Abstract
Purpose
Increasing demands on skills with mounting pressures from expectations from arthroscopic anterior cruciate ligament (ACL) reconstructions requires precise knowledge of technical details by surgeons. One such element is the minimum length of graft in femoral tunnel to allow for adequate tendon-to-bone healing and early return to activities and sports. This has, however, remained an unanswered question.
Purpose
To study and compare clinico-radiological outcomes of ACL reconstructions in patients with < 20 mm of intra-femoral tunnel graft length with those measuring ≥ 20 mm.
Methods
All eligible patients undergoing arthroscopic ACL reconstruction were sequentially divided into two groups based on the intra-femoral tunnel graft lengths (A: < 20 mm, n = 27; and B: ≥ 20 mm, n = 25). Exclusions were made for those > 45 years of age, with chondral and/or multi-ligamentous injuries and with systemic pathologies. All patients were postoperatively evaluated in clinics by physical examination and functional scoring (Lysholm and modified Cincinnati scores) at 3, 6 and 12-month intervals. Graft vascularity was assessed by signal-to-noise quotient ratio (SNQR) using magnetic resonance imaging (MRI) at 3 and 12 months.
Results
No significant differences were noted in mean Lysholm and modified Cincinnati scores between the two groups at the end of 1 year. There were also no significant differences in graft maturation over time and SNQR at 3 and 12 months in the region of interest (ROI).
Conclusions
Intra-femoral tunnel graft length of less than 20 mm does not compromise early clinical and functional outcomes of ACL reconstructions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Arthroscopic anterior cruciate ligament reconstruction (ACLR) has established its place as the procedure of choice for ACL injuries [1, 2]. Its success, however, relies heavily on technical specifics. Errors of the femoral tunnel alone are implicated in over 45% failures, making it the single, biggest technical challenge [3]. The anteromedial portal (AMP) for femoral tunnel preparation allows anatomical and stable tunnel placement [4,5,6,7,8]. An undesired consequence of this, however, is a shorter length of the femoral tunnel. This leads to a shorter intra-femoral tunnel graft (IFTG) length than that would be achieved with an oblique, longer tunnel. A short IFTG can also emerge as a problem when thin grafts need quadrupling to provide larger diameters at the cost of pruning intra-tunnel graft lengths. This can raise concerns with graft-to-bone healing and graft stiffness [9, 10]. A literature search on this important aspect of tunnel lengths and its impact on healing and ligamentisation yielded very few results [9,10,11,12,13].
The optimal IFTG length has been postulated by some to be between 14 and 35 mm [14]. Others have found IFTG lengths < 20 mm to be associated with increased postoperative laxity, in the form of positive Lachman tests 2 years postoperatively (p = 0.025) [15]. Radiologically, laxity, healing and graft incorporation have been assessed with magnetic resonance imaging (MRI) [16]. Despite the above findings, there is clearly, a lack of consensus on the ideal IFTG length at present. Also, the minimum IFTG length to prevent postoperative laxity and failure remains an open debate. While two animal studies have looked at addressing this important lacuna, only one prospective human study, thus far, has attempted to assess the impact of a short graft inside the femoral tunnel [9, 10, 12, 13].
This study attempted to cater to this gap in knowledge about the optimum length of graft within the femoral tunnel and to address doubts regarding a minimum “safe length” of the same. A single-surgeon, single-technique, prospective study was designed comparing the functional and radiological outcomes of ACLR at 3, 6 and 12 months postoperatively among patients with IFTG lengths < 20 mm and ≥ 20 mm. Radiological assessment of healing and ligamentisation were assessed by comparing preoperative MRI with postoperative scans done at 3 and 12 months. It was hypothesised that there would be similar outcomes in both groups of patients.
Materials and methods
Rationale, settings and ethical clearances Using a PICO (Patient, Intervention, Comparison and Outcome) strategy, the research question was formulated and literature review performed across various databases (Medline, PubMed, EMBASE and CINAHL) [11]. A hospital-based, prospective cohort study was conducted between November 2016 to November 2017 after obtaining necessary Ethical Committee clearance (vide clearance letter no. EC/09/15/896). Informed consents were duly obtained from all eligible candidates. A 22-point Strengthening the Reporting of Observational studies in Epidemiology (STROBE) checklist was employed to construct the research and for manuscript preparation.
Patient selection and evaluation Fifty-two consecutive patients requiring ACL reconstruction were prospectively enrolled during the study period. Exclusions were made for those > 45 years of age, with chondral and/or multi-ligamentous injuries and with systemic pathologies. Pre- and postoperative (3, 6 and 12 months) clinical evaluation of the knee employed the standard Lachman, anterior drawer, pivot and McMurray tests, while functional assessment was performed using the Lysholm knee scoring system [17] and the modified Cincinnati knee rating systems [18].
Patients underwent pre- and postoperative (3 and 12 months) MRI scans (Siemens Verio 3 T MRI) with 3 mm slices (0.5 mm gap between each) in sagittal and coronal planes using proton density fast spin-echo (PDFSE) and turbo inversion recovery magnetization (TIRM). Graft incorporation and healing in terms of load-to-failure, stiffness, and tensile strength were assessed on MRI by measuring the signal-to-noise quotient ratio (SNQR = signal from ROI in graft/ signal from background) [16]. Regions of interest (ROI) included femoral insertion, mid-substance, tibial insertion, tibial tunnel, PCL and background signal.
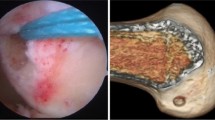
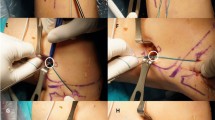
Surgical routine, follow-up and evaluation A single senior surgeon at a multispecialty tertiary care teaching centre performed all procedures using principles of anatomical single bundle ACL reconstruction. Under tourniquet control, ipsilateral semitendinosus and gracilis (STG) grafts were harvested, Bunnel-stitched, tensioned and doubled. Tunnel measurements were made and femoral tunnels drilled through the anteromedial portal (AMP). Graft length was calculated using the equation; intra-femoral tunnel graft length (IFTG) = length of femoral tunnel drilled—size of closed loop used. Femoral fixation was performed using continuous closed loop fixation device with minimum size of 15 mm. Thus, in patients with femoral tunnels measuring < 35 mm, the consequent IFTG was < 20 mm. This group of patients was classified as Group A and those with IFTGs ≥ 20 were labelled as Group B. Tibial fixation was performed using bioabsorbable interference screws with the knee in 30º flexion. Routine closure was performed and knee brace for 2 weeks allowed controlled movement. All patients were clinically reviewed at 3 months, 6 months and 1 year and MRIs were performed.
Groups A (length < 20 mm) and B (length ≥ 20 mm) consisted of 27 and 25 patients, respectively. Subjective assessment of all the patients was made at decided interval and functional scores were calculated. At the end of one year Lysholm scores, modified Cincinnati scores and SNQRs of both groups were compared.
Rehabilitation protocol A 3-phase approach was adopted. The first phase involved protecting the graft with a knee brace for 6 weeks with partial weight-bearing mobilisation with crutches for 3 weeks. Constant supervision of a dedicated physiotherapy team was provided. Gentle progression to full ROM was carried out from week 2 onwards with a focus on stretching and strengthening exercises. Phase 2 focussed on restoring full muscle strength with progression to full weight-bearing without a brace. While protecting the graft, full ROM and quadriceps, hamstrings and hip strengthening exercises were advocated. Emphasis was also laid on stability and balance training. The final phase lasted several months and focussed on the restoration of unimpeded ROM, muscle strength and agility for return to full activity and sports.
Statistical analysis The sample size was determined by using the Lysholm score to compare the effectiveness between the two groups. Assuming significance at a 10-point difference, a minimum of 25 patients/group were required to detect statistical significance. This was based on an effect size of 1.0 at alpha 0.05, power of 90% and assuming a dropout rate of 20%. The formula for the calculated sample size is given below
Zα/2: critical value of normal distribution at α/2,
Zβ: critical value of normal distribution at β.
σ1 and σ2: standard deviations of the two groups.
M1 and M2: means of the two groups.
Statistical Package for Social Sciences (SPSS) version 20.0 (IBM SPSS Armonk, NY: IBM Corp) was employed and data were checked for normality. Normally distributed continuous variables were compared using the unpaired t test, whereas the Mann–Whitney U test was used for those variables that were not normally distributed. Categorical variables were analysed using either the chi-square test or Fisher’s exact test. For all statistical tests, a p value < 0.05 was taken to indicate a statistically significant difference.
Results
Group A with IFTG lengths < 20 mm had 27 patients while group B with IFTG lengths ≥ 20 mm had 25 patients (Fig. 1). Both the groups were followed up for 1 year and one patient in each group was lost to follow-up (after 6 months). Both groups were comparable w.r.t. demographic characters (Table 1). Patients with greater IFTG lengths (group B) had higher mean body weights (p = 0.014). Around half of the patients in both groups had sporting activities responsible for the mechanism of injuries. The remainder had either a road traffic accident as a cause of injury or other miscellaneous events. The incidence of meniscal injury was similar in both groups [16 patients (59.3%) in group A and 12 patients (48%) in group B] (Table 2).
Shorter IFTG lengths in group A patients corresponded with significantly shorter mean femoral tunnel lengths (p < 0.0001). Total graft lengths, femoral and tibial tunnel diameters were, however, not significantly different between both groups. Similar mean femoral tunnel diameters of 7.35 mm in group A and 7.64 mm in group B (p = 0.23) and mean tibial tunnel diameters (7.94 mm in group A and 8.06 mm in group B, p = 0.60) implied that graft lengths were not compromised in any group to enhance graft diameter (Table 3). At 3 months, two patients (7.4%) in group A and one patient (4%) in group B had grade 1 Lachman, which increased at 12 months to four patients (14.8%) in group A and three patients (12%) in group B, though pivot test was negative in all patients at all times postoperatively. This could suggest mild graft relaxation with time (Fig. 2).
Lysholm scores were comparable in both groups at all times (p = 0.06, 0.49 and 0.32 at 3 m, 6 m and 12 m, respectively) and at the end of 1 year both groups had excellent function (Fig. 2). Similar trends were reflected in modified Cincinnati scores between both groups at 3, 6 and 12 months (P3 = 0.631, P6 = 0.162, P12 = 0.867) (Fig. 3). Functional assessment of ROM at 1 year showed very good outcomes in both groups with ROM ≥ 130º in all except two patients in each group. These four patients had values ranging between 125 and 130º. None of the patients had an ROM of < 125º.
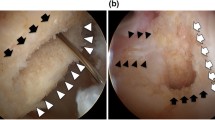
MRI at 3 months and 1 year revealed that all femoral and tibial tunnels were placed in anatomically acceptable positions. There was no evidence of cyclops lesions, graft impingement and cystic or mucoid degeneration (Fig. 4). In all the 102 scans performed, grafts were found to be intact with no discontinuities. Both groups were comparable in terms of the SNQR at all the ROIs (Table 4) at 3 and 12-months postoperatively. Specifically analysing the femoral tunnel signal in both groups, the mean SNQR at 3 months in group A was 1.41 and at 12 months was 1.31 (p = 0.68). These respective values for group B were 1.12 and 0.97 (p = 0.61) (Fig. 5) showing a fall (not statistically significant though) in signal in both groups from 3 to 12 months. This could possibly indicate withdrawal of vascularity and the onset of graft maturation at the femoral tunnels (Figs. 6 and 7).
Discussion
This study addresses the specific scepticism of small intra-femoral tunnel graft lengths and whether not crossing the number 20 mm would result in early failure of surgery. Important findings suggest comparable survivorship, function and outcomes of ACLRs done in patients irrespective of graft lengths. This asserts that as long as sound surgical principles and uniform techniques are followed, outcomes are not affected by IFTG lengths.
Thus far, only two human studies have tried looking at this peculiar, yet frequently encountered situation. They have tried defining the appropriate graft length in the femoral tunnel. The Multi-centre Orthopaedic Outcome Network (MOON) cohort study [14] was the first human study that retrospectively reviewed the data. They used both ST4 and STG grafts performed by three different surgeons using both transtibial and transportal techniques. Assessing them only on clinical data retrospectively, they inferred no difference in outcome after 2 years between patients with IFTG lengths of greater than or less than 25 mm. Another retrospective study by Guglielmetti et al. [15] compared clinical outcomes of different hamstring graft lengths (≤ 1.5 vs > 1.5 cm; ≤ 2 vs > 2 cm; ≤ 2.5 vs > 2.5 cm; and ≤ 1.5 vs > 2.5 cm.) in the femoral tunnel and reported no difference in clinical outcomes at 24 months. A potential confounding factor in the study was the employment of two different femoral drilling techniques (transtibial and transportal). This could have led to different anatomical dimensions with each approach and, thereby, different intra-tunnel lengths and outcomes of the hamstring grafts.
There have been a few animal studies evaluating this interesting conflict. Zantop et al. [9] in a goat model showed no difference in stiffness, ultimate failure load and kinematics between femoral graft lengths of 15 and 25 mm. Yuan et al. [13] in beagles looked at various graft lengths within femoral tunnels varying from 5 to 25 mm. They showed that maximum tensile strength and stiffness increase with time irrespective of graft length for specimens with graft length inside the bone tunnel of 17 mm or more. In this aspect, the findings of the present study are in agreement with those of Yuan et al. [13] as clinical and radiological outcomes were similar in patients with IFTG lengths of > 20 mm and of 15–20 mm. The hypothesis of the present study, therefore, proved true as the outcomes between both groups were comparable at different time frames.
Ours was a prospective study looking at the femoral tunnel length issue more precisely by keeping variables as minimum as possible to minimise bias. The surgeon, surgical technique including the choice of graft, graft harvest, tunnel drilling, graft fixation, rehabilitation and follow-up protocols were similar for all patients. Additionally, clinical results were recorded at similar intervals and examination findings were radiologically validated with MRI at 3 and 12 months. Outcomes in both groups of patients were similar at the follow-up periods leading up to and including the 1-year mark. This corroborated well with the assessment of laxity using the Lachman and pivot tests, and the Lysholm and modified Cincinnati scores. Also, MRI evaluation at 12 months in both groups showed signal quality synonymous with good graft survival without complications like ruptures, infections or cyclops lesions. SNQRs between the two groups at the femoral and tibial insertions and mid-substance were comparable at 3 and 12 months (Table 4). These indicate similar developments in graft vascularity, cellular proliferation and graft maturation between groups A (< 20 mm) and B (≥ 20 mm) [19,20,21,22].
Pertaining specifically to the femoral tunnel, the mean SNQRs at the femoral insertion dipped by 8–14% from the 3rd month to the 1-year mark in in both groups. This regression of vascularity, cellular proliferation and finally decreased signal intensity signifying the onset of maturation. This finding echoes the observation of Murakami et al. [23] who concluded that decreasing MR signal intensity over time was useful in evaluating graft maturation. These observations have been seconded by a Chinese study by Li et al. [24] who compared MRI findings of ACL reconstruction with allografts and autografts and concluded that the latter had superior maturity 2 years postoperatively based on lower SNQRs in different ROIs. Thus, high graft signal intensity (high SNQR) is an established indicator of inferior graft maturity. In both groups, in the present study, the mean 12-month values of the SNQRs were lower than the 3-month ones. Although these did not reach statistical significance, a longer follow-up of 2 years or more, similar to the findings of Li et al., could have illustrated statistical significance by a further drop in the respective SNQRs [24].
The tibial insertion at 12 months in both groups demonstrated increased SNQRs which would suggest persistent vascular and proliferative changes at this time. Kulczycka et al. [19] and White et al. [25] in their studies discussed that the maturation phase of a graft can range from 1 to 3 years. It is perhaps in this phase that the grafts had been subject to the 12-month MRI in our patients. Notwithstanding this, functional results between both groups were excellent and comparable at 1-year follow-ups and thus group A patients (IFTG lengths < 20 mm) were found to be enjoying similar satisfactory outcomes as group B (IFTG lengths ≥ 20 mm). This is in agreement with the findings of Li et al., Saupe et al. and Biercevicz et al. [26,27,28]. They concluded that MRI-based graft maturity may not accurately predict clinical and functional outcomes in 1-year follow-ups and that the functional result and return to sporting activities may still be good even when MRI variables does not show full maturity at 1 year post-ligament reconstruction.
The above-noted disparity in graft maturation at the femoral and tibial ends can be possibly explained by the difference in nature of graft fixation. Suspensory fixation of the femoral grafts allows 360º contact with host bone leading to enhanced Sharpey’s fibre formation when compared to the interference screw fixation on the tibial side. The graft in the latter only receives limited contact with the host tibial tunnel. This phenomenon has been observed by Colombet et al. and Smith et al. [29, 30] who concluded that suspensory fixation provides more favourable conditions for graft incorporation and maturation vis-à-vis interference screws. The MRI signal intensity was consequently superior with the former technique.
The limitations of the present study would include small sample size and a short follow-up period. Graft maturation, signified by a fall in the SNQRs on MRI, is a continuous process that may still be occurring at 12 months postoperatively. A longer follow-up could have, perhaps, allowed for a stronger statistical correlation between the falling SNQRs and graft maturity.
Based on the findings of this study, the authors believe that graft lengths between 13 and 25 mm have similar functional and radiological outcomes up to 1-year after surgery. Further work in this regard would be required with larger participants so that outcomes with even smaller graft lengths can be effectively compared and translated into clinical practice.
Conclusion
The findings of this study illustrate that satisfactory outcomes are expected with ACLR irrespective of intra-femoral tunnel graft length of the hamstring graft. The clinical findings have been validated with radiological data. Longer follow-ups will help ascertain complete ligamentisation and further consolidate the basis established by this study.
Data availability
Yes.
Code availability
Custom.
References
Alentorn-Geli E, Samitier G, Álvarez P, Steinbacher G, Cugat R (2010) Anteromedial portal versus transtibial drilling techniques in ACL reconstruction: a blinded cross-sectional study at two- to five-year follow-up. Int Orthop 34:747–754. https://doi.org/10.1007/s00264-010-1000-1
Duquin T, Wind W, Fineberg M et al (2009) Current trends in anterior cruciate ligament reconstruction. J Knee Surg 22:7–12. https://doi.org/10.1055/s-0030-1247719
Morgan J, Dahm D, Levy B, Stuart M (2012) Femoral tunnel malposition in ACL revision reconstruction. J Knee Surg 25:361–368. https://doi.org/10.1055/s-0031-1299662
Harner C, Honkamp N, Ranawat A (2008) Anteromedial Portal Technique for Creating the Anterior Cruciate Ligament Femoral Tunnel. Arthroscopy 24:113–115. https://doi.org/10.1016/j.arthro.2007.07.019
Bedi A, Altchek D (2009) The “Footprint” anterior cruciate ligament technique: an anatomic approach to anterior cruciate ligament reconstruction. Arthroscopy 25:1128–1138. https://doi.org/10.1016/j.arthro.2009.03.008
Bedi A, Musahl V, Steuber V et al (2011) Transtibial versus anteromedial portal reaming in anterior cruciate ligament reconstruction: an anatomic and biomechanical evaluation of surgical technique. Arthroscopy 27:380–390. https://doi.org/10.1016/j.arthro.2010.07.018
Gavriilidis I, Motsis E, Pakos E et al (2008) Transtibial versus anteromedial portal of the femoral tunnel in ACL reconstruction: a cadaveric study. Knee 15:364–367. https://doi.org/10.1016/j.knee.2008.05.004
Hussein M, van Eck C, Cretnik A et al (2012) Individualized anterior cruciate ligament surgery. Am J Sports Med 40:1781–1788. https://doi.org/10.1177/0363546512446928
Zantop T, Ferretti M, Bell K et al (2008) Effect of tunnel-graft length on the biomechanics of anterior cruciate ligament-reconstructed knees. Am J Sports Med 36:2158–2166. https://doi.org/10.1177/0363546508320572
Cavaignac E, Marot V, Faruch M et al (2017) Hamstring graft incorporation according to the length of the graft inside tunnels. Am J Sports Med 46:348–356. https://doi.org/10.1177/0363546517733472
Richardson WS, Wilson MC, Nishikawa J, Hayward RS (1995) The well-built clinical question: a key to evidence-based decisions. ACP J Club 123(3):A12–A13
Yamazaki S, Yasuda K, Tomita F et al (2006) The effect of intraosseous graft length on tendon-bone healing in anterior cruciate ligament reconstruction using flexor tendon. Knee Surg Sports Traumatol Arthrosc 14:1086–1093. https://doi.org/10.1007/s00167-006-0110-1
Yuan F, Zhou W, Cai J et al (2013) Optimal graft length for anterior cruciate ligament reconstruction: a biomechanical study in beagles. Orthopedics 36:e588–e592. https://doi.org/10.3928/01477447-20130426-20
Mariscalco M, Magnussen R, Mitchell J et al (2014) How much hamstring graft needs to be in the femoral tunnel? A MOON cohort study. Eur Orthop Traumatol 6:9–13. https://doi.org/10.1007/s12570-014-0275-x
Guglielmetti L, Shimba L, do Santos L, et al (2017) The influence of femoral tunnel length on graft rupture after anterior cruciate ligament reconstruction. J Orthop Traumatol 18:243–250. https://doi.org/10.1007/s10195-017-0448-9
Weiler A, Peters G, Mäurer J et al (2001) Biomechanical properties and vascularity of an anterior cruciate ligament graft can be predicted by contrast-enhanced magnetic resonance imaging. Am J Sports Med 29:751–761. https://doi.org/10.1177/03635465010290061401
Tegner Y, Lysholm J (1985) Rating systems in the evaluation of knee ligament injuries. Clin Orthop Relat Res 198:43–49
Agel J, LaPrade R (2009) Assessment of Differences Between the Modified Cincinnati and International Knee Documentation Committee Patient Outcome Scores. Am J Sports Med 37:2151–2157. https://doi.org/10.1177/0363546509337698
Kulczycka P, Larbi A, Malghem J et al (2015) Imaging ACL reconstructions and their complications. Diag Interv Imaging 96:11–19. https://doi.org/10.1016/j.diii.2014.04.007
Grassi A, Bailey J, Signorelli C et al (2016) Magnetic resonance imaging after anterior cruciate ligament reconstruction: a practical guide. World J Orthop 7:638. https://doi.org/10.5312/wjo.v7.i10.638
Janssen R, Scheffler S (2013) Intra-articular remodelling of hamstring tendon grafts after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 22:2102–2108. https://doi.org/10.1007/s00167-013-2634-5
Ntoulia A, Papadopoulou F, Ristanis S et al (2011) Revascularization process of the bone-patellar tendon–bone autograft evaluated by contrast-enhanced magnetic resonance imaging 6 and 12 months after anterior cruciate ligament reconstruction. Am J Sports Med 39:1478–1486. https://doi.org/10.1177/0363546511398039
Murakami Y, Sumen Y, Ochi M et al (1999) Appearance of anterior cruciate ligament autografts in their tibial bone tunnels on oblique axial MRI. Magn Reson Imaging 17:679–687. https://doi.org/10.1016/s0730-725x(99)00007-7
Li H, Tao H, Cho S et al (2012) Difference in graft maturity of the reconstructed anterior cruciate ligament 2 years postoperatively. Am J Sports Med 40:1519–1526. https://doi.org/10.1177/0363546512443050
White L, Kramer J, Recht M (2005) MR imaging evaluation of the postoperative knee: ligaments, menisci, and articular cartilage. Skeletal Radiol 34:431–452. https://doi.org/10.1007/s00256-005-0914-y
Li H, Chen J, Li H et al (2016) MRI-based ACL graft maturity does not predict clinical and functional outcomes during the first year after ACL reconstruction. Knee Surg Sports Traumatol Arthrosc 25:3171–3178. https://doi.org/10.1007/s00167-016-4252-5
Saupe N, White L, Chiavaras M et al (2008) Anterior cruciate ligament reconstruction grafts: MR imaging features at long-term follow-up—correlation with functional and clinical evaluation. Radiology 249:581–590. https://doi.org/10.1148/radiol.2492071651
Biercevicz A, Akelman M, Fadale P et al (2014) MRI volume and signal intensity of ACL graft predict clinical, functional, and patient-oriented outcome measures after ACL reconstruction. Am J Sports Med 43:693–699. https://doi.org/10.1177/0363546514561435
Colombet P, Graveleau N, Jambou S (2016) Incorporation of hamstring grafts within the tibial tunnel after anterior cruciate ligament reconstruction. Am J Sports Med 44:2838–2845. https://doi.org/10.1177/0363546516656181
Smith P, Stannard J, Pfeiffer F et al (2016) Suspensory versus interference screw fixation for arthroscopic anterior cruciate ligament reconstruction in a translational large-animal model. Arthroscopy 32:1086–1097. https://doi.org/10.1016/j.arthro.2015.11.026
Acknowledgements
No other contributors, no sources of funding, no assistance received
Funding
None received.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Drs. Prateek Gupta, Ashis Acharya, Vishesh Khanna and Amit Mourya certify that they have no commercial associations that might pose a conflict of interest in connection with the submitted article.
Ethical approval.
Ethical review committee statement—included: Ethical Committee clearance obtained (vide clearance letter no. EC/09/15/896).
Consent to participate.
Informed consents were duly obtained from all eligible candidates.
Consent for publication.
Obtained.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gupta, P.K., Acharya, A., Khanna, V. et al. Intra-femoral tunnel graft lengths less than 20 mm do not predispose to early graft failure, inferior outcomes or poor function. A prospective clinico-radiological comparative study. Musculoskelet Surg 107, 179–186 (2023). https://doi.org/10.1007/s12306-022-00740-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12306-022-00740-3