Abstract
The Japanese Breast Cancer Society initiated the breast cancer registry in 1975, which transitioned to the National Clinical Database-Breast Cancer Registry in 2012. This annual report presents data from 2020 and analyzes the ten-year mortality rates for those aged 65 and older. We analyzed data from 93,784 breast cancer (BC) cases registered in 2020 and assessed 10-year mortality rates for 36,279 elderly patients diagnosed between 2008 and 2012. In 2020, 99.4% of BC cases were females with a median age of 61. Most (65%) were diagnosed at early stages (Stage 0 or I). Breast-conserving surgery rates varied with stages: 58.5% at cStage I, 30.8% at cStage II, and 13.1% at cStage III. Sentinel lymph node biopsy was done in 73.6% of cases, followed by radiotherapy in 70% of those post-conserving surgery and chemotherapy in 21.1% post-surgery. Pathology showed that 63.4% had tumors under 2.0 cm, 11.7% had pTis tumors, and 77.3% had no axillary lymph node metastasis. ER positivity was seen in 75.1%, HER2 in 14.3%, and 30% had a Ki67 positivity rate above 30%. Across all stages and subtypes, there was a trend where the 10-year mortality rates increased for individuals older than 65 years. In Stage I, many deaths were not directly linked to BC and, for those with HER2-type and triple-negative BC, breast cancer-related deaths increased with age. Within Stage II, patients older than 70 years with luminal-type BC often experienced deaths not directly linked to BC, whereas patients below 80 years with HER2-type and triple-negative BC, likely had breast cancer-related deaths. In Stage III, breast cancer-related deaths were more common, particularly in HER2 and triple-negative BC. Our prognostic analysis underscores distinct mortality patterns by stage, subtype, and age in elderly BC patients. It highlights the importance of personalized treatment strategies, considering subtype-specific aggressiveness, age-related factors, and comorbidities.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Preface
The Japanese Breast Cancer Society (JBCS) initiated a breast cancer registry in 1975. In 2004, this registry transitioned to a web-based system in collaboration with the non-profit organization Japan Clinical Research Support Unit and the Public Health Research Foundation, Tokyo, Japan. In 2012, the National Clinical Database (NCD) took over the management of this registry. Further details of this system have been outlined in a prior publication [1]. Facilities across Japan participating in the NCD register patients diagnosed with new-onset breast cancer (BC). Since the inception of its current platform, the National Clinical Database-Breast Cancer Registry (NCD-BCR), in 2012, the registry has documented 945,398 BC cases through 2019 [2, 3].
Utilizing NCD-BCR data, institutions can obtain certification for surgical specialty training programs, and surgeons can either acquire or renew their professional licenses. The data has also been the basis for numerous retrospective observational studies [4,5,6,7,8,9,10,11,12,13,14,15,16,17]. Together, the JBCS and NCD Office aim to improve quality of medical care by relaying quality indicator data to BC treatment providers.
Japan has the most aged population in the world. In 2020, the Japanese Ministry of Health, Labour and Welfare reported that life expectancy was 24.8 years for 65-year-old women and 16.1 years for 75-year-old women. Therefore, it is important to evaluate the prognosis for elderly patients with BC in Japan when determining the most appropriate treatment approach.
This annual NCD-BCR report presents an overview of 93,784 BC cases registered in 2020 from 1364 institutes. Additionally, we provide an analysis of 10-year mortality rates for 36,279 elderly patients, over 65 years of age, who underwent surgery for Stage I-III BC between 2008 and 2012.
Demographics and patients’ characteristics
Of 93,784 BC cases, 93,193 (99.4%) were female. The median age at cancer diagnosis was 61 years with an interquartile range from 49 to 72 years. Moreover, 44% (41,149 patients) were aged over 65 (Table 1). The distribution of surgical procedures across various regions in Japan for female patients is as follows: 4.4% in Hokkaido, 6.4% in Tohoku, 35.0% in Kanto, 15.8% in Chubu, 5.7% in Chugoku, 3.1% in Shikoku, and 11.6% in Kyushu/Okinawa. Synchronous bilateral BC was observed in 6262 cases (6.7%), and a family history of BC was noted in 16,052 cases (17.2%). Regarding clinical Stage, 57.5% (53,531) of the female patients were diagnosed with early-stage BC of Stage 0 or I.
Pathology
The pathological assessment of surgical specimens for 89,783 patients with BC regardless of preoperative therapy revealed that 10,480 (11.7%) were finally diagnosed as pTis. The most frequent range of invasive tumor size was from 1.1 to 2.0 cm (26,899 patients, 30.0%). Of the 81,549 patients who received surgery on axillary lymph nodes, 77.3% had no axillary lymph node metastasis (pN0), while 6.1% (4952 patients) had metastasis in more than four axillary nodes. Of the 89,853 patients who received breast surgery, 75.1% were ER (estrogen receptor) positive (more than 10% positive cells), 62.1% were PgR (progesterone receptor) positive, and 14.3% were HER2 (Human Epidermal Growth Factor Receptor 2) positive. Out of the 75,370 cases where Ki67 was measured after the breast surgery, 24.8% (22,316 patients) exhibited a Ki67 positivity rate of more than 30%.
Breast cancer treatment
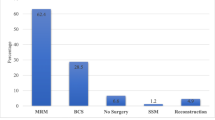
The types of surgeries categorized by cStage are presented in Table 2. Out of the 92,649 patients who underwent surgery, 39,875 (43.0%) had a partial mastectomy. The rates of breast-conserving surgery were 58.5% for cStage I, 30.8% for cStage II, and 13.1% for cStage III. A total of 68,156 patients (73.6%) underwent a sentinel lymph node biopsy (SLNB), and among them, 7164 (7.7%) subsequently had axillary node dissection. Among 48,040 patients without distant metastasis who received mastectomy, a total of 5563 (11.6%) patients underwent breast reconstruction procedures (Table 3).
The number of patients without distant metastasis who underwent postoperative radiotherapy is outlined in Table 4. Among the 39,216 patients who had breast-conserving surgery, 27,457 (70.0%) received whole breast irradiation, while 2080 (5.3%) were treated with radiotherapy to both the whole breast and regional lymph nodes. Of the 48,411 patients who had a mastectomy, 4808 (9.9%) received radiotherapy to the chest wall and regional lymph nodes, and 588 (1.2%) received radiotherapy only to the chest wall.
The types of systemic therapies administered either before or after surgery to patients without distant metastasis are detailed in Table 5. Of the 89,853 patients who underwent surgery, 10,773 (12.0%) were given neoadjuvant chemotherapy. Post-surgery, 18,971 patients (21.1%) received chemotherapy, and 56,365 (62.7%) were treated with endocrine therapy.
Ten-year mortality of elderly patients after surgery for breast cancer
In addition to presenting the characteristics of breast cancer patients registered in 2020, we conducted an analysis of the ten-year mortality rates for elderly BC patients diagnosed between 2008 and 2012. This analysis focused on patients with available clinical data, including age, ER, PgR, and HER2 status (Supplement figure). We utilized cStage information for patients who underwent neoadjuvant therapy and pStage information for those who did not. The BC subtypes were categorized as follows: luminal-type (ER or PgR positive with over 10% cell positivity), HER2-type (HER2 positive with any ER/PgR status), and triple-negative (ER and PgR negative with HER2 negative status). Patients with synchronous or metachronous bilateral BC were excluded from the analysis.
In Fig. 1 and supplementary tables, we present the ten-year mortality rates of elderly BC patients stratified by Stage, subtype, and age. In all stages and subtypes, 10-year mortality rates increased with age. For Stage I BC, it is noteworthy that the incidence of deaths due to non-breast cancer-related causes surpassed that of breast cancer-related deaths among individuals aged 65 and older, regardless of subtype. In HER2-type and triple-negative BC, there was a progressive increase in the likelihood of death due to BC as age advanced. In Stage II BC, patients aged 70 years or older with luminal-type BC were likely to die from causes other than BC. In contrast, patients under 80 years old with HER2-type and triple-negative BC tended to have a higher rate of death from BC compared to other causes. In Stage III, the risk of breast cancer-related mortality was notably elevated, surpassing the risk of death from other causes especially for HER2 and triple-negative BC in most age groups.
Our study reveals distinct mortality patterns by stage and subtype in elderly breast cancer patients and underscores the need for personalized treatment strategies that consider subtype-specific aggressiveness and age-related health factors. As elderly populations continue to grow, healthcare systems must adapt to meet the evolving needs of elderly breast cancer patients, aiming for optimal outcomes and quality of life.
Postscript
The detailed data regarding the treatment for BC, available within the Japanese Breast Cancer Registry, is a result of voluntary contributions by healthcare professionals, including physicians, across Japan. We sincerely acknowledge and appreciate their dedication in inputting this information. It is through their collective efforts that we can conduct data analysis, publish research papers, and ultimately make a more significant impact on the lives of BC patients.
We anticipate that the annual report derived from this registry will provide valuable support to physicians and scientists in comprehending the evolving trends in BC characteristics and treatment modalities in Japan.
Data availability
The data that support the findings of this study are not openly available due to the nature of the clinical data used. The clinical data are derived from the registry, which is not an open database. The data were accessed by a designated statistician through an application process approved by academic societies. Therefore, we are unable to offer the original clinical data.
References
Kubo M, et al. Annual report of the Japanese Breast Cancer Society Registry for 2016. Breast Cancer. 2020;27(4):511–8.
Hayashi N, et al. Annual report of the Japanese Breast Cancer Registry for 2017. Breast Cancer. 2020;27(5):803–9.
Tada K, et al. Characteristics of female breast cancer in japan: annual report of the National Clinical Database in 2018. Breast Cancer. 2023;30(2):157–66.
Iwamoto T, et al. Distinct breast cancer characteristics between screen- and self-detected breast cancers recorded in the Japanese Breast Cancer Registry. Breast Cancer Res Treat. 2016;156(3):485–94.
Kataoka A, et al. Young adult breast cancer patients have a poor prognosis independent of prognostic clinicopathological factors: a study from the Japanese Breast Cancer Registry. Breast Cancer Res Treat. 2016;160(1):163–72.
Kawai M, et al. Body mass index and survival after diagnosis of invasive breast cancer: a study based on the Japanese National Clinical Database-Breast Cancer Registry. Cancer Med. 2016;5(6):1328–40.
Niikura N, et al. Changes in tumor expression of HER2 and hormone receptors status after neoadjuvant chemotherapy in 21,755 patients from the Japanese breast cancer registry. Ann Oncol. 2016;27(3):480–7.
Kubo M, et al. A population-based recurrence risk management study of patients with pT1 node-negative HER2+ breast cancer: a National Clinical Database study. Breast Cancer Res Treat. 2019;178(3):647–56.
Miyashita M, et al. Role of postmastectomy radiotherapy after neoadjuvant chemotherapy in breast cancer patients: a study from the Japanese Breast Cancer Registry. Ann Surg Oncol. 2019;26(8):2475–85.
Hojo T, et al. Taxane-based combinations as adjuvant chemotherapy for node-positive ER-positive breast cancer based on 2004–2009 data from the Breast Cancer Registry of the Japanese Breast Cancer Society. Breast Cancer. 2020;27(1):85–91.
Ogiya R, et al. Breast cancer survival among Japanese individuals and US residents of Japanese and other origins: a comparative registry-based study. Breast Cancer Res Treat. 2020;184(2):585–96.
Yamada A, et al. Systemic therapy and prognosis of older patients with Stage II/III breast cancer: a large-scale analysis of the Japanese Breast Cancer Registry. Eur J Cancer. 2021;154:157–66.
Aihara T, et al. Prognosis and effectiveness of chemotherapy for medullary breast carcinoma. Breast Cancer Res Treat. 2022;196(3):635–45.
Shimomura A, et al. Clinicopathological features of male patients with breast cancer based on a nationwide registry database in Japan. Breast Cancer. 2022;29(6):985–92.
Terada M, et al. Surgical treatment trends and identification of primary breast tumors after surgery in occult breast cancer: a study based on the Japanese National Clinical Database-Breast Cancer Registry. Breast Cancer. 2022;29(4):698–708.
Yotsumoto D, et al. Trends in adjuvant therapy after breast-conserving surgery for ductal carcinoma in situ of breast: a retrospective cohort study using the National Breast Cancer Registry of Japan. Breast Cancer. 2022;29(1):1–8.
Adachi Y, et al. Analysis of prognosis in different subtypes of invasive lobular carcinoma using the Japanese National Cancer Database-Breast Cancer Registry. Breast Cancer Res Treat. 2023;201(3):397–408.
Acknowledgements
The authors thank all the affiliated institutes participating in the Breast Cancer Registry of the JBCS for their efforts to register the patients’ data.
Funding
This work was funded by the Registration Committee of the Japanese Breast Cancer Society.
Author information
Authors and Affiliations
Contributions
Study concept and design: YS, MM, NN, and TK. Assembly of data: HK and YS. Manuscript writing: YS. Critical revision of the manuscript for important intellectual content: The Registration Committee of the JBCS (NN, YS, TK, TI, NS, KT, MN, NH, MY, CW). Final approval of manuscript: All.
Corresponding author
Ethics declarations
Conflict of interest
YS have received honorariums from Pfizer, Astra Zeneca, Daiichi Sankyo, Eisai, Eli Lily, Chugai Chugai Pharmaceutical Ltd, MSD, and Nihon Kayaku. HK have received honorariums from Chugai Pharmaceutical Ltd and consultation fee from EPS corporation. HK and NK are affiliated to HQA, a social collaborative department supported by National CLinlical Database, Johnson & Johnson KK, Nipro corporation and Intuitive Surgical Sarl. NN have received honorariums from Chugai, Eli Lilly, MSD, Daiichi Sankyo, AstraZeneca, and Pfizer. MM have received honorariums from Chugai, AstraZeneca, Eli Lilly, Pfizer, MSD, Taiho, Daiichi Sankyo and Eisai. TK have received grants from Pfizer Co. Ltd, Kanzawa Medical Research Foundation and Japan Kampo Medicines Manufacturers Association. TI have received grants from Pfizer. MN have received honorariums from AstraZeneca, Eli Lilly, Pfizer, Novartis, Chugai, Taiho, Daiichi Sankyo, Eisai, Kyowa-Kirin, MSD, Myriad genetics, and Denka. NH have received grants from MSD, Chugai, and Konica Minolta Japan and honorariums from Eli Lilly, Astrazeneca, taiho, Eizai, ExactScience, Daiichi-Sankyo, Novartis, Pfizer, and Chugai. MY have received consulting fees from Eli Lilly and Chugai and honorariums from Agilent technologies, Chugai, Ono Yakuhin, MSD and Daiichi Sankyo. CW have received a honorarium from Chugai. MT have received research grants from Chugai, Takeda, Pfizer, Taiho, JBCRG assoc., KBCRN assoc., Eisai, Eli-Lilly and companies, Daiichi-Sankyo, AstraZeneca, Astellas, Shimadzu, Yakult, Nippon Kayaku, AFI technology, Luxonus, Shionogi, GL Science, and Sanwa Shurui. MT have received honorariums from Chugai, Takeda, Pfizer, Kyowa-Kirin, Taiho, Eisai, Daiichi-Sankyo, AstraZeneca, Eli Lilly and companies, MSD, Exact Science, Novartis, Shimadzu, Yakult, Nippon Kayaku, Devicore Medical Japan, and Sysmex. SS have received grants from Taiho, Eisai, Chugai, Takeda, and Daiichi Sankyo and honorariums from Chugai/Roche Astra Zeneca, Eli Lilly, Pfizer, Kyowa Kirin, Daiichi Sankyo and MSD.
Ethical approval
This article does not contain any studies with animals performed by any of the authors. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Sagara, Y., Kumamaru, H., Niikura, N. et al. 2020 Annual Report of National Clinical Database-Breast Cancer Registry: 10-year mortality of elderly breast cancer patients in Japan. Breast Cancer 31, 179–184 (2024). https://doi.org/10.1007/s12282-023-01532-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12282-023-01532-8