Abstract
Introduction
The patient experience with prostate cancer differs throughout the disease continuum, with health-related quality of life (HRQoL) and symptoms worsening as the disease progresses. To understand the prostate cancer experience, it is important to understand the experience of same-aged men without prostate cancer as a basis for comparison. This study provides the US population reference values for six patient-reported outcome (PRO) questionnaires.
Methods
A cross-sectional online survey, including several PRO questionnaires, was administered in 2019 to a representative sample of US adults. The male sample (N = 876) was raked by age to have similar characteristics of men in key advanced prostate cancer trials (mean/median age: 67.5/70.0 years), with the majority being white and non-Hispanic.
Results
Results from six PRO questionnaires (Brief Pain Inventory; Quality of Life in Neurological Disorders 2.0 Cognitive Short Form; PRO Measurement Information System Fatigue-Short Form; Functional Assessment of Cancer Therapy-General; European Organization for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; and European Organization for Research and Treatment of Cancer Quality of Life Questionnaire Prostate Cancer Module) indicated that the US representative sample of men have good role, physical, and emotional functioning but slightly impaired social, functional, and overall well-being. In addition, they have normal cognitive function, few financial problems, minimal pain and fatigue, minimal urinary and bowel symptoms, and limited use of incontinence aids.
Conclusions
The availability of the reference values for these PRO questionnaires will enable researchers to compare the HRQoL of patients with advanced prostate cancer in the US with that of the general US population and allow for a better interpretation of those scores. Registration numbers of advanced prostate cancer trials: NCT02677896, NCT02003924, NCT01212991, NCT00974311.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Baseline patient-reported outcome (PRO) scores of patients with advanced prostate cancer across multiple clinical trials indicate a moderate health-related quality of life (HRQoL) and low symptom burden, particularly for those patients who have not yet developed metastasis and/or castration resistance or received chemotherapy |
PRO scores from a representative population of men are needed to better interpret PRO scores from patients with advanced prostate cancer (metastatic or castration-resistant prostate cancer [CRPC]) |
What was learned from the study? |
Responses to the online survey of PROs indicated that in the US the general population of men, raked by age to have a similar age distribution (median age of 70) of men in key prostate cancer trials across the advanced prostate cancer continuum (metastatic hormone-sensitive prostate cancer [mHSPC] and CRPC), had normal physical and emotional well-being and slightly impaired social, functional, and overall well-being |
This sample from the general population also reported low levels of pain, low urinary, bowel, and hormone treatment-related symptoms and use of incontinence aids, as well as normal cognitive function and minimal fatigue scores which were consistent with the US “average” scores |
The findings collectively indicate a similarity in overall well-being between the general population and patients with prostate cancer but some differences in disease and treatment-related symptoms |
Introduction
Prostate cancer is the second most common cancer in American men (after skin cancer), with 191,930 new cases and 33,330 deaths estimated in 2020 [1]. Typically, in most men, the cancer is localized in the prostate when it is first diagnosed, and patients can undergo definitive primary therapy such as radical prostatectomy or external-beam radiotherapy, with curative intent. A biochemical relapse/reoccurrence (i.e., an increase in prostate-specific antigen levels) with or without the presence of metastasis may occur, however, leading most patients to initiate androgen deprivation therapy (ADT). With time, prostate cancer cells develop mechanisms that allow themselves to grow despite castration levels of testosterone, and the disease becomes castration-resistant prostate cancer (CRPC) [2].
The patient experience with prostate cancer differs throughout the disease continuum, with many symptoms worsening as the disease progresses toward metastatic hormone-sensitive prostate cancer (mHSPC), metastatic CRPC (mCRPC), and/or CRPC [3,4,5]. Baseline characteristics for patients with these stages of advanced prostate cancer across multiple clinical trials demonstrate a moderate health-related quality of life (HRQoL) [6] and low symptom burden [7,8,9,10,11,12,13]. However, patient-reported outcomes (PROs) in trials with patients with mCRPC who had progressed during or after chemotherapy [13, 14] suggest that these patients have a more impaired HRQoL and functioning and higher symptom burden than patients with less advanced prostate cancer.
In the clinical trial setting, the important and relevant treatment-related symptoms and impacts on HRQoL that advanced prostate cancer has on patients are typically assessed. These often include pain, fatigue, urinary symptoms, gastrointestinal symptoms, and impaired functioning (e.g., social, physical, cognitive, emotional) and are commonly measured through many different PRO questionnaires such as the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire Core 30 (EORTC QLQ-C30) and the Functional Assessment of Cancer Therapy-General (FACT-G) questionnaire [4, 6, 15,16,17]. To better characterize the symptom and HRQoL burden of patients with advanced prostate cancer, this population should be compared with a population of men with similar demographic characteristics. This comparison provides insights into the incremental burden of advanced prostate cancer. Additionally, understanding population rates of symptoms and HRQoL scores allows for the evaluation of treatments and the potential to impact symptom burden and HRQoL relative to those without advanced prostate cancer.
The main objective of this study was to provide a set of US population reference values for several symptom and HRQoL PRO questionnaires when the reference population is iterative proportional fitted (“raking”) to reflect men with advanced prostate cancer in the pivotal enzalutamide trials (ARCHES [NCT02677896] [7], PROSPER [NCT02003924] [10], PREVAIL [NCT01212991] [12], and AFFIRM [NCT00974311] [14]; Supplemental Table 1). Raking these populations allows for the comparison of symptoms and HRQoL between patients with advanced prostate cancer and those of national norms, a particularly valuable comparison when interpreting results from and between clinical trials. Additionally, many of the symptoms and HRQoL assessed with PRO questionnaires are likely to be impacted by age (e.g., fatigue, pain, or physical functioning); thus, reference values from a population of men with similar age characteristics are important in the setting of prostate cancer. The availability of national reference values also supports the interpretability of the PRO questionnaires used in advanced prostate cancer clinical trials and illuminates whether patients are able to maintain or normalize symptom and HRQoL scores relative to the general population.
Methods
Study Design
A cross-sectional online survey was administered in January and February 2019. The selection criteria included men and women aged ≥ 18 years in the US general population. Respondents were recruited by Critical Mix (Connecticut, USA), which had an existing respondent panel mirroring the US Census. The panelists were invited by email and phone alerts to complete the online survey. Participants were compensated for completing the survey via a pre-specified reward structure that is a function of survey length and complexity.
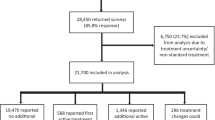
A total of 1615 men and women completed the online survey. The total included 1104 respondents representative of the US adult population (by age, sex, and region) and an enriched sample of 511 male respondents aged ≥ 65 years. These 511 men were sampled from the same patient panel and recruitment was restricted to men ≥ 65 years of age. Given the focus on prostate cancer, the analysis set was restricted to the men who completed the online survey. The final sample of 876 men was used for this study after the sample was raked to be similar to the age distribution of men who participated in these advanced prostate cancer clinical trials (i.e., 65.4% aged 41–74 years and 34.6% aged ≥ 75 years; see Fig. 1 for study sample flow diagram).
The online survey included three sections. In the first section, respondents were introduced to the survey and informed that the purpose of the research was to collect HRQoL data from a general US population and that the survey would take approximately 30–45 min to complete. The second section included questions about their demographics and comorbidities. The third section included nine PRO questionnaires to be completed by all respondents (except the prostate cancer specific questionnaire [EORTC QLQ-PR25], which was not given to female respondents). This survey was deemed exempt by the Institutional Review Board, as all responses were anonymized, aggregated, and untraceable to the respondents by Ethical and Independent Review Services (#21149). The study complies with the Declaration of Helsinki, and informed consent was obtained from all participants.
PRO Questionnaires
Nine PRO questionnaires, commonly administered in prostate cancer clinical trials, were administered (Supplemental Table S2). Of these, the results of the generic questionnaires (Brief Pain Inventory Short Form [BPI-SF], Quality of Life in Neurological Disorders 2.0 Cognitive Function Short Form [Neuro-QoL 2.0 Cognitive Function Short Form], and PRO Measurement Information System Fatigue-Short Form 4a [PROMIS Fatigue-SF4a]) and cancer-related questionnaires (FACT-G, EORTC QLQ-C30, and EORTC QLQ-PR25) are discussed in this publication. The other three questionnaires (EQ-5D-5L, Veterans Rand [VR-12], and Work Productivity and Activity Impairment [WPAI]) are not included in this report, as these are not commonly used to measure HRQoL in patients with prostate cancer.
BPI-SF consists of nine items and measures pain intensity and interference with daily functioning. Pain intensity and interference items are rated on a Likert scale of 0–10 (pain intensity: no pain [0] to pain as bad as you can imagine [10]; pain interference: no interference [0] to completely interferes [10]). Higher scores indicate greater pain [18]. Only pain intensity was administered in the study.
The Neuro-QoL 2.0 Cognitive Function Short Form consists of eight items and measures the cognitive function of patients with a neurological condition or disorder. Each item is rated on a Likert scale of 1–5 (1 = very often, 2 = often, 3 = sometimes, 4 = rarely, and 5 = never). Raw scores range from 8 to 40, and standardized T-scores are generated with a mean of 50 and standard deviation of 10. Higher T-scores indicate better cognitive function. Standardized T-scores > 45 are considered normal cognitive function. Standardized T-scores < 30, 30–40, and 41–45 are considered severe, moderate, and mild cognitive dysfunction, respectively [19].
PROMIS Fatigue-SF4a consists of four items and measures fatigue. Each item is rated on a Likert scale of 1–5 (1 = never, 2 = rarely, 3 = sometimes, 4 = often, and 5 = always). Raw scores range from 4 to 20, and standardized T-scores are generated with a mean of 50 and standard deviation of 10. Higher scores indicate greater fatigue. Standardized T-scores < 55, 55–60, 61–70, and > 70 are considered normal, mild, moderate, and severe fatigue, respectively [20].
FACT-G consists of 27 items and measures the HRQoL of patients with cancer. There are four domain scores and a total score. The domain scores include physical well-being (PWB) [seven items], social well-being (SWB) [seven items], emotional well-being (EWB) [six items], and functional well-being (FWB) [seven items]. Each item is rated on a Likert scale of 0 to 4 (0 = not at all, 1 = a little bit, 2 = somewhat, 3 = quite a bit, and 4 = very much). PWB, SWB, and FWB domain scores range from 0 to 28, and EWB score ranges from 0 to 24. The FACT-G scores range from 0 to 108. For all domain and total scores, higher scores indicate better HRQoL [21].
EORTC QLQ-C30 is a general cancer questionnaire. It consists of 30 items that measure the HRQoL of patients with cancer. The EORTC QLQ-C30 has five functioning scales (physical, role, cognitive, emotional, and social), three symptom scales (fatigue, nausea/vomiting, and pain), and additional single symptom items (dyspnea, insomnia, appetite loss, constipation, diarrhea, and financial difficulties). Each item is rated on a Likert scale of 1–4 (1 = not at all, 2 = a little bit, 3 = quite a bit, 4 = very much). The EORTC QLQ-C30 questionnaire also contains two global questions that use a 7-point scale scoring with anchors (1 = very poor, 7 = excellent) to generate a global health status/QoL score. All functioning, symptoms, and global health status/QoL domain scores range from 0 to 100. Higher functioning and global health status/QoL scores indicate better HRQoL, while higher symptom scores indicate more severe symptoms [22].
EORTC QLQ-PR25 is the prostate cancer module of the EORC QLQ-C30. It consists of 25 items that measure the HRQoL of patients with prostate cancer. The EORTC QLQ-PR25 has two multi-item functioning domains of sexual activity (two items) and sexual functioning (four items); three multi-item symptom domains of urinary symptoms (eight items), bowel symptoms (four items), and hormone treatment–related symptoms (six items); and a single item symptom domain measuring the degree to which wearing incontinence aids has been problematic. There is also a modified urinary symptoms domain, which includes the three disease-related symptom items of the eight-item domain, and an alternative urinary symptoms domain, which includes the five disease and treatment-related symptom items (symptom impact items are omitted) of the eight-item domain. Each item is rated on a Likert scale of 1–4 (1 = not at all, 2 = a little bit, 3 = quite a bit, and 4 = very much). All functioning and symptom domain scores range from 0 to 100. Higher functioning scores indicate better HRQoL, while higher symptom scores indicate more severe symptoms [23].
Statistical Analysis
The reference values to represent a population with advanced prostate cancer were derived by raking the nationally representative data from the male respondents. This raking was based on the age distribution (41–74 years vs. ≥ 75 years) from four pivotal phase 3 enzalutamide clinical trials in mHSPC (ARCHES), nonmetastatic castration-resistant prostate cancer (nmCRPC) [PROSPER], and mCRPC chemo-naive (PREVAIL) or post-chemo (AFFIRM). The demographics of patients from these four trials are presented in the Supplemental Materials (Supplementary Table S1). The PRO scores for each questionnaire were calculated based on its respective standard scoring guidelines. The raked PRO scores were summarized using descriptive statistics. A crosstabulation of the PROMIS Fatigue-SF4a and Neuro-QoL 2.0 Cognitive Function Short Form mean reference values was also conducted to summarize the percentage of respondents in this sample with above average fatigue and/or below average cognitive function.
Results
Baseline Characteristics
A total of 876 men were included in the sample raked on age for advanced prostate cancer. The mean age of the raked sample was 67.5 years, with 65.4% of men < 75 years of age and 34.6% of men ≥ 75 years. The median age of the raked sample was 70 years. Eighty-six percent of the men were of White race and 97.0% were non-Hispanic. Fifty-eight percent earned an education of associate’s degree or higher. Sampled men were located in all geographic regions of the US. Twenty-four percent of men were from the Northeast; 26.3%, from the Midwest; 32.3%, from the South; and 17.3%, from the West. The most common comorbidities they had at the time of their survey completion were hypertension (372 [42.5%]), arthritis or other rheumatologic diseases (206 [23.6%]), back pain (189 [21.6%]), and diabetes (192 [21.9%]) (Table 1).
Reference Scores by Questionnaire
The mean (SD) reference values for BPI-SF pain at its worst, its least, and its average were 2.6 (2.6), 1.7 (2.1), and 2.2 (2.3), respectively, out of a maximum score of 10, denoting low pain (31% reported no pain on average; Table 2). The mean (SD) reference values for the eight items of the Neuro-QoL 2.0 Cognitive Function Short Form ranged from 4.3 to 4.7 out of a maximum score of 5, denoting rarely experiencing cognitive impairment. The mean (SD) reference value for the Neuro-QoL 2.0 Cognitive Function Short Form raw score was 35.9 (5.3) out of a maximum score of 40, and the mean (SD) T-score was 55.1 (8.5), denoting high-normal range cognition (Table 2). Eighty-seven percent of respondents had normal cognitive function (T-scores > 45; Table 3). The mean (SD) reference value for PROMIS Fatigue-SF4a raw score was 7.7 (3.8) out of a maximum of 20, and the mean (SD) T-score was 45.8 (10.1), denoting low-normal levels of fatigue (Table 2). Eighty percent of respondents had normal levels of fatigue (T scores < 55; Table 4).
The mean (SD) reference value for the FACT-G total score was 78.8 (16.6) out of a maximum score of 108. The FACT-G domain with the highest reference value was PWB with a mean (SD) of 24.8 (4.0). The mean (SD) values for SWB, EWB, and FWB were 17.2 (6.9), 19.5 (4.4), and 18.8 (6.2), respectively. On an item level, the mean (SD) reference values for items GP1 (“I have a lack of energy”) and GP7 (“I am forced to spend time in bed”) were 1.0 (1.05) and 0.2 (0.6), respectively, out of a maximum of 4, denoting low levels of fatigue (Table 2).
The mean (SD) reference value for the EORTC QLQ-C30 global health status/QoL was 69.1 (20.7) out of a maximum score of 100. The EORTC QLQ-C30 domain with the highest reference value was cognitive functioning with a mean (SD) of 88.4 (18.0). The mean (SD) reference values for physical, emotional, role, and social functioning were 82.8 (21.9), 85.2 (19.5), 86.2 (22.7), and 87.7 (23.5), respectively. The symptoms with the highest values (i.e., more burden) were fatigue (22.4 [22.1]), insomnia (22.0 [27.7]), pain (21.6 [22.6]), dyspnea (15.4 [24.4]), and financial difficulties (10.4 [24.5]). The mean values for all other symptoms were < 10, denoting little burden (constipation: 9.8 [20.2], diarrhea: 7.4 [17.2], appetite loss: 6.1 [17.0], nausea/vomiting: 3.3 [11.1]) (Table 2).
The mean (SD) reference values for the EORTC QLQ-PR25 sexual activity and sexual functioning domains were 36.6 (26.5) and 75.3 (21.5), respectively, out of a maximum score of 100. The EORTC QLQ-PR25 domain with the highest mean (SD) value denoting the greatest burden was urinary symptoms (13.7 [15.6]). The mean (SD) reference values for the alternative domains for urinary symptoms were 15.8 (16.8) for the five-item urinary symptoms domain and 22.6 (22.6) for the modified three-item urinary symptoms domain. The mean (SD) reference values for hormone treatment–related symptoms, use of incontinence aids, and bowel symptoms were 8.0 (12.3), 4.9 (17.3), and 4.7 (11.4), respectively (Table 2).
A cross-tabulation of the PROMIS Fatigue-SF4a and Neuro-QoL 2.0 Cognitive Function Short Form mean reference values indicated that 74% of the raked general population (with a median age of 70 years and mean age of 67.5 years) had normal fatigue (described as score < 55) and normal cognitive function (described as score > 45; Table 5). Eight percent of the raked general population had above average fatigue (described as score ≥ 55) and below average cognitive function (described as score ≤ 45; Table 5). Six percent had normal fatigue and below average cognitive function, and 13% had above average fatigue and normal cognitive function (Table 5).
Discussion
The reference values were based on a nationally representative demographic sample (Critical Mix [Connecticut, United States]) and raked by the age of men with advanced prostate cancer in the enzalutamide pivotal trials (with a median and mean age of 70 and 67.5 years, respectively). The BPI-SF mean reference values indicated low pain at its worst, its least, and its average (scores were 3%, 2%, and 2% of the scale range, respectively). Despite the advanced age of the sample, the mean value for the Neuro-QoL 2.0 Cognitive Function Short Form was 55 (i.e., above the average of 50 [better cognitive function]), while the mean value for the PROMIS Fatigue-SF4a was 46 (i.e., below the average of 50 [less fatigue]). These results for Neuro-QoL 2.0 Cognitive Function Short Form and PROMIS Fatigue-SF4a in this study aligned with the instruments’ reference values. Both instruments have T-scores of 50 as the average score for the general US population, with an SD of 10.
Again, despite the advanced age of the raked sample, both EORTC QLQ-C30 and FACT-G suggested moderate HRQoL with a mean health status/QoL score of 69 and a FACT-G total score of 78.78 (moderate HRQoL was defined as 51–79 [6]). Generally, the two overall scores are comparable, measuring multiple aspects of HRQoL, despite fundamental differences in composition and scoring, making comparisons difficult.
In terms of functioning, participants reported high functioning across the different domains, but some differences were observed between FACT-G and EORTC QLQ-C30. Emotional functioning was similar between FACT-G (mean score of 19.5 [81% of the scale range]) and EORTC QLQ-C30 (mean score of 85.2 [85.2% of the scale range]) but social functioning was worse in FACT-G (mean score of 17.2 [61% of the scale range]) than in EORTC QLQ-C30 (mean score 87.7 [87.7% of the scale range]). Physical functioning was similar between FACT-G (mean score of 24.8 [89% of the scale range]) and EORTC QLQ-C30 (mean score of 82.8 [82.8% of the scale range]). Functional well-being was moderate in FACT-G (score was 67% of the scale range), and role and cognitive functioning were good in EORTC QLQ-C30 (scores were 86.2 and 88.4, respectively).
The mean reference value for the fatigue-related GP items (GP1 and GP7) suggested very low fatigue impairment (GP1 mean [SD] score: 0.98 [1.05]; GP7 mean [SD] score: 0.18 [0.58]). These item-level mean reference values allow for a further examination of the impact of fatigue in patients with advanced prostate cancer compared to its impact in a representative population.
The results of EORTC QLQ-C30 show little symptomatic burden. The symptoms with the highest scores (15.4–22.4) were fatigue, pain, dyspnea, and insomnia. Higher scores for these symptoms than for the others are probably due to the advanced age of the raked population, as these symptoms are commonly observed in the elderly population [24,25,26,27].
The EORTC QLQ-PR25 mean reference values indicated low urinary, bowel, and hormone treatment-related symptoms and a low use of incontinence aids (scores were 14%, 5%, 8%, and 5% of the scale range, respectively).
Although the men in this sample are advanced in age, the findings collectively suggest they have good role, physical, and emotional functioning but slightly impaired social, functional, and overall well-being; low pain, dyspnea, insomnia, and urinary, bowel, and hormone treatment–related symptoms; a low use of incontinence aids; normal cognitive function and fatigue; and very low nausea/vomiting, appetite loss, constipation, diarrhea, and financial problems. These data are similar to the HRQoL reference values reported in EORTC QLQ-C30 for the general US population, thus validating these findings [28]. More specifically, the EORTC QLQ-C30 reference values obtained in this study are similar and in some instances slightly higher than the reference values obtained for the US population using the same questionnaire in a large-scale global survey study [28]. The mean (SD) reference value for the global health status/QoL in this study was 69.1 (20.7) compared to 63.9 (22.9) in the US population in the large-scale survey study [28]. A trend for worse health state/QoL in Nolte et al., 2019, despite their sample including younger respondents (19.9% of respondents were ≥ 70 years of age), may be driven by their female respondent cohort. In Nolte et al. [28], women had slightly worse PRO scores than men did, with the largest differences observed for insomnia, fatigue and pain.
The HRQoL results reported in the general US population in this study are comparable to the HRQoL results of patients with prostate cancer reported in the literature. Males with asymptomatic nonmetastatic prostate cancer tend to have low pain and moderate QoL [11], similar to what those in the general US population in this study [29] had. As prostate cancer advances to mCRPC in patients, however, they tend to have moderate to severe pain and poorer QoL [29, 30]. Many men with nmCRPC and chemo-naïve mCRPC also tend to have some fatigue, which can be moderate to severe in intensity and can impact their QoL [31].
The availability of the reference values for these PROs is very important. The reference values enable researchers to compare the self-reported HRQoL of patients with advanced prostate cancer in the US with the HRQoL reported in the general US population and allow for a better interpretation of those PRO scores. These reference values also allow for a better understanding and interpretation of patients’ HRQoL at baseline and post-treatment, and this improved understanding and interpretation enables a keener evaluation of the impact of treatment.
Furthermore, the US reference values allow for the interpretation of cognitive impairment and fatigue in the advanced prostate cancer clinical trials compared to the national norms of cognitive impairment and fatigue. This is important for prostate cancer, as cognitive impairment and fatigue are common treatment-related impacts [32,33,34] and the specific interaction between cognitive impairment and fatigue has not been thoroughly explored. The results of the crosstabulation of the PROMIS Fatigue-SF4a and Neuro-QoL 2.0 Cognitive Function Short Form mean reference values indicate that within the general population a large percentage of the raked male population (median age of 70 years) have above average fatigue and/or below average cognitive function (227 [26%]). Researchers can take these results into account when they evaluate fatigue and cognitive impairment in patients with advanced prostate cancer. Some evidence suggests that the high rates of cognitive impairment and fatigue observed in males in advanced prostate cancer trials may in part result from their advanced age (not necessarily solely resulting from the cancer itself or the treatment [32,33,34]). Finally, the reference values can help define HRQoL cut-off scores for certain subgroups in advanced prostate cancer clinical trial populations such as mHSPC and mCRPC. It can be evaluated, for example, how far from the reference values are patients with mCRPC vs. mHSPC.
Study Strengths and Limitations
There are some limitations to this study. First, the sample for it was raked against the age of patients participating in global prostate cancer clinical trials (not exclusively US patients). The US reference values may not therefore be generalizable to advanced prostate cancer patient populations in other countries. Second, the sample for this study was raked against patients from prostate cancer clinical trials who are usually healthier and have higher functioning than the general prostate cancer population (e.g., clinical trials typically include patients with an Eastern Cooperative Oncology Group status of 0 or 1 and exclude patients with certain comorbidities such as cardiovascular disease). Third, respondents may have had respondent burden while completing the lengthy survey. Fourth, there may be a selection bias with more healthy and educated people with sufficient knowledge of the internet responding to the online survey.
This study has several strengths, however. First, it’s the first to provide reference values for several PRO questionnaires that are commonly administered in prostate cancer trials to men of similar ages to those of typical patients with prostate cancer. While reference values have been reported in other studies, given that many of the domains assessed with these questionnaires are likely to be impacted by age (e.g., fatigue, pain, or physical functioning), age-raked reference values are important in the setting of prostate cancer. Second, administering the survey online was an efficient, cost-effective method of obtaining large, representative samples of the general population. This method has advantages over in-person or telephone interviews, as it yields higher response rates and avoids interviewer bias. Third, the online survey was administered in a scientifically robust manner. For example, respondents were required to complete each question before they could complete the following one; therefore, there were no missing data for the survey. The survey was also designed to prevent respondents from answering too fast or randomly, a design based on Critical Mix’s built-in processes and technologies. Furthermore, the “Not Applicable” response option was added to selected questions so that respondents who were not being treated for advanced prostate cancer would be able to indicate that a question did not apply to them. Fourth, the results of this study were aligned to those reported in the general population for EORTC QLQ-C30, PROMIS Fatigue-SF4a, and Neuro-QoL 2.0 Cognitive Function Short Form, and their alignment demonstrates that the data were robust and respondents were attentive. Although the survey included nine questionnaires, the mean time to complete the survey was 24 min.
Conclusions
This study presented a set of reference values for several PRO questionnaires (BPI-SF, Neuro-QoL 2.0 Cognitive Function Short Form, PROMIS Fatigue-SF4a, FACT-G, EORTC QLQ-C30, and EORTC QLQ-PR25) that are commonly used in prostate cancer trials from a US population that was gender- and age-raked to patients with prostate cancer. The availability of these reference values is valuable because it will enable researchers to compare the HRQoL of patients with advanced prostate cancer in the US with the HRQoL of the general US population. This comparison will allow for a better interpretation of these PRO scores, a better characterization of the incremental burden of advanced prostate cancer, and a better assessment of treatment benefits on HRQoL.
References
National Cancer Institute. SEER Cancer statistics factsheets: prostate cancer. 2015. http://seer.cancer.gov/statfacts/html/prost.html. Accessed Jan 2016.
Aggarwal RR, Feng FY, Small EJ. Emerging categories of disease in advanced prostate cancer and their therapeutic implications. Oncology (Williston Park). 2017;31:467–74.
Tomaszewski EL, Moise P, Krupnick RN, et al. Symptoms and impacts in non-metastatic castration-resistant prostate cancer: qualitative study findings. Patient. 2017;10:567–78.
Holmstrom S, Naidoo S, Turnbull J, Hawryluk E, Paty J, Morlock R. Symptoms and impacts in metastatic castration-resistant prostate cancer: qualitative findings from patient and physician interviews. Patient. 2019;12:57–67.
Nussbaum N, George DJ, Abernethy AP, et al. Patient experience in the treatment of metastatic castration-resistant prostate cancer: state of the science. Prostate Cancer Prostatic Dis. 2016;19:111–21.
Naidoo S, Paty J, Fickley C, et al. Enhancing interpretation of patient-reported outcome scores through intuitive metrics: an example from prostate cancer. Value Health. 2016;19(3):A160.
Armstrong AJ, Szmulewitz RZ, Petrylak DP, et al. ARCHES: a randomized, phase III study of androgen deprivation therapy with enzalutamide or placebo in men with metastatic hormone-sensitive prostate cancer. J Clin Oncol. 2019;37:2974–86.
Chi KN, Protheroe A, Rodriguez-Antolin A, et al. Patient-reported outcomes following abiraterone acetate plus prednisone added to androgen deprivation therapy in patients with newly diagnosed metastatic castration-naive prostate cancer (LATITUDE): an international, randomised phase 3 trial. Lancet Oncol. 2018;19:194–206.
Agarwal N, McQuarrie K, Bjartell A, et al. Health-related quality of life after apalutamide treatment in patients with metastatic castration-sensitive prostate cancer (TITAN): a randomised, placebo-controlled, phase 3 study. Lancet Oncol. 2019;20:1518–30.
Sternberg CN, Fizazi K, Saad F, et al. Enzalutamide and survival in nonmetastatic, castration-resistant prostate cancer. N Engl J Med. 2020;382:2197–206.
Saad F, Cella D, Basch E, et al. Effect of apalutamide on health-related quality of life in patients with non-metastatic castration-resistant prostate cancer: an analysis of the SPARTAN randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 2018;19:1404–16.
Beer TM, Armstrong AJ, Rathkopf DE, et al. Enzalutamide in metastatic prostate cancer before chemotherapy. N Engl J Med. 2014;371:424–33.
Cella D, Traina S, Li T, et al. Relationship between patient-reported outcomes and clinical outcomes in metastatic castration-resistant prostate cancer: post hoc analysis of COU-AA-301 and COU-AA-302. Ann Oncol. 2018;29:392–7.
Scher HI, Fizazi K, Saad F, et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N Engl J Med. 2012;367:1187–97.
Kretschmer A, Ploussard G, Heidegger I, et al. Health-related quality of life in patients with advanced prostate cancer: a systematic review. Eur Urol Focus. 2021;7:742–51.
Van Hemelrijck M, Sparano F, Moris L, et al. Harnessing the patient voice in prostate cancer research: systematic review on the use of patient-reported outcomes in randomized controlled trials to support clinical decision-making. Cancer Med. 2020;9:4039–58.
Cella DF, Tulsky DS, Gray G, et al. The functional assessment of cancer therapy scale: development and validation of the general measure. J Clin Oncol. 1993;11:570–9.
Daut RL, Cleeland CS, Flanery RC. Development of the Wisconsin Brief Pain Questionnaire to assess pain in cancer and other diseases. Pain. 1983;17:197–210.
National Institute of Neurological Disorders and Stroke (NINDS)User Manual for the Quality of Life in Neurological DisordersA(Neuro-QoL)Q4 Measures, Version 2.0. http://www.healthmeasures.net. Accessed Mar 2015.
Patient-Reported Outcomes Measures Information System.Scoring Manual for the PROMIS Fatigue Measure. http://www.healthmeasures.net. Accessed Feb 2019.
Cella D.Manual of the Functional Assessment of Chronic Illness Therapy (FACIT) Measurement System, Version 4.1. Center on Outcomes, Research & Education (CORE); Evanston, IL (2004). http://www.facit.org Accessed 14 June 2022.
Fayers PM, Aaronson NK, Bjordal K, Groenvold M, Curran D, Bottomley A. The EORTC QLQ-C30 scoring manual. 3rd ed. Brussels: European Organisation for Research and Treatment of Cancer; 2001.
van Andel G, Bottomley A, Fosså SD, et al. An international field study of the EORTC QLQ-PR25: a questionnaire for assessing the health-related quality of life of patients with prostate cancer. Eur J Cancer. 2008;44:2418–24.
Moreh E, Jacobs JM, Stessman J. Fatigue, function, and mortality in older adults. J Gerontol A Biol Sci Med Sci. 2010;65:887–95.
Zis P, Daskalaki A, Bountouni I, Sykioti P, Varrassi G, Paladini A. Depression and chronic pain in the elderly: links and management challenges. Clin Interv Aging. 2017;12:709–20.
Mahler DA. Evaluation of dyspnea in the elderly. Clin Geriatr Med. 2017;33:503–21.
Patel D, Steinberg J, Patel P. Insomnia in the elderly: a review. J Clin Sleep Med. 2018;14:1017–24.
Nolte S, Liegl G, Petersen MA, et al. General population normative data for the EORTC QLQ-C30 health-related quality of life questionnaire based on 15,386 persons across 13 European countries, Canada and the Unites States. Eur J Cancer. 2019;107:153–63.
Curran D, Fossa S, Aaronson N, Kiebert G, Keuppens F, Hall R. Baseline quality of life of patients with advanced prostate cancer. European Organization for Research and Treatment of Cancer (EORTC), Genito-Urinary Tract Cancer Cooperative Group (GUT-CCG). Eur J Cancer. 1997;33:1809–14.
Jenkins V, Solis-Trapala I, Payne H, et al. Treatment experiences, information needs, pain and quality of life in men with metastatic castrate-resistant prostate cancer: results from the EXTREQOL study. Clin Oncol (R Coll Radiol). 2019;31:99–107.
Rodriguez Antolin A, Martinez-Pineiro L, Jimenez Romero ME, et al. Prevalence of fatigue and impact on quality of life in castration-resistant prostate cancer patients: the VITAL study. BMC Urol. 2019;19:92.
Ryan C, Wefel JS, Morgans AK. A review of prostate cancer treatment impact on the CNS and cognitive function. Prostate Cancer Prostatic Dis. 2020;23:207–19.
Feng LR, Regan J, Shrader JA, et al. Cognitive and motor aspects of cancer-related fatigue. Cancer Med. 2019;8:5840–9.
Wang X, Hui Y, Wang S, et al. Comparison of effectiveness and safety outcomes of abiraterone versus enzalutamide in patients with metastatic castration-resistant prostate cancer: a systematic review and meta-analysis. J Pharm Pharm Sci. 2020;23:451–61.
Acknowledgements
We thank the participants of the study.
Funding
This study and the Journal’s Rapid Service and Open Access Fee was funded by Astellas Pharma Inc.
Medical Writing, Editorial, and Other Assistance
The authors would like to thank Mok Oh for his guidance in the development of this manuscript. Medical writing assistance was provided by Veleka Allen, PhD, from IQVIA. Editorial assistance was provided by Stephanie Rippon, MBio, and Nicholas Strange, BA, from Complete HealthVizion, which was funded by both Astellas Pharma Inc. and Pfizer Inc.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
AG, RM and JT contributed substantially to the study design. The study data was acquired by RM, JT and JR. AG, DC, JT and JR analyzed the study data. All authors made significant contributions to interpreting the study data.
Disclosures
David Cella, Arijit Ganguli, James Turnbull, Jeffrey Rohay and Robert Morlock all have nothing to disclose.
Compliance with Ethics Guidelines
This survey was deemed Institutional Review Board–exempt, as all responses were anonymized, aggregated, and untraceable to the respondents by Ethical and Independent Review Services. The study complies with the Declaration of Helsinki and informed consent was obtained from all participants.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Cella, D., Ganguli, A., Turnbull, J. et al. US Population Reference Values for Health-Related Quality of Life Questionnaires Based on Demographics of Patients with Prostate Cancer. Adv Ther 39, 3696–3710 (2022). https://doi.org/10.1007/s12325-022-02204-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-022-02204-3