Abstract
Extensive evidence supports the claim that the serum neurofilament light chain (sNfL) can be used as a biomarker to monitor disease severity in patients with spinocerebellar ataxia type 3 (SCA3). However, little is known about the associations between sNfL levels and neurochemical alterations in SCA3 patients. In this study, we performed a cross-sectional study to analyze the association between sNfL and brain metabolic changes in SCA3 patients. The severity of ataxia was assessed by using the Scale for the Assessment and Rating of Ataxia (SARA) and the International Cooperative Ataxia Rating Scale (ICARS). The sNfL levels and brain metabolic changes, represented by N-acetyl aspartate (NAA)/creatine (Cr) and choline complex (Cho)/Cr ratios, were measured by a single-molecule array and proton magnetic resonance spectroscopy, respectively. In this cohort, we observed consistently elevated sNfL levels and reduced brain metabolites in the cerebellar hemispheres, dentate nucleus, and cerebellar vermis. However, this correlation was further validated in the cerebellar cortex after analysis using pairwise comparisons and a Bonferroni correction. Taken together, our results further confirmed that sNfL levels were increased in SCA3 patients and were negatively correlated with metabolic changes in the cerebellar cortex. Our data also support the idea that sNfL levels are a promising potential complementary biomarker for patients with SCA3.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Spinocerebellar ataxia type 3 (SCA3) is the most common autosomal dominant ataxia worldwide and is caused by an abnormal CAG expansion at exon 10 of the ATXN3 gene [1]. The number of CAG trinucleotide repeats in ATXN3 ranges from 12 to 44 in the normal population, whereas at least one expanded allele ranging between 52 and 87 CAG repeats is defined as an expanded repeat that is responsible for SCA3 [2]. SCA3 is a disease characterized by clear clinical heterogeneity and mainly involves progressive cerebellar ataxia, pyramidal signs, and extrapyramidal syndrome. Non-characteristic manifestations, such as spastic paraplegia phenotypes, cognitive and psychiatric disturbances, and sleep disorders, have also been observed [3].
The expansion of CAG repeats in the ATXN3 gene produces an abnormally folded protein, which leads to selective neuronal cell death and neurochemical alterations. For instance, decreased acetylcholine and N-acetyl aspartate (NAA) are visualized in SCA3 patients by using magnetic resonance spectroscopy (MRS) [4,5,6]. NAA is one of the most important brain metabolites determined by proton MRS and has also been used as a biomarker of neuronal integrity and viability [7, 8]. MRS can reveal early metabolic/cellular changes in various spinocerebellar ataxia types (SCAs) that are likely to occur before cerebellar signs become obvious; in some cases, they even occur before brain atrophy. This observation indicates that the measurement of neurochemical changes in brain tissue by MRS might be superior to clinical observation in specific circumstances [4].
Preclinical studies of promising drugs to treat ATXN3 expansion have emerged. These studies include disease-modifying therapies, for instance, using antisense oligonucleotides (ASOs) against ATXN3 mRNA to reduce the expression of abnormal proteins in SCA3 animal models and patient-derived cell models [9,10,11]. The use of the CRISPR/Cas9 system to remove the expanded ATXN3 allele has been achieved in induced pluripotent stem cells (iPSCs) of SCA3 patients [12]. However, biomarkers used to evaluate the efficacy of gene therapy in humans have not been adequately assessed. In parallel with the rapid development of these potential therapeutics, biomarkers that can be used to measure target engagement, disease onset, and disease progression need to be established to evaluate the treatment effect of clinical trials. Previous studies have suggested that the serum neurofilament light chain (sNfL) is a promising marker of target engagement in SCA3, as sNfL is detectable in the presymptomatic and symptomatic SCA3 cohort [13,14,15,16]. sNfL levels and cerebellar metabolites of NAA/creatine (Cr) ratios have also been found to be biomarkers of neuro-axonal damage in SCA3 patients, but the relationship between them is still unclear. Therefore, this study assessed whether there is a significant association between cerebellar NAA/Cr values and indicators of neurodegeneration, as reflected by sNfL levels.
Methods
Subjects
This study was approved by the Ethics Committee of the Affiliated Hospital of Hangzhou Normal University. Informed consent was obtained from all subjects before they were recruited for the study. In our study, 20 unrelated patients with genetically confirmed ATXN3 repeat expansion and 14 healthy controls were recruited consecutively between March 2020 and May 2022 from the Department of Neurology at the Affiliated Hospital of Hangzhou Normal University. These normal participants were matched for age and sex at the group level. Serum samples were obtained from 20 patients with ataxia features caused by ATXN3 repeat expansion and 14 normal individuals. The Scale for the Assessment and Rating of Ataxia (SARA) and the International Cooperative Ataxia Rating Scale (ICARS) were used to measure the severity of ataxia symptoms at the time of serum collection.
Proton Magnetic Resonance Spectroscopy (1H-MRS)
All participants underwent identical MR procedures using a Siemens 1.5-T magnetic resonance imaging (MRI) scanner. 1H-MRS measurements were then performed to determine NAA/Cr and choline complex (Cho)/Cr ratio in local cerebellar metabolites [17]. The multivoxel 1H-MRS sequence was acquired using the following parameters: repetition time (TR) = 4280 ms, echo time (TE) = 135 ms, and voxel size = 6.3 mm × 6.3 mm × 15 mm. We selected three target region of interest (ROI) placements in the cerebellum—the bilateral cerebellar hemispheres, the dentate nucleus, and the cerebellar vermis—in sequence by one voxel. The localizations of the three ROI placements are presented in Supplemental Fig. 1. Each ROI was examined bilaterally for the presence of lateralization. The local cerebellar metabolites demonstrating NAA/Cr and Cho/Cr ratio were detected via a proton MR wave.
sNfL Levels
Serum samples were taken from all participants and collected according to standardized local procedures. The serum samples were aliquoted and immediately stored at − 80 °C. sNfL concentrations were detected using a single-molecule array (Simoa) method and then quantified on the Simoa HD-1 Analyzer platform (GBIO, Hangzhou, China) using a Simoa NF-light Advantage Kit (Quanterix, Massachusetts, USA) [18]. All sNfL concentrations were within the linear ranges of this assay.
Statistical Analysis
Statistical analyses were performed using SPSS 25.0 for Mac (Armonk, NY), and graphs were drafted using GraphPad Prism 7 (La Jolla, CA). Test statistics were considered significant at p < 0.05 (two-tailed). The normality of variables was determined using the Shapiro–Wilk test, and we tested the homogeneity of variances using Levene’s. For variables in line with the assumptions of normality and homogeneity of variances, the groups were compared using Student’s t test with a Bonferroni correction for multiple comparisons, if applicable. For variables not in line with the assumptions of normality, the groups were compared using the nonparametric Mann–Whitney U test with a Bonferroni correction for multiple comparisons, if applicable.
Correlation analyses were performed to investigate the relationships between sNfL levels and metabolite changes in the dentate nucleus, cerebellar vermis, and cerebellar hemisphere. Specifically, cerebellar metabolite changes were fitted with the individual sNfL level as a covariate while adjusting for the effects of age. Then, correction for multiple comparisons at the cluster level at p < 0.05 was used to perform the correlation analyses between the sNfL and cerebellar metabolite changes. Finally, the moderation/mediation model was used to test whether the cerebellar metabolite levels would moderate the relationship between sNfL levels and ataxia severity by using Hayes’s algorithm with bootstrapping [19].
Results
Demographic and Clinical Data
In total, 20 SCA3 patients and 14 healthy controls were recruited in this study. The sociodemographic, clinical, and biomarker characteristics of all participants are summarized in Table 1. No significant differences in age or sex were observed between the SCA3 patients and the controls (p > 0.05). The mean age for serum collection in patients with SCA3 was 39.2 years; the mean age at disease onset was 32.5 years, but this age varied widely (23–64 years). The mean CAG repeat length of the SCA3 patients was 74.6. The mean SARA and ICARS scores of all SCA3 carriers were 12.54 and 34.35, respectively.
Brain Metabolite Levels
MRS was performed to measure the NAA/Cr ratios, and we found that both ratios were significantly reduced in the cerebellar cortex, cerebellar vermis, and dentate nucleus of the SCA3 patients compared to the normal controls (cerebellar cortex: p = 0.001, cerebellar vermis: p = 0.005; dentate nucleus: p < 0.001). The NAA/Cho ratios significantly reduced in the cerebellar vermis (p = 0.003) and dentate nucleus (p < 0.001), and no significant difference was identified in the cerebellar cortex. The Cho/Cr ratios in the three regions of interest (ROIs) did not differ between SCA3 patients and normal controls (p > 0.05; Fig. 1; Supplemental Table 1). In addition, no significant differences in the NAA/Cr, Cho/Cr, or NAA/Cho ratios were observed in the three ROIs of the left and right cerebellum (p > 0.05).
Brain metabolite levels of the MRS images in the controls and patient group. (A–C) The values of NAA/Cr ratios were significantly reduced in the cerebellar cortex, cerebellar vermis, and dentate nucleus in the ATXN3 expansion carriers compared with that in the normal controls. (D–F) The values of the Cho/Cr ratios in the three ROIs did not differ between SCA3 patients and normal controls. (G) The values of the NAA/Cho ratio in the cerebellar cortex did not differ between SCA3 patients and normal controls. (H–I) The values of the NAA/Cho ratios were significantly reduced in the cerebellar vermis and dentate nucleus in the ATXN3 expansion carriers compared with that in the normal controls. p values from the independent samples t test(A–B, D–I)and Mann–Whitney U tests(C)are displayed as follows: *p ≤ 0.05; **p ≤ 0.01; ***p ≤ 0.001; ns, not significant. NAA, N-acetyl aspartate; Cr, creatine; Cho, choline complex; NC, normal controls; SCA3, Spinocerebellar ataxia type 3 patients
In all SCA3 carriers, the total SARA scores were negatively correlated with the NAA/Cr ratios (dentate nucleus: r = − 0.689, p = 0.001; cerebellar vermis: r = − 0.553, p = 0.011; cerebellar hemisphere: r = − 0.493, p = 0.027), but they were not correlated with the NAA/Cho ratios in the three ROIs. Similar negative correlations between the total ICARS scores and the NAA/Cr ratios in the three ROIs were observed (dentate nucleus: r = − 0.691, p = 0.001; cerebellar vermis: r = − 0.537, p = 0.015; cerebellar hemisphere: r = − 0.486, p = 0.03), and the ICARS scores did not correlate with the NAA/Cho ratios in the three ROIs. In addition, the Cho/Cr ratios in the cerebellar hemisphere and cerebellar vermis did not correlate with the total SARA and ICARS scores; however, they did negatively correlate with these scores in the dentate nucleus (SARA: r = − 0.576, p = 0.008, ICARS: r = − 0.506, p = 0.023). To confirm the robustness of the primary findings, the Bonferroni correction was applied to calculate an adjusted p value threshold (p < 0.016); the NAA/Cr values in the dentate nucleus and cerebellar vermis were still negatively correlated with the SARA and ICARS scores by this standard, whereas the NAA/Cr values in the cerebellar hemisphere were not correlated with the SARA or ICARS scores. The Cho/Cr ratios in the cerebellar hemisphere were also still negatively correlated with the SARA and ICARS scores after the Bonferroni test. No significant associations were found between the values of the NAA/Cr and NAA/Cho ratios and age at disease onset, disease duration, or CAG repeats in patients with SCA3 (p > 0.05).
sNfL Levels
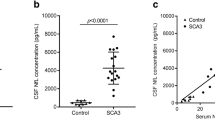
At the time of serum collection, the ATXN3 expansion carriers had higher sNfL levels (mean: 35.33 pg/mL) than the healthy controls (mean: 7.43 pg/mL, p = 0.001; Table 1). This observation was still statistically significant after correction for age (post hoc Bonferroni-corrected p = 0.04). In participants with SCA3, sNfL levels were not associated with age at serum collection, age at onset, disease duration, or CAG repeats. The sNfL levels were correlated with the SARA and ICARS scores (SARA: r = 0.472, p = 0.036; ICARS: r = 0.465, p = 0.039). sNfL did not correlate with posture and gait, limb kinetic function, or oculomotor disorders in any subgroup of ICARS scores, but it was significantly correlated with speech disorders (r = 0.620, p = 0.004). We also used age as a covariate because of its general association with sNfL levels. Statistical values demonstrated the significant correlation between the sNfL concentration and the SARA and ICARS scores in the SCA3 patients (SARA: r = 0.520, p = 0.023; ICARS: r = 0.525, p = 0.021). The sNfL concentration was also positively correlated with the ICARS subgroup, including speech disorders (r = 0.674, p = 0.002) and limb kinetic function (r = 0.526, p = 0.021).
Associations of sNFL Levels and Brain Metabolite Levels
In all SCA3 patients, increased sNfL levels were associated with decreased cerebellar hemisphere, cerebellar vermis, and dentate nucleus NAA/Cr and NAA/Cho levels when age was included as a covariate. Higher sNfL levels were correlated with lower NAA/Cr ratios in the dentate nucleus (r = − 0.482, p = 0.036), cerebellar vermis (r = − 0.521, p = 0.022), and cerebellar hemisphere (r = − 582, p = 0.009). Higher sNfL values did not correlate with the levels of NAA/Cho in the dentate nucleus (r = − 0.361, p = 0.129) but were negatively correlated with the NAA/Cho levels in the cerebellar vermis (r = − 0.599, p = 0.007) and cerebellar hemisphere (r = − 0.677, p = 0.001) (Fig. 2). No significant correlations between the Cho/Cr ratios in the three ROIs and sNfL emerged. By Bonferroni's adjustment (p < 0.016), higher sNfL levels were still significantly associated with lower NAA/Cho levels in the cerebellar hemisphere and cerebellar vermis. Moreover, sNfL levels correlated with lower NAA/Cr levels in the cerebellar hemisphere, whereas they showed no significant correlation in the dentate nucleus and cerebellar vermis region. No significant association was found between sNfL levels and brain metabolite levels (NAA/Cr, Cho/Cr, and NAA/Cho ratios) of three ROIs in the normal controls by either the Bonferroni-corrected threshold of p < 0.016 or the uncorrected threshold (Table 2).
Associations of sNfL levels with brain metabolite levels. (A–C) Higher sNfL levels were correlated with lower NAA/Cr ratios in the cerebellar cortex, dentate nucleus, and cerebellar vermis. (D–F) No significant correlations between the Cho/Cr ratios in the three ROIs and sNfL emerged. (G–H) Higher sNfL levels were correlated with lower NAA/Cho ratios in the cerebellar cortex and cerebellar vermis. (I) Higher sNfL values did not correlate with the levels of NAA/Cho in the dentate nucleus. NAA, N-acetyl aspartate; Cr, creatine; Cho, choline complex; sNfL, serum neurofilament light chain; NC, normal controls; SCA3, Spinocerebellar ataxia type 3 patients
Mediation Model of the Effect of MRS Metabolite Values/Changes on Ataxia Severity
We evaluated whether cerebellar metabolite levels in SCA3 patients were a moderator of the mediation of the relationship between sNfL levels and ataxia symptoms. We used two separate models with either the NAA/Cr ratio or NAA/Cho ratio as the mediator. The analysis revealed a significant NAA/Cr ratios-moderated effect of sNfL levels predicting ataxia severity (Model 1a predicted sNfL levels; Model 1b predicted ICARS scores). While the direct effect (sNfL levels ⟶ ICARS scores) was not significant, the indirect effect (sNfL levels ⟶ NAA/Cr ratios ⟶ ICARS scores) was statistically significant for the cerebellar cortex of SCA3 participants (Table 3). The pathways summarize the relationships found between the variables (Fig. 3). We also tested whether the NAA/Cho ratios in the cerebellar cortex were mediators, and no significance was identified (Supplemental Table 2; Supplemental Fig. 2). Additionally, the metabolite levels, including the NAA/Cr and NAA/Cho ratios, in the dentate nucleus and cerebellar vermis were also tested, and no significant differences were found in our cohort (Supplemental Tables 3, 4, 5 and 6).
Mediation model of the effect of NAA/Cr rations in cerebellum on the ICARS scores, with age as a covariate. (a–f) Unstandardized regression coefficients for each path; statistically significant pathways are shown in bold and marked with solid lines. NAA/Cr, N-acetyl aspartate/creatine; sNfL, neurofilament light chain; ICARS, International Cooperative Ataxia Rating Scale
Discussion
In the current study, the sNfL and brain metabolite levels of a cohort of 20 SCA3 patients were evaluated to examine the associations between the two measurements. Compared to the control individuals, we found higher sNfL levels and lower cerebellar metabolite levels, including NAA/Cr and NAA/Cho ratios, in the SCA3 patients. Both measures, cerebellar metabolite and sNfL levels, are correlated with the disease severity of SCA3 patients. In addition, elevated sNfL levels were associated with lower cerebellar metabolite levels; in particular, low NAA/Cr values are known to indicate cerebellar degeneration in ATXN3 repeat expansion carriers. As the correlations of sNfL levels and cerebellar metabolites capture abnormalities in SCA3 patients, they may serve as complementary biomarkers for disease detection and future treatment monitoring.
In SCA3, polyQ-expanded ATXN3 protein leads to neurotoxicity and causes selective neuronal cell death along with metabolic/cellular changes detectable by MRS, including decreases in acetylcholine and NAA [5]. The NAA/Cr ratio has been found to be a good neuroimaging biomarker and can be used to monitor the clinical progression of SCA3 [4, 20]. Our findings demonstrated decreased NAA/Cr and NAA/Cho ratios in the dentate nucleus, cerebellar vermis, and cerebellar hemisphere, suggesting that the degree of neuronal dysfunction in SCA3 patients would be consistent with previous findings [5, 21]. Associations between the severity of ataxia and metabolic/cellular changes have been described as reflected by NAA/Cr ratios. Our findings also showed that ICARS scores were negatively correlated with NAA/Cr ratios in the three ROIs of the dentate nucleus, cerebellar vermis, and cerebellar hemisphere in 20 SCA3 patients, which is in line with previous results [5, 22]. Furthermore, we found that SARA scores had an inverse relationship with NAA/Cr ratios in these ROIs. These results further confirmed that the metabolic alterations in the cerebellum were correlated with the predominant clinical features in SCA3 patients and suggested that NAA is a good marker for SCA3 [4, 5].
Neurofilament light protein can be released in significant quantities following neuro-axonal damage or neurodegeneration. Elevated sNfL levels are considered a biomarker of clinical disease severity in SCA3 [13, 23, 24]. Higher sNfL levels not only correlate with ataxic symptoms and non-ataxic signs in SCA3 patients with clinically observable ataxia but also show similar correlations in preclinical SCA3 individuals [14, 24]. Our findings were consistent with a previously published study on SCA3, which also found that sNfL was elevated in patients with SCA3 and that sNfL levels correlated with ataxia symptoms [14]. This correlation between elevated sNfL levels and clinical severity persisted after accounting for the potential confounder of age.
In our patients with SCA3, both higher sNfL levels and decreased metabolites in the cerebellum were significantly correlated with clinical severity, as reflected by the ICARS scores. Therefore, we hypothesized that the elevation of sNfL values might be correlated with a lower cerebellar NAA/Cr ratios. Our study also explored the association between sNfL and MRI features in a subgroup of ataxic SCA3 patients. First, the ROI analysis showed that certain cerebellar regions with a lower NAA/Cr ratio tended to be significantly associated with higher sNfL levels. These included regions in the dentate nucleus, cerebellar vermis, and cerebellar hemisphere. Furthermore, higher sNfL levels were also associated with a reduced NAA/Cho ratio in the cerebellar vermis and cerebellar hemisphere, but a similar association was not found in the dentate nucleus in our analyses. Third, we unexpectedly failed to find a significant relationship between increased sNfL and the Cho/Cr ratio in the cerebellum. One potential explanation for this is that the Cho/Cr ratio was not observed to decrease in our patients. This finding suggests that the elevation of sNfL was mainly associated with neuronal dysfunction rather than cell membrane or neurotransmitter metabolism impairment in our SCA3 patients, likely indicating the subtle impairment of neurofilaments in the cerebellum. Regarding these findings, further studies are needed to explore the association between the NAA/Cr ratio and sNfL levels in SCA3.
Both higher sNfL levels and decreased cerebellar metabolites have been suggested to reflect neuronal dysfunction not only in SCA3 but also in other degenerative diseases [25,26,27,28]. In our study, we found both measures, NAA/Cr ratio and sNfL levels, to be significant predictors of clinical severity, which was reflected by ICARS scores. However, it is difficult to estimate the relevance of sNfL levels because changes in these levels may result from shifts in the NAA/Cr or NAA/Cho ratios or from both metabolites simultaneously. In this study, a mediation model based on a multiple regression approach helped clarify how prominent the contribution of each component was. Our results indicated that the prediction of clinical severity according to sNfL levels is more likely to be driven by changes in the NAA/Cr ratio but not by changes in the NAA/Cho ratio. sNfL levels and cerebellar metabolites of the NAA/Cr ratio are both biomarkers of neuro-axonal damage in patients with SCA3. Serum sNfL levels often reflect neuronal damage in the whole brain, while the NAA/Cr ratio reflects damage in part of the cerebellum in SCA3, which may explain why the NAA/Cr ratio acts as a mediator.
Several potential limitations restrict the interpretability of our findings. First, the limited sample size was not sufficient to draw an affirmative conclusion; thus, the findings need to be further validated using a larger cohort. Second, information on asymptomatic SCA3 patients was not available, making it difficult to analyze the correlation between cerebellar metabolites and sNfL levels. Third, not only should the neurochemical values of NAA, Cho, and Cr be included, but glutamine (Gln), glutamate (Glu), and Tau should also be determined, as this could help to further explore their correlation with sNfL in future research. Finally, while the mediated moderation analysis in our present study provides evidence of an associated effect, we need to acknowledge that the effects were only significant for ICARS (but not SARA) scores and need to be replicated in future research.
In summary, we used a multimodal approach combining serum biomarker and cerebellar metabolite levels to investigate the association between sNfL levels and indicators of neurodegeneration in 20 SCA3 patients. To the best of our knowledge, this is the first study to examine whether sNfL concentrations correlate with cerebellar metabolites as reflected by the NAA/Cr ratios in ATXN3 mutation carriers. Our results confirmed that sNfL levels are increased in SCA3 patients and that they are correlated with clinical disease severity, reflected as ICARS and SARA scores, and brain metabolic changes. We demonstrated that sNfL and cerebellar metabolites are promising complementary biomarkers that can capture the effects of the expansion of the ATXN3 mutation. In addition, our multiple regression models revealed that a decrease in the NAA/Cr ratio mediated this predictive relationship. Additional longitudinal studies are strongly warranted to build on these results in the future.
Data Availability
Data available upon request to the authors.
References
Kawaguchi Y, Okamoto T, Taniwaki M, Aizawa M, Inoue M, et al. CAG expansions in a novel gene for Machado-Joseph disease at chromosome 14q321. Nature genetics. 1994;8:221–8.
Maciel P, Gaspar C, DeStefano AL, Silveira I, Coutinho P, et al. Correlation between CAG repeat length and clinical features in Machado-Joseph disease. Am J Hum Genet. 1995;57:54–61.
Yuan X, Ou R, Hou Y, Chen X, Cao B, et al. Extra-cerebellar signs and non-motor features in chinese patients with spinocerebellar ataxia type 3. Front Neurol. 2019;10:110. https://doi.org/10.3389/fneur.2019.00110.
Wang P-S, Chen H-C, Wu H-M, Lirng J-F, Wu Y-T, Soong B-W. Association between proton magnetic resonance spectroscopy measurements and CAG repeat number in patients with spinocerebellar ataxias 2, 3, or 6. PLoS One. 2012;7:e47479. https://doi.org/10.1371/journal.pone.0047479.
Peng H, Liang X, Long Z, Chen Z, Shi Y, et al. Gene-related cerebellar neurodegeneration in SCA3/MJD: a case-controlled imaging-genetic study. Front Neurol. 2019;10:1025. https://doi.org/10.3389/fneur.2019.01025.
Chen H-C, Lirng J-F, Soong B-W, Guo WY, Wu H-M, et al. The merit of proton magnetic resonance spectroscopy in the longitudinal assessment of spinocerebellar ataxias and multiple system atrophy-cerebellar type. Cerebellum Ataxias. 2014;1:17. https://doi.org/10.1186/s40673-014-0017-4.
Tsai G, Coyle JT. N-acetylaspartate in neuropsychiatric disorders. Prog Neurobiol. 1995;46:531–40.
Oppenheimer SM, Bryan RN, Conturo TE, Soher BJ, Preziosi TJ, Barker PB. Proton magnetic resonance spectroscopy and gadolinium-DTPA perfusion imaging of asymptomatic MRI white matter lesions. Magn Reson Med. 1995;33:61–8.
McLoughlin HS, Moore LR, Chopra R, Komlo R, McKenzie M, et al. Oligonucleotide therapy mitigates disease in spinocerebellar ataxia type 3 mice. Ann Neurol. 2018;84:64–77. https://doi.org/10.1002/ana.25264.
Hauser S, Helm J, Kraft M, Korneck M, Hübener-Schmid J and Schöls L. Allele-specific targeting of mutant ataxin-3 by antisense oligonucleotides in SCA3-iPSC-derived neurons. Mol Ther Nucleic Acids. 2022; 27. https://doi.org/10.1016/j.omtn.2021.11.015.
Costa MdC, Radzwion M, McLoughlin HS, Ashraf NS, Fischer S, et al. In vivo molecular signatures of cerebellar pathology in spinocerebellar ataxia type 3. Mov Disord: official journal of the Movement Disorder Society. 2020;35:1774–86. https://doi.org/10.1002/mds.28140.
He L, Wang S, Peng L, Zhao H, Li S, et al. CRISPR/Cas9 mediated gene correction ameliorates abnormal phenotypes in spinocerebellar ataxia type 3 patient-derived induced pluripotent stem cells. Transl Psychiatry. 2021;11:479. https://doi.org/10.1038/s41398-021-01605-2.
Li Q-F, Dong Y, Yang L, Xie J-J, Ma Y, et al. Neurofilament light chain is a promising serum biomarker in spinocerebellar ataxia type 3. Mol Neurodegener. 2019;14:39. https://doi.org/10.1186/s13024-019-0338-0.
Peng Y, Zhang Y, Chen Z, Peng H, Wan N, et al. Association of serum neurofilament light and disease severity in patients with spinocerebellar ataxia type 3. Neurol. 2020;95:e2977–87. https://doi.org/10.1212/WNL.0000000000010671.
Garcia-Moreno H, Prudencio M, Thomas-Black G, Solanky N, Jansen-West KR, et al. Tau and neurofilament light-chain as fluid biomarkers in spinocerebellar ataxia type 3. Eur J Neurol. 2022. https://doi.org/10.1111/ene.15373
Wilke C, Bender F, Hayer SN, Brockmann K, Schöls L, et al. Serum neurofilament light is increased in multiple system atrophy of cerebellar type and in repeat-expansion spinocerebellar ataxias: a pilot study. J Neurol. 2018;265:1618–24. https://doi.org/10.1007/s00415-018-8893-9.
Krahe J, Binkofski F, Schulz JB, Reetz K, Romanzetti S. Neurochemical profiles in hereditary ataxias: a meta-analysis of magnetic resonance spectroscopy studies. Neurosci Biobehav Rev. 2020;108:854–65. https://doi.org/10.1016/j.neubiorev.2019.12.019.
Rohrer JD, Woollacott IOC, Dick KM, Brotherhood E, Gordon E, et al. Serum neurofilament light chain protein is a measure of disease intensity in frontotemporal dementia. Neurol. 2016;87:1329–36. https://doi.org/10.1212/WNL.0000000000003154.
Hayes AF, Rockwood NJ. Regression-based statistical mediation and moderation analysis in clinical research: observations, recommendations, and implementation. Behav Res Ther. 2017;98:39–57. https://doi.org/10.1016/j.brat.2016.11.001.
Adanyeguh IM, Henry P-G, Nguyen TM, Rinaldi D, Jauffret C, et al. In vivo neurometabolic profiling in patients with spinocerebellar ataxia types 1, 2, 3, and 7. Mov Disord: official journal of the Movement Disorder Society. 2015;30:662–70. https://doi.org/10.1002/mds.26181.
Huang S-R, Wu Y-T, Jao C-W, Soong B-W, Lirng J-F, et al. CAG repeat length does not associate with the rate of cerebellar degeneration in spinocerebellar ataxia type 3. Neuroimage Clin. 2017;13:97–105. https://doi.org/10.1016/j.nicl.2016.11.007.
Lirng J-F, Wang P-S, Chen H-C, Soong B-W, Guo WY, et al. Differences between spinocerebellar ataxias and multiple system atrophy-cerebellar type on proton magnetic resonance spectroscopy. PLoS One. 2012;7:e47925. https://doi.org/10.1371/journal.pone.0047925.
Coarelli G, Darios F, Petit E, Dorgham K, Adanyeguh I, et al. Plasma neurofilament light chain predicts cerebellar atrophy and clinical progression in spinocerebellar ataxia. Neurobiol Dis. 2021;153:105311. https://doi.org/10.1016/j.nbd.2021.105311.
Wilke C, Haas E, Reetz K, Faber J, Garcia-Moreno H, et al. Neurofilaments in spinocerebellar ataxia type 3: blood biomarkers at the preataxic and ataxic stage in humans and mice. EMBO Mol Med. 2020;12:e11803. https://doi.org/10.15252/emmm.201911803.
Illán-Gala I, Lleo A, Karydas A, Staffaroni AM, Zetterberg H, et al. Plasma Tau and neurofilament light in frontotemporal lobar degeneration and Alzheimer disease. Neurol. 2021;96:e671–83. https://doi.org/10.1212/WNL.0000000000011226.
van der Ende EL, Meeter LH, Poos JM, Panman JL, Jiskoot LC, et al. Serum neurofilament light chain in genetic frontotemporal dementia: a longitudinal, multicentre cohort study. Lancet Neurol. 2019;18:1103–11. https://doi.org/10.1016/S1474-4422(19)30354-0.
Elahi FM, Casaletto KB, La Joie R, Walters SM, Harvey D, et al. Plasma biomarkers of astrocytic and neuronal dysfunction in early- and late-onset Alzheimer’s disease. Alzheimers Dement. 2020;16:681–95. https://doi.org/10.1016/j.jalz.2019.09.004.
Aamodt WW, Waligorska T, Shen J, Tropea TF, Siderowf A, et al. Neurofilament light chain as a biomarker for cognitive decline in Parkinson disease. Mov Disord: official journal of the Movement Disorder Society. 2021;36:2945–50. https://doi.org/10.1002/mds.28779.
Acknowledgements
The local medical ethics committee of the affiliated hospital of Hangzhou normal university approved the study, and written informed consent was obtained from all participants.
Funding
This work was supported by the grant, 2021KY898 (YCC), from the Medical and Health Science and Technology Project of Zhejiang Province.
Author information
Authors and Affiliations
Contributions
Y.C. Chen designed the work. Y.C. Chen, Y. Jin, Z.Y. Hu, M.Q. Qiu, D. Li, Q.S. Cai, C.J. Tao, D.N. Lou, L. Qi, S.D. Chen, H. Yu, and Z.M. Gao initiated the project. Y.C. Chen, Y. Jin, Z.Y. Hu, M.Q. Qiu, D. Li, Q.S. Cai, C.J. Tao, D.N. Lou, L. Qi, S.D. Chen, H. Yu, and Z.M. Gao collected and analyzed the data. Y.C. Chen wrote the manuscript. H. Yu and Z.M. Gao commented and revised on the manuscript. Y.C. Chen supervised all aspects of the project. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Hao Yu and Zhongming Gao shared last authorship.
Supplementary Information
Below is the link to the electronic supplementary material.


Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, Y., Jin, Y., Hu, Z. et al. Association Between Serum Neurofilament Light Chain and Neurochemistry Deficits in Patients with Spinocerebellar Ataxia Type 3. Cerebellum 23, 92–100 (2024). https://doi.org/10.1007/s12311-022-01507-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-022-01507-z