Abstract
Ovarian cancer is considered to be the most lethal type of gynecological cancer. During the advanced stages of ovarian cancer, an accumulation of ascites is observed. Fucosylation has been classified as an abnormal post-translational modification that is present in many diseases, including ovarian cancer. Ovarian cancer cells that are cultured with ascites stimulation change their morphology; concomitantly, the fucosylation process is altered. However, it is not known which fucosylated proteins are modified. The goal of this work was to identify the differentially fucosylated proteins that are expressed by ovarian cancer cell lines that are cultured with ovarian cancer patients’ ascites. Aleuria aurantia lectin was used to detect fucosylation, and some changes were observed, especially in the cell membrane. Affinity chromatography and mass spectrometry (MALDI-TOF) were used to identify 6 fucosylated proteins. Four proteins (Intermediate filament family orphan 1 [IFFO1], PHD finger protein 20-like protein 1 [PHF20L1], immunoglobulin gamma 1 heavy chain variable region partial [IGHV1–2], and Zinc finger protein 224 [ZNF224]) were obtained from cell cultures stimulated with ascites, and the other two proteins (Peregrin [BRPF1] and Dystrobrevin alpha [DTNA]) were obtained under normal culture conditions. The fucosylated state of some of these proteins was further analyzed. The experimental results show that the ascites of ovarian cancer patients modulated the fucosylation process. The PHD finger protein 20-like protein 1, Zinc finger protein 224 and Peregrin proteins colocalize with fucosylation at different levels.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ovarian cancer is considered to be the most lethal type of gynecological cancer, despite ranking eighth in worldwide cancer incidence. In Mexico, the reported incidence in 2018 was 4759 cases and a mortality of 2765 cases [1]. Ovarian cancer can be classified into different histotypes; nevertheless, epithelial ovarian cancer is the most common and the most lethal [2]. In the majority of ovarian cancer cases, the cancer metastasizes into the abdominal cavity, and the presence of tumor cells is frequently associated with ascites accumulation [3]. This ascites accumulation is observed during the advanced stages [4] and can contribute to transcoelomic metastasis.
Ascites is a highly heterogeneous fluid that contains a wide variety of soluble factors and a mixture of cell populations, including tumoral, stromal and immune cells [5, 6]. The composition of ascites from various origins has been analyzed by different groups, and these analyses have demonstrated their complexity and heterogeneity [7, 8]. Thus, given the diversity of molecules found in it, ascites is a stimulus that could alter various biological processes, among which, fucosylation may be included. Typically, fucosylation and sialylation are terminal carbohydrate chain-modifications of glycoproteins (which means that fucose and sialyl are frequently located at the end of the carbohydrate chain) [9, 10]. Fucosylation and sialylation mediate biological functions and have also been implicated in cancer [9, 11, 12].
An increase in the fucosylation of tumor cells contributes to several abnormal characteristics, such as a decrease in cellular adhesion and uncontrolled tumor growth [13]. However, there is limited information on the function of fucosylation and/or the fucosylated status of glycoproteins during the development and progression of ovarian cancer. More recently, Hu et al. [14] showed that when the CD147 glycoprotein was modified by Lewis Y antigen, the glycoprotein improved the survival capacity of ovarian cancer cells by restricting autophagic cell death; thus, CD147 plays an important role in the malignant progression of ovarian cancer [14]. Additional studies on this antigen have shown that the overexpression of Lewis Y antigen promotes the human epididymis protein 4-mediated invasion and metastasis of ovarian cancer cells [15], p27 degradation by regulating ubiquitin-proteasome activity in the CAOV-3 and SKOV-3 cell lines [16], and chemotherapeutic resistance in ovarian cancer [17]. A few examples of modified fucosylated proteins are haptoglobin in several cancers, including ovarian cancer [18,19,20], transferrin and alpha-fetoprotein in liver cancer [21, 22] and thyroglobulin in thyroid cancer [23]. In particular, previous results from our group have shown that the level of fucosylation of haptoglobin increases with stage progression [19] and was found to be highly fucosylated in different subtypes of epithelial ovarian cancer [20].
It has been confirmed that ascites can change the cellular phenotype, and the ascites compounds that are produced by or in response to malignant cells may be a good alternative to the microenvironment in which malignant cells are immersed [24]; these components could participate in the altered regulation of different processes, including fucosylation. For this reason, the goal of our work was to determine the differentially fucosylated proteins in ovarian cancer cell lines cultured with ascites stimulation. Experimental results have shown that the ascites of ovarian cancer modulate the fucosylation process since the profiles of the fucosylated proteins were altered in the ascites-treated SKOV-3 and OVCAR-3 cell lines. In particular, among the differentially fucosylated proteins, PHD finger protein 20-like protein 1, Zinc finger protein 224, and Peregrin were detected. Together, our results suggest that ascites may favor tumor progression through the modification of cellular processes, such as fucosylation.
Materials and Methods
Study Model
The SKOV-3 (ATCC HTB-77) and OVCAR3 (ATCC HTB 161) cell lines were used. The SKOV-3 model has been used previously in our laboratory as described [24]. Briefly, SKOV-3 cells were cultured in McCoy’s 5A (Corning, 10–050-CVR) culture medium (15 mL) supplemented with 10% fetal bovine serum (Corning, 35–010-CV) and 1% penicillin/streptomycin (PAA, P11–010) at 37 °C and 5% CO2. The culture was grown until the cells reached 75% confluence. Then, the medium was discarded for the subsequent addition of ascites (15 mL). The cells were maintained under the ascites stimulus at 37 °C and 5% CO2 for the indicated time periods. The cells that were maintained in the medium were used as a control. OVCAR-3 culture was grown in RPMI 1640 (Corning, 10–004-CM) culture medium supplemented with 10% fetal bovine serum and bovine insulin (Millipore, I0516). The culture was grown until the cells reached 75% confluence. Then, the medium was discarded for the subsequent addition of ascites (2 mL). The cells were maintained under the ascites stimulus at 37 °C and 5% CO2 for 24 h. The cells that were maintained in medium were used as a control. The OVCAR-3 cell line was employed only for use in immunofluorescence assays.
Biologicals Samples
Epithelial ovarian cancer (EOC) ascites were obtained from patients diagnosed with epithelial ovarian cancer at the Instituto Nacional de Cancerología (INCan) under the approval of the Scientific and Bioethical Committees (009/029/GOI) (CB/549/09); before obtaining the samples, informed consent letters were signed by the patients. The samples were used based on the Declaration of Helsinki about the ethical principles of medical research involving human samples. The clinical and pathological characteristics of the epithelial ovarian cancer patients are described in Table 1.
Whole ascites were collected by medical personnel. Ascites samples contaminated with blood were excluded from the study. Approximately 100 mL was centrifuged at 1000 rpm for 10 min to recover the supernatant and the cell pellet. Only cell-free ascites (supernatants) were further employed for experiments and were stored at −70 °C until use. Before use, ascites were defrosted and warmed at room temperature.
Treatment of SKOV-3 Cells with Ovarian Cancer Ascites A03 and Cellular Protein Extraction
At the end of the treatment time (24 h) of SKOV-3 cells with ascites 03 and the control condition, the cultures were washed with sterile 1x phosphate-buffered saline (PBS) and were subsequently recovered using Cell Stripper Solution (Corning, 25–056-CI). Cell lysis was carried out using RIPA buffer (5 mM Tris-HCl, 2 mM EDTA, 50 mM NaCl and 1% Nonidet P-40) with a cocktail of protease and phosphatase inhibitors (1 mg/ml of aprotinin and leupeptin, and 1 mM of PMSF, NaF, and Na3VO4). Finally, the samples were quantified using a DC Protein Assay Kit (Bio-Rad, 500–0114).
Affinity Chromatography with Aleuria Aurantia Lectin (AAL)
The purification of the fucosylated proteins was performed using AAL coupled to an agarose resin (Vector Labs, B-1393). The total protein extracts of SKOV-3 cells obtained from the control or experimental conditions were incubated with AAL for 16 h at 4 °C; afterward, the unbound proteins were removed by washing with TBS-T (contained Tween 20), while the fucosylated proteins bound to the resin were eluted with 200 μl elution buffer for AAL (Vector Labs, ES3100). The recovered fucosylated proteins were dialyzed using a dialysis membrane with a molecular weight cutoff of 8–12 kDa (Spectrum laboratories, 3787-F15). Dialysis was performed with 1x PBS while stirring at 4 °C for 48 h, and the 1x PBS was changed four times.
SDS-PAGE and Overlay
The total protein extracts (50 μg) and fractions that were washed and eluted from the affinity chromatography column were visualized by 10% SDS PAGE. The gel was stained with a Silver Stain Plus Kit (Bio-Rad, 161–0462, −0463, −0464). A replicate of the gel was transferred to a nitrocellulose membrane. The nitrocellulose membrane was incubated with Aleuria aurantia lectin (Vector Labs, B-1395 1:2000 dilution in PBS) for 2 h at room temperature. Afterward, the membrane was washed twice with PBS-T (contained 1% Tween), and streptavidin alkaline phosphatase (Vector Labs, S5512) was added to the membrane, which was incubated for 1 h at room temperature. Again, the membrane was washed twice with PBS-T (contained 1% Tween). Finally, it was incubated with the NBT/BCIP substrate (Vector Labs, SK5400) for development.
Confocal Microscopy Analysis
Briefly, SKOV-3 and OVCAR-3 cells were treated for different time periods with different ascites. Afterward, the cells in the ascites or culture medium conditions were fixed using 4% p-formaldehyde (Sigma Aldrich, 6148) for 1 h at 37 °C. The cultures were washed three times with 1x PBS and were then blocked with 10% fetal bovine serum (Corning, 35–010-CV) for 1 h at 37 °C; previously, some samples were permeabilized with 0.2% Triton X-100, and the rest were maintained until the next step. In the case of the colocalization assays, the corresponding primary antibody was added (anti-PHD finger protein 20-like protein 1, Sigma Aldrich HPA028417; anti-Zinc finger protein 224, Santa Cruz Biotechnology, sc-293,394; anti-Peregrin, Santa Cruz Biotechnology sc-81,059) in a 1:50 dilution and incubated for 16 h at 4 °C. Then, the samples were washed three times with 1x PBS. The cultures were incubated with AAL-biotin (1:50 dilution, Vector Labs, B-1395) for 1 h at 4 °C. Subsequently, the samples were washed three times with 1x PBS, and then streptavidin-fluorescein (1:200 dilution, Life Technologies, 43–4311) was added to the cultures, and the cultures were incubated for 1 h at room temperature. The corresponding secondary antibodies (anti-mouse Alexa Fluor 594, Invitrogen, A21203 at a 1:100 dilution and anti-rabbit Alexa Fluor 647, Abcam, ab150063 at a 1:300 dilution) were added together with streptavidin-fluorescein. Negative controls were included for each condition and were incubated only with streptavidin-fluorescein and with or without the corresponding secondary antibodies. The nuclei were stained with DAPI (1:50 dilution, Molecular Probes, D1306) for 5 min at room temperature. Finally, the samples were mounted with VectaShield® mounting medium (Vector Laboratories, H-1000). The analysis and quantification of fluorescence (arbitrary units of fluorescence) were performed using a Carl Zeiss Microscope (Carl Zeiss LMS 700) and the microscope software (ZEN 2012, Carl Zeiss Microscopy).
Mass Spectrometry (MALDI-TOF) and Protein Identification
The samples were processed as described previously [24] with some modifications. Briefly, peptide digestion was performed using mass grade trypsin (Sigma-Aldrich, T 6567). The peptide mass and identity were obtained using an Ultraflex TOF/TOF (Bruker Daltonics, Germany) that was configured in a delayed extraction mode and in reflectron mode. The peaks produced by the autolysis of trypsin and the contamination derivatives were excluded from the list. The monoisotopic masses of the spectrum were used for protein identification using the MASCOT v2.2 search engine (www.matrixscience.com). The search was carried out in the NCBI and/or SwissProt DB. The search parameters were as follows: peptide tolerance, 0.5–1 Da; and species, Homo sapiens. The maximum number of enzymatic cuts lost was set at 1.
Bioinformatics Analyses
Bioinformatic analyses were performed using The Human Protein Atlas, NetNGlyc and STRING 10.0 for protein-protein interaction (PPI) networks and to propose the possible functions exerted under our experimental conditions.
Statistical Analysis
For confocal microscopy images analysis, one-way ANOVA in GraphPad Prism 7 was performed to determine changes between each ascites time compared. A Bonferroni post hoc test was applied to compare multiple groups. Pearson correlation was used for colocalizing analyses.
The correlation analysis was performed for nonparametric samples due to the size of the sample; therefore, the Spearman correlation coefficient (r) was used, and the confidence intervals were established at 85.7% due to the characteristics of the population. The association was determined for clinical characteristics, such as the levels of CA125 (U/ml), the histological subtype, and the clinical stage. Additionally, we sought the association with the progression-free period (PFS) and the overall survival (OS) of the patients included in the study. Both the clinical characteristics and the association with survival were made based on the level of fucosylation of the cell membranes at 24 h; the fucosylation was induced by each of the 13 ascites in the SKOV-3 and OVCAR3 ovarian cancer cell lines. SPSS Ver 23, Inc., Chicago, III, US was employed to analyze the data.
Results
Membrane Fucosylation is Altered by Ascites Stimulation
To determine if ascites were involved in the induction of fucosylated proteins, SKOV-3 cells were stimulated with different ascites that emulate the natural microenvironment for malignant cells. Changes in protein fucosylation were observed. The expression level of fucosylated proteins in ascites-treated cells compared to those in the control conditions was analyzed by confocal microscopy. SKOV-3 cells cultured in control conditions showed a homogeneous distribution of fucosylation (Fig. 1, culture medium, nonpermeabilized cells). When SKOV-3 cells were permeabilized, they showed a perinuclear label for this post-translational modification that seemed to colocalize with the nuclear label (Fig. 1, culture medium, permeabilized cells). The morphology and fucosylation intensity changed under ascites stimulation. Membrane fucosylation (Fig. 1, nonpermeabilized, A01, A02) decreased at 24 h, and this effect was more evident when the cells were stimulated with ascites A01, but the level of fucosylation was reestablished at 72 h (Fig. 1; Supplementary Fig. 1). However, fucosylation was not completely lost; in the permeabilized cells, this glycoprotein modification was observed within the cytoplasm at all times of stimulation (Fig. 1, permeabilized cells, A01, A02). To confirm that the effect of ascites on the fucosylation level was universal, we analyzed the effects of different ascites (10 different samples) on SKOV-3 cells (Supplementary Fig. 2a) and on the OVCAR-3 cell line that was stimulated for 24 h (Supplementary Fig. 2b); we also analyzed the effects of two different ascites on Caco-2 and BEAS-2B cells that were stimulated for 24 h (Supplementary Fig. 2d). We found that ascites stimulated the fucosylation of all cells analyzed. The OVCAR-3 cell line cultured without ascites showed a very discreet level of fucosylation; however, when ascites-stimulated cells were analyzed, 8 out of 10 ascites significantly increased the membrane fucosylation levels (Supplementary Fig. 2b). Morphology was also modified by ascites, which induced the rounding of the cells and the formation of spheroids (Supplementary Fig. 2c). These fucosylation levels and the cell morphology were also modified in Caco-2 cells and BEAS-2B cells (Supplementary Fig. 2d). Taken together, these results confirm that in general, ascites have the ability to modulate the fucosylation process in different cell populations.
Fucosylation is modified due to ascites stimulation. SKOV-3 cells were treated for different time periods with ascites from EOC patients (A01 and A02) and were analyzed by confocal microscopy. Non-permeabilized cells were employed to analyze membrane fucosylation, and the permeabilized cells were used to analyze cytoplasmic fucosylation. Fucosylation was detected by biotinylated Aleuria aurantia lectin (1:200 dilution) and FITC-conjugated streptavidin (1:200 dilution). The nuclei were stained with DAPI. The selected images are representative of three independent biological replicates
When correlation analyses were performed, no significant correlations were found (Table 2); however, for SKOV-3, an inversely proportional tendency was found that correlated with the levels of fucosylation and the advanced clinical stages of the disease (CS III, IV) with an r = −0.432. The levels of the CA125 tumor marker also showed a slight, but direct, tendency to correlate with the levels of fucosylation and had an r = 0.309. On the other hand, the OVCAR3 cell line showed a stronger and directly proportional tendency to correlate between the fucosylation levels and the clinical stage with an r = 0.528. In this cell line, a directly proportional tendency of association between CA125 and the fucosylation levels was also obtained. In our study population, fucosylation did not show any correlation with patient survival; however, in all cases, it will be necessary to increase the sample size to determine or rule out the existence of an association between these variables.
SKOV-3 Cells that Are Stimulated with Ascites from Patients with EOC Have a Differentially Fucosylation Protein Profile
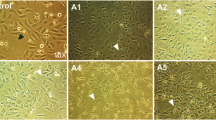
Under normal culture conditions, SKOV-3 cells showed a characteristic morphology, i.e., large cells with an abundant cytoplasm [25] (Fig. 2, culture medium, light microscopy). When fucosylation was evaluated with Aleuria aurantia lectin (AAL) in cells maintained in culture medium, fucosylation was strongly detected in the high molecular weight proteins (Fig. 2, 10% SDS-PAGE; overlay-AAL, upper panel). On the other hand, ascites-treated cells showed a much more accentuated spindle-shaped morphology (Fig. 2, ascites, light microscopy), as has been previously reported [24]. These morphological changes were observed with the 13 different ascites tested in this work. When overlay assays were performed, high molecular weight fucosylated proteins were detected (Fig. 2, 10% SDS-PAGE; overlay-AAL, lower panel). Although there were some similar bands in both conditions (black arrowheads, 10% SDS-PAGE, upper and lower panel), there were also some marked differences (asterisks, 10% SDS-PAGE, upper and lower panel). When comparing the pattern of fucosylated proteins, there were some proteins detected by AAL that could not be seen in the purification column (E1, 10% SDS-PAGE) (arrowheads, 10% SDS-PAGE and overlay-AAL, upper and lower panels). In contrast, some bands that appeared in the purification assay (E1, 10% SDS-PAGE) were not observed with AAL (dot, SDS-PAGE and overlay-AAL, upper and lower panels), suggesting that these proteins were not fucosylated.
Purification of fucosylated proteins by affinity chromatography with Aleuria aurantia lectin. SKOV-3 cells show a characteristic morphology under control conditions (Light microscopy, right panel, 2a). In SKOV-3 cells that were stimulated with ascites (light microscopy, left panel, 2a), abundant mucus was observed (black arrow, light microscopy, right panel, 2a), some large cells (black arrowhead, light microscopy, right panel, 2a) and membranes with large protrusions (white arrow, light microscopy, right panel, 2a). Fucosylated proteins were purified by Aleuria aurantia lectin affinity chromatography; different fractions were obtained and analyzed by 10% SDS-PAGE (2b). The overlay assay shows fucosylation in TE, UP, E1, and W1 fractions (2c). Black arrowheads (▶): similarities between the control condition (left panel) and the ascites-treated condition (right panel). Asterisks (*): differences between the control and experimental conditions. Arrowhead (<): similarities in fucosylation between the overlay test and the polyacrylamide gel. Arrows (↑): fucosylated proteins that are present in the overlay but are not present in the polyacrylamide gel. Dot (•): proteins that are present in the polyacrylamide gel but are not present in the overlay. TE: total extract; UP: unbound proteins; E1: eluate 1; W1-W6: washes
Identification of Fucosylated Proteins
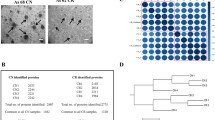
To identify which proteins had altered fucosylation as a result of ascites treatment, experiments were performed to purify the fucosylated proteins by affinity chromatography, and the profiles of the fucosylated proteins were obtained. In general, under ascites stimulation, more fucosylated proteins were observed; this pattern was conserved among all three biological replicates that were run in triplicate (Fig. 3). The protein profile showed changes, especially in some high molecular weight proteins (100–150 kDa) and in some low molecular weight proteins (15–25 kDa). The high molecular weight proteins were more visible in the control conditions (culture medium), and the low molecular weight proteins were more enriched in the experimental conditions (ascites); these results are highlighted in the extended right panels (Fig. 3). Thus, for identification purposes, the proteins that were present in the highlighted sections were chosen.
Fucosylated proteins in SKOV-3 cells under control conditions and treated with ascites. Whole protein extracts of SKOV-3 cells (W) stimulated with ascites or in medium are shown in the first two lanes. The subsequent lanes show the protein elution patterns of the fucosylated proteins corresponding to E1 obtained from the control conditions (M) and the experimental conditions (A); the proteins were selected from three independent biological replicates (assay 1–3). The amplified areas highlight the differences in the conditions studied and the histogram of the density of the amplified bands (darker lines correspond to the control conditions, while-gray lines correspond to the experimental conditions). Protein bands labeled with numbers 1–8 were selected for MS identification
Enriched fucosylated proteins that showed differences between both conditions were selected for their identification by MALDI-TOF (Fig. 3, numbers in circle, assay 2). The five following proteins were identified: Intermediate filament family orphan 1 [IFFO1], PHD finger protein 20-like protein 1 [PHF20L1], Peregrin [BRPF1], Dystrobrevin alpha [DTNA], Zinc finger protein 224 [ZNF224], and an immunoglobulin fragment (immunoglobulin gamma 1 heavy chain variable region, partial [IGHV1–2]). The main mass spectrometry data are shown in Table 3. To determine the post-translational modifications that might affect the proteins, different databases were reviewed; it has been reported that although some proteins can be phosphorylated or acetylated, fucosylation and glycosylation have not been described. However, in silico analyses in the database NetNGlyc showed many probable glycosylation sites, and this makes fucosylation possible (Supplementary Fig. 3).
Fucosylation Levels are Different among Proteins
To validate that the proteins identified by mass spectrometry were indeed fucosylated, three of them were further analyzed in both the SKOV-3 and OVCAR-3 cell lines. By confocal microscopy, it was possible to confirm that they were fucosylated by colocalization with AAL (Fig. 4, upper panel). All of the proteins analyzed showed colocalization at different levels, and all of them showed even more colocalization when the cells were incubated with ascites (Fig. 4, intermediate panel), including Peregrin (a protein identified from the control conditions). These results confirm the fucosylated state of these proteins and suggest that ascites stimulates the fucosylation of the three proteins (Pearson correlation coefficient was >0.5 in ascites conditions). The same three proteins in OVCAR-3 cells were analyzed, confirming that they were also fucosylated and showed colocalization with AAL (Fig. 4, lower panel).
Colocalization between identified proteins and fucosylation. For the colocalizing assay, ascites 02 was employed for SKOV-3 cells, and ascites A04 was employed for OVCAR-3 cells. Three of the proteins that were previously identified were analyzed (in red). Aleuria aurantia lectin was employed to follow fucosylation and is visualized in green. Merged images are shown, and the colocalization images are shown where the white spots indicate the points of colocalization between each protein and fucosylation. Pearson correlation was performed using the Zen 2.3 program
Protein-Protein Interaction (PPI) Network Analyses of Fucosylated Proteins in SKOV-3 Cells
To understand the possible role of the identified proteins during tumor progression, the STRING database was used to generate an interaction network (Fig. 5). In addition, a detailed search of all the identified proteins was performed. The proteins that were identified to have a structural function were Intermediate filament family orphan 1 and Dystrobrevin alpha; it was determined that the remaining proteins PHD finger protein 20-like protein 1, Peregrin and Zinc finger protein 224 act as transcription regulators. An analysis of expression was performed in The Human Protein Atlas database (https://www.proteinatlas.org); this analysis indicated that most proteins, except for PHD finger protein 20-like protein 1 and Zinc finger protein 224 (which have either moderate or high expression levels), have low protein expression levels in different types of cancer tissues, including ovarian cancer.
Discussion
Ascites, the accumulation of fluid in the abdominopelvic cavity, typically manifests in many types of advanced cancer, such as colon, pancreatic, stomach, breast, lymphoma, lung, and ovarian cancer [26, 27]. Usually, in the case of ovarian cancer, ascites contains growth factors, cytokines, and lipid mediators among other molecules [28, 29]. Together, these components favor the tumor, and some spheroids and cells can move through it, favoring metastasis [30, 31]. Actually, the presence of ascites favors conditions that promote proliferation, invasion, anti-apoptosis, chemoresistance and tumor heterogeneity [32,33,34]; these characteristics are because the cellular components then promote tumor growth, angiogenesis, and attenuate apoptosis [35,36,37,38].
The elements that conform to the different ascites are crucial to understanding how these ascites regulate the cells; in this case, the SKOV-3 and OVCAR-3 cell lines. However, it is very difficult to define the composition of each of these ascites. It is important to mention that IL-6 is usually found in ovarian cancer ascites [39]. Moreover, IL-6 and IL-8 are among the most abundant interleukins in ascites [40]. This is relevant because there are some reports that have indicated that the increased levels of fucosylation could be related to the higher levels of IL-6 in pancreatic cancer cells [41]. IL-6 signaling in ovarian cancer can regulate tumor cell proliferation, invasion and angiogenesis [42,43,44]. Additionally, an elevation of TGF-β, IL-6 and IL-8 act as oncogenic stimuli that modulate the process of epithelial mesenchymal transition, allowing invasion to occur more easily [35, 45]. To confirm this hypothesis, it will be necessary to determine the IL-6 levels in our ascites samples.
As reported by Toledo-Leyva et al. (2018) [24], the presence of ascites changes the phenotype of SKOV-3 cells, and this acquired phenotype (mesenchymal) is associated with increased invasion and migration properties [46]. Fucosylation is also modified, and this was demonstrated in this work, using cell-free ascites; therefore, we can affirm that the alterations observed in the fucosylation patterns are related to the soluble factors present in ascites and not to overgrowth of ascites tumor cells. However, we cannot discard the role of soluble immune modulators such as IL-6 or TGF-β. Most likely, not all ascites will induce the same change; however, at least for ascites with time-tested kinetics, there seemed to be a tendency to decrease the membrane-associated fucosylation levels (Fig. 1), whereas an increase in the cytoplasmic signal was observed (Fig. 1).
However, in OVCAR-3 cells, increased membrane fucosylation was observed (Supplementary fig. 2); this could be related to the morphology and cell phenotype; indeed, it has been reported that fucosylation could vary among cell and tissue types [47]. Although the fucosylation profile might be different, PHD finger protein 20-like protein 1and Zinc finger protein 224 and Peregrin fucosylated proteins were also found in OVCAR3 cells (Fig. 4). Nevertheless, it is possible that if the membrane proteins were analyzed, we could find important differences between these two cell lines in terms of their fucosylated proteins. Thus, fucosylation can be an important biochemical process for malignant cells.
We decided to use the SKOV-3 cell line because it is the most representative cell line of ovarian cancer, and many articles have used it as a reference. On the other hand, we noticed that the fucosylation of OVCAR-3 was mainly maintained in the membrane under all conditions of stimulation with ascites; however, when the SKOV-3 cells were permeabilized, the fucosylation label was distributed throughout the cytoplasm. The proteins identified by mass spectrometry were analyzed in OVCAR-3, and it was found that at the fucosylation colocalization points, the protein was located at the plasma membrane; this is a situation that is not expected for proteins with a nuclear function; thus all of these results must be analyzed in depth. There are reports on nuclear and cytoplasmic glycosylation. Several studies have also strongly indicated that high-mobility-group proteins, important structural components of chromatin, are glycoproteins [48]. The increased fucosylation of proteins with transcriptional regulation functions would increase the protein size; this could become an issue when the proteins are being transported inside the nucleus to perform their functions. However, it has been demonstrated that the neoglycoproteins BSA-fucose, BSA-mannose and BSA-glucose are rapidly transported inside the nuclei, whereas BSA itself is not [49]. This implies that fucosylation facilitates the translocation to the nuclei and maybe also towards other organelles. Sugar-mediated nuclear transport appears distinct from the basic peptide-mediated NLS pathway [49]. This must be corroborated with additional experiments.
According to the background in ovarian cancer and other types of cancer, the fucosylation process is altered. Therefore, we decided to evaluate the total level of fucosylation both, in permeabilized and non-permeabilized cells. Typically this type of glycosylation is normally established in the secretory pathway and then presented on the cell surface in mammals. We consider that regardless of whether fucosylation occurs in the secretory pathway, the visualization of the level of intracellular fucosylation could provide information on the total levels of fucosylation. Moreover, given that we are studying tumor cells, we believe that intracellular fucosylation could reflect alterations in the mechanisms of membrane secretion of fucosylated proteins, which do not move efficiently towards the cell surface. However, we found that at least for SKOV-3 cells, at 24 h the level of membrane fucosylation (nonpermeabilized cells) was very low; therefore intracellular fucosylation (permeabilized cells) was sought, all under the same experimental conditions. Our results suggest that while what we see inside the cell are proteins that have the membrane as their final destination, for some reason they do not reach it. In the same way, experiments with permeabilized cells allowed us to observe that although membrane fucosylation did not increase drastically, total fucosylation did so, suggesting an exacerbated fucosylation process and/or a deficiency of the fucosylated protein secretory system.
Nevertheless, why is fucosylation important? Protein fucosylation affects immune cell recognition, cell interactions, and general biology [50,51,52]. FX is an important enzyme for the de novo synthesis of fucose, and its deletion in mice induced a defect that impaired the self-renewal capacity of bone marrow hematopoietic stem cells [53]. In healthy conditions in the human body, this post-translational modification is an essential component of several blood group antigens, and it is implicated in host microbe interactions and in numerous ontogenetic events [54,55,56,57,58]. It also regulates cell adhesion molecules and growth factor receptors [59, 60]. Moreover, it has been implicated in pathological conditions, including cancer, where it has been shown to have a strong impact in different ways; for example, it has been associated with multidrug resistance in breast cancer cells [61], cell proliferation [62], cell migration and tumor formation [63]. In addition, there are reports that suggest that fucosylated glycans in neuroepithelial cells are required to guide the migration of neurons in the developing zebrafish hindbrain [64].
Additionally, some reports have indicated that a deficiency in fucosylation leads to TRAIL-induced apoptosis resistance [65]. TRAIL induces apoptosis in many types of human cancer cells but does not induce apoptosis in normal cells. Therefore, a decrease in membrane fucosylation could disfavor cell apoptosis as an evasion strategy.
Considering that there is enough literature describing the heterogeneity among ascitic fluids, the characterization of their content was beyond our reach. The purpose was to show that, in general terms, most of these ascitic fluids induce changes in the cells, which are relevant for the development and progression of the tumor. Of the 10 ascitic fluids analyzed, eight increased the level of fucosylation. These ascites are rich in cells from many sources including immunological, tumor, stromal, and other. However, as described in the materials and methods section, we processed these ascites to obtain ascites without cells before incubation with the cell lines. SKOV-3 cells that were stimulated with ascites for 72 h had a higher expression of fucosylation that correlated with the change in the phenotype acquired by these cells under ascites stimuli (epithelial-to-mesenchymal transition), which may increase the cellular migration. In fact, there are reports indicating that fucosylation is important during migration [64]. On the other hand, OVCAR-3 cells were also susceptible to ascitic fluids and their stimulation induced an increase in fucosylation levels. Moreover, OVCAR-3 cells showed a very different morphology (Supplementary Fig. 1c) compared with that of SKOV-3 cells stimulated with the same ascites. OVCAR-3 cells were rounded by ascites and, in some cases, the formation of spheroids was observed. This could also be related to a higher migration capacity and may lead to the establishment of a metastatic tumor. Additionally, high fucosylation in OVCAR-3 could be an important feature that confers resistance to cisplatin [66, 67].
OVCAR-3 cells were obtained from the malignant ascites of a patient with progressive adenocarcinoma of the ovary, and the cultured cells exhibit androgen and estrogen receptors (ATCC HTB 161). SKOV-3 is an ovarian cancer cell line derived from the ascites of a 64-year-old Caucasian female with an ovarian serous cystadenocarcinoma (ATCC HTB 77). Both are ovarian cancer cell lines, but it has been described that there may be cell-type and tissue-specific variations in glycosylation [68, 69].
At this time we do not dare to provide clinical relevance to our results. We only present experimental results that could become clinically relevant once a larger number of samples can be analyzed. Moreover, when a correlation analysis was made between the fucosylation and the different clinical characteristics of the patients, no statistically significant differences were found, probably because the number of samples is insufficient. However, we found an interesting association between fucosylation and the clinical stage.
The identification of fucosylated proteins that are associated with particular diseases (in this case, ovarian cancer) opens the possibility to analyze them for future applications. For example, some of the fucosylated proteins have been employed as biomarkers, such as haptoglobin and alpha fetoprotein, which are proteins that undergo central fucosylation, and that are used as tumor markers in hepatocellular cancer [70, 71]. Moreover, changes in the fucosylation of particular proteins could affect their interaction with other molecules; this would, in turn, alter their participation in different processes, including cell adhesion, differentiation, and signaling pathways. It is also known that these fucosylated antigens facilitate the invasion and spread of tumor cells [72,73,74,75]. In relation to cancer, a study conducted in 2016 by Mehta et al. [76] showed that the increase in central fucosylation is associated with the dedifferentiation of primary hepatocytes and with the appearance of markers that are indicative of the transition of the cells from an epithelial to a mesenchymal state [76]. Another study reported that the inhibition of the fucosylation of annexin A4 inhibited cell proliferation in ovarian clear cell carcinoma [77].
Studies with the serum of patients with ovarian cancer, where the type of glycans found in the glycoproteins was evaluated, found an increased expression level of tri- and tetra-antennal oligosaccharides with variations in sialylation and fucosylation [78]. Previous work in our laboratory has shown that high levels of fucosylation occur in biopsies from the tumors of patients diagnosed with EOC [20]. At the same time, other groups have highlighted the relevance of fucosylation in ovarian cancer (described above). Thus, we hypothesize that the altered fucosylation of the proteins identified here affects their function. In this context, it is known that some receptors and ligands lose their affinity, and this leads to alterations in the signaling pathways in which they participate [79]. Therefore, a more detailed analysis is necessary regarding the effects of fucosylation on the structure and function of the identified proteins, which are mainly transcriptional regulators; this implies that the proteins need to bind to specific protein domains to exert their functions [80].
The analysis of protein through the design of the network (Fig. 5) was to support the possible role of the identified proteins during the progression of the tumor instead of using them to understand the process of fucosylation itself. Alterations at the glycan level in some proteins have been used for therapeutic purposes, and this is one of the current strategies for cancer treatment [81, 82]. The deletion of a specific glycan or the modification of glycan chains with fucose or sialic acid on the antibodies enhances antibody-dependent cellular cytotoxicity, which is a key pathway in the elimination of cancerous tissue [83,84,85].
In conclusion, our results suggest that ascites from ovarian cancer patients are able to induce fucosylation on different cell types. Therefore, our results may be the foundation for further studies that will allow us to obtain evidence that supports the use of this information in the search for diagnostic biomarkers; our results may also be a foundation for the discovery of possible therapeutic targets or biomarkers for cancer prognosis and progression.
References
GLOBOCAN, 2018. Estimated cancer incidence and prevalence woldwide in 2018. [online] Available at: http://globocan.iarc.fr [Last access: 24th March 2019]
Auersperg N, Wong AS, Choi KC, Kang SK, Leung PC (2001) Ovarian surface epithelium: biology, endocrinology, and pathology. Endocr Rev 22(2):255–288
Cvetkovic D (2003) Early events in ovarian oncogenesis. Reprod Biol Endocrinol 1(68):68. https://doi.org/10.1186/1477-7827-1-68
Odicino F, Pecorelli S, Zigliani L, Creasman W (2008) History of the FIGO cancer staging system. Int J Gynecol Obstet 101(2):205–210. https://doi.org/10.1016/j.ijgo.2007.11.004
Pasquet M, Golzio M, Mery E, Rafii A, Benabbou N, Mirshahi P, Hennebelle I, Bourin P, Allal B, Teissie J, Mirshahi M, Couderc B (2010) Hospicells (ascites-derived stromal cells) promote tumorigenicity and angiogenesis. Int J Cancer 126(9):2090–2101. https://doi.org/10.1002/ijc.24886
Lane D, Bachvarov D, Rancourt C, Piche A (2014) Role of malignant ascites on human mesothelial cells and their gene expression profiles. BMC Cancer 14:288. https://doi.org/10.1186/1471-2407-14-288
Shender VO, Pavlyukov MS, Ziganshin RH, Arapidi GP, Kovalchuk SI, Anikanov NA, Altukhov IA, Alexeev DG, Butenko IO, Shavarda AL, Khomyakova EB, Evtushenko E, Ashrafyan LA, Antonova IB, Kuznetcov IN, Gorbachev AY, Shakhparonov MI, Govorun VM (2014) Proteome–metabolome profiling of ovarian Cancer ascites reveals novel components involved in intercellular communication. Mol Cell Proteomics 13(12):3558–3571. https://doi.org/10.1074/mcp.M114.041194
Bery A, Leung F, Smith CR, Diamandis EP, Kulasingam V (2014) Deciphering the ovarian cancer ascites fluid peptidome. Clin Proteomics 11(1):13. https://doi.org/10.1186/1559-0275-11-13
Pinho S, Reis C (2015) Glycosylation in cancer: mechanism and clinical implications. Nat Rev Cancer 15(9):540–555. https://doi.org/10.1016/j.neo.2018.06.001
Miyoshi E, Moriwaki K, Nakagawa T (2008) Biological function of fucosylation in cancer biology. J Biochem 143(6):725–729. https://doi.org/10.1093/jb/mvn011
Shah M, Telang S, Shah P, Patel P (2008a) Tissue and serum α2-3-and α2-6-linkage specific sialylation changes in oral carcinogenesis. Glycoconj J 25(3):279–290. https://doi.org/10.1007/s10719-007-9086-4
Shah M, Telang S, Raval G, Shah P, Patel PS (2008b) Serum fucosylation changes in oral cancer and oral precancerous conditions. Cancer J 113(2):336–346. https://doi.org/10.1002/cncr.23556
Vajaria B, Patel P (2016) Glycosylation: a halmark of cancer? Glycoconj J 34(2):147–156. https://doi.org/10.1007/s10719-016-9755-2
Hu Z, Cai M, Deng L, Zhu L, Gao J, Tan M, Liu J, Lin B (2016) The fucosylated CD147 enhances the autophagy in epithelial ovarian cancer cells. Oncotarget 7(50):82921–82932. https://doi.org/10.18632/oncotarget.13289
Zhuang H, Hu Z, Tan M, Zhu L, Liu J, Liu D, Yan L, Lin B (2014) Overexpression of Lewis y antigen promotes human epididymis protein 4-mediated invasion and metastasis of ovarian cancer cells. Biochimie 105:91–98. https://doi.org/10.1016/j.biochi.2014.06.022
Cai M, Jin S, Deng L, Zhu L, Hu Z, Liu D, Liu J, Tan M, Gao J, Wang H, Lin B (2017) Lewis y antigen promotes p27 degradation by regulating ubiquitin-proteasome activity. Oncotarget 8(66):110064–110076. https://doi.org/10.18632/oncotarget.22617
Liu J, Zheng M, Qi Y, Wang H, Liu M, Liu Q, Lin B (2018) Lewis(y) antigen-mediated positive feedback loop induces and promotes chemotherapeutic resistance in ovarian cancer. Int J Oncol 53(4):1774–1786. https://doi.org/10.3892/ijo.2018.4496
Thomson S, Dargan E, Turner GA (1992) Increased fucosylation and other carbohydrate changes in haptoglobin in ovarian cancer. Cancer Lett 66:43–48
Garibay-Cerdenares OL, Hernández-Ramírez VI, Osorio-Trujillo JC, Hernández-Ortíz M, Gallardo-Rincón D, Cantú de León D, Encarnación-Guevara S, Villegas-Pineda JC, Talamás-Rohana P (2014) Proteomic identification of fucosylated haptoglobin alpha isoforms in ascitic fluids and its localization in ovarian carcinoma tissues from Mexican patients. J Ovarian Res 7:27. https://doi.org/10.1186/1757-2215-7-27
Villegas-Pineda JC, Garibay-Cerdenares OL, Hernández-Ramírez VI, Gallardo-Rincón D, Cantú de León D, Pérez-Montiel-Gómez MD, Talamás-Rohana P (2015) Integrins and haptoglobin: molecules overexpressed in ovarian cancer. Pathol Res Pract 211(12):973–981. https://doi.org/10.1016/j.prp.2015.10.002
Yarnashita K, Korde N, Endo T, Iwaki Y, Kobata A (1989) Altered glycosylation of serum transferrin of patients with hepatocellular carcinoma. J Biol Chem 264:2415–2423
Aoyagi Y, Isernura M, Yosizawa Z, Suzuki Y, Sekine C, Ono T, Ichida F (1985) Fucosylation of serum o-fetoprotein in patients with primary hepatocellular carcinoma. Biochim Biophs Acta 830:217–223
Yamamoto K, Tsujii T, Tarutani O, Osawa T (1984) Structural changes of carbohydrate chains of human thyroglobulin accompanying tranformations of thyroid glands. Eur J Biochem 143:133–144
Toledo-Leyva A, Villegas-Pineda JC, Encarnación-Guevara S, Gallardo-Rincón D, Talamás Rohana P (2018) Effect of ovarian cancer ascites on SKOV-3 cells proteome: new proteins associated with aggressive phenotype in epithelial ovarian cancer. Proteome Sci 16:3. https://doi.org/10.1186/s12953-018-0133-9
Carduner L, Picot CR, Leroy-Dudal J, Blay L, Kellouche S, Carreiras F (2014) Cell cycle arrest or survival signaling through αv integrins, activation of PKC and ERK1/2 lead to anoikis resistance of ovarian cancer spheroids. Exp Cell Res 320(2):329–342. https://doi.org/10.1016/j.yexcr.2013.11.011
Runyon BA (1994) Care of patients with ascites. N Engl J Med 330:337–342. https://doi.org/10.1056/NEJM199402033300508
Shen-Gunther J, Mannel RS (2002) Ascites as a predictor of ovarian malignancy. Gynecol Oncol 87(1):77–83
Offner FA, Obrist P, Stadlmann S, Feichtinger H, Klingler P, Herold M, Zwierzina H, Hittmair A, Mikuz G, Abendstein B, Zeimet A, Marth C (1995) IL-6 secretion by human peritoneal mesothelial and ovarian cancer cells. Cytokine 7(6):542–547
Matte I, Lane D, Laplante C, Rancourt C, Piche A (2012) Profiling of cytokines in human epithelial ovarian cancer ascites. Am J Cancer Res 2(5):566–580
Matte I, Lane D, Bachvarov D, Rancourt C, Piche A (2014) Role of malignant ascites on human mesothelial cells and their gene expression profiles. BMC Cancer 14:288. https://doi.org/10.1186/1471-2407-14-288
Matte I, Lane D, Laplante C, Garde-Granger P, Rancourt C, Piche A (2015) Ovarian cancer ascites enhance the migration of patient-derived peritoneal mesothelial cells via cMet pathway through HGF-dependent and -independent mechanisms. Int J Cancer 137(2):289–298. https://doi.org/10.1002/ijc.29385
Bhowmick NA, Neilson EG, Moses HL (2004) Stromal fibroblasts in cancer initiation and progression. Nature 432(7015):332–337
Wels J, Kaplan RN, Rafii S, Lyden D (2008) Migratory neighbors and distant invaders: tumor-associated niche cells. Genes Dev 22(5):559–574. https://doi.org/10.1101/gad.1636908
Villegas-Pineda JC, Toledo-Leyva A, Osorio-Trujillo C, Hernández-Ramírez VI, Talamás-Rohana P (2017) The translational blocking of α5 and α6 integrin subunits affects migration and invasion, and increases sensitivity to carboplatin of SKOV-3 ovarian cancer cell line. Exp Cell Res 351(2):127–134. https://doi.org/10.1016/j.yexcr.2017.01.010
So KA, Min KJ, Hong JH, Lee JK (2015) Interleukin-6 expression by interactions between gynecologic cancer cells and human mesenchymal stem cells promotes epithelial–mesenchymal transition. Int J Oncol 47(4):1451–1459. https://doi.org/10.3892/ijo.2015.3122
Lane D, Matte I, Rancourt C, Piche A (2012) Osteoprotegerin (OPG) protects ovarian cancer cells from TRAIL-induced apoptosis but does not contribute to malignant ascites-mediated attenuation of TRAIL-induced apoptosis. J Ovarian Res 5(1):34. https://doi.org/10.1186/1757-2215-5-34
Reid PE, Brown NJ, Holen I (2009) Breast cancer cells stimulate osteoprotegerin (OPG) production by endothelial cells through direct cell contact. Mol Cancer 8:49. https://doi.org/10.1186/1476-4598-8-49
Yin J, Zeng F, Wu N, Kang K, Yang Z, Yang H (2015) Interleukin-8 promotes human ovarian cancer cell migration by epithelial-mesenchymal transition induction in vitro. Clin Transl Oncol 17(5):365–370. https://doi.org/10.1007/s12094-014-1240-4
Lane D1, Matte I, Rancourt C, Piché A (2011) Prognostic significance of IL-6 and IL-8 ascites levels in ovarian cancer patients. BMC Cancer 30(11):210. https://doi.org/10.1186/1471-2407-11-210
Giuntoli RL, Webb TJ, Zoso A, Rogers O, Diaz-Montes TP, Bristow RE, Oelke M (2009) Ovarian cancer-associated ascites demonstrates altered immune environment: implications for antitumor immunity. Anticancer Res 29:2875–2884
Terao N, Takamatsu S, Minehira T, Sobajima T, Nakayama K, Kamada Y, Miyoshi E (2015) Fucosylation is a common glycosylation type in pancreatic cancer stem cell-like phenotypes. World J Gastroenterol 21(13):3876–3887. https://doi.org/10.3748/wjg.v21.i13.3876
Syed V, Ulinski G, Mok SC, Ho SM (2002) Reproductive hormone-induced, STAT3-mediated interleukin 6 action in normal and malignant human ovarian surface epithelial cells. J Natl Cancer Inst 94:617–629
Obata NH, Tamakoshi K, Shibata K, Kikkawa F, Tomoda Y (1997) Effects of interleukin-6 on in vitro cell attachment, migration and invasion of human ovarian carcinoma. Anticancer Res 17:337–342
Nilsson MB, Langley RR, Fidler IJ (2005) Interleukin-6, secreted by human ovarian carcinoma cells, is a potent proangiogenic cytokine. Cancer Res 65:10794–10800. https://doi.org/10.1158/0008-5472.CAN-05-0623
Wang Y, Xu RC, Zhang XL, Niu XL, Qu Y, Li LZ, Meng XY (2012) Interleukin-8 secretion by ovarian cancer cells increases anchorage-independent growth, proliferation, angiogenic potential, adhesion and invasion. Cytokine 59(1):145–155. https://doi.org/10.1016/j.cyto.2012.04.013
Soon H, Moon A (2010) Epithelial-mesenchymal transition and cell invasion. Toxicol Res 26(4):245–252. https://doi.org/10.5487/TR.2010.26.4.245
Smith PL, Myers JT, Rogers CE, Zhou L, Petryniak B, Becker DJ, Homeister JW, Lowe JB (2002) Conditional control of selectin ligand expression and global fucosylation events in mice with a targeted mutation at the FX locus. J Cell Biol 158:801–815
Chao YB, Scovell WM, Yan SB (1994) High mobility group protein, HMG-1, contains insignificant glycosyl modification. Protein Sci 3(12):2452–2454. https://doi.org/10.1002/pro.5560031230
Hart GW, Haltiwanger RS, Holt GD, Kelly WG (1989) Glycosylation in the nucleus and cytoplasm. Annu Rev Biochem 58:841–874
Feizi T (2000) Carbohydrate-mediated recognition systems in innate immunity. Immunol Rev 173:79–88
Marth JD, Grewal PK (2008) Mammalian glycosylation in immunity. Nat Rev Immunol 8:874–887
Pickard JM, Maurice CF, Kinnebrew MA, Abt MC, Schenten D, Golovkina TV, Bogatyrev SR, Ismagilov RF, Pamer EG, Turnbaugh PJ, Chervonsky AV (2014) Rapid fucosylation of intestinal epithelium sustains host–commensal symbiosis in sickness. Nature 514:638–641. https://doi.org/10.1038/nature13823
Myers J, Huang Y, Wei L, Yan Q, Huang A, Zhou L (2010) Fucose-deficient hematopoietic stem cells have decreased self-renewal and aberrant marrow niche occupancy. Transfusion 50:2660–2669. https://doi.org/10.1111/j.1537-2995.2010.02745.x
Soochi K, Boyun K, Sang Song Y (2016) Ascites modulates cancer cell behavior, contributing to tumor heterogeneity in ovarian cancer. Cancer Sci 107(9):1173–1178. https://doi.org/10.1111/cas.12987
Larsen RD, Ernst LK, Nair RP, Lowe JB (1990) Molecular cloning, sequence, and expression of a human GDP-L-fucose:beta-D-galactoside 2-alpha-L-fucosyltransferase cDNA that can form the H blood group antigen. Proc Natl Acad Sci U S A 87:6674–6678
Kelly RJ, Ernst LK, Larsen RD, Bryant JG, Robinson JS, Lowe JB (1994) Molecular basis for H blood group deficiency in Bombay (oh) and Para-Bombay individuals. Proc Natl Acad Sci U S A 91:5843–5847
Hooper LV, Gordon JI (2001) Glycans as legislators of host-microbial interactions: spanning the spectrum from symbiosis to pathogenicity. Glycobiol 11:1R–10R. https://doi.org/10.1093/glycob/11.2.1R
Tonetti M, Sturla L, Bisso A, Benatti U, De Flora A (1996) Synthesis of GDP-L fucose by the human FX protein. J Biol Chem 271(44):27274–27279
Lowe JB (1997) Selectin ligands, leukocyte trafficking, and fucosyltransferase genes. Kidney Int 51(5):1418–1426
Wang Y, Shao L, Shi S, Harris RJ, Spellman MW, Stanley P, Haltiwanger RS (2001) Modification of epidermal growth factor-like repeats with O-fucose. Molecular cloning and expression of a novel GDP-fucose protein O-fucosyltransferase. J Biol Chem 276(43):40338–40345
Feng X, Zhao L, Gao S, Song X, Dong W, Zhao Y, Zhou H, Cheng L, Miao X, Jia L (2016) Increased fucosylation has a pivotal role in multidrug resistance of breast cancer cells through miR-224-3p targeting FUT4. Gene 578(2):232–241. https://doi.org/10.1016/j.gene.2015.12.028
Carrascal MA, Silva M, Ramalho JS, Pen C, Martins M, Pascoal C, Amaral C, Serrano I, Oliveira MJ, Sackstein R, Videira PA (2017) Inhibition of fucosylation in human invasive ductal carcinoma reduces E-selectin ligand expression, cell proliferation and ERK1/2 and p38 MAPK activation. Mol Oncol 2(5):579–593. https://doi.org/10.1002/1878-0261.12163
Zhou Y, Fukuda T, Hang Q, Hou S, Isaji T, Kameyama A, Gu J (2017) Inhibition of fucosylation by 2-fluorofucose suppresses human liver cancer HepG2 cell proliferation and migration as well as tumor formation. Sci Rep 7(1):11563. https://doi.org/10.1038/s41598-017-11911-9
Ohata S, Kinoshita S, Aoki R, Tanaka H, Wada H, Tsuruoka-Kinoshita S, Tsuboi T, Watabe S, Okamoto H (2009) Neuroepithelial cells require fucosylated glycans to guide the migration of vagus motor neuron progenitors in the developing zebrafish hindbrain. Development 136(10):1653–1663. https://doi.org/10.1242/dev.033290
Moriwaki K, Narisada M, Imai T, Shinzaki S, Miyoshi E (2010) The effect of epigenetic regulation of fucosylation on TRAIL-induced apoptosis. Glycoconj J 27(7–9):649–659. https://doi.org/10.1007/s10719-010-9310-5
Blanas A, Sahasrabudhe NM, Rodríguez E, van Kooyk Y, van Vliet SJ (2018) Fucosylated antigens in Cancer: an Alliance toward tumor progression, metastasis, and resistance to chemotherapy. Front Oncol 23(8):39. https://doi.org/10.3389/fonc.2018.00039
Lv X, Song J, Xue K, Li Z, Li M, Zahid D, Cao H, Wang L, Song W, Ma T, Gu J, Li W (2019) Core fucosylation of copper transporter 1 plays a crucial role in cisplatin-resistance of epithelial ovarian cancer by regulating drug uptake. Mol Carcinog 58:1–14. https://doi.org/10.1002/mc.22971
Anugraham M, Jacob F, Nixdorf S, Everest-Dass AV, Heinzelmann-Schwarz V, Packer NH (2014) Specific glycosylation of membrane proteins in epithelial ovarian cancer cell lines: glycan structures reflect gene expression and DNA methylation status. Mol Cell Proteomics 13(9):2213–2232. https://doi.org/10.1074/mcp.M113.037085
Medzihradszky KF, Kaasik K, Chalkley RJ (2010) Tissue-specific glycosylation at the Glycopeptide level. Mol Cell Proteomics 14(8):2103–2110. https://doi.org/10.1074/mcp.M115.050393
Aoyagi Y, Isemura M, Yosizawa Z, Suzuki Y, Sekine C, Ono T, Ichida F (1985) Fucosylation of serum alpha fetoprotein in patients with primary hepatocellular carcinoma. Biochim Biophys Acta 830(3):217–223
Noda K, Miyoshi E, Kitada T, Nakahara S, Gao CX, Honke K, Shiratori Y, Moriwaki H, Sasaki Y, Kasahara A, Hori M, Hayashi N, Taniguchi N (2002) The enzymatic basis for the conversion of non-fucosylated to fucosylated alpha-fetoprotein by acyclic retinoid treatment in human hepatoma cells: activation of alpha 1,6 fucosyltransferase. Tumor Biol 23(4):202–211
Yan LM, Lin B, Zhu LC, Hao YY, Qi Y, Wang CZ, Gao S, Liu SC, Zhang SL, Iwamori M (2010) Enhancement of the adhesive and spreading potentials of ovarian carcinoma rmg-1 cells due to increased expression of integrin alpha5beta1 with the Lewis y-structure on transfection of the alpha1,2-fucosyltransferase gene. Biochim 92(7):852–857. https://doi.org/10.1016/j.biochi.2010.02.012
Gao L, Yan L, Lin B, Gao J, Liang X, Wang Y, Liu J, Zhang S, Iwamori M (2011) Enhancive effects of Lewis y antigen on cd44-mediated adhesion and spreading of human ovarian cancer cell line rmg-i. J Exp Clin Cancer Res 30:15. https://doi.org/10.1186/1756-9966-30-15
Wang X, Gu J, Ihara H, Miyoshi E, Honke K, Taniguchi N (2006) Core fucosylation regulates epidermal growth factor receptor-mediated intracellular signaling. J Biol Chem 281(5):2572–2577
Orczyk-Pawilowicz M (2007) The role of fucosylation of glycoconjugates in health and disease. Postepy Hig Med Dosw (Online) 61:240–252
Mehta A, Comunale MA, Rawat S, Casciano JC, Lamontagne J, Herrera H, Ramanathan A, Betesh L, Wang M, Norton P, Steel LF, Bouchard MJ (2016) Intrinsic hepatocyte dedifferentiation is accompanied by upregulation of mesenchymal markers, protein sialylation and core alpha 1,6 linked fucosylation. Sci Rep 6:27965. https://doi.org/10.1038/srep27965
Wang H, Deng L, Cai M, Zhuang H, Zhu L, Hao Y, Gao J, Liu J, Li X, Lin B (2016) Annexin A4 fucosylation enhances its interaction with the NF-kB p50 and promotes tumor progression of ovarian clear cell carcinoma. Oncotarget 8(64):108093–108107. https://doi.org/10.18632/oncotarget.10226
Mitra I, Alley WR Jr, Goetz JA, Vasseur JA, Novotny MV, Jacobson SC (2013) Comparative profiling of N-glycans isolated from serum samples of ovarian cancer patients and analyzed by microchip electrophoresis. Proteome Res 12(10):4490–4496. https://doi.org/10.1021/pr400549e
Wang Y, Fukuda T, Isaji T, Lu J, Im S, Hang Q, Gu W, Hou S, Ohtsubo K, Gu J (2015) Loss of α1,6-fucosyltransferase inhibits chemical-induced hepatocellular carcinoma and tumorigenesis by down-regulating several cell signaling pathways. FASEB J 29(8):3217–3227. https://doi.org/10.1096/fj.15-270710
Phillips T (2008) Regulation of transcription and gene expression in eukaryotes. Nature Education 1(1):199
Prendergast JM, Galvao da Silva AP, Eavarone DA, Ghaderi D, Zhang M, Brady D, Wicks J, DeSander J, Behrens J, Rueda BR (2017) Novel anti-Sialyl-Tn monoclonal antibodies and antibody-drug conjugates demonstrate tumor specificity and anti-tumor activity. MAbs 9(4):615–627. https://doi.org/10.1080/19420862.2017.1290752
Bassoy EY, Kasahara A, Chiusolo V, Jacquemin G, Boydell E, Zamorano S, Riccadonna C, Pellegatta S, Hulo N, Dutoit V, Derouazi M, Dietrich PY, Walker PR, Martinvalet D (2017) ER-mitochondria contacts control surface glycan expression and sensitivity to killer lymphocytes in glioma stem-like cells. EMBO 36(11):1493–1512. https://doi.org/10.15252/embj.201695429
Sato Y, Isaji T, Tajiri M, Yoshida-Yamamoto S, Yoshinaka T, Somehara T, Fukuda T, Wada Y, Gu J (2009) An N-glycosylation site on the β-propeller domain of the integrin α5 subunit plays key roles in both its function and site-specific modification by β1, 4-N-acetylglucosaminyltransferase III. J Biol Chem 284(18):11873–11881. https://doi.org/10.1074/jbc.M807660200
Shields RL, Lai J, Keck R, O'Connell LY, Hong K, Meng YG, Weikert SH, Presta LG (2002) Lack of fucose on human IgG1 N-linked oligosaccharide improves binding to human fc gamma RIII and antibody-dependent cellular toxicity. J Biol Chem 277(30):26733–26740
Shinkawa T, Nakamura K, Yamane N, Shoji-Hosaka E, Kanda Y, Sakurada M, Uchida K, Anazawa H, Satoh M, Yamasaki M, Hanai N, Shitara K (2003) The absence of fucose but not the presence of galactose or bisecting N-acetylglucosamine of human IgG1 complex-type oligosaccharides shows the critical role of enhancing antibody-dependent cellular cytotoxicity. J Biol Chem 278(5):3466–3473
Acknowledgments
We gratefully acknowledge Gabriel Martinez Batallar for mass spectrometry analysis at the laboratory of Dr. Sergio Encarnación Guevara from Centro de Ciencias Genómicas, UNAM, Mexico; Jessica Márquez Dueñas for her help in the purchase and procurement of materials.
Funding
This project was supported by a grant for Health Research (233739), from CONACYT and the Health Ministry, México; DRAA was a recipient of a PhD fellowship also from CONACYT, México (338858).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Figure 1
Time course of fucosylation of SKOV-3 cells treated with ascites 01 and 02. The graphs show the time course of fucosylation (arbitrary units of fluorescence expressed as fluorescence intensity) in nonpermeabilized (left panel) and permeabilized (right panel) cells. The data of three independent experiments were analyzed by one-way ANOVA in GraphPad Prism 7. (JPG 45 kb)
Supplementary Figure 2
Ascites modulate fucosylation expression in different cell lines. Membrane fucosylation was analyzed in SKOV-3 (a), OVCAR-3 (b), and Caco-2 and BEAS-2B (d) cell lines. Ascites from 10 different ovarian cancer patients were employed to stimulate the cells for 24 h. The samples were analyzed by confocal microscopy. Fucosylation was detected by biotinylated Aleuria aurantia lectin (1:200 dilution) and FITC-conjugated streptavidin (1:200 dilution). The nuclei were stained with DAPI. The selected images are representative of two independent biological replicates. The graphs show the level of fucosylation (arbitrary units of fluorescence expressed as fluorescence intensity) in SKOV-3 cells (a, lower panel) and OVCAR-3 cells (b, lower panel). The data of two independent experiments were analyzed by one-way ANOVA in GraphPad Prism 7. (JPG 2277 kb)
Supplementary Figure 3
Schematic representation of probable sites of N-glycosylation. By in silico analysis, the probable N-glycosylation sites were predicted using the NetNGlyc database. Each of the analyzed proteins is represented (each canonical isoform); the red arrows indicate the site in which the probable glycosylation may occur. The position and the value assigned are indicated next to the arrows. A potential crossing 0.5 threshold predicts glycosylation. (JPG 112 kb)
Supplementary Table 1
(DOCX 17 kb)
Rights and permissions
About this article
Cite this article
Alberto-Aguilar, D.R., Hernández-Ramírez, V.I., Osorio-Trujillo, J.C. et al. Ascites from Ovarian Cancer Induces Novel Fucosylated Proteins. Cancer Microenvironment 12, 181–195 (2019). https://doi.org/10.1007/s12307-019-00227-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12307-019-00227-z