Abstract
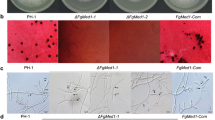
Fusarium graminearum is a filamentous fungal plant pathogen that infects major cereal crops. The fungus produces both sexual and asexual spores in order to endure unfavorable environmental conditions and increase their numbers and distribution across plants. In a model filamentous fungus, Aspergillus nidulans, early induction of conidiogenesis is orchestrated by the fluffy genes. The objectives of this study were to characterize fluffy gene homologs involved in conidiogenesis and their mechanism of action in F. graminearum. We characterized five fluffy gene homologs in F. graminearum and found that FlbD is the only conserved regulator for conidiogenesis in A. nidulans and F. graminearum. Deletion of fgflbD prevented hyphal differentiation and the formation of perithecia. Successful interspecies complementation using A. nidulans flbD demonstrated that the molecular mechanisms responsible for FlbD functions are conserved in F. graminearum. Moreover, abaA-wetA pathway is positively regulated by FgFlbD during conidiogenesis in F. graminearum. Deleting fgflbD abolished morphological effects of abaA overexpression, which suggests that additional factors for FgFlbD or an AbaA-independent pathway for conidiogenesis are required for F. graminearum conidiation. Importantly, this study led to the construction of a genetic pathway of F. graminearum conidiogenesis and provides new insights into the genetics of conidiogenesis in fungi.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adams T.H., Wieser J.K., and Yu J.H. 1998. Asexual sporulation in Aspergillus nidulans. Microbiol. Mol. Biol. Rev. 62, 35–54.
Alexopoulos C.J., Mims C.W., and Blackwell M. 1996. Introductory mycology John Wiley & Sons, New York, N.Y., USA.
Aramayo R., Peleg Y., Addison R., and Metzenberg R. 1996. Asm-1 +, a Neurospora crassa gene related to transcriptional regulators of fungal development. Genetics 144, 991–1003.
Arratia-Quijada J., Sánchez O., Scazzocchio C., and Aguirre J. 2012. FlbD, a Myb transcription factor of Aspergillus nidulans is uniquely involved in both asexual and sexual differentiation. Eukaryot. Cell 11, 1132–1142.
Bailey L.A. and Ebbole D.J. 1998. The fluffy gene of Neurospora crassa encodes a Gal4p-type C6 zinc cluster protein required for conidial development. Genetics 148, 1813–1820.
Bowden R.L. and Leslie J.F. 1992. Nitrate-nonutilizing mutants of Gibberella zeae (Fusarium graminearum) and their use in determining vegetative compatibility. Exp. Mycol. 16, 308–315.
Bowden R.L. and Leslie J.F. 1999. Sexual recombination in Gibberella zeae. Phytopathology 89, 182–188.
Cappellini R.A. and Peterson J.L. 1965. Macroconidium formation in submerged cultures by a non-sporulating strain of Gibberella zeae. Mycologia 57, 962–966.
Chang M.H., Chae K.S., Han D.M., and Jahng K.Y. 2004. The GanB Ga-protein negatively regulates asexual sporulation and plays a positive role in conidial germination in Aspergillus nidulans. Genetics 167, 1305–1315.
Correll J.C., Klittich C.J.R., and Leslie J.F. 1987. Nitrate nonutilizing mutants of Fusarium oxysporum and their use in vegetative compatibility tests. Phytopathology 77, 1640–1646.
Ebbole D.J. 2010. The conidium, pp. 577–590. In Borkovich K.A. and Ebbole D.J. (eds.), Cellular and molecular biology of filamentous fungi, ASM Press, Washington, D.C., USA.
Etxebeste O., Garzia A., Espeso E.A., and Ugalde U. 2010. Aspergillus nidulans asexual development: making the most of cellular modules. Trends Microbiol. 18, 569–576.
Fischer R. and Kües U. 2006. Asexual sporulation in mycelial fungi, pp. 263–292. In Kües U. and Fischer R. (eds.), The Mycota I growth, differentiation and sexuality, Springer Berlin Heidelberg, Germany.
Garzia A., Etxebeste O., Herrero-Garcia E., Fischer R., Espeso E.A., and Ugalde U. 2009. Aspergillus nidulans FlbE is an upstream developmental activator of conidiation functionally associated with the putative transcription factor FlbB. Mol. Microbiol. 71, 172–184.
Garzia A., Etxebeste O., Herrero-García E., Ugalde U., and Espeso E.A. 2010. The concerted action of bZip and cMyb transcription factors FlbB and FlbD induces brlA expression and asexual development in Aspergillus nidulans. Mol. Microbiol. 75, 1314–1324.
Gasch A.P., Moses A.M., Chiang D.Y., Fraser H.B., Berardini M., and Eisen M.B. 2004. Conservation and evolution of Cis-regulatory systems in ascomycete fungi. PLoS Biol. 2, e398.
Guenther J.C. and Trail F. 2005. The development and differentiation of Gibberella zeae (anamorph: Fusarium graminearum) during colonization of wheat. Mycologia 97, 229–237.
Han Y.K., Kim M.D., Lee S.H., Yun S.H., and Lee Y.W. 2007. A novel F-box protein involved in sexual development and pathogenesis in Gibberella zeae. Mol. Microbiol. 63, 768–779.
Harris S.D. 2005. Morphogenesis in germinating Fusarium graminearum macroconidia. Mycologia 97, 880–887.
Hong S.Y., So J., Lee J., Min K., Son H., Park C., Yun S.H., and Lee Y.W. 2010. Functional analyses of two syntaxin-like SNARE genes, GzSYN1 and GzSYN2, in the ascomycete Gibberella zeae. Fungal Genet. Biol. 47, 364–372.
Horwitz B.A., Sharon A., Lu S.W., Ritter V., Sandrock T.M., Yoder O.C., and Turgeon B.G. 1999. AG protein alpha subunit from Cochliobolus heterostrophus involved in mating and appressorium formation. Fungal Genet. Biol. 26, 19–32.
Kim J.E., Han K.H., Jin J., Kim H., Kim J.C., Yun S.H., and Lee Y.W. 2005. Putative polyketide synthase and laccase genes for biosynthesis of aurofusarin in Gibberella zeae. Appl. Environ. Microbiol. 71, 1701–1708.
Kwon N.J., Garzia A., Espeso E.A., Ugalde U., and Yu J.H. 2010a. FlbC is a putative nuclear C2H2 transcription factor regulating development in Aspergillus nidulans. Mol. Microbiol. 77, 1203–1219.
Kwon N.J., Shin K.S., and Yu J.H. 2010b. Characterization of the developmental regulator FlbE in Aspergillus fumigatus and Aspergillus nidulans. Fungal Genet. Biol. 47, 981–993.
Lau G.W. and Hamer J.E. 1998. Acropetal: A genetic locus required for conidiophore architecture and pathogenicity in the rice blast fungus. Fungal Genet. Biol. 24, 228–239.
Lee B.N. and Adams T.H. 1994. The Aspergillus nidulans fluG gene is required for production of an extracellular developmental signal and is related to prokaryotic glutamine synthetase I. Genes Dev. 8, 641–651.
Lee J.Y., Kim L.H., Kim H.E., Park J.S., Han K.H., and Han D.M. 2013. A putative APSES transcription factor is necessary for normal growth and development of Aspergillus nidulans. J. Microbiol. 51, 800–806.
Lee S.H., Lee J., Lee S., Park E.H., Kim K.W., Kim M.D., Yun S.H., and Lee Y.W. 2009. GzSNF1 is required for normal sexual and asexual development in the ascomycete Gibberella zeae. Eukaryot. Cell 8, 116–127.
Lee J., Lee T., Lee Y.W., Yun S.H., and Turgeon B.G. 2003. Shifting fungal reproductive mode by manipulation of mating type genes: obligatory heterothallism of Gibberella zeae. Mol. Microbiol. 50, 145–152.
Lengeler K.B., Davidson R.C., D’souza C., Harashima T., Shen W.C., Wang P., Pan X., Waugh M., and Heitman J. 2000. Signal transduction cascades regulating fungal development and virulence. Microbiol. Mol. Biol. Rev. 64, 746–785.
Leslie J.F. and Summerell B.A. 2006. The Fusarium laboratory manual. Blackwell Pub., Ames, I.A., USA.
Lin Y., Son H., Lee J., Min K., Choi G.J., Kim J.C., and Lee Y.W. 2011. A putative transcription factor MYT1 is required for female fertility in the ascomycete Gibberella zeae. PLoS ONE 6, e25586.
Livak K.J. and Schmittgen T.D. 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25, 402–408.
Lysøe E., Pasquali M., Breakspear A., and Kistler H.C. 2011. The transcription factor FgStuAp influences spore development, pathogenicity, and secondary metabolism in Fusarium graminearum. Mol. Plant-Microbe Interact. 24, 54–67.
Namiki F., Matsunaga M., Okuda M., Inoue I., Nishi K., Fujita Y., and Tsuge T. 2001. Mutation of an arginine biosynthesis gene causes reduced pathogenicity in Fusarium oxysporum f. sp melonis. Mol. Plant-Microbe Interact. 14, 580–584.
Ohara T., Inoue I., Namiki F., Kunoh H., and Tsuge T. 2004. REN1 is required for development of microconidia and macroconidia, but not of chlamydospores, in the plant pathogenic fungus Fusarium oxysporum. Genetics 166, 113–124.
Ohara T. and Tsuge T. 2004. FoSTUA, encoding a basic helixloop- helix protein, differentially regulates development of three kinds of asexual spores, macroconidia, microconidia, and chlamydospores, in the fungal plant pathogen Fusarium oxysporum. Eukaryot. Cell 3, 1412–1422.
Park A.R., Cho A.R., Seo J.A., Min K., Son H., Lee J., Choi G.J., Kim J.C., and Lee Y.W. 2012. Functional analyses of regulators of G protein signaling in Gibberella zeae. Fungal Genet. Biol. 49, 511–520.
Park H.S. and Yu J.H. 2012. Genetic control of asexual sporulation in filamentous fungi. Curr. Opin. Microbiol. 15, 669–677.
Rodríguez-Urra A.B., Jiménez C., Nieto M.I., Rodríguez J., Hayashi H., and Ugalde U. 2012. Signaling the induction of sporulation involves the interaction of two secondary metabolites in Aspergillus nidulans. ACS Chem. Biol. 7, 599–606.
Sambrook J. and Russell D.W. 2001. Molecular cloning: a laboratory manual, 4nd ed Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y., USA.
Seo J.A., Guan Y., and Yu J.H. 2003. Suppressor mutations bypass the requirement of fluG for asexual sporulation and sterigmatocystin production in Aspergillus nidulans. Genetics 165, 1083–1093.
Seo J.A., Guan Y., and Yu J.H. 2006. FluG-dependent asexual development in Aspergillus nidulans occurs via derepression. Genetics 172, 1535–1544.
Shen W.C., Wieser J., Adams T.H., and Ebbole D.J. 1998. The Neurospora rca-1 gene complements an Aspergillus flbD sporulation mutant but has no identifiable role in Neurospora sporulation. Genetics 148, 1031–1041.
Shimizu K. and Keller N.P. 2001. Genetic involvement of a cAMPdependent protein kinase in a G protein signaling pathway regulating morphological and chemical transitions in Aspergillus nidulans. Genetics 157, 591–600.
Soid-Raggi G., Sánchez O., and Aguirre J. 2006. TmpA, a member of a novel family of putative membrane flavoproteins, regulates asexual development in Aspergillus nidulans. Mol. Microbiol. 59, 854–869.
Son H., Kim M.G., Min K., Seo Y.S., Lim J.Y., Choi G.J., Kim J.C., Chae S.K., and Lee Y.W. 2013. AbaA regulates conidiogenesis in the ascomycete fungus Fusarium graminearum. PLoS ONE 8, e729815.
Son H., Kim M.G., Min K., Seo Y.S., Lim J.Y., Choi G.J., Kim J.C., Chae S.K., and Lee Y.W. 2014. WetA is required for conidiogenesis and conidia maturation in the ascomycete fungus Fusarium graminearum. Eukaryot. Cell 13, 87–98.
Son H., Lee J., Park A.R., and Lee Y.W. 2011a. ATP citrate lyase is required for normal sexual and asexual development in Gibberella zeae. Fungal Genet. Biol. 48, 408–417.
Son H., Seo Y.S., Min K., Park A.R., Lee J., Jin J.M., Lin Y., Cao P., Hong S.Y., Kim E.K., and et al. 2011b. A phenome-based functional analysis of transcription factors in the cereal head blight fungus, Fusarium graminearum. PLoS Pathog. 7, e100230.
Trail F., Xu H., Loranger R., and Gadoury D. 2002. Physiological and environmental aspects of ascospore discharge in Gibberella zeae (anamorph Fusarium graminearum). Mycologia 94, 181–189.
Tuch B.B., Li H., and Johnson A.D. 2008. Evolution of eukaryotic transcription circuits. Science 319, 1797–1799.
Wieser J. and Adams T.H. 1995. flbD encodes a Myb-like DNAbinding protein that coordinates initiation of Aspergillus nidulans conidiophore development. Genes Dev. 9, 491–502.
Wong P., Walter M., Lee W., Mannhaupt G., Münsterkötter M., Mewes H.W., Adam G., and Güldener U. 2011. FGDB: revisiting the genome annotation of the plant pathogen Fusarium graminearum. Nucleic Acids Res. 39, D637–D639.
Yu J.H. 2006. Heterotrimeric G protein signaling and RGSs in Aspergillus niduians. J. Microbiol. 44, 145–154.
Yu J.H., Hamari Z., Han K.H., Seo J.A., Reyes-Dominguez Y., and Scazzocchio C. 2004. Double-joint PCR: a PCR-based molecular tool for gene manipulations in filamentous fungi. Fungal Genet. Biol. 41, 973–981.
Yu J.H., Wieser J., and Adams T.H. 1996. The Aspergillus FlbA RGS domain protein antagonizes G protein signaling to block proliferation and allow development. EMBO J. 15, 5184–5190.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplemental material for this article may be found at http://www.springerlink.com/content/120956.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Son, H., Kim, MG., Chae, SK. et al. FgFlbD regulates hyphal differentiation required for sexual and asexual reproduction in the ascomycete fungus Fusarium graminearum . J Microbiol. 52, 930–939 (2014). https://doi.org/10.1007/s12275-014-4384-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-014-4384-6