Abstract
A 50-year-old woman was referred to our hospital with elevated serum amylase levels. Physical examination revealed no jaundice or abdominal tenderness. Serum IgG4 was negative. Computed tomography revealed a localized pancreatic duct narrowing in the pancreatic head, with caudal pancreatic duct dilation and an intraductal papillary mucinous neoplasm. Pancreatic enlargement was not observed. Endoscopic ultrasonography (EUS) showed a small hypoechoic mass. Although EUS-guided, fine-needle aspiration was performed, no diagnosis was established. Endoscopic retrograde pancreatography showed a localized narrowing in the main pancreatic duct of the pancreatic head. A biopsy of the narrowing was performed through the minor papilla because of difficult access from the major papilla. The specimen showed the infiltration of numerous IgG4-positive plasma cells, suggesting type 1 autoimmune pancreatitis (AIP). Six months later, magnetic resonance cholangiopancreatography revealed improvement in the narrowing without specific treatment. The patient presented with localized narrowing of the pancreatic duct and caudal duct dilation, which was distinct from pancreatic cancer. Diagnostic difficulties arose from negative serum IgG4 results, the lack of typical imaging characteristics of AIP, and failure to meet the AIP criteria according to the relevant Japanese and international guidelines. However, AIP was suspected and surgery was successfully avoided through a biopsy.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Autoimmune pancreatitis (AIP) is a distinct form of pancreatitis clinically characterized by the frequent presentation of obstructive jaundice with or without a pancreatic mass [1]. AIP is classified into types 1 and 2, with type 1 being the most common type in Japan [2]. Type 1 AIP is histologically characterized by the infiltration of IgG4-positive plasma cells [3, 4] and is considered a pancreatic lesion of IgG4-related systemic disease. However, approximately 20% of patients with AIP do not have elevated serum IgG4 levels [5]. In such cases, particularly when typical pancreatic enlargement is not observed, the diagnosis of AIP can be challenging because of the difficulty in obtaining an adequate specimen for diagnosis, and sometimes, patients may even undergo surgery under the misdiagnosis of pancreatic cancer. Here we report a case of suspected serum-IgG4-negative type 1 AIP without pancreatic enlargement, characterized by localized pancreatic duct narrowing. A pancreatic duct biopsy performed through the minor papilla strongly suggested but did not definitively confirm the diagnosis.
Case report
A 50-year-old woman was initially referred to our hospital in 2015 for an elevated serum amylase level. She had a history of undergoing maintenance hemodialysis for chronic kidney disease secondary to immunoglobulin A nephropathy and hypertension. The patient had no subjective symptoms. A physical examination revealed no jaundice or abdominal tenderness. Her serum amylase level was 564 IU/L with pancreatic-type dominance. No elevated inflammatory markers or tumor markers were noted. Serum IgG4 was negative (24 mg/dL). There were no extrapancreatic lesions suggestive of IgG4-related disease.
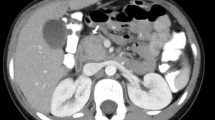
Abdominal computed tomography (CT) showed a localized pancreatic duct narrowing in the pancreatic head with caudal pancreatic duct dilation. No neoplastic lesions near the narrowing, pancreatic enlargement, capsule-like rim, or uniformly delayed enhancement in the late phase of the CT scan were observed (Fig. 1). Similarly, magnetic resonance cholangiopancreatography (MRCP) revealed localized pancreatic duct narrowing in the pancreatic head with several 10 mm cysts without nodules in the pancreatic head and tail suggestive of an intraductal papillary mucinous neoplasm (IPMN) without malignant findings (Fig. 2).
Endoscopic ultrasonography (EUS) revealed findings indicative of pancreatic duct wall thickening at the narrowing of the pancreatic duct, appearing as a hypoechoic, tumor-like mass (Fig. 3a–c), and EUS-guided fine-needle aspiration (EUS-FNA) cytology and biopsy were performed (Fig. 3d). No malignant findings were detected; however, a definitive diagnosis could not be made because the specimen was insufficient.
Endoscopic ultrasound image. a, b The pancreatic duct is narrowed at the pancreatic head, showing wall thickening and presenting a tumor-like appearance (red arrow). c cystic lesions suggestive of intraductal papillary mucinous neoplasm were detected at the caudal side of the pancreatic duct narrowing (green arrows). d endoscopic ultrasonography-guided fine needle aspiration was performed on a hypoechoic mass lesion (yellow arrowheads)
Endoscopic retrograde cholangiopancreatography (ERCP) showed a localized pancreatic duct narrowing at the pancreatic head that was difficult to approach from the major papilla owing to its loop shape. Therefore, brush cytology and pancreatic duct biopsy were performed through the minor papilla after balloon dilation (Fig. 4). The brush cytology results were Class III, suggestive of IPMN. The biopsy specimen revealed infiltration of lymphocytes and IgG4-positive plasma cells, with a count of 40 cells per high-power field (HPF) (Fig. 5). The biopsy specimens did not contain any IPMN components. These findings met only one major criterion from the Japanese AIP guidelines at the time of diagnosis, namely “irregular narrowing of the main pancreatic duct observed via endoscopic retrograde pancreatography (ERP),” and 2 minor criteria, namely “more than 10 infiltrating IgG4-plasma cells per HPF” and “no malignant cells detected using EUS-FNA” [6]. However, the criteria were not met for a definitive diagnosis of AIP according to the guidelines.
Endoscopic retrograde pancreatography image. a endoscopic retrograde pancreatography showed a localized narrowing of the pancreatic head (red arrow). The pancreatic duct narrowing was difficult to approach from the major papilla due to loop formation (green circle). b pancreatic duct biopsy for the narrowing was performed through the minor papilla
Although a definitive diagnosis based on these guidelines was not possible, AIP was strongly suspected. In the international guidelines [2] as well, while meeting some criteria such as “localized narrowing of the pancreatic duct without significant dilation upstream” and “only 2 criteria of Level 2 in pancreatic histology: Periductal lymphoplasmacytic infiltration without granulocytic infiltration, and abundant (> 10 cells/HPF) IgG4-positive cells” was not sufficient to definitively diagnose AIP. Although a definitive diagnosis based on these guidelines was not possible because multiple criteria were met and malignant findings were ruled out, AIP was strongly suspected.
As the patient was asymptomatic, a decision was made to monitor without steroid treatment. After 6 months, the narrowing improved on MRCP, suggesting a high probability of AIP as the clinical diagnosis (Fig. 6). She underwent annual magnetic resonance imaging examinations following spontaneous improvement of the pancreatic ductal narrowing. No recurrence of the narrowing has been noted to date. While the IPMN has shown a gradual increase in its size over 8 years, no malignant features such as cystic nodules have been observed, and we continue to monitor its progression.
Discussion
Most AIP cases in Japan are type 1, which is considered a pancreatic lesion associated with IgG4-related systemic disease [2]. Therefore, the serum IgG4 levels are typically elevated. However, in 20% of AIP cases, patients may be IgG4-negative [5], necessitating a comprehensive diagnosis based on imaging findings and other criteria. In our case, the serum IgG4 levels were within the normal range. Additionally, imaging findings were limited to localized narrowing of the pancreatic duct and a small tumor-like structure, which is not typical of pancreatic cancer; however, pancreatic cancer could not be ruled out, making the diagnosis extremely difficult.
According to the Japanese guidelines for AIP, which focus mainly on type 1 AIP, in cases without pancreatic enlargement, a definitive diagnosis is only possible with histological examination and may not even meet the criteria for probable diagnosis [7]. However, the histological diagnosis of AIP is challenging because of the difficulty in acquiring sufficient tissue samples without surgery. In a prospective multicenter study in Japan, the diagnostic rates of EUS-FNA for levels 1 and 2 were 43.4% and 15.1%, respectively, according to the International Consensus Diagnostic Criteria [8]. In our case, because the diagnosis of AIP could not be confirmed using EUS-FNA, we attempted to perform ERCP to obtain tissue samples. However, a standard pancreatic duct biopsy through the major papilla was difficult because of the loop-shaped pancreatic duct. Therefore, the biopsy was performed through the minor papilla, and the presence of IgG4-positive plasma cells was confirmed.
In general, cannulation of the minor papilla presents challenges, with success rates ranging from 73.5% to 83.3% [9,10,11]. Even when successful, the subsequent insertion of stiff instruments such as biopsy forceps is often difficult. Therefore, reports on diagnostic rates for pancreatic duct biopsies via the minor papilla are limited, and no cases have been documented in which AIP has been diagnosed. Diagnosing AIP using a pancreatic duct biopsy is challenging and its precise diagnostic rate remains unclear. It was initially difficult to diagnose AIP through a standard pancreatic duct biopsy; however, we were able to perform a biopsy via the minor papilla.
Several studies have reported the presence of IgG4-positive plasma cells in pancreatic neoplastic lesions [12, 13]. Consequently, identifying IgG4-positive plasma cells in a lesion via pancreatic duct biopsy through the papilla is unable to exclude the possibility of a pancreatic neoplasm. Therefore, we used cytology and EUS-FNA alongside the pancreatic duct biopsy to verify the absence of malignancy. Although no malignant findings were observed in any of the samples, we informed the patient that malignancy could not be entirely excluded; thus, he opted for follow-up imaging.
Another factor that complicated the diagnosis in this case was the coexisting of IPMN. In the clinical diagnostic criteria for AIP, “diffuse or localized irregular narrowing of the main pancreatic duct characteristic of ERP” is adopted [6, 7]. Even in localized lesions, the main pancreatic duct upstream of the narrow region is often not significantly dilated [14]. In this case, pancreatic cancer was initially suspected due to a localized pancreatic duct narrowing, pancreatic mass, and caudal pancreatic duct dilation. This case was atypical for AIP because the caudal pancreatic duct was dilated, possibly because of the presence of an IPMN on the caudal side.
Some reports indicate a high possibility of pancreatic cancer coexisting with IPMN [15,16,17], necessitating the initial consideration of pancreatic cancer in our case, with both IPMN and pancreatic duct narrowing. Furthermore, brush cytology of the narrowing revealed Class III findings and suspected IPMN, making it necessary to rule out a tumorous lesion within the pancreatic duct narrowing. However, histological analysis of the biopsy specimen from the narrowing did not indicate IPMN; instead, IgG4-positive cells were observed. Therefore, the Class III findings were considered unrelated to the pancreatic duct narrowing but due to the presence of a coexistent IPMN.
Identification of IgG4-positive plasma cells plays a crucial role in the diagnosis of IgG4-related diseases (IgG4-RD), including AIP. Notably, while the comprehensive IgG4-RD guidelines [18] require a ratio of IgG4/IgG-positive cells > 40% and > 10 IgG4-positive plasma cells/HPF, the guidelines for AIP have adapted to accommodate smaller biopsy samples, such as those obtained via needle biopsy. According to these AIP guidelines, the presence of > 10 IgG4-positive cells per HPF is sufficient for a diagnosis, even if the IgG4/IgG ratio does not exceed 40%. In this case, although the ratio criterion was not satisfied, the presence of > 10 cells/HPF, a crucial element in the current AIP guidelines, was satisfied.
Most patients with type 1 AIP reportedly respond well to steroids [19]. Their use is recommended in symptomatic cases such as obstructive jaundice caused by biliary narrowing, abdominal and back pain, and the presence of extrapancreatic lesions [20]. In addition, due to its favorable response, it is considered useful for treatment as well as diagnosis, and a response to steroids is among the current diagnostic criteria. A 2-week steroid trial and subsequent response assessment can also confirm the diagnosis of AIP without negative consequences in cases of resectable pancreatic cancer [21]. Although the patient in this case was asymptomatic and not absolutely indicated for treatment, diagnostic steroid use could have facilitated the differentiation from pancreatic cancer. In fact, we offered this option to the patient, but she refused it due to the underlying chronic kidney disease requiring dialysis and concern about the side effects of steroids. As a result, in this case, despite the lack of steroid therapy, the pancreatic duct narrowing improved.
A reported 65% of patients without steroid treatment experienced spontaneous remission. Additionally, IgG4 seronegativity was an independent predictor of spontaneous remission in type 1 AIP, indicating that steroid therapy might not always be required in patients with IgG4 seronegativity [22]. This case aligned with that finding. Considering the spontaneously improved pancreatic duct narrowing, relevant examination findings, including a positive IgG4 pancreatic duct biopsy, and the fact that no tumors appeared during long-term follow-up, we clinically diagnosed the condition as type 1 AIP.
In conclusion, diagnosing this case was difficult because of the normal IgG4 levels and the lack of pancreatic enlargement, which did not align with the AIP guidelines. Nonetheless, the pancreatic duct biopsy results raised the suspicion of AIP; thus, surgery was avoided. AIP should be considered when atypical pancreatic duct narrowing is observed even in the absence of elevated IgG4 levels or pancreatic enlargement. Additionally, when EUS-FNA is not feasible, pancreatic duct biopsy, especially through the minor papilla, can lead to a diagnosis, despite its challenges.
References
Masamune A, Kikuta K, Hamada S, et al. Nationwide epidemiological survey of autoimmune pancreatitis in Japan in 2016. J Gastroenterol. 2020;55:462–70.
Shimosegawa T, Chari ST, Frulloni L, et al. International consensus diagnostic criteria for autoimmune pancreatitis: guidelines of the international association of pancreatology. Pancreas. 2011;40:352–8.
Kawaguchi K, Koike M, Tsuruta K, et al. Lymphoplasmacytic sclerosing pancreatitis with cholangitis: a variant of primary sclerosing cholangitis extensively involving pancreas. Hum Pathol. 1991;22:387–95.
Uchida K, Okazaki K. Clinical and pathophysiological aspects of type 1 autoimmune pancreatitis. J Gastroenterol. 2018;53:475–83.
Kamisawa T, Takuma K, Tabata T, et al. Serum IgG4-negative autoimmune pancreatitis. J Gastroenterol. 2011;46:108–16.
Japan Pancreas Society, the Research Committee of Intractable Pancreatic Disease (Principal investigator: Tooru Shimosegawa) provided by the Ministry of Health, Labour and Welfare of Japan. Japanese Guidelines. For management of autoimmune pancreatitis. Suizo. 2013;2013(28):715–84.
The Japan Pancreas Society, the Research Committee of Intractable Diseases for IgG4-related disease supported by the Japanese Ministry of Health. Labor and welfare. amendment of the japanese consensus guidelines for autoimmune pancreatitis. Suizo. 2020;35:465–550.
Kanno A, Masamune A, Fujishima F, et al. Diagnosis of autoimmune pancreatitis by EUS-guided FNA using a 22-gauge needle: a prospective multicenter study. Gastrointest Endosc. 2016;84:797-804.e1.
Lans JI, Geenen JE, Johanson JF, et al. Endoscopic therapy in patients with pancreas divisum and acute pancreatitis: a prospective, randomized, controlled clinical trial. Gastrointest Endosc. 1992;38:430–4.
Jacob L, Geenen JE, Catalano MF, et al. Clinical presentation and short-term outcome of endoscopic therapy of patients with symptomatic incomplete pancreas divisum. Gastrointest Endosc. 1999;49:53–7.
Ertan A. Long-term results after endoscopic pancreatic stent placement without pancreatic papillotomy in acute recurrent pancreatitis due to pancreas divisum. Gastrointest Endosc. 2000;52:9–14.
Kamisawa T, Chen PY, Tu Y, et al. Pancreatic cancer with a high serum IgG4 concentration. World J Gastroenterol. 2006;12:6225–8.
Liu Q, Niu Z, Li Y, et al. Immunoglobulin G4 (IgG4)-positive plasma cell infiltration is associated with the clinicopathologic traits and prognosis of pancreatic cancer after curative resection. Cancer Immunol Immunother. 2016;65:931–40.
Nishino T, Oyama H, Toki F, et al. Differentiation between autoimmune pancreatitis and pancreatic carcinoma based on endoscopic retrograde cholangiopancreatography findings. J Gastroenterol. 2010;45:988–96.
Yamaguchi K, Ohuchida J, Ohtsuka T, et al. Intraductal papillary-mucinous tumor of the pancreas concomitant with ductal carcinoma of the pancreas. Pancreatology. 2002;2:484–90.
Tada M, Kawabe T, Arizumi M, et al. Pancreatic cancer in patients with pancreatic cystic lesions: a prospective study in 197 patients. Clin Gastroenterol Hepatol. 2006;4:1265–70.
Uehara H, Nakaizumi A, Ishikawa O, et al. Development of ductal carcinoma of the pancreas during follow-up of branch duct intraductal papillary mucinous neoplasm of the pancreas. Gut. 2008;57:1561–5.
Umehara H, Okazaki K, Kawa S, et al. Research program for intractable disease by the ministry of health, labor and welfare MHLW Japan. The 2020 revised comprehensive diagnostic RCD criteria for IgG4-RD. Mod Rheumatol. 2021;31:529–33.
Kamisawa T, Shimosegawa T, Okazaki K, et al. Standard steroid treatment for autoimmune pancreatitis. Gut. 2009;58:1504–7.
Hart PA, Kamisawa T, Brugge WR, et al. Long-term outcomes of autoimmune pancreatitis: a multicentre, international analysis. Gut. 2013;62:1771–6.
Moon SH, Kim MH, Park DH, et al. Is a 2-week steroid trial after initial negative investigation for malignancy useful in differentiating autoimmune pancreatitis from pancreatic cancer? Prospect Outcome Stud Gut. 2008;57:1704–12.
Kubota K, Watanabe S, Uchiyama T, et al. Factors predictive of relapse and spontaneous remission of autoimmune pancreatitis patients treated/not treated with corticosteroids. J Gastroenterol. 2011;46:834–42.
Acknowledgements
We would like to thank Editage (www.editage.com) for English language editing.
Author information
Authors and Affiliations
Contributions
KK (Karen Kimura) contributed to the planning, literature review, and writing of the article. Kazuya Koizumi (KK), MS, MM, JK, and ST provided professional suggestions for conducting case reports. All the authors commented on the draft and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflict of interest related to this work.
Informed consent
Informed consent was obtained from this patient.
Human and animal rights
All the procedures were performed in accordance with the ethical standards of the Declaration of Helsinki and its amendments.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kimura, K., Koizumi, K., Masuda, S. et al. A suspected case of serum IgG4-negative type 1 autoimmune pancreatitis detected due to localized pancreatic duct narrowing: a case report. Clin J Gastroenterol (2024). https://doi.org/10.1007/s12328-024-01993-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12328-024-01993-0