Abstract
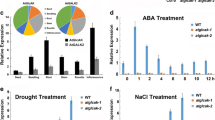
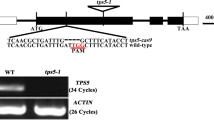
Sugars such as sucrose or glucose function both as building materials for biosynthesis, and as signaling molecules that modulate gene expression. Compared to studies of sugar signaling in bacteria, yeast and animals, knowledge of the signaling pathways in plants is still poorly understood. Here, we investigated the effect of the disruption and overexpression of an Arabidopsis thaliana adenosine 5’-phosphosulfate reductase 2, AtAPR2, on plant responses to glucose stresses. AtAPR2 encodes an enzyme of the sulfate assimilation pathway and it is a member of a three gene family that also includes AtAPR1 and AtAPR3. Expression of AtAPR1, AtAPR2 and AtAPR3 were strongly induced by glucose treatment. Overexpression of AtAPR2 resulted in enhanced cotyledon greening and fresh weight increase when plants were treated with high glucose. By contrast, a T-DNA insertion mutant (atapr2-2) line showed delayed greening and fresh weight growth inhibition in response to glucose and also the non-metabolizable analog 2-deoxyglucose. The expression of three glucose responsive genes, Hexokinase 1 (HXK1), Phenylalanine ammonia lyase 1 (PAL1) and Pathogenesis related gene 5 (PR5), was elevated in AtAPR2-overexpressing and WT plants in response to glucose treatment, but in the atapr2-2 mutant line the transcript level for these genes decreased. Furthermore, AtAPR2-overexpressing plants displayed delayed flowering under long day condition. The data implicates AtAPR2 as a component controlling flowering time and glucose response in Arabidopsis thaliana, although the exact function of AtAPR2 is not clear.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Berendt U, Haverkamp T, Prior A, Schwenn J. D (1995) Reaction mechanism of thioredoxin: 3’-phospho-adenylylsulfate reductase investigated by site-directed mutagenesis. Eur J Biochem 233:347–356
Bick JA, Dennis JJ, Zylstra GJ, Nowack J, Leustek T (2000) Identification of a new class of 5’-adenylylsulfate (APS) reductase from sulfate-assimilating bacteria. J Bacteriol 182:135–142
Brunold C (1990) Reduction of sulfate to sulfide. In: Rennenberg H, Brunold C, De Kok LJ, Stulen I (eds) Sulfur nutrition and sulfur assimilation in higher plants: fundamental, environmental and agricultural aspects. SPB Academic Publishing, The Hague pp 13–31, ISBN 90-5103-038-X
Carroll KS, Gao H, Chen H, Stout CD, Leary JA, Bertozzi CR (2005) A conserved mechanism for sulfonucleotide reduction. PLoS Biol 3 e250
Chartron J, Carroll KS, Shiau C, Gao H, Leary JA, Bertozzi CR, Stout CD (2006) Substrate recognition, protein dynamics, and iron-sulfur cluster in Pseudomonas aeruginosa adenosine-5’-phosphsulfate reductase. J Mol Biol 364:162–169
Chartron J, Shiao C, Stout CD, Carroll KS (2007) 3’-Phosphoadenosine-5’-phosphosulfate reductase in complex with thioredoxin: A structural snapshot in the catalytic cycle. Biochem 46:3942–3951
Earley KW, Haag JR, Pontes O, Opper K, Juehne T, Song K, Pikaard CS. (2006) Gateway-compatible vectors for plant functional genomics and proteomics. Plant J 45:616–629
Fujiki Y, Ito M, Nishida I, Watanabe A (2000) Multiple signaling pathways in gene expression during sugar starvation. Pharmacological analysis of din gene expression in suspension-cultured cells of Arabidopsis. Plant Physiol 124:1139–1148
Funck D, Claub K, Frommer WB, Hellmann H.A. (2012) The Arabidopsis CstF64-like RSR1/ESP1 protein participates in glucose signaling and flowering time control. Front Plant Sci 3:1–13
Gibson SI (2005) Control of plant development and gene expression by sugar signaling. Curr Opin Plant Biol 8:93–102
Graham IA (1996) Carbohydrate control of gene expression in higher plants. Res Microbiol 147:572–580
Hesse H, Trachsel N, Suter M, Kopriva S, von Ballmoos P, Rennenberg H, Brunold C (2003) Effect of glucose on assimilatory sulphate reduction in Arabidopsis thaliana roots. J Exp Bot 54:1701–1709
Kim SK, Rahman A, Bick JA, Conover RC, Johnson MK, Mason JT, Hirasawa M, Leustek, T, Knaff DB (2004) Properties of the cysteine residues and iron-sulfur cluster of the assimilatory 5’-adenylylsulfate reductase from Pseudomonas aeruginosa. Biochem 43:13478–13486
Kim SK, Rahman A, Conover RC, Johnson MK, Mason JT, Hirasawa M, Moore ML, Leustek T, Knaff DB (2006) Properties of the cysteine residues and the iron-sulfur cluster of the assimilatory 5’-adenylyl sulfate reductase from Enteromorpha intestinalis. Biochem 45:5010–5018
Kim SK, Rahman A, Mason JT, Hirasawa M, Conover RC, Johnson MK, Miginiac-Maslow M, Keryer E, Knaff DB, Leustek T (2005) The interaction of 5’-adenylylsulfate reductase from Pseudomonas aeruginosa with its substrates. Biochim Biophys Acta 1710:103–112
Koch KE (1996) Carbohydrate modulated gene expression in plants. Annu Rev Plant Physiol Plant Mol Biol 47:509–540
Kopriva S, Suter M, von Ballmoos P, Hesse H, Krähenbühl U, Rennenberg H, Brunold C (2002) Interaction of sulfate assimilation with carbon and nitrogen metabolism in Lemna minor. Plant Physiol 130:1406–1413
Krapp A, Hofmann B, Schafer C, Stitt M (1993) Regulation of the expression of rbcS and other photosynthetic genes by carbohydrates: a mechanism for the sink regulation of photosynthesis? Plant J 3:817–828
Kruse C, Jost R, Lipschis M, Kopp B, Hartmann M, Hell R (2007) Sulfur-enhanced defence: effects of sulfur metabolism, nitrogen supply, and pathogen lifestyle. Plant Biol 9:608–619
Leustek T, Martin MN, Bick JA, Davies JP (2000) Pathways and regulation of sulfur metabolism revealed through molecular and genetic studies. Ann Rev Plant Physiol Plant Mol Biol 51:141–165
Lillig CH, Prior A, Schwenn JD, Aslund F, Ritz D, Vlamis-Gardikas A, Holmgren A (1999) New thioredoxins and glutaredoxins as electron donors of 3'-phosphoadenylylsulfate reductase. J Biol Chem 274:7695–7698
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2T-DDC method. Methods 25:402–408
Loreti E, de Bellis L, Alpi A, Perata P (2001) Why and how do plants sense sugars? Ann Bot 88:803–812
Mugford SG, Lee BR, Koprivova A, Matthewman C, Kopriva S (2011) Control of sulfur partitioning between primary and secondary metabolism. Plant J 65:96–105
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Nambara E, Marion-Poll A (2005) Abscisic acid biosynthesis and catabolism. Annu Rev Plant Biol 56:165–185
Noctor G, Arisi AM, Jouanin L, Kunert KJ, Rennenberg H, Foyer CH (1998) Glutathione: biosynthesis, metabolism and relationship to stress tolerance explored in transformed plants. J Exp Bot 49:623–647
Rausch T, Wachter A (2005) Sulfur metabolism: a versatile platform for launching defence operations. Trends Plant Sci 10:503–509
Rolland F, Baena-Gonzalez E, Sheen J (2006) Sugar sensing and signaling in plants: conserved and novel mechanisms. Annu Rev Plant Biol 57:675–709
Rolland F, Moore B, Sheen J (2002) Sugar sensing and signaling in higher plants. Plant Cell 14:S185–S205
Saito K (2004) Sulfur assimilatory metabolism. The long and smelling road. Plant Physiol 136:2443–2450
Savage H, Montoya G, Svensson C, Schwenn JD, Sinning I (1997) Crystal structure of phosphoadenylyl sulphate (PAPS) reductase: A new family of adenine nucleotide alpha hydrolases. Structure 5:895–906
Schwenn JD, Krone FA, Husmann K (1988) Yeast PAPS reductase: Properties and requirements of the purified enzyme. Arch Microbiol 150:313–319
Setya A, Murillo M, Leustek T (1996) Sulfate reduction in higher plants: Molecular evidence for a novel 5'-adenylylphosphosulfate (APS) reductase. Proc Natl Acad Sci (USA) 93:13383–13388
Smeekens S (2000) Sugar-induced signal transduction in plants. Annu Rev Plant Physiol Plant Mol Biol 51:49–81
Tsakraklides GP, Martin M, Chalam, R, Tarczynski M, Schmidt A, Leustek T (2002) Sulfate reduction is increased in transgenic Arabidopsis thaliana expressing 5’-adenylylsulfate reductase from Pseudomonas aeruginosa. Plant J 32: 879–889
Weber M, Suter M, Brunold C, Kopriva S (2000) Sulfate assimilation in higher plants characterization of a stable intermediate in the adenosine 5'-phosphosulfate reductase reaction. Eur J Biochem 267:3647–3653
Xiao W, Sheen J, Jang JC (2000) The role of hexokinase in plant sugar signal transduction and growth and development. Plant Mol Biol 44:451–461
Zhang B, Pasini R, Dan H, Joshi N, Zhao Y, Leustek T, Zheng Z-L (2014) Aberrant gene expression in mutants of the Arabidopsis sulfate transporter SULTR1;2 suggests a possible regulatory role for this sulfate transporter in response to sulfur nutrient status. Plant J 77:185–197
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Chung, JS., Lee, HN., Leustek, T. et al. The Arabidopsis thaliana adenosine 5’-phosphosulfate reductase 2 (AtAPR2) participates in flowering time and glucose response. J. Plant Biol. 58, 128–136 (2015). https://doi.org/10.1007/s12374-014-0514-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12374-014-0514-2