Abstract
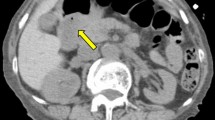
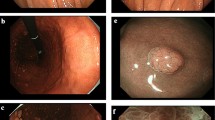
A 71-year-old woman was given a barium meal examination as part of a workup for recurring melena and iron deficiency anemia (IDA), and it revealed a large duodenal polyp measuring 60 mm in diameter. Subsequent upper gastrointestinal endoscopy showed a large pedunculated polyp in the duodenal bulb. Magnifying endoscopy with narrow-band imaging (NBI) showed that the lesion consisted of a regular enlarged intervening part between crypts of the epithelium that resembled gastric mucosa. Although the lesion was suspected of being benign, it was resected by endoscopic submucosal dissection (ESD) to prevent the progression of the IDA. The pathology examination revealed the proliferation of mildly irregular-shaped or dilated glands lined by cuboidal cells and low columnar cells in the submucosa. The gastric glands were immunohistochemically positive for MUC6, suggesting pyloric gland differentiation. The lesion was covered by a foveolar-type epithelium, and we made a diagnosis of pyloric gland adenoma (PGA). PGAs are most common in the stomach, and they are rare in the duodenum, where endoscopic treatment is technically challenging. Here we report a case of large duodenal PGA successfully resected by ESD. Since part of PGAs has been reported to be associated with adenocarcinoma, minimally invasive treatment strategies are desirable to reduce the risk of progression to carcinoma.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pyloric gland adenomas (PGAs) are rare neoplasms of the gastrointestinal tract that exhibit gastric pyloric gland differentiation [1]. PGAs are most common in the stomach [2], and they are rare in the duodenum, where endoscopic treatment is technically challenging [3]. Since part of PGAs has been reported to be associated with adenocarcinoma [4], surgical intervention is desirable to reduce the risk of progression to carcinoma. Recent advances in endoscopic modalities have made it possible to treat even large lesions located in the duodenum by minimally invasive procedures. Here we report a case of large duodenal PGA successfully resected by endoscopic submucosal dissection (ESD) without any adverse events.
Case report
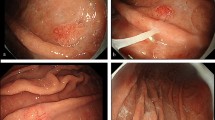
A 71-year-old woman came to our hospital for a workup for recurring melena and anemia. Her anemia improved in response to oral iron supplementation but became severe again when she stopped taking the supplement. There was no history of Helicobacter pylori infection. Laboratory studies showed a hemoglobin concentration 8.7 d/dL, revealing mild iron deficiency anemia (IDA), but an immunoassay fecal occult blood test was negative. A barium meal examination revealed a large duodenal polyp, measuring 60 mm in diameter that filled the duodenal bulb (Fig. 1). Subsequent upper gastrointestinal endoscopy showed a large pedunculated polyp in the duodenal bulb (Fig. 2). Magnifying endoscopy with narrow-band imaging (NBI) showed that the lesion consisted of a regular enlarged intervening part between crypts of the epithelium that resembled gastric mucosa. Since colonoscopy and capsule endoscopy did not detect any other findings besides the duodenal polyp, the polyp was concluded to be the cause of the IDA. The patient’s hemoglobin concentration temporarily improved to 11.3 g/dL in response to oral iron supplementation. Although the lesion was suspected of being a benign polyp, we decided to resect it endoscopically to control IDA. We attempted to perform endoscopic mucosal resection (EMR) first, but did not succeed because of the narrow working space in the duodenal bulb and its thick stalk. Then we attempted ESD and succeeded in achieving en bloc resection (Fig. 3). After en bloc resection, the lesion was divided into some pieces by a snare to get it through the esophagogastric junction. Pathological examination revealed proliferation of mildly irregular-shaped or dilated glands lined by cuboidal cells and low columnar cells mainly in the submucosa (Fig. 4). The gastric glands were immunohistochemically positive for MUC6, suggesting pyloric gland differentiation. The surface of the lesion was covered by non-neoplastic epithelium that exhibited foveolar metaplasia, and we made a diagnosis of PGA. The patient was discharged from the hospital 6 days postoperatively, and her hemoglobin concentration gradually improved to 13 g/dL on oral iron supplementation. The anemia and melena have not recurred since the iron supplement was discontinued.
a Pathological examination revealed that the PGA was mainly located in the submucosa (HE, × 2). b The PGA consisted of tightly packed pyloric tubules lined by cuboidal to low columnar cells (HE, × 4). c Irregular eosinophilic mucous glands extending from deep in the mucosa to the submucosa expressed MUC6 (MUC6, × 4). d Surface layer composed of foveolar-type epithelium that weakly expressed MUC5AC (MUC5AC, × 4)
Discussion
Thanks to recent advancements in endoscopic technology and screening routines, more duodenal adenomas are being found [5,6,7]. However, because duodenal PGAs are rare, no treatment strategies have been established [8].
Duodenal PGAs resemble gastric PGAs and are characterized by gastric pyloric gland differentiation. Histopathologically, PGAs consist of tightly packed pyloric-gland-like tubules lined by cuboidal to low columnar cells containing small round nuclei and clear or eosinophilic cytoplasm [9]. The tumor glands express MUC6, and the surface layer is composed of a foveolar epithelium that expresses MUC5AC immunohistochemically. In our patient irregular eosinophilic mucous glands expressing MUC6 covered by a tubular epithelium that weakly expressed MUC5AC was observed from deep in the mucosa to the submucosa and the lesion was diagnosed as a PGA. Brunner’s glands were found around the PGA, and a PGA that arose from Brunner’s glands has also been reported [10]. The results of a multicenter study that included 57 cases of duodenal PGA showed that the PGAs occurred predominantly in elderly patients with a median age of 73.5 years and that they were more common in females (55%) [11]. The most common site of the duodenal PGAs was the proximal duodenum (69%), and most patients presented with a protruding polypoid lesion (98%). The mean size of the lesion was 14.8 mm. The results of the study showed that large duodenal PGAs frequently exhibited advanced pathology, and that carcinomas arose from approximately 20% of all PGAs. These findings highlighted the need to resect PGAs.
Since endoscopic resection in the duodenum entails a high risk of severe adverse events, large duodenal neoplasms often require surgical resection [12], and duodenal lesions tend to be treated conservatively. After the attempt to perform EMR failed in our patient because of the narrow working space and its thick stalk, we performed ESD and succeeded in achieving en bloc resection without any adverse events. Since the line of dissection can be seen more accurately during ESD even in narrow working spaces, it can serve as an alternative option for treating large pedunculated duodenal polyps.
A search of the literature did not retrieve any reports of cases of a large pedunculated PGA filling the duodenal bulb that was resected by ESD. In conclusion, we demonstrated that a large duodenal PGA can be successfully resected by ESD without any adverse events.
References
Pezhouh MK, Park JY. Gastric pyloric gland adenoma. Arch Pathol Lab Med. 2015;139:823–6.
Bakotic BW, Robinson MJ, Sturm PD, et al. Pyloric gland adenoma of the main pancreatic duct. Am J Surg Pathol. 1999;23(2):227–31.
Yahagi N, Kato M, Ochiai Y, et al. Outcomes of endoscopic resection for superficial duodenal epithelial neoplasia. Gastrointest Endosc. 2018;88(4):676–82.
Chen ZM, Scudiere JR, Abraham SC, et al. Pyloric gland adenoma: An entity distinct from gastric foveolar type adenoma. Am J Surg Pathol. 2009;33(2):186–93.
Endo M, Abiko Y, Oana S, et al. Usefulness of endoscopic treatment for duodenal adenoma. Dig Endosc. 2010;22:360–5.
Tsuji S, Doyama H, Tsuji K, et al. Preoperative endoscopic diagnosis of superficial non-ampullary duodenal epithelial tumors, including magnifying endoscopy. World J Gastroenterol. 2015;21:11832–41.
Maruoka D, Arai M, Ishigami H, et al. Cold polypectomy for nonampullary duodenal adenoma. Endoscopy. 2015;47(Suppl 1):E477–8.
Mitsuishi T, Hamatani S, Hirooka S, et al. Clinicopathological characteristics of duodenal epithelial neoplasms: Focus on tumors with a gastric mucin phenotype (pyloric gland-type tumors). PLoS ONE. 2017;12(4):e0174985.
Kushima R, Mukaisho K, Vieth M, et al. Pyloric-gland-type adenomas of the stomach. Stomach Intestine. 2003;38:1377–87.
Kushima R. “Jūnishichō: Igata no sennshu gan” [Gastric-type Adenoma and Adenocarcinoma of the Duodenum]. Stomach and Intestine. 2020;55:425–8 ((in Japanese)).
Miller GC, Kumarasinghe MP, Borowsky J, et al. Clinicopathological features of pyloric gland adenomas of the duodenum: a multicentre study of 57 cases. Histopathology. 2020;76:404–10.
Kushima R, Rüthlein HJ, Stolte M, et al. “Pyloric gland-type adenoma” arising in heterotopic gastric mucosa of the duodenum, with dysplastic progression of the gastric type. Virchows Arch. 1999;435:452–7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Ken Ohata, Shunya Takayanagi, Eiji Sakai, Takashi Muramoto, and Hirotsugu Hashimoto declare that they have no conflict of interest.
Human rights
All procedures followed have been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed consent
Informed consent was obtained from all patients for being included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Takayanagi, S., Sakai, E., Muramoto, T. et al. Large duodenal pyloric gland adenoma successfully resected by endoscopic submucosal dissection. Clin J Gastroenterol 14, 538–541 (2021). https://doi.org/10.1007/s12328-021-01367-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12328-021-01367-w