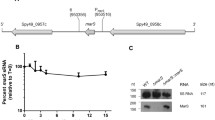
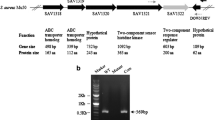
Abstract
The pathogenesis of Staphylococcus aureus, from local infections to systemic dissemination, is mediated by a battery of virulence factors that are regulated by intricate mechanisms, which include regulatory proteins and small RNAs (sRNAs) as key regulatory molecules. We have investigated the involvement of sRNA RsaF, in the regulation of pathogenicity genes hyaluronate lyase (hysA) and serine proteaselike protein D (splD), by employing S. aureus strains with disruption and overexpression of rsaF. Staphylococcus aureus strain with disruption of rsaF exhibited marked down-regulation of hysA transcripts by 0.2 to 0.0002 fold, and hyaluronate lyase activity by 0.2–0.1 fold, as well as increased biofilm formation, during growth from log phase to stationery phase. These mutants also displayed down-regulation of splD transcripts by 0.8 to 0.005 fold, and reduced activity of multiple proteases by zymography. Conversely, overexpression of rsaF resulted in a 2- to 4- fold increase in hysA mRNA levels and hyaluronidase activity. Both hysA and splD mRNAs demonstrated an increased stability in RsaF+ strains. In silico RNA-RNA interaction indicated a direct base pairing of RsaF with hysA and splD mRNAs, which was established in electrophoretic mobility shift assays. The findings demonstrate a positive regulatory role for small RNA RsaF in the expression of the virulence factors, HysA and SplD.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Arnaud, M., Chastanet, A., and Débarbouillé, M. 2004. New vector for efficient allelic replacement in naturally nontransformable, low-GC-content, Gram-positive bacteria. Appl. Environ. Microbiol. 70, 6887–6891.
Augagneur, Y., King, A.N., Germain-Amiot, N., Sassi, M., Fitzgerald, J.W., Sahukhal, G.S., Elasri, M.O., Felden, B., and Brinsmade, S.R. 2020. Analysis of the CodY RNome reveals RsaD as a stressresponsive riboregulator of overflow metabolism in Staphylococcus aureus. Mol. Microbiol. 113, 309–325.
Baba, T., Bae, T., Schneewind, O., Takeuchi, F., and Hiramatsu, K. 2008. Genome sequence of Staphylococcus aureus strain Newman and comparative analysis of staphylococcal genomes: polymorphism and evolution of two major pathogenicity islands. J. Bacteriol. 190, 300–310.
Bohn, C., Rigoulay, C., Chabelskaya, S., Sharma, C.M., Marchais, A., Skorski, P., Borezée-Durant, E., Barbet, R., Jacquet, E., Jacq, A., et al. 2010. Experimental discovery of small RNAs in Staphylococcus aureus reveals a riboregulator of central metabolism. Nucleic Acids Res. 38, 6620–6636.
Boisset, S., Geissmann, T., Huntzinger, E., Fechter, P., Bendridi, N., Possedko, M., Chevalier, C., Helfer, A.C., Benito, Y., Jacquier, A., et al. 2007. Staphylococcus aureus RNAIII coordinately represses the synthesis of virulence factors and the transcription regulator Rot by an antisense mechanism. Genes Dev. 21, 1353–1366.
Bonnin, R.A. and Bouloc, P. 2015. RNA degradation in Staphylococcus aureus: diversity of ribonucleases and their impact. Int. J. Genomics 2015, 395753.
Brinsmade, S.R., Kleijn, R.J., Sauer, U., and Sonenshein, A.L. 2010. Regulation of CodY activity through modulation of intracellular branched-chain amino acid pools. J. Bacteriol. 192, 6357–6368.
Bronner, S., Monteil, H., and Prévost, G. 2004. Regulation of virulence determinants in Staphylococcus aureus: complexity and applications. FEMS Microbiol. Rev. 28, 183–200.
Buchad, H. and Nair, M. 2021. The small RNA SprX regulates the autolysin regulator WalR in Staphylococcus aureus. Microbiol. Res. 250, 126785.
Chabelskaya, S., Gaillot, O., and Felden, B. 2010. A Staphylococcus aureus small RNA is required for bacterial virulence and regulates the expression of an immune-evasion molecule. PLoS Pathog. 6, e1000927.
Chakravarty, S. and Massé, E. 2019. RNA-dependent regulation of virulence in pathogenic bacteria. Front. Cell. Infect. Microbiol. 9, 337.
Chomczynski, P. and Sacchi, N. 2006. The single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction: twenty-something years on. Nat. Protoc. 1, 581–585.
Felden, B., Vandenesch, F., Bouloc, P., and Romby, P. 2011. The Staphylococcus aureus RNome and its commitment to virulence. PLoS Pathog. 7, e1002006.
Geisinger, E., Adhikari, R.P., Jin, R., Ross, H.F., and Novick, R.P. 2006. Inhibition of rot translation by RNAIII, a key feature of agr function. Mol. Microbiol. 61, 1038–1048.
Geissmann, T., Chevalier, C., Cros, M.J., Boisset, S., Fechter, P., Noirot, C., Schrenzel, J., François, P., Vandenesch, F., Gaspin, C., et al. 2009. A search for small noncoding RNAs in Staphylococcus aureus reveals a conserved sequence motif for regulation. Nucleic Acids Res. 37, 7239–7257.
Gimza, B.D., Larias, M.I., Budny, B.G., and Shaw, L.N. 2019. Mapping the global network of extracellular protease regulation in Staphylococcus aureus. mSphere 4, e00676–19.
González Plaza, J.J. 2020. Small RNAs as fundamental players in the transference of information during bacterial infectious diseases. Front. Mol. Biosci. 7, 101.
Gordon, R.J. and Lowy, F.D. 2008. Pathogenesis of methicillin-resistant Staphylococcus aureus infection. Clin. Infect. Dis. 46, S350–S359.
Hart, M.E., Tsang, L.H., Deck, J., Daily, S.T., Jones, R.C., Liu, H., Hu, H., Hart, M.J., and Smeltzer, M.S. 2013. Hyaluronidase expression and biofilm involvement in Staphylococcus aureus UAMS-1 and its sarA, agr and sarA agr regulatory mutants. Microbiology 159, 782–791.
Hochbaum, A.I., Kolodkin-Gal, I., Foulston, L., Kolter, R., Aizenberg, J., and Losick, R. 2011. Inhibitory effects of D-amino acids on Staphylococcus aureus biofilm development. J. Bacteriol. 193, 5616–5622.
Holmqvist, E. and Vogel, J. 2018. RNA-binding proteins in bacteria. Nat. Rev. Microbiol. 16, 601–615.
Hynes, W.L. and Walton, S.L. 2000. Hyaluronidases of Gram-positive bacteria. FEMS Microbiol. Lett. 183, 201–207.
Ibberson, C.B., Jones, C.L., Singh, S., Wise, M.C., Hart, M.E., Zurawski, D.V., and Horswill, A.R. 2014. Staphylococcus aureus hyaluronidase is a CodY-regulated virulence factor. Infect. Immun. 82, 4253–4264.
Ibberson, C.B., Parlet, C.P., Kwiecinski, J., Crosby, H.A., Meyerholz, D.K., and Horswill, A.R. 2016. Hyaluronan modulation impacts Staphylococcus aureus biofilm infection. Infect. Immun. 84, 1917–1929.
Jenul, C. and Horswill, A.R. 2019. Regulation of Staphylococcus aureus virulence. Microbiol. Spectr. 7, 7.2.29.
Kathirvel, M., Buchad, H., and Nair, M. 2016. Enhancement of the pathogenicity of Staphylococcus aureus strain Newman by a small noncoding RNA SprX1. Med. Microbiol. Immunol. 205, 563–574.
Krysko, O., Teufelberger, A., Van Nevel, S., Krysko, D.V., and Bachert, C. 2019. Protease/antiprotease network in allergy: The role of Staphylococcus aureus protease-like proteins. Allergy 74, 2077–2086.
Lalaouna, D., Baude, J., Wu, Z., Tomasini, A., Chicher, J., Marzi, S., Vandenesch, F., Romby, P., Caldelari, I., and Moreau, K. 2019. RsaC sRNA modulates the oxidative stress response of Staphylococcus aureus during manganese starvation. Nucleic Acids Res. 47, 9871–9887.
Lantz, M.S. and Ciborowski, P. 1994. Zymographic techniques for detection and characterization of microbial proteases. Methods Enzymol. 235, 563–594.
Le Pabic, H., Germain-Amiot, N., Bordeau, V., and Felden, B. 2015. A bacterial regulatory RNA attenuates virulence, spread and human host cell phagocytosis. Nucleic Acids Res. 43, 9232–9248.
Le Scornet, A. and Redder, P. 2019. Post-transcriptional control of virulence gene expression in Staphylococcus aureus. Biochim. Biophys. Acta Gene Regul. Mech. 1862, 734–741.
Livak, K.J. and Schmittgen, T.D. 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCt method. Methods 25, 402–408.
Makris, G., Wright, J.D., Ingham, E., and Holland, K.T. 2004. The hyaluronate lyase of Staphylococcus aureus - a virulence factor? Microbiology 150, 2005–2013.
Manna, A.C., Kim, S., Cengher, L., Corvaglia, A., Leo, S., Francois, P., and Cheung, A.L. 2018. Small RNA teg49 is derived from a sarA transcript and regulates virulence genes independent of SarA in Staphylococcus aureus. Infect. Immun. 86, e00635–17.
Mitchell, G., Fugère, A., Pépin Gaudreau, K., Brouillette, E., Frost, E.H., Cantin, A.M., and Malouin, F. 2013. SigB is a dominant regulator of virulence in Staphylococcus aureus small-colony variants. PLoS ONE 8, e65018.
Monk, I.R., Shah, I.M., Xu, M., Tan, M.W., and Foster, T.J. 2012. Transforming the untransformable: application of direct transformation to manipulate genetically Staphylococcus aureus and Staphylococcus epidermidis. mBio 3, e00277–11.
Morfeldt, E., Taylor, D., von Gabain, A., and Arvidson, S. 1995. Activation of alpha-toxin translation in Staphylococcus aureus by the trans-encoded antisense RNA, RNAIII. EMBO J. 14, 4569–4577.
Nair, D., Memmi, G., Hernandez, D., Bard, J., Beaume, M., Gill, S., Francois, P., and Cheung, A.L. 2011. Whole-genome sequencing of Staphylococcus aureus strain RN4220, a key laboratory strain used in virulence research, identifies mutations that affect not only virulence factors but also the fitness of the strain. J. Bacteriol. 193, 2332–2335.
Nilsen, T.W. 2013. Gel purification of RNA. Cold Spring Harb. Protoc. 2013, 180–183.
Paharik, A.E., Salgado-Pabon, W., Meyerholz, D.K., White, M.J., Schlievert, P.M., and Horswill, A.R. 2016. The Spl serine proteases modulate Staphylococcus aureus protein production and virulence in a rabbit model of pneumonia. mSphere 1, e00208–16.
Pain, A., Ott, A., Amine, H., Rochat, T., Bouloc, P., and Gautheret, D. 2015. An assessment of bacterial small RNA target prediction programs. RNA Biol. 12, 509–513.
Papenfort, K. and Vanderpool, C.K. 2015. Target activation by regulatory RNAs in bacteria. FEMS Microbiol. Rev. 39, 362–378.
Papenfort, K. and Vogel, J. 2010. Regulatory RNA in bacterial pathogens. Cell Host Microbe 8, 116–127.
Pietrocola, G., Nobile, G., Rindi, S., and Speziale, P. 2017. Staphylococcus aureus manipulates innate immunity through own and host-expressed proteases. Front. Cell. Infect. Microbiol. 7, 166.
Pichon, C. and Felden, B. 2005. Small RNA genes expressed from Staphylococcus aureus genomic and pathogenicity islands with specific expression among pathogenic strains. Proc. Natl. Acad. Sci. USA 102, 14249–14254.
Ramirez-Peña, E., Treviño, J., Liu, Z., Perez, N., and Sumby, P. 2010. The group A Streptococcus small regulatory RNA FasX enhances streptokinase activity by increasing the stability of the ska mRNA transcript. Mol. Microbiol. 78, 1332–1347.
Reed, S.B., Wesson, C.A., Liou, L.E., Trumble, W.R., Schlievert, P.M., Bohach, G.A., and Bayles, K.W. 2001. Molecular characterization of a novel Staphylococcus aureus serine protease operon. Infect. Immun. 69, 1521–1527.
Romilly, C., Lays, C., Tomasini, A., Caldelari, I., Benito, Y., Hammann, P., Geissmann, T., Boisset, S., Romby, P., and Vandenesch, F. 2014. A non-coding RNA promotes bacterial persistence and decreases virulence by regulating a regulator in Staphylococcus aureus. PLoS Pathog. 10, e1003979.
Sambrook, J. and Russel, D.W. 2001. Molecular Cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Lab Press, New York, USA.
Shaw, L., Golonka, E., Potempa, J., and Foster, S.J. 2004. The role and regulation of the extracellular proteases of Staphylococcus aureus. Microbiology 150, 217–228.
Stach, N., Kaszycki, P., Wladyka, B., and Dubin, G. 2018. Extracellular proteases of Staphylococcus spp. In Savini, V. (ed.), Pettoman Travelling Staphylococci. pp. 135–145. Academic Press, San Diego, California, USA.
Stentzel, S., Teufelberger, A., Nordengrün, M., Kolata, J., Schmidt, F., van Crombruggen, K., Michalik, S., Kumpfmüller, J., Tischer, S., Schweder, T., et al. 2017. Staphylococcal serine protease-like proteins are pacemakers of allergic airway reactions to Staphylococcus aureus. J. Allergy Clin. Immunol. 139, 492–500.
Tam, K. and Torres, V.J. 2019. Staphylococcus aureus secreted toxins and extracellular enzymes. Microbiol. Spectr. 7, 7.2.16.
Tan, L., Li, S.R., Jiang, B., Hu, X.M., and Li, S. 2018. Therapeutic targeting of the Staphylococcus aureus accessory gene regulator (agr) system. Front. Microbiol. 9, 55.
Thammavongsa, V., Kim, H.K., Missiakas, D., and Schneewind, O. 2015. Staphylococcal manipulation of host immune responses. Nat. Rev. Microbiol. 13, 529–543.
Toledo-Arana, A., Repoila, F., and Cossart, P. 2007. Small noncoding RNAs controlling pathogenesis. Curr. Opin. Microbiol. 10, 182–188.
Tomasini, A., Moreau, K., Chicher, J., Geissmann, T., Vandenesch, F., Romby, P., Marzi, S., and Caldelari, I. 2017. The RNA targetome of Staphylococcus aureus non-coding RNA RsaA: impact on cell surface properties and defense mechanisms. Nucleic Acids Res. 45, 6746–6760.
Tong, S.Y.C., Davis, J.S., Eichenberger, E., Holland, T.L., and Fowler, V.G. Jr. 2015. Staphylococcus aureus infections: epidemiology, pathophysiology, clinical manifestations, and management. Clin. Microbiol. Rev. 28, 603–661.
Tuchscherr, L., Bischoff, M., Lattar, S.M., Noto Llana, M., Pförtner, H., Niemann, S., Geraci, J., Van de Vyver, H., Fraunholz, M.J., Cheung, A.L., et al. 2015. Sigma factor SigB is crucial to mediate Staphylococcus aureus adaptation during chronic infections. PLoS Pathog. 11, e1004870.
Vockenhuber, M.P. and Suess, B. 2012. Streptomyces coelicolor sRNA scr5239 inhibits agarase expression by direct base pairing to the dagA coding region. Microbiology 158, 424–435.
Zapf, R.L., Wiemels, R.E., Keogh, R.A., Holzschu, D.L., Howell, K.M., Trzeciak, E., Caillet, A.R., King, K.A., Selhorst, S.A., Naldrett, M.J., et al. 2019. The small RNA Teg41 regulates expression of the Alpha phenol-soluble modulins and is required for virulence in Staphylococcus aureus. mBio 10, e02484–18.
Zdzalik, M., Kalinska, M., Wysocka, M., Stec-Niemczyk, J., Cichon, P., Stach, N., Gruba, N., Stennicke, H.R., Jabaiah, A., Markiewicz, M., et al. 2013. Biochemical and structural characterization of SplD protease from Staphylococcus aureus. PLoS ONE 8, e76812.
Zhao, X., Koestler, B.J., Waters, C.M., and Hammer, B.K. 2013. Post-transcriptional activation of a diguanylate cyclase by quorum sensing small RNAs promotes biofilm formation in Vibrio cholerae. Mol. Microbiol. 89, 989–1002.
Zmantar, T., Kouidhi, B., Miladi, H., Mahdouani, K., and Bakhrouf, A. 2010. A microtiter plate assay for Staphylococcus aureus biofilm quantification at various pH levels and hydrogen peroxide supplementation. New Microbiol. 33, 137–145.
Acknowledgements
NP was supported by BSR Fellowship in Science F.4-1/2006-(BSR)/7-128/2007(BSR) by University Grant Commission, Government of India, New Delhi.
The authors thank Professors Timothy Foster and Ian. R. Monk, Trinity College, Dublin for S. aureus and E. coli strains. We thank Professor Cristiane Wolz, University of Tubingen, Tubingen, Germany for supplying pMAD plasmid. The pCN40 plasmid was obtained from Network on antimicrobial resistance in Staphylococcus aureus (NARSA) program supported under NIAD, NIH contract No. HHSN272200700055C. We thank DBT-MSUB-ILSPARE, Dr. Vikram Sarabhai Science Block, Maharaja Sayajirao University of Baroda for the Confocal microscopy facility.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interests.
Additional information
Supplemental material for this article may be found at http://www.springerlink.com/content/120956.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Patel, N., Nair, M. The small RNA RsaF regulates the expression of secreted virulence factors in Staphylococcus aureus Newman. J Microbiol. 59, 920–930 (2021). https://doi.org/10.1007/s12275-021-1205-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-021-1205-6