Abstract
This study aimed to explore the role of the cerebellum on the somatosensory temporal discrimination (STD) process. Twenty healthy volunteers were enrolled in the study. Each participant was included in three different transcranial direct current stimulation (tDCS) sessions as anodal, cathodal, and sham sessions in randomised order. Anodal and cathodal cerebellar tDCS (cTDCS) were given for 20 min at an intensity of 2 mA. The results of the study indicate no effect of cTDCS on STD. Although the study results show that cTDCS does not affect STD, because of the restricted sample size of the study, it is useful and necessary to investigate this relationship more in depth in a larger healthy subject population using different cTDCS methodologies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Almost a century ago, in 1917, Gordon Holmes reported finding no abnormalities of any sensory modalities in his case series of soldiers with serious head injuries and cerebellum damage during World War I [1]. Many researchers and clinicians subsequently confirmed this critical observation, which led to the underestimation of the effect of the cerebellum on the somatosensory system for many years. However, some electrophysiological findings were obtained from patients with cerebellar damage [2,3,4], and the analysis of the effect of cerebellar repetitive Transcranial Magnetic Stimulation (rTMS) on sensory processing in normal individuals [5,6,7] gradually revealed the role of the cerebellum in processing somatosensory information.
The Somatosensory Temporal Discrimination Threshold (STDT) describes the shortest time interval for perceiving a pair of tactile stimuli to be perceived as separate. The findings obtained from a functional magnetic resonance imaging study [8] show that the prefrontal cortex, the right postcentral gyrus, the inferior parietal lobule, the basal ganglia, and the cerebellum are active during the Somatosensory Temporal Discrimination (STD) task. Since precise timing is crucial, not only for cognition but also for motor behaviour, it is not a surprise to detect STD abnormalities in movement disorders related to basal ganglia and associated cortical area involvement. Indeed, impaired STDT has been demonstrated in detail in Parkinson’s disease and adult-onset focal dystonia (for a review, see [9] and [10] respectively). Increased STD has also been demonstrated in patients with cerebellar atrophy [2].
This study aimed to explore the role of the cerebellum on the STD process. In compliance with this, the excitability of the cerebellum was changed by using cerebellar transcranial direct current stimulation (cTDCS), and it was investigated whether there was any STDT change as a result. Here, we hypothesise that increased cerebellar activity with anodal cTDCS will reduce STDT while cathodal cTDCS results in increased STDT.
Method
Participants
Twenty healthy volunteers (10 males, aged between 26 and 54 years of age) without systemic and neurological disease were the subjects. All participants provided written, informed consent approved by the local Research Ethics Committee.
STD procedure
Before the placement of the Ag–AgCl skin electrodes (10-mm diameter surface), the skin was cleaned with alcohol. The electrodes were placed on the dorsum of the right hand at a distance from each other of 1 cm, and the anode was placed distal to the cathode. The square-wave electrical pulses of 0.2 ms duration were given using a constant current stimulator. The stimulation started from 1 mA in intensity and increased in 0.2 mA steps. When the stimulation was felt, it was repeated three times and recorded as the minimal sensory threshold if the stimulus was perceived consistently. After the sensory threshold was determined, a stimulation intensity 1.5 times higher than the threshold was calculated, and electrical stimulation was applied at this intensity throughout the study. The paired stimuli started at 5 ms interstimulus intervals (ISI) and increased in 5 ms steps. The interval between the stimuli was 10–15 s. The first of three consecutive ISIs where the participants perceived two temporally distinct stimuli was recorded as the ascending STDT (aSTDT). Paired stimuli were then delivered to the subjects, decreasing in 5 ms steps until the subjects recognised two separate stimuli as one stimulus three times consecutively. The first ISI value at which the participants perceived two distinct stimuli as one stimulus three times sequentially was recorded as the descending STDT (dSTDT). The arithmetic mean of aSTDT and dSTDT was calculated as a mean STDT value. The subject’s compliance was checked according to whether the subject perceived random single stimuli during the double stimulation paradigm.
A Nihon Kohden EMG device (Neuropack MEB-5504 k, Japan) was used for electrical stimulation. The triggering of the stimulus and the setting of the ISIs were arranged using CED Micro 1401 Data Acquisition Unit (CED1401, UK) and signal software (Signal V4.0, CED, UK). The same researcher (EEA, IA, or ET) performed three different STD measurements for the same subject.
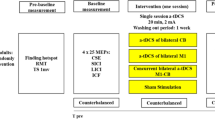
cTDCS
The cerebellum’s modulation with weak cTDCS was performed using electrodes placed between two sponges soaked in saline solution. The active electrode was 5 × 5 cm (current density: 0.08 mA/cm2), whereas the reference electrode was 9 × 5 cm (current density: 0.044 mA/cm2). The active electrode was placed 1.5 cm below and 3 cm lateral to the right inion, while the reference electrode was located on the buccal muscle. Anodal, cathodal, and sham cTDCS were used. Anodal and cathodal cTDCS were given for 20 min at an intensity of 2 mA, with a fade-in and fade-out of 20 s. Sham cTDCS was delivered identically; however, the current intensity was decreased to 0 mA after 30 s. The Soterix Medical 1 × 1 transcranial direct current stimulation (tDCS) Device (Soterix Medical Inc., New York, NY, USA) was used for cTDCS. Each participant was included in three different tDCS sessions as anodal, cathodal, and sham sessions with a randomised order, at least 3 days apart. STDT measurements were made before and immediately after cTDCS. The different researchers applied STD and tDCS procedures; the subject was unaware of what cTDCS protocol was applied and the researcher performing the STDT measurements was also blind to the applied cTDCS condition.
Statistical analysis
The mean and the standard deviation were calculated for each measurement. Data distribution of individual means was established using the Shapiro–Wilk test. The effect size was calculated by Cohen’s d value based on two groups with the same group sizes:
where:\(SD\text{pooled= }\sqrt{\text{((}{SD}_{1}^{2}\text{+}{SD}_{2}^{2}\text{)/2)}}\)
Equivalence tests were performed using the TOSTER package in the open-source programming language R.
The repeated measure ANOVA test was performed to detect the cTDCS effect on STDT. Pearson correlation analysis was used to analyse the correlation between variables. The results were considered significant when P < 0.05. The R Version 4.0.2 (R Foundation for Statistical Computing) program was used for statistical analysis.
Results
The Cohen’s d values of the cathodal, anodal, and sham sessions were 0.008, 0.031, and 0.022 respectively. Based on the Cohen’s benchmarks (small (d = 0.2), medium (d = 0.5), and large (d = 0.8)) [11], the results indicate the small effect of cTDCS on STDT across three sessions.
The equivalence test was non-significant for the cathodal (t(38) = 0.124, p = 0.549), anodal (t(38) = 0.99, p = 0.5), and sham (t(38) = − 0.005, p = 0.502) sessions.
All but one participant attended all three sessions, with none reporting an issue that would require ending the session. One participant dropped out of the study without attending the sham cTDCS session.
STD normative data
The baseline STD values obtained before the cTDCS procedure from the three different sessions are shown in Fig. 1. There were no significant differences between the three baseline STD measurements (p > 0.05). Moreover, there was a high correlation between the three baseline STD values (Fig. 2).
Correlogram of the three baseline STDT measurements. S1, S2, and S3 represent three different pre-cTDCS (anodal, cathodal, and sham) STDT measurements of the subjects, in randomised order. The histograms show the distribution of each variable. Correlation coefficients showed correlations between three measures (S1, S2, and S3). Each significance level is associated with a symbol: p-values (0, 0.001, 0.01) < = > symbols (“***”, “**”)
The mean STDT values from the three different cTDCS sessions are shown in Fig. 3.
A repeated-measures 2-way ANOVA on the combined data with factors “STIMULUS POLARITY (anodal × cathodal × sham)” and “TIME(before × after cTDCS)” showed that neither STIMULUS POLARITY F(2,112) = 0.25, p = 0.778, nor TIME f(1,112) = 0.00, p = 0.987 had a significant effect on STDT. There was no significant STIMULUS POLARITY × TIME interaction (p > 0.05).
Discussion
The results of the study suggest that STD is a measure of sensory processing with good repeatability. Indeed, the three mean baseline STD values obtained from the three different operators were very close to each other and strongly correlated.
As a model, Pavlovian eyelid responses are a useful and straightforward system for investigating the role of the cerebellum in time perception. In this paradigm, the conditioned stimulus (CS), which does not cause eyeblink response, is paired with eyeblink — eliciting unconditioned stimulus (US) with a specific time interval. After paired stimulus train, CS produced an eyeblink response alone. The evidence implies that classical conditioning of eyeblink response forms in the interpositus nucleus of the cerebellum [12]. While the intermediate cerebellum regulates the motor aspect of timing, the lateral cerebellar hemispheres regulate the cognitive or perceptual components of timing along with the frontoparietal association cortex connections. Indeed, rTMS studies have presented findings suggesting that the lateral cerebellum plays a role in time processing. Two different studies in which lateral cerebellum excitability changes with 1 Hz rTMS reveal that it impairs time perception at long ISIs (hundreds of milliseconds) after rTMS [7, 13]. Although these studies suggest that the lateral cerebellum may play a role in STD, one point that should not be overlooked is that the ISIs in these studies were longer (several hundred milliseconds) than normative STD values. Another study in which continuous theta burst stimulation (cTBS) inhibited the lateral cerebellum showed that cerebellar modulation did not affect STDT values [14].
This study’s result, which falsified our hypothesis, is that cTDCS does not affect STDT. Although this result does not demonstrate the effect of cTDCS on STDT, we think it is necessary to be cautious about concluding that the cerebellum does not affect STD based on this finding. Considering the STD abnormalities detected in patients with cerebellar involvement [2], a possibility that should be considered is the inability to stimulate/modulate the cerebellar structures involved in STD with rTMS [14] or cTDCS. Although it is accepted that cTDCS changes cerebellum excitability depending on polarity, the findings that have been obtained by studies focusing on this topic are not very consistent. In particular, it has been shown [15] that the cerebello-brain inhibition (CBI) obtained by stimulating the cerebellum and motor cortex (M1) with a specific time interval using TMS increases with anodal cTDCS and decreases with cathodal cTDCS. However, later studies make inconsistent findings, such as reduced CBI with anodal cTDCS [16] or decreased CBI after both anodal and cathodal cTDCS [17] were reported. Heterogeneous effects of cTDCS on different nervous system domains, such as motor functions and cognitive functions, have also been observed (for a review, see [18]). Inconsistent findings may be related to cTDCS protocol differences such as stimulation intensity, stimulation duration, and electrode montage. The results of the equivalence test in this study indicate it would be better to study a larger population to analyse the effect of cTDCS on STDT. The limited number of recruited subjects is the major limitation of the study.
In conclusion, although our results show that cTDCS does not affect STD, we think it is useful and necessary to investigate this relationship more in depth in a larger healthy subject population using different cTDCS methodologies (such as various electrode montages, longer stimulation duration, and online analysis).
References
Gordon H. The symptoms of acute cerebellar injuries due to gunshot injuries. Brain. 1917;40:461–535. https://doi.org/10.1093/brain/40.4.461.
Manganelli F, Dubbioso R, Pisciotta C, Antenora A, Nolano M, De Michele G, Filla A, Berardelli A, Santoro L. Somatosensory temporal discrimination threshold is increased in patients with cerebellar atrophy. Cerebellum. 2013;12:456–9. https://doi.org/10.1007/s12311-012-0435-x.
Restuccia D, Della Marca G, Valeriani M, Leggio MG, Molinari M. Cerebellar damage impairs detection of somatosensory input changes. A somatosensory mismatch-negativity study. Brain. 2007;130:276–87. https://doi.org/10.1093/brain/awl236.
Restuccia D, Valeriani M, Barba C, Le Pera D, Capecci M, Filippini V, Molinari M. Functional changes of the primary somatosensory cortex in patients with unilateral cerebellar lesions. Brain. 2001;124:757–68. https://doi.org/10.1093/brain/124.4.757.
Cattaneo Z, Renzi C, Casali S, Silvanto J, Vecchi T, Papagno C, D’Angelo E. Cerebellar vermis plays a causal role in visual motion discrimination. Cortex. 2014;58:272–80. https://doi.org/10.1016/j.cortex.2014.01.012.
Grube M, Lee KH, Griffiths TD, Barker AT, Woodruff PW. Transcranial magnetic theta-burst stimulation of the human cerebellum distinguishes absolute, duration-based from relative, beat-based perception of subsecond time intervals. Front Psychol. 2010;1:171. https://doi.org/10.3389/fpsyg.2010.00171.
Lee KH, Egleston PN, Brown WH, Gregory AN, Barker AT, Woodruff PW. The role of the cerebellum in subsecond time perception: evidence from repetitive transcranial magnetic stimulation. J Cogn Neurosci. 2007;19:147–57. https://doi.org/10.1162/jocn.2007.19.1.147.
Pastor MA, Day BL, Macaluso E, Friston KJ, Frackowiak RS. The functional neuroanatomy of temporal discrimination. J Neurosci. 2004;24:2585–91. https://doi.org/10.1523/JNEUROSCI.4210-03.2004.
Conte A, McGovern EM, Narasimham S, Beck R, Killian O, O’Riordan S, Reilly RB, Hutchinson M. Temporal discrimination: mechanisms and relevance to adult-onset dystonia. Front Neurol. 2017;8:625. https://doi.org/10.3389/fneur.2017.00625.
Lee MS, Lee MJ, Conte A, Berardelli A. Abnormal somatosensory temporal discrimination in Parkinson’s disease: pathophysiological correlates and role in motor control deficits. Clin Neurophysiol. 2018;129:442–7. https://doi.org/10.1016/j.clinph.2017.11.022.
Cohen J. Statistical Power Analysis for the Behavioral Sciences. New York: Routledge Academic; 1988.
Hu B, Chen H, Feng H, Zeng Y, Yang L, Fan ZL, Wu YM, Sui JF. Disrupted topography of the acquired trace-conditioned eyeblink responses in guinea pigs after suppression of cerebellar cortical inhibition to the interpositus nucleus. Brain Res. 2010;1337:41–55. https://doi.org/10.1016/j.brainres.2010.03.089.
Koch G, Oliveri M, Torriero S, Salerno S, Lo Gerfo E, Caltagirone C. Repetitive TMS of cerebellum interferes with millisecond time processing. Exp Brain Res. 2007;179:291–9. https://doi.org/10.1007/s00221-006-0791-1.
Conte A, Rocchi L, Nardella A, Dispenza S, Scontrini A, Khan N, Berardelli A. Theta-burst stimulation-induced plasticity over primary somatosensory cortex changes somatosensory temporal discrimination in healthy humans. PLoS One. 2012;7:e32979. https://doi.org/10.1371/journal.pone.0032979.
Galea JM, Jayaram G, Ajagbe L, Celnik P. Modulation of cerebellar excitability by polarity-specific noninvasive direct current stimulation. J Neurosci. 2009;29:9115–22. https://doi.org/10.1523/JNEUROSCI.2184-09.2009.
Doeltgen SH, Young J, Bradnam LV. Anodal direct current stimulation of the cerebellum reduces cerebellar brain inhibition but does not influence afferent input from the hand or face in healthy adults. Cerebellum. 2016;15:466–74. https://doi.org/10.1007/s12311-015-0713-5.
Batsikadze G, Rezaee Z, Chang DI, Gerwig M, Herlitze S, Dutta A, Nitsche MA, Timmann D. Effects of cerebellar transcranial direct current stimulation on cerebellar-brain inhibition in humans: a systematic evaluation. Brain Stimul. 2019;12:1177–86. https://doi.org/10.1016/j.brs.2019.04.010.
van Dun K, Bodranghien F, Manto M, Marien P. Targeting the cerebellum by noninvasive neurostimulation: a review. Cerebellum. 2017;16:695–741. https://doi.org/10.1007/s12311-016-0840-7.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ataoğlu, E.E., Türksoy, E., Aslan, İ. et al. Cerebellar Transcranial Direct Current Stimulation Does Not Alter Somatosensory Temporal Discrimination Threshold. Cerebellum 21, 920–925 (2022). https://doi.org/10.1007/s12311-021-01340-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-021-01340-w