Abstract
Follicular lymphoma is a hematolymphoid neoplasm that originates from germinal center B cells. It is made up of a combination of small cleaved centrocytes and a varying quantity of larger non-cleaved centroblasts to describe the clinical, microscopic, immunohistochemical, and molecular features of oral follicular lymphomas. Follicular lymphomas affecting the oral cavity were retrieved from pathology files. Immunohistochemistry was performed to confirm the diagnosis, and fluorescence in situ hybridization (FISH) was employed to detect rearrangements in BCL2, BCL6, and MYC genes. Clinical and follow-up data were obtained from the patient’s medical and pathology files. Twenty cases were obtained. There was an equal sex distribution (10 males: 10 females) and a mean age of 60.9 years (range: 10–83 years-old). Lesions presented as asymptomatic swellings, usually in the palate (10 cases) and the buccal mucosa (7 cases). Five patients presented with concomitant nodal involvement. Microscopic evaluation depicted the follicular growth pattern with diffuse areas in six cases. Grades 1 and 2 follicular lymphomas represented 12 cases, while grade 3A neoplasms accounted for other 8 cases. Two cases showed rearrangements in MYC, BCL2, and BCL6 genes, while single BCL2 translocation was found in eight cases. Two cases had no translocation. Three patients deceased and the 2-year overall survival achieved 88%. Follicular lymphoma affecting the oral cavity is uncommon, usually affects the palate as a non-ulcerated swelling and the presence of a systemic disease most always be ruled out.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Follicular lymphoma (FL) is a hematolymphoid neoplasm derived from germinal center B cells consisting of a mixture of small-sized cleaved centrocytes and a variable amount of large-sized non-cleaved centroblasts [1,2,3,4]. The incidence of FLs is higher in Western countries, with 2 to 4 cases per 100,000 people in the USA and Europe, representing 20–25% of all non-Hodgkin lymphomas (NHL) [1, 5]. FL is less commonly seen in Asian countries and Brazil, accounting for 10 to 20% of all cases diagnosed in Central and South Americas [6, 7].
The pathogenesis of this neoplasm centers around the translocation t(14;18)(q32; q21), leading to the IGH/BCL2 fusion, resulting in the overexpression of the anti-apoptotic protein Bcl2. This translocation is observed in approximately 90% of FLs, but its presence can be as low as 50 to 75% in high-grade cases [4, 8, 9]. Moreover, unusual cases may exhibit translocations in MYC, BCL2, and/or BCL6 genes. However, since the immune morphology is consistent with FL, these cases are not reclassified as diffuse large B-cell lymphoma/high-grade B-cell lymphoma with MYC and BCL2 rearrangements (DLBCL/HGBL-MYC/BCL2), but are rather called double/triple-hit FL [10,11,12,13]. Although rare, recognition of these cases is important to refine the biological heterogeneity of FL, and also because they may exhibit poorer outcomes [14, 15].
FLs most commonly affect the lymph nodes, possibly developing extranodal disseminations, although primary extranodal cases may also be diagnosed [16]. In addition to the classic/systemic disease, several other variants of follicular lymphoma that carry specific clinical presentations, biological behavior, and/or microscopic features are recognized by the WHO classification of lymphoid tumors and the International Consensus Classification (ICC) position papers, and should be excluded in cases affecting the oral cavity [10, 12, 13].
In the head and neck, Waldeyer’s ring and salivary glands are the most affected extranodal sites [17], and FL represents 7.2% of all B-cell neoplasms in the oral cavity [18]. However, most cases affecting this subsite are reported in the literature as single case reports or included in larger head and neck series that also include nodal diseases. Therefore, detailed analyses of cases affecting the oral cavity are limited. This study aimed to investigate the clinical, microscopic, and molecular characteristics of FL affecting the oral cavity.
Material and methods
Ethics statement
This study was approved by the Ethics Committee of the Piracicaba Dental School, University of Campinas, Piracicaba, Brazil (protocol no. 67128417.4.0000.5418) and the Ethics Committee of the Federal University of Minas Gerais, Belo Horizonte, Brazil (protocol no. 10723019.0.1001.514). All procedures were in accordance with the ethical standards of the responsible committees on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
Study design
Cases diagnosed as FL affecting the oral cavity were retrospectively retrieved from the pathology files of the authors’ institutions. Formalin-fixed paraffin-embedded (FFPE) tissue blocks were obtained, and new 4-μm histological sections were cut and stained with hematoxylin and eosin (H&E) for microscopic description, diagnostic confirmation, and histological grading following the latest classifications of hematolymphoid neoplasms [12, 13]. Tumors were further classified according to the proportion of follicular areas as follows: follicular when showing a >75% follicular pattern, follicular and diffuse when showing a 25–75% follicular pattern, focally diffuse when showing a <25% follicular pattern, and diffuse when 0% follicular pattern was seen [19].
Clinical features were retrieved from the patients' pathology reports or medical files, and included age, sex, tumor location, clinical presentation, time of evolution, treatment employed, systemic involvement, status at last follow-up (dead or alive), and time of follow-up. The overall survival rate was defined as the period from the date of diagnosis to the date of the patient’s demise (from any cause) or last follow-up.
Immunohistochemistry
Immunohistochemistry was performed on 3-μm sections of FFPE tissues that were dewaxed with xylene and hydrated in a descending ethanol series. The endogenous peroxidase activity was blocked with 10% hydrogen peroxide in a single bath for 15 min. After washing in PBS buffer (pH 7.4), the sections were incubated for 2 h with primary antibodies and then exposed to high-sensitive horseradish peroxidase reagents (ADVANCE, Dako, Carpinteria, CA, USA) and diaminobenzidine tetrahydrochloride (DAB, Sigma-Aldrich, St Louis, MO, USA). The slides were counterstained with Carazzi hematoxylin for 3 min. Positive controls were used for each antibody, while the negative control was obtained by omitting the primary antibody.
Cases were submitted to the following primary antibodies: CD20, CD3, CD23, CD10, Bcl6, Bcl2, cyclin D1, and Ki-67, with additional reactions carried out, if necessary, to confirm the diagnoses. To screen for a possible large B-cell lymphoma with IRF4 rearrangement (LBCL-IRF4), MUM1 protein was also investigated in high-grade cases.
Fluorescence in situ hybridization (FISH) for MYC, BCL2, and BCL6 rearrangements
FISH was performed on 4-μm tissue microarray sections using dual-color break-apart probes (MYC/8q24 and BCL6/3q27) and dual-color fusion translocation probes (BCL2/18q21) (Abbott Laboratories, Des Plaines, IL). Cells were hybridized with MYC, BCL2, and BCL6 probes, and nuclei were counterstained with 4′,6-diamidino-2-phenylindole antifade. Fluorescence was detected using an Olympus BX41 fluorescence microscope (OlympusTM, Japan) with excitation filters for 4′,6-diamidino-2-phenylindole (260 nm), and rhodamine (570 nm). For each case, up to 200 interphase nuclei were analyzed using an ASI image analysis system (Applied Spectral ImagingTM, Israel). The spots in nuclei were scored as previously described [20].
Statistical analysis
Clinicopathological, immunohistochemical, and FISH data were descriptively analyzed, with categorical variables presented as absolute numbers and percentages, whereas continuous variables were presented as mean, standard deviation (SD), and range. The overall survival rate was obtained using the Kaplan–Meier method. SPSS version 22.0 (IBM, Germany) was used for the statistical analyses.
Results
Final cohort composition
Following the current guidelines of the World Health Organization classification of hematolymphoid tumors, the essential criteria used in this study to classify tumors as follicular lymphomas consisted of a B-cell neoplasm composed of varying proportions of centrocytes (CC) and/or centroblasts (CB)/large transformed cells, and immunophenotype compatible with germinal center B-cell origin demonstrated by the expression of CD10 or BCL6. Although considered a desirable finding, all cases in our series had at least partial follicular growth pattern and the investigation of MYC, BCL2, and BCL6 was also used to further support the diagnoses. Twenty-seven cases were initially retrieved and all cases were carefully reviewed; however, two cases were excluded as they could not be reliably differentiated from marginal zone lymphoma of the mucosa-associated lymphoid tissue (MALT lymphoma), two cases were excluded due to the lack of clinical information, one case was diagnosed in the parotid gland and two cases were reclassified as mantle cell lymphoma due to cyclin D1 positivity. Therefore, 20 cases of oral FL were confirmed and included in the current series.
Clinical features and treatment
During the period investigated, the total number of cases diagnosed and the number of oral follicular lymphomas diagnosed in each institution were the following: School of Dentistry, Universidade Federal de Minas Gerais (from 2000 to 2023) 31,126 cases of oral pathology and four oral follicular lymphomas; Oral pathology service of the João de Barros Barreto University Hospital (from 2007 to 2022) 10,559 cases of oral pathology and four oral follicular lymphomas; Piracicaba Dental Scool, University of Campinas (from 2000 to 2022) 42,581 cases of oral pathology and six oral follicular lymphomas; Getúlio Sales Diagnósticos Pathology laboratory (from 2010 to 2023) 2417 cases of nodal/extranodal lymphomas and four oral follicular lymphomas; School of Dentistry, University of Pretoria (from 2000 to 2020) 26,000 cases of oral pathology and one oral follicular lymphoma; and Araçatuba Institute of Pathology (from 2020 to 2022) 63,572 cases, 158 nodal/extranodal lymphomas, and one oral follicular lymphoma.
Detailed clinicopathologic features are available in Table 1. There was an equal sex distribution (10 males: 10 females). The mean age was 60.9 years (range: 10–83 years), with a peak incidence in the eighth decade of life. The most commonly affected subsites were the palate (10 cases; 50%) and buccal mucosa (7 cases; 35%). Five cases (25%) were known to affect the cervical lymph nodes simultaneously, and associated B-symptoms were reported by four patients (20%). All cases presented as slow-growing tumors, more commonly asymptomatic (11 cases; 55%), although pain was reported in four cases (20%). Associated surface ulceration was not seen in any case, and tumors usually presented as normal-colored swellings. Telangectasia was reported in some cases. The mean duration of symptoms reported by patients was 6.5 months (range 1–24 months), although in three cases the lesions increased more rapidly in the month before biopsy and histopathologic diagnosis (Fig. 1). Data on treatment and status at last follow-up were available for eight and 11 patients, respectively. Chemotherapy alone was used for five patients, while combination chemo- and radiotherapy was applied for three patients. One patient refused any form of treatment. Because patients were treated in different hospitals from many regions of Brazil, detailing the specific chemotherapy protocols was unfortunately not possible.
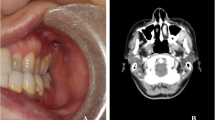
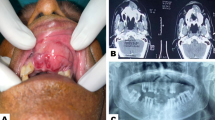
Clinical presentation of follicular lymphomas. A A 71-year-old male patient presenting with a one-month history of swelling involving the hard palate. B A 37-year-old female patient exhibiting an asymptomatic swelling in the hard palate. C A 78-year-old female patient presenting with an irregular swelling affecting the left buccal mucosa. D An 83-year-old female patient exhibiting a large tumor in the left cheek submucosa
Partial remission of the tumor was observed in seven cases (63.3%), total remission was seen in one patient (9.1%), and in one case there was no remission up to the last patient’s appointment (9.1%). At last follow-up, eight patients (72.7%) were alive, while three (27.3%) were deceased due to unknown causes (Table 1). The mean follow-up time was 18 months, ranging from 1 to 56 months, and a descriptive Kaplan–Meier curve showed a 2-year survival rate of 88%.
Microscopic findings
All included cases of FL presented with microscopic features that characterize this entity when diagnosed in the lymph nodes. Most cases (14 cases; 70%) exhibited a predominantly follicular growth pattern, while six cases also demonstrated some areas of diffuse proliferation. The so-called back-to-back pattern of the neoplastic follicles was present in all cases in different proportions, frequently surrounded by a fibrotic stroma. The follicles usually exhibited a poorly defined morphology, with irregular contour, size variation, irregular mantle zone (frequently absent), lack of tingible body macrophages in the neoplastic follicles, and lack of normal zonation expected in the germinal center. Tumor cells were predominantly of small size with hyperchromatic nuclei and an irregular nuclear contour consistent with centrocytes. Larger cells with prominent nucleoli consistent with centroblasts were seen in all cases in varied amounts. Twelve cases had lower than 15 centroblasts/HPF and were classified as low-grade FLs (grades 1 and 2), while eight cases with more than 15 centroblasts/HPF were also observed and classified as high-grade follicular lymphomas, but always with the presence of centrocytes, representing grade 3A FLs.
All cases exhibited a strong and diffuse reactivity to the mature B-cell marker CD20 in the neoplastic cells concentrated in the follicles. CD3 revealed the presence of reactive T cells predominantly in the interfollicular region, with a variable amount of T cells within the neoplastic follicles. Tumor cells were also positive for Bcl6 (13 cases; 65%) and/or CD10 (18 cases; 90%), confirming a germinal center origin. Follicular dendritic cells (FDC) were highlighted by CD23 in all 15 cases where the immunohistochemical stain was performed. CD23 also highlighted the follicular structures in some tumor areas where this pattern was not evident on routine H&E staining and neoplastic lymphoid cells were also stained for CD23 in one case. Reactivity to Bcl2 protein in the neoplastic germinal centers was seen in 18 cases, with only two high-grade FLs being negative. Cyclin D1 and MUM1 were negative in all cases evaluated. Ki67 proliferative index varied and was higher in grade 3A tumors (mean proliferation index of 32.5%) than in grade 1 and 2 tumors (mean proliferation index of 20.5%). The Ki67-positive cells were distributed inside and outside the follicles (higher inside the follicles), and did not reveal the proliferative zonation usually observed in normal germinal centers (Fig. 2).
Microscopic, immunohistochemistry, and fluorescence in situ hybridization of follicular lymphomas in the oral cavity. A Infiltration of neoplastic cells showing both a follicular and diffuse microscopic pattern (H&E, 50×). B The neoplastic follicles are predominantly composed of small-sized centrocytes. The clear and dark zones of normal germinal centers cannot be readily identified. There are no tingible body macrophages. (H&E, 100×). C Neoplastic cells comprising predominantly small centrocytes and rare large-sized centroblasts (H&E, 400×). D CD20 protein expression showing a predominance of B-lymphocytes within the follicles (DAB, 50×). E CD3 expression showing predominantly interfollicular reactive T cells (DAB, 50×). F CD10 protein expression demonstrating germinal center origin (DAB, 50×). G Bcl6 expression mirroring the CD10 expression pattern (DAB, 50×). H Bcl2 expression in the neoplastic follicles (DAB, 50×). I Ki-67 expression showing the proliferation index of the analyzed case (DAB, 100×). J Negative break-apart FISH for the BCL6 gene. K Positive break-apart FISH indicating a translocation involving the BCL2 gene. L Negative break-apart FISH for the MYC gene
Rearrangement status of BCL2, BCL6, and MYC genes
Fluorescence in situ hybridization was performed on 12 cases of FL to determine the rearrangement of BCL2, BCL6, and MYC genes, while the other eight cases did not have available tissue for analysis. The results obtained are presented in Table 2. A single rearrangement of the BCL2 gene was seen in eight cases (66.6%), two cases exhibited rearrangement in MYC, BCL2, and BCL6 simutaneously (16.6%) (Fig. 3), and the remaining two cases (20%) did not show any rearrangements (16.6%).
Microscopic, immunohistochemical, and fluorescence in situ hybridization findings of a triple-hit follicular lymphoma case. A Proliferation of poorly defined neoplastic follicles that lack a mantle zone and normal zonation (H&E, 50×). B The neoplastic follicles are composed of centrocytes and less than 15 centroblasts/HPF, denoting a low-grade tumor (H&E, 10×). C CD20 expression showing strong reactivity in a vaguely nodular pattern, indicating mature B-cell differentiation (DAB, 50×). D CD3 expression showing surrounding reactive T cells (DAB, 50×). E CD10 protein expression demonstrating germinal center origin (DAB, 50×). F CD23 expression highlighting the residual follicular dendritic cell meshworks (DAB, 50×). G Bcl6 expression mirroring the CD10 expression pattern (DAB, 50×). H Bcl2 expression in the neoplastic follicles (DAB, 50×). I Ki-67 expression showing the proliferation index of the analyzed case (DAB, 200×). J Positive break-apart FISH indicating a translocation involving the BCL6 gene. K Positive break-apart FISH indicating a translocation involving the BCL2 gene. L Positive break-apart FISH indicating a translocation involving the MYC gene
Discussion
Follicular lymphoma was initially described in 1925 as a benign disorder with an indolent clinical behavior, being rapidly recognized as a malignancy in the following years [21]. It is now defined as a neoplasm derived from follicle center B cells with at least a partial follicular growth pattern, and is considered the second most common NHL subtype [6, 22]. The head and neck region is one of the most affected locations, but extranodal FLs involving the oral cavity are exceedingly rare [16, 18, 22]. The current series demonstrated that cases of FL affecting the oral cavity might represent either primary or disseminated diseases. The tumor masses usually manifest as non-ulcerated swellings, more frequently in the palate and the buccal mucosa, and the majority of oral FLs are histologically classified as low-grade tumors. Moreover, BCL2 rearrangement was present in the majority of the cases, although unusual examples exhibited rearrangements in BCL2, BCL6, and MYC genes.
While most FLs predominantly manifest in peripheral lymph nodes, our study highlights the rarity and distinct characteristics of oral FLs, with primary extranodal occurrences more common in areas such as the gastrointestinal tract, soft tissue, breast, and ocular adnexa [22]. The literature reveals a limited number of oral FL reports, with a notable study identifying 24 instances mainly affecting the palate, averaging 64 years of age and an equal gender distribution [3]. Our findings align with Argyris et al. [23] documentation of eight oral cases, but emphasize the palate and buccal mucosa as primary sites in our cohort, with a mean age of 60.9 years. These data, compared to existing literature, indicate subtle demographic differences. Clinically, our cases consistently presented as asymptomatic, non-ulcerated swellings, reflecting an indolent progression, which might pose challenges in early identification.
FLs commonly demonstrate at least a partial follicular growth pattern [24]. However, in some instances, as shown in six cases of our cohort, this pattern was not clearly discernible. The diffuse proliferation of tumor cells might obscure the neoplastic follicles, making the correct diagnosis more difficult. Employing follicular dendritic cell markers, like CD21, CD23, and/or CD35, and complementing them with the expression of CD10 and Bcl6 can lead to an accurate diagnosis [10, 12]. The World Health Organization used to recommend microscopic grading of FLs based on centroblast numbers, but its last edition withdrew this recommendation given the weak evidence regarding its utility in the era of new chemotherapeutic regimens and its questionable reproducibility [13, 25]. Conversely, the ICC position paper still recommends the use of microscopic grading, with evidences suggesting that grade 3B FLs have a worse prognosis compared to grades 1, 2, and 3A [12]. Our cohort displayed an even distribution between low- and high-grade tumors. Noticeably, grade 3A FLs more often demonstrated the loss of markers like CD10, Bcl6, and/or Bcl2 [20, 25]. While grade 3B FLs, predominantly featuring centroblasts, have been renamed to follicular large B-cell lymphoma in the latest World Health Organization classification [13, 26], differentiating it from diffuse large B-cell lymphoma is difficult, given their similar prognosis and clinical behavior [20]. Notably, no high-grade cases in our study represented grade 3B FL.
The chromosomal abnormality t(14;18), leading to the translocation between IGH and BCL2 genes, has been viewed as the defining genetic hallmark of FL, being identified in a vast majority, about 85–90% of systemic nodal FL cases [13, 26]. In the current series, eight cases showed the characteristic BCL2 rearrangement, while two cases lacked BCL2 rearrangement. A comparison to systemic nodal FL shows that while 10–20% of the low-grade tumors and approximately 50% of their grade 3 neoplasms lack this translocation [10, 27], our oral FLs data showed only two cases lacking BCL2 rearrangement. Moreover, while structural alterations involving BCL6 are usually linked with follicular large B-cell lymphomas, our study showed two cases with rearrangements not only in BCL2, but also in BCL6 and MYC genes. Labeling them as “triple hit FLs” seems fitting given that despite these genetic rearrangements, their immunomorphology still aligns more with FL than with diffuse large B-cell lymphomas (DLBCL). The existence of such “triple hit FLs” in the literature has been previously documented; however, their prognostic significance remains debatable. For instance, while Miyaoka et al. [11] indicated a predominantly indolent clinical behavior, Ziemba et al. [14] documented a spectrum of presentations, with some of these FL cases manifesting aggressive clinical courses. This was observed in our cohort, with one of the triple-hit FL patients unfortunately succumbing to the disease after 5 months of follow-up.
More recently, large B-cell lymphoma with IRF4 rearrangement has been better characterized and defined as a distinct entity. These tumors commonly exhibit a follicular growth pattern, more often affecting the Waldeyer’s ring of young patients and are associated with a better prognosis [28]. As a consequence of IRF4 rearrangement, tumor cells overexpress MUM1 protein, which was used as a screening approach in this series, although no case was shown to overexpress this protein in the analyzed cases in the current sample [28, 29].
In this series, five patients affected by oral FLs also exhibited manifestations of the disease in cervical lymph nodes. This suggests that oral manifestations could represent an extranodal extension of the tumor. Therefore, all cases diagnosed in the oral cavity should prompt clinicians to investigate patients for systemic disease [20, 22]. Moreover, in addition to classic FLs, the current terminology used by the fifth edition of the World Health Organization classification of haematolymphoid tumors [13], several other well-known variants or subtypes of FLs should also be excluded from the diagnosis of oral lesions, such as duodenal-type FL, which commonly affects younger patients; testicular-type FL, associated with a favorable prognosis and absence of BCL2 rearrangement and Bcl2 protein expression; and pediatric-type FL, which often affects the lymph nodes of children or young adults and also lacks BCL2 rearrangements [10, 30]. Although we have included one patient aged 10 years old in our series, this tumor expressed Bcl2 protein, carried rearrangements in BCL2, BCL6, and MYC genes, and exhibited a highly aggressive clinical behavior; therefore, we favored a diagnosis of a triple-hit FL for this patient. Moreover, the extranodal presentation of a pediatric-type FL is excluded by definition, which further supports our diagnosis of a triple-hit FL [13].
The fifth edition of the World Health Organization classification of haematolymphoid tumors further recognizes in situ follicular B-cell neoplasm, primary cutaneous follicle center lymphoma, and follicular lymphomas with uncommon features like those with “blastoid” or “large centrocyte” cytological features and those with a predominantly diffuse growth pattern. In addition to these variants, the ICC position paper also describes the BCL2 rearrangement-negative, CD23-positive follicle center lymphoma that frequently carries STAT6 rearrangements [12, 13, 26, 31].
Significant improvements in treating patients with FLs have led to improved survival rates, with rituximab-based schemes being the most frequently used therapeutic approaches. However, many clinical and pathological factors may impact patients’ prognosis, including advanced age (> 60 years), hemoglobin concentration < 12g/dL, elevated serum lactate dehydrogenase, disseminated diseases, and the presence of more than four lymph nodes affected by the neoplasm [12, 16]. These features account for the follicular lymphoma international prognostic index (FLIPI). Moreover, grade 3B tumors and high-grade transformation foci are associated with more aggressive tumor behavior. Relapsed and refractory FLs are also common and are usually associated with a poorer response to first-line chemotherapy regimens [32]. In the current series, out of the 11 patients with available follow-up data, three deceased at last appointment, one of them having presented with simutaneous involvement of cervical lymph nodes.
In conclusion, our data support that FL affecting the oral cavity is uncommon and usually affects the palate as a non-ulcerated swelling. Importantly, the presence of a systemic disease should be investigated in all cases. We also describe the existence of triple-hit FLs in this anatomic site, which can be further explored in specific future investigations.
Data availability
The data that support the findings of this study are available from the corresponding author upon request.
References
Apostolidis J, Mokhtar N, Al Omari R, Darweesh M, Al Hashmi H (2020) Follicular lymphoma: update on management and emerging therapies at the dawn of the new decade. Hematol Oncol 38:213–222
Carbone A, Roulland S, Gloghini A, Younes A, von Keudell G, López-Guillermo A, Fitzgibbon J (2019) Follicular lymphoma. Nature reviews. Disease primers 5:83
Wagner VP, Rodrigues-Fernandes CI, Carvalho MVR, Dos Santos JN, Barra MB, Hunter KD et al (2021) Mantle cell lymphoma, malt lymphoma, small lymphocytic lymphoma, and follicular lymphoma of the oral cavity: an update. J Oral Pathol Med 50:622–630
Kumar E, Pickard L, Okosun J (2021) Pathogenesis of follicular lymphoma: genetics to the microenvironment to clinical translation. Br J Haematol 194:810–821
Casulo C (2021) Upfront identification of high-risk follicular lymphoma. Hematol Oncol 39:88–93
Laurini JA, Perry AM, Boilesen E, Diebold J, Maclennan KA, Müller-Hermelink HK et al (2012) Classification of non-Hodgkin lymphoma in Central and South America: a review of 1028 cases. Blood 120:4795–4801
de Moraes T, Hungria V, Chiattone C, Pavlovsky M, Abenoza LM, Agreda GP, Armenta J et al (2019) Epidemiology of hematologic malignancies in real-world settings: findings from the Hemato-Oncology Latin America observational registry study. J Glob Oncol 5:1–19
Freedman A, Jacobsen E (2020) Follicular lymphoma: 2020 update on diagnosis and management. Am J Hematol 95:316–327
Jóna Á, Kenyeres A, Barna S, Illés Á, Simon Z (2022) Clinical and biological prognostic factors in follicular lymphoma patients. PLoS One 17:e0272787
Jaffe ES, Harris NL, Swerdlow SH, Ott G, Nathwani BN, de Jong D et al (2017) Follicular lymphoma. In: Swerdlow SH, Campo E, Harris NL et al (eds) World Health Organization classification of tumours of hematopoietic and lymphoid tissues, 4th edn. IARC Press, Lyon
Miyaoka M, Kikuti YY, Carreras J, Ikoma H, Hiraiwa S, Ichiki A et al (2018) Clinicopathological and genomic analysis of double-hit follicular lymphoma: comparison with high-grade B-cell lymphoma with MYC and BCL2 and/or BCL6 rearrangements. Mod Pathol 31:313–326
Campo E, Jaffe ES, Cook JR, Quintanilla-Martinez L, Swerdlow SH, Anderson KC et al (2022) The international consensus classification of mature lymphoid neoplasms: a report from the clinical advisory committee. Blood 140:1229–1253
Xerri L, Medeiros JL, Klapper W, Louissaint A Jr, Watanabe T, Karube K et al (2022) Follicular Lymphoma. In: Naresh KN, Ott G, Jong D, Dave S (eds) World Health Organization classification of tumours of hematopoietic and lymphoid tissues (online), 5th edn. IARC Press, Lyon
Ziemba JB, Wolf Z, Weinstock M, Asakrah S (2020) Double-hit and triple-hit follicular lymphoma. Am J Clin Pathol 153:672–685
Chaudhary S, Brown N, Song JY et al (2021) Relative frequency and clinicopathologic characteristics of MYC-rearranged follicular lymphoma. Hum Pathol 114:19–27
Lin Z, Zha J, Yi S, Li Z, Ping L, He X et al (2023) Clinical characteristics and outcomes of follicular lymphoma patients with extranodal involvement: analysis of a series of 1090 cases in China. Clin Transl Oncol 25:1821–1829
Beasley MJ (2012) Lymphoma of the thyroid and head and neck. Clin Oncol (R Coll Radiol) 24:345–351
de Arruda JAA, Schuch LF, Conte Neto N, de Souza LL, Rodrigues-Fernandes CI, Abreu LG et al (2021) Oral and oropharyngeal lymphomas: a multi-institutional collaborative study. J Oral Pathol Med 50:603–612
Fratoni S, Zanelli M, Zizzo M, Sanguedolce F, Aimola V, Cerrone G, Ricci L, Filosa A, Martino G, Ascani S (2020) The broad landscape of follicular lymphoma: Part I. Pathologica 112:1–16
Horn H, Ziepert M, Becher C, Barth TFE, Bernd H-W, Feller AC et al (2013) MYC status in concert with BCL2 and BCL6 expression predicts outcome in diffuse large B-cell lymphoma. Blood 121:2253–2263
van Besien K, Schouten H (2007) Follicular lymphoma: a historical overview. Leuk Lymphoma 48:232–243
Sorrentino A, Ferragina F, Barca I, Arrotta A, Cristofaro MG (2022) Extra-nodal lymphomas of the head and neck and oral cavity: a retrospective study. Curr Oncol (Toronto, Ont.) 29:7189–7197
Argyris PP, Dolan M, Piperi E, Tosios KI, Pambuccian SE, Koutlas IG (2013) Oral follicular lymphomas. A short report of 8 cases with assessment of the IGH/BCL2 gene fusion with fluorescence in situ hybridization. Oral Surg Oral Med Oral Pathol Oral Radiol 116:343–347
Ansell SM (2013) Malignant B cells at the helm in follicular lymphoma. J Clin Oncol 31:2641–2642
Cree IA, Tan PH, Travis WD, Wesseling P, Yagi Y, White VA et al (2021) Counting mitoses: SI(ze) matters. Mod Pathol 34:1651–1657
Alaggio R, Amador C, Anagnostopoulos I, Attygalle AD, IBO A, Berti E et al (2022) The 5th edition of the World Health Organization classification of haematolymphoid tumours: lymphoid neoplasms. Leukemia 36:1720–1748
Takata K, Miyata-Takata T, Sato Y, Yoshino T (2014) Pathology of follicular lymphoma. J Clin Exp Hematop 54:3–9
Hesius EAM, van Laar L, Oosterveld M et al (2023) Reclassification of diffuse large B cell lymphoma to large B cell lymphoma with IRF4 rearrangement in an adult population. Histopathology 82:1013–1020
Karube K, Guo Y, Suzumiya J, Sugita Y, Nomura Y, Yamamoto K et al (2007) CD10-MUM1+ follicular lymphoma lacks BCL2 gene translocation and shows characteristic biologic and clinical features. Blood 109:3076–3079
Ferry JA (2010) Recent advances in follicular lymphoma: pediatric, extranodal, and follicular lymphoma in situ. Surg Pathol Clin 3:877–906
Siddiqi IN, Friedman J, Barry-Holson KQ, Ma C, Thodima V, Kang I et al (2016) Characterization of a variant of t(14;18) negative nodal diffuse follicular lymphoma with CD23 expression, 1p36/TNFRSF14 abnormalities, and STAT6 mutations. Mod Pathol 29:570–581
Yang G, Mills M, Kim Y, Figura NB, Doyle C, Oliver D et al (2020) Enhancement of the Follicular Lymphoma International Prognostic Index (FLIPI) with lymphopenia (FLIPI-L): a predictor for overall survival and histologic transformation. Blood Cancer J 9:104
Funding
This study was supported by the São Paulo State Research Foundation (FAPESP #22/03123-5), the Minas Gerais State Research Foundation (FAPEMIG), the Rio de Janeiro State Research Foundation (FAPERJ), the National Council for Scientific and Technological Development (CNPq), and the Coordination for the Improvement of Higher Education Personnel (CAPES, 001).
Author information
Authors and Affiliations
Contributions
Study concept and design: FPF. Data acquisition: LLS, CVBLC, MSPS, CDS, BABA, JCCXJ, LR, PAV, FPF. Data analysis and interpretation: LLS, CVBLC, MSPS, BABA, HARP, RAM, LR, WvH, RMRB, PAV, GRAM, FPF. Statistical analysis: LLS. Manuscript preparation: LLS, CVBLC, GRAM, FPF. Manuscript editing: all authors. Manuscript review: all authors.
Corresponding author
Ethics declarations
Ethical approval
The study was approved by the Ethics Committee of the Piracicaba Dental School, University of Campinas, Piracicaba, Brazil (protocol no. 67128417.4.0000.5418) and the Ethics Committee of the Federal University of Minas Gerais, Belo Horizonte, Brazil (protocol no. 10723019.0.1001.514).
Consent for publication
Consent for publication was obtained for every individual person’s data included in the study.
Informed Consent
This work is covered by protocol approved by the Ethics Committee of the Piracicaba Dental School, University of Campinas, Piracicaba, Brazil (protocol no. 67128417.4.0000.5418) and the Ethics Committee of the Federal University of Minas Gerais, Belo Horizonte, Brazil (protocol no. 10723019.0.1001.514) and it does not require informed consent due to the retrospective nature of the work and the lack of data that could potentially identify the patient.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Souza, L.L., Cáceres, C.V.B.L., Sant’Ana, M.S.P. et al. Oral follicular lymphoma: a clinicopathologic and molecular study. J Hematopathol 16, 199–208 (2023). https://doi.org/10.1007/s12308-023-00563-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12308-023-00563-6