Abstract
Introduction
A trial-based assessment was completed to evaluate the cost-effectiveness of ceritinib as a first-line treatment for advanced non-small cell lung cancer (NSCLC) with rearrangement of anaplastic lymphoma kinase.
Methods
Based on the disease situation of advanced NSCLC, a Markov model was constructed to estimate the costs and benefits of ceritinib and platinum-based chemotherapy. The cost information and health utilities were obtained from published literature. The incremental cost-effectiveness ratio was calculated. The stability of the model was verified by sensitivity analyses.
Results
The base case analysis results indicated that compared with platinum-based chemotherapy, ceritinib therapy would increase benefits in a 5-, 10- and 15-year time horizon, with extra costs of $230,661.61, $149,321.52 and $136,414.43 per quality-adjusted life-year gained, respectively. The most sensitive parameter in the model analysis was the cost of ceritinib. Probabilistic sensitivity analysis suggested that at the current price of ceritinib, the chance of ceritinib being cost-effective was 0 at the willingness-to-pay threshold of $27,142.85 per quality-adjusted life-year (three times the per capita gross domestic product of China).
Conclusion
As a first-line treatment for advanced NSCLC with rearrangement of anaplastic lymphoma kinase, ceritinib is unlikely to be cost-effective at the current price from the Chinese healthcare perspective. To meet the treatment demands of patients, it may be a better option to reduce the price or provide appropriate drug assistance policies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lung cancer, which has the highest incidence among cancers, is the main cause of death among men and women in 87 countries and 26 countries, respectively [1, 2]. In 2013, approximately 1.8 million people worldwide were newly diagnosed with lung cancer, and 1.6 million died of lung cancer [3]. In China, the incidence rate of lung cancer was 57.70/100,000, the mortality was 46.92/100,000 and both rank first [4]. Moreover, one study showed that the average economic burden of lung cancer was $43,336 (US dollars throughout) per patient in the urban areas of China; the economic burden in the first year after diagnosis was $30,277 per capita, which accounted for 171% of household annual income [5]. More than 85% of patients with lung cancer have non-small cell lung cancer (NSCLC) [6]. Generally, patients with advanced NSCLC and good performance status benefit from chemotherapy and usually receive platinum-based regimens [7]. However, the clinical outcome of chemotherapy is not particularly good, and the response rate is only approximately 20% [8].
After anaplastic lymphoma kinase (ALK) rearrangements were discovered, small-molecule ALK tyrosine kinase inhibitors became an active research field [9]. Crizotinib, which is an important drug for treating ALK-rearranged NSCLC, will eventually produce drug resistance [10]. As a new second-generation ALK inhibitor, ceritinib was initially used to treat advanced NSCLC in patients after the progress of crizotinib treatment [11]. Recently, a clinical trial (ASCEND-4) from 134 centres in 28 countries showed that compared with platinum-based chemotherapy, ceritinib first-line treatment significantly prolonged the deterioration time for patients with advanced ALK-rearranged NSCLC. The results indicated that the median progression-free survival (PFS) was 16.6 months after ceritinib treatment, whereas it was 8.1 months in the platinum-based chemotherapy group [12]. Therefore, ceritinib should be considered as a first-line treatment for ALK rearrangement patients with advanced non-squamous NSCLC. However, in China, the cost-effectiveness of ceritinib in treating advanced NSCLC is unclear.
Our study was designed to evaluate the long-term cost-effectiveness of ceritinib versus platinum-based chemotherapy for advanced ALK-rearranged NSCLC from the perspective of Chinese healthcare.
Methods
Economic Model
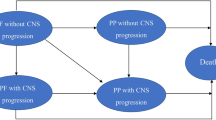
On the basis of the clinical trial and considering the actual clinical situation, we established a Markov model with TreeAge Pro 2011 to assess the economic outcome of ceritinib versus platinum-based chemotherapy for patients with advanced ALK-rearranged NSCLC. Because our economic evaluation was based on a mathematical model to simulate the patient’s lifetime, it did not require the approval of the independent ethics committee. The Markov model (Fig. 1) was utilized to predict health and economic outcomes consisting of three states: PFS, progressed survival and death. All patients first entered a state of PFS with the first-line treatments. Once the disease progresses, the patient could receive second-line treatment. Patients who experienced multiple progressions were considered incurable, no longer received treatment and remained in the state until death [13]. The model cycle was 3 weeks, consistent with the treatment cycle. During each model cycle, patients moved to the next health state or stayed in the current state according to a specific probability until a time horizon termination of 15 years. The probability of progressive disease or death was calculated according to the results of the ASCEND-4 clinical trial.
a Abbreviated decision tree and Markov model of drug regimens used to compare two strategies for treating advanced non-small cell lung cancer with rearrangement of anaplastic lymphoma kinase. b Influence diagram shows a network of three health states linked by transitional variables. NSCLC non-small cell lung cancer
Two potential competing strategies were compared by simulating a population with advanced ALK-rearranged NSCLC: pemetrexed combined with cisplatin/carboplatin followed by pemetrexed maintenance or initial use of targeted therapy with ceritinib. After disease progression, docetaxel, pemetrexed, gefitinib or erlotinib was administered. Hospice treatment was assumed to undergo best supportive care (BSC).
The primary outcome measures are quality-adjusted life-years (QALYs) and cost. The incremental cost-effectiveness ratios (ICERs), which indicate the incremental cost of each additional QALY, were calculated. In line with the World Health Organization (WHO) proposal [14], the willingness-to-pay (WTP) threshold of $27,142.85 used in the cost-effectiveness analysis was equal to three times the per capita gross domestic product (GDP) of China in 2017. All cost and utility values are discounted to 2017 using a 5% annual rate in accordance with Chinese pharmacoeconomics guidelines [15]. The exchange rate is 6.594 RMB for 1 US dollar (December 2017) [16].
This article is based on previously conducted studies and model techniques and does not contain any studies with human participants or animals performed by any of the authors.
Clinical Inputs
The inputs of transition parameters and proportions were based on the ASCEND-4 clinical trial, which is displayed in Fig. 2. The ASCEND-4 trial compared the first-line treatments of ceritinib with platinum-based chemotherapy followed by pemetrexed maintenance for advanced ALK-rearranged NSCLC, and the results indicated that the median PFS was 16.6 months and 8.1 months, respectively.
The survival rates of both treatment regimens were derived from the ASCEND-4 trial report. First, the PFS and overall survival (OS) state probability were extracted by the GetData Graph Digitizer software (version 2.25) from the published Kaplan–Meier curves of PFS and OS in the ASCEND-4 trial. Then, these data were used to fit the survival curves with Weibull survival models using R software (version 3.2.2). The obtained values of scale (λ) and shape (γ) parameters are demonstrated in Fig. 2. The transition probability of disease progression and death at cycle t in the model was estimated as follows: P(t) = 1 − exp[λ(t − 1)γ − λtγ].
Cost and Utility Data
Since this analysis was carried out from the perspective of the Chinese healthcare system, only direct costs, including drug, administration, end-of-life treatment and management of serious adverse events (SAEs), were considered (Table 1).
As observed in the supplementary appendix of the ASCEND-4 trial, ceritinib was administered to patients with ALK-rearranged NSCLC at 750 mg per day before disease progression. However, the ASCEND-8 trial indicated that relative to 750 mg, ceritinib at a dose of 450 mg had consistent efficacy and less gastrointestinal toxicity [17, 18]. Based on the results of the ASCEND-8, the recommended daily dose of ceritinib has been changed to 450 mg in the USA, European Union and other countries worldwide [19, 20]. Therefore, drug-treatment costs for the ceritinib arm in the state of PFS were based on the recommended dose of 450 mg daily with food. Platinum-based chemotherapy, which is pemetrexed (500 mg/m2) combined with cisplatin (75 mg/m2) or carboplatin (AUC 5–6), was given every 3 weeks. After four cycles of combination chemotherapy, if there was no disease progression, patients would undergo maintenance treatment (pemetrexed 500 mg/m2). As a result of the lack of a detailed clinical trial plan, the cost of follow-up treatment was derived from our previous study, which is an empirically assessed cost of treating NSCLC in China [21].
All costs related to chemotherapy, BSC and management of SAEs are derived from the published literature in China [22,23,24,25]. SAEs included in the model were considered only grade 3 or 4 toxicity with an incidence rate greater than 5% and significant differences between the two groups (Table 1) [26]. The management cost of SAEs is the unit cost of each SAE multiplied by its corresponding occurrence rate. We assumed that the body surface area was 1.72 m2 to estimate the antineoplastic agent dosage [22].
The health utility was obtained from published literature (Table 1) [27]. QALYs were obtained by combined life years with utility. We used utilities of 0.71 and 0.67 for patients in the disease progression-free state and disease progression state, respectively.
Sensitivity Analyses
One-way and probabilistic sensitivity analyses were performed by setting the upper and lower limits and a specific distribution for each parameter to reflect the influence of the uncertainty of the model parameters on the results of the study. One-way sensitivity analyses fixed other parameter values and changed only one parameter to explore the effect of this parameter on the results. In order to consider uncertainty, we set a range of ± 30% for all parameters, except for the discount rates, the range of which is derived from the Chinese guideline on pharmacoeconomic evaluations [15]. In the probabilistic sensitivity analysis, parameters were set in a distribution (Table 1), and the Monte Carlo method was used to simulate 1000 random times. The probability and utility used the beta distribution; and lognormal distribution was adopted for all input costs (Table 1). The cost-effectiveness acceptability curves and the scatter plot obtained by probabilistic sensitivity analysis describe the sensitive range of WTP and the cost-effective probability of ceritinib therapy compared with platinum-based chemotherapy.
Results
Base Case Analysis
Weibull models were used to fit the survival curves of PFS and OS from the ASCEND-4 trial (Fig. 2) and show that the decision analysis model established in this study can reflect the clinical effects very well. The base case results (Table 2) suggested that compared with chemotherapy, the ceritinib therapy would add an extra 0.33, 0.59 and 0.65 QALY in a 5-, 10- and 15-year time span, and the ICERs were $230,661.61, $149,321.52 and $136,414.43 per QALY, respectively.
Uncertainty Analyses
Figure 3 presents the results of one-way sensitivity analyses. The price of ceritinib had the greatest influence on the result, and then the utility of PFS, cost of pemetrexed, body surface area and the discount rate. The other parameters did not significantly modify the ICER. All of these parameters that differ in scope did not cause ICER to be lower than the WTP threshold ($27,142.85).
The probability sensitivity analysis reflects the influence of the overall change of all parameters on the results. The ICER scatter plot (Fig. 4) of 1000 simulations showed that all ICERs were distributed above the WTP threshold ($27,142.85/QALY) line, so the probability that ceritinib was cost-effective was 0.
Acceptability curves (Fig. 5) indicated that the cost-effectiveness likelihood of ceritinib arm increased with increasing WTP thresholds, and the sensitive range was about $90,000 to $300,000. It suggested that if the WTP exceeded $138,000 (per capita GDP $46,000), more than 50% of advanced ALK-rearranged NSCLC, with first-line treatment of ceritinib therapy, could achieve cost-effectiveness. At WTPs exceeding $300,000, the cost-effectiveness likelihood of first-line treatment with ceritinib was almost 100%.
Discussion
In the past few years, cancer treatment has improved greatly [28]. However, the ensuing problems have also raised people’s concern, such as the cost of certain new anti-tumour drugs, which involve the value of the drug, the economic level of the people, the funds that the healthcare system provides for them and so on [29]. The high cost of treatment and the rapid rise in the prices of anticancer drugs have created a heavy burden and raised issues with regard to the long-term sustainability of patients and healthcare systems [30]. For example, in the USA, the costs of cancer treatment have increased more than 10 times per year from 2000 ($5000–10,000) to 2012 (> $100,000), while the average income of American households has decreased by 8% over the last 10 years [31]. Therefore, it is necessary to ensure the affordability and accessibility of cancer drugs [32].
Our study is the first economic evaluation of ceritinib for the first-line treatment of patients with advanced ALK-positive NSCLC from the perspective of China. The base case analysis showed that ICERs for ceritinib versus platinum-based chemotherapy were $230,661.61, $149,321.52 and $136,414.43 per QALY at 5, 10 and 15 years, respectively. The results of the Monte Carlo simulation showed that all ICERs were above the WTP threshold (Fig. 4), and ceritinib was not cost-effective at the current cost. However, one-way sensitivity analysis results revealed that the price of ceritinib was the most significant parameter to the evaluation results, so the best option is to lower the price of ceritinib.
Multiple studies have been conducted to assess the cost-effectiveness of ceritinib in treating patients with ALK-positive NSCLC. A Canadian study demonstrated that ceritinib was cost-effective compared with pemetrexed, BSC and historical control as second-line treatment [23]. A Mexican study revealed that ceritinib was a cost-effective therapy compared with current therapies for chemotherapy-experienced patients [33]. A UK-based analysis showed that ceritinib would be a cost-effective option compared with other alternatives (crizotinib, docetaxel and pemetrexed) for previously treated patients [34]. A study by Zhou et al. demonstrated that ceritinib was cost-effective compared with crizotinib and platinum doublet chemotherapy among previously untreated patients with metastatic NSCLC from the perspective of US payers [35].
Because each country’s healthcare system, costs and models used are different, the conclusions drawn are different. Ceritinib has been recommended as a first-line treatment for advanced ALK-rearranged NSCLC in the 2019 National Comprehensive Cancer Network (NCCN) guidelines [7]. However, as a new treatment option, ceritinib places a heavy burden on patients ($67.46/150 mg). Therefore, it is necessary to make an economic assessment of ceritinib, especially in developing countries, such as China, where the population is more than 13.9 billion and the resources of the healthcare system are insufficient [5, 36, 37]. With the gradual development of pharmacoeconomics, it is becoming more and more widely used in the medical field. The relevant documents in China have proposed to use pharmacoeconomic evaluation in drug pricing [38]. Drug pricing based on the evidence-based basis of pharmacoeconomics will help to control the excessive growth of drug costs. According to the theory, methods and research results of pharmacoeconomics, evidence-based evidence can be provided for the selection of medical insurance catalogue drugs, which is conducive to reducing medical expenses and optimizing the allocation of health resources. Use of a formulary is a widely accepted method for the management of drugs for medical insurance in China. Drugs selected in the formulary must satisfy some criteria, including being clinically needed, safe, effective, convenient to use and reasonably priced [39]. In recent years, pharmacoeconomic evaluation data are also listed as required documents in selection of the National Basic Medical Insurance Drug Formulary List, which is a primary reimbursement formulary in China [40]. Although the pharmacoeconomics has attracted great interest from the Chinese government, it is not implemented nationwide in China [41].
There are several limitations in this study. First, subsequent therapy costs after disease progression are estimated on the basis of the ASCEND-4 trial, NCCN guidelines and expert opinion, which may be different from clinical practice in China. However, the sensitivity analyses indicated that they did not have much effect on the outcome. Second, we did not consider the costs for all adverse events (AEs); because some of the AE costs were negligible, there was little impact on the results. Thirdly, what we mean by progression of first-line treatment is progression-free disease on second-line treatment. According to a study by Chouaid et al. [27], while there is a slight numerical difference between the utility of progressive disease on first-line treatment and the utility of progression-free disease on second-line treatment, differences in utility values were considered clinically irrelevant. During the course of disease progression, the health state utility may go well below 0.70. Therefore in the current study, we used the utility of 0.67 for patients in the disease progression state. In order to address the uncertainty of utilities, we have set a wider range (± 30%) to perform sensitivity analyses. Although the utility of PFS impacted the results greatly, all of the parameters did not cause ICER to be lower than the WTP threshold. Finally, as a result of the lack of a clear WTP threshold in China, according to the recommendation from the WHO [14], the current study used triple the per capita GDP of China as the WTP threshold, which might vary with different factors. However, with long-term follow-up of these events, the uncertainty of these parameters would be further reduced.
Conclusion
From the Chinese healthcare perspective, ceritinib, as the first-line treatment for advanced NSCLC with ALK rearrangement, is unlikely to be cost-effectiveness according to our analysis. To meet a broad range of treatment demands for a vast number of patients, the medical decision-making department should consider making appropriate price cuts or require the enterprises to provide appropriate drug donation policies.
References
Global Burden of Disease Cancer Collaboration, Fitzmaurice C, Allen C, et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 32 cancer groups, 1990 to 2015: a systematic analysis for the global burden of disease study. JAMA Oncol. 2017;3(4):524–48.
Zaidi Z, Hamdi Cherif M. PS01.07: Geographical distribution of lung cancer mortality worldwide: topic: pathology. J Thorac Oncol. 2016;11(11S):S273–4.
Wong MCS, Lao XQ, Ho KF, Goggins WB, Tse SLA. Incidence and mortality of lung cancer: global trends and association with socioeconomic status. Sci Rep. 2017;7(1):14300.
Chen W, Zheng R, Zhang S, et al. Cancer incidence and mortality in China in 2013: an analysis based on urbanization level. Chin J Cancer Res. 2017;29(1):1–10.
Zhang X, Liu S, Liu Y, et al. Economic burden for lung cancer survivors in urban China. Int J Environ Res Public Health. 2017;14(3):308.
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61(2):69–90.
Nation Comprehensive Cancer Network. Non-small cell lung cancer [version 2.2019]. https://www.nccn.org/. Accessed 13 Aug 2019.
Tan DS, Araujo A, Zhang J, et al. Comparative efficacy of ceritinib and crizotinib as initial ALK-targeted therapies in previously treated advanced NSCLC: an adjusted comparison with external controls. J Thorac Oncol. 2016;11(9):1550–7.
Amin AD, Li L, Rajan SS, et al. TKI sensitivity patterns of novel kinase-domain mutations suggest therapeutic opportunities for patients with resistant ALK+ tumors. Oncotarget. 2016;7(17):23715–29.
Zhu Q, Hu H, Weng DS, et al. Pooled safety analyses of ALK-TKI inhibitor in ALK-positive NSCLC. BMC Cancer. 2017;17(1):412.
Li S, Qi X, Huang Y, Liu D, Zhou F, Zhou C. Ceritinib (LDK378): a potent alternative to crizotinib for ALK-rearranged non-small-cell lung cancer. Clin Lung Cancer. 2015;16(2):86–91.
Soria JC, Tan DSW, Chiari R, et al. First-line ceritinib versus platinum-based chemotherapy in advanced ALK-rearranged non-small-cell lung cancer (ASCEND-4): a randomised, open-label, phase 3 study. Lancet. 2017;389(10072):917–29.
Wan X, Zhang Y, Tan C, et al. First-line nivolumab plus ipilimumab vs sunitinib for metastatic renal cell carcinoma: a cost-effectiveness analysis. JAMA Oncol. 2019;5(4):491–6.
Murray CJ, Evans DB, Acharya A, Baltussen RM. Development of WHO guidelines on generalized cost-effectiveness analysis. Health Econ. 2000;9(3):235–51.
China Center for Health Economic Research. China guidelines for pharmacoeconomic evaluations (Version 2011) [in Chinese] (2011). https://tools.ispor.org/peguidelines/. Accessed 13 Aug 2019.
The People’s Bank of China. Exchange rate. http://www.pbc.gov.cn. Accessed 13 Aug 2019.
Cho BC, Kim DW, Bearz A, et al. ASCEND-8: a randomized phase 1 study of ceritinib, 450 mg or 600 mg, taken with a low-fat meal versus 750 mg in fasted state in patients with anaplastic lymphoma kinase (ALK)-rearranged metastatic non-small cell lung cancer (NSCLC). J Thorac Oncol. 2017;12:1357–67.
Cho BC, Obermannova R, Bearz A, et al. Efficacy and safety of ceritinib (450 mg/d or 600 mg/d) with food versus 750-mg/d fasted in patients with ALK receptor tyrosine kinase (ALK)-positive NSCLC: primary efficacy results from the ASCEND-8 study. J Thorac Oncol. 2019;14:1255–65.
Novartis Pharmaceuticals Corporation. Zykadia dosing and administration. https://www.hcp.novartis.com/products/zykadia/alk-nsclc/dosing-administration. Accessed 13 Aug 2019.
European Medical Agency. Zykadia European public assessment report. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/003819/WC500187504.pdf. Accessed 13 Aug 2019.
Zeng X, Karnon J, Wang S, et al. The cost of treating advanced non-small cell lung cancer: estimates from the Chinese experience. PLoS One. 2012;7(10):e48323.
Zeng X, Peng L, Li J, et al. Cost-effectiveness of continuation maintenance pemetrexed after cisplatin and pemetrexed chemotherapy for advanced nonsquamous non-small-cell lung cancer: estimates from the perspective of the Chinese health care system. Clin Ther. 2013;35(1):54–65.
Hurry M, Zhou ZY, Zhang J, et al. Cost-effectiveness of ceritinib in patients previously treated with crizotinib in anaplastic lymphoma kinase positive (ALK+) non-small cell lung cancer in Canada. J Med Econ. 2016;19(10):936–44.
Lu S, Ye M, Ding L, Tan F, Fu J, Wu B. Cost-effectiveness of gefitinib, icotinib, and pemetrexed-based chemotherapy as first-line treatments for advanced non-small cell lung cancer in China. Oncotarget. 2017;8(6):9996–10006.
Chongqing T, Liubao P, Xiaohui Z, et al. Cost-utility analysis of the newly recommended adjuvant chemotherapy for resectable gastric cancer patients in the 2011 Chinese National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology: Gastric Cancer. Pharmacoeconomics. 2014;32(3):235–43.
Wan XM, Peng LB, Ma JA, Li YJ. Economic evaluation of nivolumab as a second-line treatment for advanced renal cell carcinoma from US and Chinese perspectives. Cancer. 2017;123(14):2634–41.
Chouaid C, Agulnik J, Goker E, et al. Health-related quality of life and utility in patients with advanced non-small-cell lung cancer: a prospective cross-sectional patient survey in a real-world setting. J Thorac Oncol. 2013;8(8):997–1003.
Novello S, Barlesi F, Califano R, et al. Metastatic non-small-cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2016;27(Suppl 5):v1–27.
Barron A, Wilsdon T. Challenging perceptions about oncology product pricing in breast and colorectal cancer. Pharmaceut Med. 2016;30(6):321–6.
Migliorino MR, Santo A, Romano G, et al. Economic burden of patients affected by non-small cell lung cancer (NSCLC): the LIFE study. J Cancer Res Clin Oncol. 2017;143(5):783–91.
Ocana A, Amir E, Tannock IF. Toward value-based pricing to boost cancer research and innovation. Cancer Res. 2016;76(11):3127–9.
Salmasi S, Lee KS, Ming LC, et al. Pricing appraisal of anti-cancer drugs in the south east Asian, western Pacific and east Mediterranean region. BMC Cancer. 2017;17(1):903.
Balu S, Cerezo-Camacho O, Smith NJ, Beckerman R. Cost-effectiveness of ceritinib versus current therapies for chemotherapy-experienced anaplastic lymphoma kinase positive non-small cell lung cancer patients in Mexico. Value Health. 2015;18(7):A821.
Zhou Z, Zhang J, Fan L, et al. Cost-effectiveness of ceritinib in the treatment of previously treated anaplastic lymphoma kinase-positive (ALK+) non-small cell lung cancer in the United Kingdom. Value Health. 2015;18(7):A455–6.
Zhou ZY, Mutebi A, Han S, et al. Cost-effectiveness of ceritinib in previously untreated anaplastic lymphoma kinase-positive metastatic non-small cell lung cancer in the United States. J Med Econ. 2018;21(6):577–86.
National Bureau of Statistics of China. China statistical year book 2018. http://www.stats.gov.cn/tjsj/ndsj/2018/indexeh.htm. Accessed 13 Aug 2019.
Shi J, Yao Y, Liu G. Modeling individual health care expenditures in China: evidence to assist payment reform in public insurance. Health Econ. 2018;27:1945–62.
Gao SN, Liu GQ. The application of pharmacoeconomics in the field of medicine and health [in Chinese]. Drug Econ. 2017;12(8):16–8.
Peng J, Tan C, Zeng X, Liu S. Cost-effectiveness analysis of capecitabine monotherapy versus capecitabine plus oxaliplatin in elderly patients with advanced gastric cancer. PLoS One. 2018;13(6):e0199553.
Announcement of the “2019 National Medical Insurance Drug Catalogue Adjustment Work Plan”. China Health Insurance. https://www.zgylbx.com/. Accessed 13 Aug 2019.
Koh L, Glaetzer C, Chuen Li S, Zhang M. Health technology assessment, international reference pricing, and budget control tools from China’s perspective: what are the current developments and future considerations? Value Health Reg Issues. 2016;9:15–21.
Acknowledgements
Funding
This work and associated journal article processing charges were supported by the National Natural Science Foundation of China (grant numbers 81401547, 81603081); and the Key Science-Technology Research and Development Program of Hunan Province (grant number 2016JC2062). All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
Ye Peng, Fang Ma, Chongqing Tan, Xiaomin Wan, Lidan Yi, Liubao Peng and Xiaohui Zeng have nothing to disclose.
Compliance with Ethics Guidelines
Because our economic evaluation was based on a mathematical model to simulate the patient’s lifetime, it did not require the approval of the independent ethics committee. This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Enhanced Digital Features
To view enhanced digital features for this article go to https://doi.org/10.6084/m9.figshare.9873323.
Rights and permissions
About this article
Cite this article
Peng, Y., Ma, F., Tan, C. et al. Model-Based Economic Evaluation of Ceritinib and Platinum-Based Chemotherapy as First-Line Treatments for Advanced Non-Small Cell Lung Cancer in China. Adv Ther 36, 3047–3058 (2019). https://doi.org/10.1007/s12325-019-01103-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-019-01103-4