquences. Larhyss J 9:55–72
Errochdi S, EL ALAMI M, Bennas N, Belqat B, Ater M, Fdil F (2012) Étude de la qualité physicochimique et microbiologique de deux réseaux hydrographiques nord marocains: Laou et Tahaddart, Méditerranée, vol 118, pp 41–51. http://journals.openedition.org/mediterranee/6221
Forsberg C (1982) Limnological research can improve and reduce the cost of monitoring and control of water quality. Hydrobiologia 86:143–146. https://doi.org/10.1007/BF00005801
Gaikwad SR, Tarot SR, Chavan TP (2004) Diversity of Phytoplankton and zooplankton with respect to pollution status of river Tapi in north Maharastra region. J Curr Sci 5:749–754
Ghavzan NJ, Gunale VR, Trivedy RK (2006) Limnological evaluation of an urban fresh water river with special reference to phytoplankton. PollutRes 25(2):259–268
Ghermandi A, Van den Bergh JCJM, Brander LM, Nunes PALD (2008) The economic value of wetland conservation and creation: a meta-analysis. [Working Paper 79]. Fondazione Eni Enrico Mattei, Milan, Italy
Green J (1993) Diversity and dominance in planktonic rotifers. Hydrobiologia 255(256):345–352
Groga N (2012) Structure fonctionnement et dynamique du phytoplancton dans le lac de Taabo (Côte d'Ivoire). Dissertation, the National Institut of polytechnic of Toulouse (INP Toulouse)
Hamaidi-Chergui F (2012) Contribution à la connaissance des peuplements planctoniques de six plans d’eau du nord de l’Algérie. Dissertation, university of science and technologie Houari Boumedien
Jindal R, Vatsal P (2005) Plankton as biomonitors ofsaprobity. Aquacult 6(1):1–16
Kagalou I, Petridis D, Tsimarakis G (2003) Seasonal variation of water quality parameters and plankton in a shallow Greek lake. J Freshw Ecol 18(2):199–206. https://doi.org/10.1080/02705060.2003.9664485
Kajak Z (1983) Ecological characteristics of lakes in North-Eastern Poland vs. their trophic gradient. XII. Dependence of chosen indices of structure and functioning of ecosystems of different trophic status and mictic type for 42 lakes. Ekol Pol 31:495–530
Katsiapi M, Moustaka-Gouni M, Michaloudi KKA (2011) Phytoplankton and water quality in a Mediterranean drinking-water reservoir (Marathonas Reservoir, Greece). Environ Monit Assess 181:563–575. https://doi.org/10.1007/s10661-010-1851-3
LakeAccess (2006) Seeing below the surface. Three Rivers Park District, MN and University of Minnesota-Duluth, Duluth, MN 55812. http://www.lakeaccess.org/ecology/lakeecologyprim15.html. Accessed 5 May 2018
Lange-Bertalot H (1979) Pollution tolerance of diatoms as a criterion of water quality estimation. Nova Hedwig Beih 64:285–304
Loez C, Salibian A (2008) Premières données sur le phytoplancton et les caractéristiques physico chimiques du Rio Reconquista (Buenos Aires, Argentine). Rev Hydrobiol Trop 23:283–296 http://www.documentation.ird.fr/hor/fdi:34342
Margalef R (1958) Temporal succession and spatial heterogeneity in phytoplankton. In: Buzzati-Treverso AA (ed) Perspectives in Marine Biology, Berkeley
Martinelli L, Krushe AV, VIctoria RL, De camango PB, Benardes M, Ferraz FS, De Mareas JM, Ballester MM (1999) Effets of sewage on the chemical composition of Piracaba river Brasil. Water Air Soil Pollut 110:67–79. https://doi.org/10.1023/A:1005052213652
Moshood MK (2009) Phytoplankton assemblage of a small, shallow, tropical African reservoir. Rev Biol Trop 57(4):1009–1025. https://doi.org/10.15517/rbt.v57i4.5443
Mowe MAD, Mitrovic SM, Lim RP, Furey A, Yeo DCJ (2015) Tropical cyanobacterial blooms: a review of prevalence, problem taxa, toxins and influencing environmental factors. J Limnol 74:205–224
O.C.D.E. (1982) Eutrophisation des eaux: méthode de surveillance, d'évaluation et de lutte. Organisation de Coopération et de Développement Economiques, Paris
Ouhmidou M, Chahlaoui A, Kharroubi A, Chahboune M (2015) Etude de la qualité physico-chimique et bactériologique des eaux du barrage Hassan Addakhil d’Errachidia (Maroc). J Mater Environ Sci 6:1663–1671
Padisák J (1997) Cylindrospermopsis raciborskii (Woloszynska) Seenayya and Subba Raju, an expanding, highly adaptative cyanobacterium: worldwide distribution and review of its ecology. Algol Stud 107:563–593 http://real.mtak.hu/id/eprint/3229
Paturej E (2006) Assessment of the trophic state of the coastal Lake Gardno based on community structure and zooplankton-related indices. EJPAU Biol 9(2):3–14 http://www.ejpau.media.pl/volume9/issue2/art-17.html
R Development Core Team (2014) R: A language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria. Available at http://www.R-project.org
Radji R, Bandje A, Issifou L, Edorh T, Kokou K (2013) Diversité et dynamique des assemblages phytoplanctoniques dans les écosystèmes aquatiques au Sud du Togo. Afr Sc 9(2):66–77
Reynolds CS, Reynolds SN, Munawar IF, Munawar M (2000) The regulation of phytoplankton population dynamics in the world’s largest lakes. Aquat Ecosyst Health 3:1–21. https://doi.org/10.1016/S1463-4988(99)00066-4
Ribeiro KF, Duarte L, Crossetti LO (2018) Everything is not everywhere: a tale on the biogeography of cyanobacteria. Hydrobiologia 820:23–48. https://doi.org/10.1007/s10750-018-3669-x
Rodier, J., Legube, B., Merlet, N et al (2009) l’Analyse de l’eau. 9emeEd. Dunod. Paris.
Rogozin AG (2000) Specific structural features of zooplankton in lakes differing in trophic status: species populations. Ekol (Moscow) 31(6):438–443. https://doi.org/10.1023/A:1026691809887
Rolland A (2009) dynamique et diversité du phytoplancton dans le réservoir marne (bassin versant de la seine). Dissertation, University of Savoie
Schembri MA, Neilan BA, Saint CP (2001) Identification of genes implicated in toxin production in the cyanobacterium Cylindrospermopsis raciborskii. Environ Toxicol 16(5):413–421
Shannon CE, Weaver W (1949) The mathematical theory of communication. University of Illinois Press, Urbana
Soininen J, Kononen K (2004) Comparative study of monitoring South-Finnish rivers and streams using macroinvertebrate and benthic diatom community structure. Aquat Ecol 38:63–75. https://doi.org/10.1023/B:AECO.0000021004.06965.bd
Sommer U, Gliwicz ZM, Lampert W, Duncan A (1986) The PEG-model of seasonal succession of planktonic events in fresh waters. Arch Hydrobiol 106:433–471
Système d'évaluation de la qualité de l'eau (SEQ-Eau) (2014) Normes de qualité des eaux des masses d’eau naturelles. Directive cadre eau européenne MEED et agence de l’eau. Accessed 5 May 2018
Utermöhl H (1958) ―Zur Vervollkommnung der quantitativen Phytoplankton-methodik. Mitteilungen der Internationale Vereinigung fur Theoretische und Angewandte Limnologie 9:1–38
Vyas LN, Kumar HD (1968) Studies on phytoplankton and other algae of Indra Sagar tank, Udaipur, India. Hydrobiologia 31:421–434. https://doi.org/10.1007/BF00134443
Wetzel RG (2001) Limnology: lake and river ecosystems. London
Acknowledgments
The authors are grateful for RABAH MAZARI Nadia from the Algerian Institute of Forensics science and for Khaled BELHASNAT (for assisting with sample collection) from the National Superior School of Marine Science and Coastal Management, which contributed in the realization of this project.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Water resources and water management for environmental integration in the Euro-Mediterranean region
Rights and permissions
About this article
Cite this article
Arab, S., Hamil, S., Rezzaz, M.A. et al. Seasonal variation of water quality and phytoplankton dynamics and diversity in the surface water of Boukourdane Lake, Algeria. Arab J Geosci 12, 29 (2019). https://doi.org/10.1007/s12517-018-4164-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12517-018-4164-4