Abstract
Background
The Japanese Breast Cancer Society Registry started in 1975; it was transferred to the registry platform of the National Clinical Database in 2012. We provide the annual data and an analysis of the Breast Cancer Registry for 2017.
Methods
Patients’ characteristics and pathological data of the 95,203 registered Japanese breast cancer patients from 1,427 institutes in 2017 were obtained. Trends in age at diagnosis and pathological stage were determined during the most recent 6 years (2012–2017).
Results
The mean onset age was 60.2 years with bimodal peaks at 45–49 years and 65–69 years. A short-term trend of the most recent 6 years of data caused the second, older peak. At diagnosis, 32.4% of breast cancer patients were premenopausal. The distribution of stages revealed that the proportion of early stage breast cancer (stage 0–I) increased up to 60%. At the initial diagnosis, 2.2% of patients presented with metastatic disease. Sentinel node biopsy without axillary node dissection was performed without neoadjuvant chemotherapy (NAC) in 68.8%, and with NAC in 31.1%, of patients. For patients without NAC, lymph node metastasis was less than 3% if the tumor size was less than 1 cm. The proportion of node-negativity decreased to 79.5% when tumor size was 2.1–5 cm.
Conclusions
This analysis of the registry provides new information for effective treatment in clinical practice, cancer prevention, and the conduct of clinical trials. Further development of the registry and progress in collecting prognostic data will greatly enhance its scientific value.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Preface
The Japanese Breast Cancer Society (JBCS) registry was started in 1975. It was organized as a new web-based system cooperating with the non-profit organization, Japan Clinical Research Support Unit and the Public Health Research Foundation (Tokyo, Japan) from 2004. The management of Breast Cancer Registry (BCR) has transferred to the registry platform of National Clinical Database (NCD) in 2012. The details of the system have been described previously [1]. Patients who were diagnosed to have a new onset breast cancer at NCD participating facilities throughout Japan were eligible for the registry regardless of whether or not those undergo a breast surgery.
Since the BCR-NCD was started in 2012, the total number of records until 2017 has accumulated to 752,099. In the year 2017, there were 95,203 patients registered from 1,427 institutes. The BCR-NCD has been governed by the Registration Committee of JBCS. For their records, TNM classification is registered according to the 7th edition of the Union for International Cancer Control staging system, and histological classification is registered according to the General Rules for Clinical and Pathological Recording of Breast Cancer, which was further transferred to the Classification of Tumors of the Breast and Female Genital Organs [1,2,3,4].
In this report, we provide the annual data and analyze the trends of the BCR-NCD for 2017, including patients’ characteristics and breast cancer treatments. We show the clinicopathological characteristics for patients with and without neoadjuvant chemotherapy (NAC) separately, because all the pathological findings were examined from the results of surgical specimen regardless of whether or not patients had received NAC.
Key findings
Patients’ characteristics
The incidence per year of breast cancer, including ductal carcinoma in situ, was reported to be 104,379 in 2017 by the National Cancer Center [5]. Thus, 91.2% of newly diagnosed breast cancer patients were included in the JBCS registry in 2017. Patients’ characteristics of all the registered Japanese breast cancer patients are shown in Table 1. Of the 95,203 patients, 94,612 of (99.4%) were female. Figure 1 shows the distribution of onset age. The average onset age was 60.2 ± 14.1 years (mean ± standard deviation) with bimodal peaks at 45–49 years and 65–69 years (Fig. 1). A short-term trend in the most recent six years of data (2012–2017) caused the older second peak in the onset age. At diagnosis, 32.4% of breast cancer patients were premenopausal, and 14,317 patients (15.1%) had a family history of breast cancer (Table 1). The proportion of patients with Body Mass Index over 25 in Japanese population was 25.7%. 10.3% of patients had bilateral breast cancer: simultaneous for 6.3% and metachronal for 4.0%. A trend of distribution of stage was shown in Fig. 2. It revealed that the proportion of early stage breast cancer (stage 0–I) increased up to approximately 60% during the 6 years (Fig. 2). When including stage IIA, the proportion reached to 80.2%. The 12,180 of 87,724 patients (13.9%) who had surgery without NAC were diagnosed as DCIS. Overall, 2086 patients (2.2%) presented with metastatic disease at the initial diagnosis. Table 2 showed the site of distant metastasis in stage IV patients. Metastasis was found in bone for 53.3%, lung for 40.0%, and liver for 24.4% of the patients (Table 2).
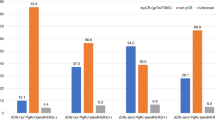
Figure 3 showed distribution of breast cancer subtype that was classified by estrogen receptor (ER) and progesterone receptor (PR) and HER2 status; ER+PR+HER2− for 48,074 patients (67.1%), ER+PR−HER2− for 5,906 patients (8.2%), ER−PR+HER2− for 249 patients (0.3%), ER+PR+HER2+ for 4,325 patients (6.0%), ER+PR−HER2+ for 1,967 patients (2.7%), ER−PR+HER2+ for 141 patients (0.2%), ER−PR−HER2+ for 4,072 patients (5.7%), and ER−PR−HER2− for 6,955 patients (9.7%) (Fig. 3). The proportion of each subtype was same as that in 2016 [1].
Breast cancer treatment
Of 90,232 patients without distant metastasis (M0), 9,320 patients (10.3%) received NAC. Table 3 showed surgical procedure for patients with or without NAC. For the 87,724 stage 0–III breast cancer patients with surgery, breast-conserving surgery was performed in 37,721 patients (48.0%) without NAC and in 3,405 patients (37.5%) with NAC (Table 3). For axillary surgery, sentinel node biopsy (SNB) without axillary lymph node dissection was performed in 54,082 patients (68.8%) without NAC and in 2,830 patients (31.1%) with NAC. Pathological findings on surgical specimen for patients with and without neoadjuvant chemotherapy are shown in Table 4. According to biomarker status, ER was negative for 23.6%, PR was negative for 34.7%, and HER2 was positive for 16.6% in patients with NAC; whereas, ER was negative for 12.5%, PR was negative for 21.1%, and HER2 was positive for 10.7% of patients without NAC (Table 4).
Pathological tumor size and lymph node status
Table 5 showed the association between pathological tumor size and number of pathological lymph node (LN) metastasis according to NAC. For patients without NAC, LN metastasis was rare at less than 3% if tumor size was equal to less than 1 cm (Table 5). However, the proportion of node-negativity decreased to 79.5% when tumor size was 2.1–5 cm (Table 5). For patients with NAC, LN metastasis remained about 40% of patients even if tumor size was equal to or smaller than 2 cm after NAC.
Postscript
Since treatment strategy of breast cancer has been developed based on intrinsic biological subtypes following the St Gallen International Expert Consensus in 2011, the proportion of patients who received chemotherapy has decreased [6]. In addition, because an annual breast cancer screening in Japan is becoming more popular, the detection rate of early stage breast cancer continues to increase. These developing data on the JBCS registry provides significant information for effective treatment in clinical practice, cancer prevention, and the conduct of clinical trial. Contribution by all medical workers are needed and greatly appreciated for further development and progress of the JBCS registry, especially in collecting prognostic data that will greatly enhance its scientific value. In addition, analyzing, discussing, and publishing on the registry will contribute to the development and progress of clinical practice for breast cancer.
References
Kubo M, Kumamaru H, Isozumi U, et al. Annual report of the Japanese Breast Cancer Society registry for 2016. Breast Cancer. 2020. https://doi.org/10.1007/s12282-020-01081-4,2019.
Sobin LH, Gospodarowicz MK, Wittekind C. TNM classification of malignant tumours. 7th ed. New York: Wiley; 2010. p. 131–141.
The Japanese Breast Cancer Society. General rules for clinical and pathological recording of breast cancer. 17th ed. Tokyo: Kanehara Shuppan; 2012.
Lakhani SR, Ellis IO, Schnitt SJ, Tan PH, van de Vijver MJ. WHO classification of tumours of the breast. 4th ed. Lyon: IARC Press; 2012.
Cancer Registry and Statistics. Cancer Information Service, National Cancer Center, Japan (Ministry of Health, Labour and Welfare, National Cancer Registry) 2020. https://ganjoho.jp/reg_stat/statistics/dl/index.html. Accessed 06 Jul 2020.
Goldhirsch A, Wood WC, Coates AS, et al. Strategies for subtypes—dealing with the diversity of breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2011. Ann Oncol. 2011;22(8):1736–47.
Acknowledgements
The authors thank all the affiliated institutes participating in the Breast Cancer Registry of the JBCS for their efforts to register the patients’ data.
Funding
This work was funded by the Registration Committee of the Japanese Breast Cancer Society.
Author information
Authors and Affiliations
Contributions
Study concept and design: NH and HJ. Assembly of data: HK and UI. Manuscript writing: NH. Critical revision of the manuscript for important intellectual content: The Registration Committee of the JBCS (HM, KT, MY, EO, MN, SA, YK, TK, KA, NN, MM, KI, MK, YY, SI, and HJ). Final approval of manuscript: SI and HJ.
Corresponding author
Ethics declarations
Conflict of interest
NH, MK, MM, YK, NN, and YY have received honorariums as a speaker or consultant/advisory role. NN and YY have received grants from Chugai Pharmaceutical Co. MM, YK. YY has received honorariums as a speaker or consultant/advisory role. YY have received grants from Eli Lilly. MM, YK, NN, and YY have received honorariums as a speaker or consultant/advisory role. YY have received grants from Eisai. NH, MM, YK, NN, and YY have received honorariums as a speaker or consultant/advisory role from AstraZeneca. NH, MM, YK, NN, YY, and HK have received honorariums as a speaker or consultant/advisory role. YY have received grants from Pfizer Japan Inc. MM, YK, and YY have received honorariums as a speaker or consultant/advisory role. YY have received grants from Taiho Pharma. MM, YK, and YY have received honorariums as a speaker or consultant/advisory role. YY has received grants from Daiichi-Sankyo. NH, YK, NN, and YY have received honorariums as a speaker or consultant/advisory role. YY have received grants from Novartis Pharma. NH, YK, and YY have received honorariums as a speaker or consultant/advisory role. YY has received grants, and MM has received non-financial support from Kyowa Hakko Kirin. YY has received grants and honorariums as a speaker or consultant/advisory role from Takeda. YY has received grants and honorariums as a speaker or consultant/advisory role from Sysmex. YY has received honorariums as a speaker or consultant/advisory role from GE Health Care Japan. NN has received grants from Nippon Mediphysics, Daiichi-Sankyo, BMS, and MSD. YY has received grants and honorariums as a speaker or consultant/advisory role from Nihon Kayaku. HK has received honorariums as a speaker or consultant/advisory role from Mitsubishi Tanabe Pharma. NH has received honorariums as a speaker or consultant/advisory role from Genomic Health Inc., Devicor Japan, and Allergan Japan. HK, HM, and UI report affiliation with the Department of Healthcare Quality Assessment at the University of Tokyo. The department is a social collaboration department supported by the National Clinical Database, Johnson & Johnson K.K., and the Nipro Corporation. YY and HJ are board members of the Japanese Breast Cancer Society. All of these declarations are outside the submitted work. The other authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with animals performed by any of the authors. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Hayashi, N., Kumamaru, H., Isozumi, U. et al. Annual report of the Japanese Breast Cancer Registry for 2017. Breast Cancer 27, 803–809 (2020). https://doi.org/10.1007/s12282-020-01139-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12282-020-01139-3