Abstract
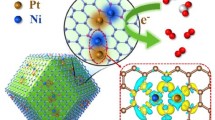
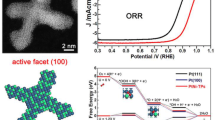
Elaborated design of catalytic systems with a specifically tailored site distance to match the intermediates could substantially improve reaction kinetics and boost catalytic activity under unfavorable reaction conditions. Considering the lower energy barriers of water splitting upon the synergy of dual sites, constructing synergistic Pt-M (M: transition metal) dual sites is an effective way to boost Pt with highly catalytic hydrogen evolution reaction (HER) performance. An unconventional “Ni(OH)2-coated high-index Pt facets” was constructed to obtain long-range Pt-Ni dual sites, in which Ni composition as a water dissociation synergistic site can protect Pt from electrolyte corrosion and ensure efficient proton donation to Pt sites. The obtained long-range Pt-Ni dual sites present 3.84 mA·cm−1 of current density, which is 7.5 times specific activity higher than that of commercial Pt/C towards alkaline HER. The enhanced HER performance is attributed to synergistic catalysis on Pt-Ni dual sites accompanied by unconventional electron coupling. This work illustrates a new strategy to construct the long-range dual sites by unconventional strategy for fundamental electrocatalytic study of alkaline HER.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Dubouis, N.; Grimaud, A. The hydrogen evolution reaction: From material to interfacial descriptors. Chem. Sci. 2019, 10, 9165–9181.
Hua, W.; Sun, H. H.; Xu, F.; Wang, J. G. A review and perspective on molybdenum-based electrocatalysts for hydrogen evolution reaction. Rare Met. 2020, 39, 335–351.
Wang, B.; Yang, F. L.; Feng, L. G. Recent advances in Co-based electrocatalysts for hydrogen evolution reaction. Small, in press, https://doi.org/10.1002/smll.202302866.
Zhou, W. J.; Jia, J.; Lu, J.; Yang, L. J.; Hou, D. M.; Li, G. Q.; Chen, S. W. Recent developments of carbon-based electrocatalysts for hydrogen evolution reaction. Nano Energy 2016, 28, 29–43.
Gan, T.; Wang, D. S. Atomically dispersed materials: Ideal catalysts in atomic era. Nano Res, in press, https://doi.org/10.1007/s12274-023-5700-4.
Lu, Q. P.; Yu, Y. F.; Ma, Q. L.; Chen, B.; Zhang, H. 2D transition-metal-dichalcogenide-nanosheet-based composites for photocatalytic and electrocatalytic hydrogen evolution reactions. Adv. Mater. 2016, 28, 1917–1933.
Meng, X. Y.; Yu, L.; Ma, C.; Nan, B.; Si, R.; Tu, Y. C.; Deng, J.; Deng, D. H.; Bao, X. H. Three-dimensionally hierarchical MoS2/graphene architecture for high-performance hydrogen evolution reaction. Nano Energy 2019, 61, 611–616.
Puangsombut, P.; Tantavichet, N. Effect of plating bath composition on chemical composition and oxygen reduction reaction activity of electrodeposited Pt-Co catalysts. Rare Met. 2019, 38, 95–106.
Turner, J. A. Sustainable hydrogen production. Science 2004, 305, 972–974.
Yin, H. J.; Tang, Z. Y. Ultrathin two-dimensional layered metal hydroxides: An emerging platform for advanced catalysis, energy conversion and storage. Chem. Soc. Rev. 2016, 45, 4873–4891.
Zhu, J.; Hu, L. S.; Zhao, P. X.; Lee, L. Y. S.; Wong, K. Y. Recent advances in electrocatalytic hydrogen evolution using nanoparticles. Chem. Rev. 2020, 120, 851–918.
Shen, J.; Wang, D.S. How to select heterogeneous CO2 reduction electrocatalyst. Nano Research Energy, in press, https://doi.org/10.26599/NRE.2023.9120096.
Bender, J. T.; Petersen, A. S.; Østergaard, F. C.; Wood, M. A.; Heffernan, S. M. J.; Milliron, D. J.; Rossmeisl, J.; Resasco, J. Understanding cation effects on the hydrogen evolution reaction. ACS Energy Lett. 2023, 8, 657–665.
Wu, S. H.; Li, C.; Wang, Y.; Zhuang, Y.; Pan, Y.; Wen, N.; Wang, S.; Zhang, Z. Z.; Ding, Z. X.; Yuan, R. S. et al. The Keto-switched photocatalysis of reconstructed covalent organic frameworks for efficient hydrogen evolution. Angew. Chem., Int. Ed. 2023, 62, e202309026.
Yoo, S. H.; Aota, L. S.; Shin, S.; El-Zoka, A. A.; Kang, P. W.; Lee, Y.; Lee, H.; Kim, S. H.; Gault, B. Dopant evolution in electrocatalysts after hydrogen oxidation reaction in an alkaline environment. ACS Energy Lett. 2023, 8, 3381–3386.
Wang, F. Q.; Zhang, W. L.; Wan, H. B.; Li, C. X.; An, W. K.; Sheng, X.; Liang, X. Y.; Wang, X. P.; Ren, Y. L.; Zheng, X. et al. Recent progress in advanced core-shell metal-based catalysts for electrochemical carbon dioxide reduction. Chin Chem Lett 2022, 33, 2259–2269.
Zheng, J.; Sheng, W. C.; Zhuang, Z. B.; Xu, B. J.; Yan, Y. S. Universal dependence of hydrogen oxidation and evolution reaction activity of platinum-group metals on pH and hydrogen binding energy. Science 2016, 2, e1501602.
Sheng, W. C.; Zhuang, Z. B.; Gao, M. R.; Zheng, J.; Chen, J. G.; Yan, Y. S. Correlating hydrogen oxidation and evolution activity on platinum at different pH with measured hydrogen binding energy. Nat. Commun. 2015, 6, 5848.
Tang, P.; Huang, P. Y.; Swallow, J. E. N.; Wang, C. B.; Gianolio, D.; Guo, H.; Warner, J. H.; Weatherup, R. S.; Pasta, M. Structure-property relationship of defect-trapped Pt single-site electrocatalysts for the hydrogen evolution reaction. ACS Catal. 2023, 13, 9558–9566.
Wang, L.; Liu, Y. N.; Chen, Z. F.; Dai, Q. Z.; Dong, C. L.; Yang, B.; Li, Z. J.; Hu, X. B.; Lei, L. C.; Hou, Y. Theory-guided design of electron-deficient ruthenium cluster for ampere-level current density electrochemical hydrogen evolution. Nano Energy 2023, 115, 108694.
Yang, W. W.; Li, M. Y.; Zhang, B. K.; Liu, Y. Z.; Zi, J. Z.; Xiao, H.; Liu, X. Y.; Lin, J. K.; Zhang, H. Y.; Chen, J. et al. Interfacial microenvironment modulation boosts efficient hydrogen evolution reaction in neutral and alkaline. Adv. Funct. Mater., in press, https://doi.org/10.1002/adfm.202304852.
Zheng, X. Z.; Shi, X. Y.; Ning, H. H.; Yang, R.; Lu, B.; Luo, Q.; Mao, S. J.; Xi, L. L.; Wang, Y. Tailoring a local acid-like microenvironment for efficient neutral hydrogen evolution. Nat. Commun. 2023, 14, 4209.
Wang, L.; Zhu, Y. H.; Zeng, Z. H.; Lin, C.; Giroux, M.; Jiang, L.; Han, Y.; Greeley, J.; Wang, C.; Jin, J. Platinum-nickel hydroxide nanocomposites for electrocatalytic reduction of water. Nano Energy 2017, 31, 456–461.
Wang, P. T.; Zhang, X.; Zhang, J.; Wan, S.; Guo, S. J.; Lu, G.; Yao, J. L.; Huang, X. Q. Precise tuning in platinum-nickel/nickel sulfide interface nanowires for synergistic hydrogen evolution catalysis. Nat. Commun. 2017, 8, 14580.
Ning, S.; Ou, H.; Li, Y.; Lv, C.; Wang, S.; Wang, D.; Ye, J. Co0–Coδ+ interface double-site-mediated C–C coupling for the photothermal conversion of CO2 into light olefins. Angew. Chem., Int. Ed. 2023, 62, e202302253.
Wang, L. G.; Wu, J. B.; Wang, S. W.; Liu, H.; Wang, Y.; Wang, D. S. The reformation of catalyst: From a trial-and-error synthesis to rational design. Nano Res, in press, https://doi.org/10.1002/smll.202302866.
Subbaraman, R.; Tripkovic, D.; Chang, K.-C.; Strmcnik, D.; Paulikas, A. P.; Hirunsit, P.; Chan, M.; Greeley, J.; Stamenkovic, V.; Markovic, N. M. Trends in activity for the water electrolyser reactions on 3d M (Ni, Co, Fe, Mn) hydr(oxy)oxide catalysts. Nat. Mater. 2012, 11, 550–557.
Li, M. F.; Duanmu, K. N.; Wan, C. Z.; Cheng, T.; Zhang, L.; Dai, S.; Chen, W. X.; Zhao, Z. P.; Li, P.; Fei, H. L. et al. Single-atom tailoring of platinum nanocatalysts for high-performance multifunctional electrocatalysis. Nat. Catal. 2019, 2, 495–503.
Zhu, S. Q.; Qin, X. P.; Xiao, F.; Yang, S. L.; Xu, Y.; Tan, Z.; Li, J. D.; Yan, J. W.; Chen, Q.; Chen, M. S. et al. The role of ruthenium in improving the kinetics of hydrogen oxidation and evolution reactions of platinum. Nat. Catal. 2021, 4, 711–718.
Wang, Y.; Wu, J.; Tang, S. H.; Yang, J. R.; Ye, C. L.; Chen, J.; Lei, Y. P.; Wang, D. S. Synergistic Fe-Se atom pairs as bifunctional oxygen electrocatalysts boost low-temperature rechargeable Zn-air battery. Angew. Chem., Int. Ed. 2023, 62, e202219191.
Wang, Y.; Zheng, M.; Li, Y. R.; Ye, C. L.; Chen, J.; Ye, J. Y.; Zhang, Q. H.; Li, J.; Zhou, Z. Y.; Fu, X. Z. et al. p-d orbital hybridization induced by a monodispersed ga site on a Pt3Mn nanocatalyst boosts ethanol electrooxidation. Angew. Chem., Int. Ed. 2022, 61, e202115735.
Qin, Y. C.; Zhang, W. L.; Wang, F. Q.; Li, J. J.; Ye, J. Y.; Sheng, X.; Li, C. X.; Liang, X. Y.; Liu, P.; Wang, X. P. et al. Extraordinary p-d hybridization interaction in heterostructural Pd-PdSe nanosheets boosts C–C bond cleavage of ethylene glycol electrooxidation. Angew. Chem., Int. Ed. 2022, 61, e202200899.
Zheng, X. B.; Yang, J. R.; Li, P.; Jiang, Z. L.; Zhu, P.; Wang, Q. S.; Wu, J. B.; Zhang, E. H.; Sun, W. P.; Dou, S. X. et al. Dual-atom support boosts nickel-catalyzed urea electrooxidation. Angew. Chem., Int. Ed. 2023, 62, e202217449.
Li, R. Z.; Zhang, Z. D.; Liang, X.; Shen, J.; Wang, J.; Sun, W. M.; Wang, D. S.; Jiang, J. C.; Li, Y. D. Polystyrene waste thermochemical hydrogenation to ethylbenzene by a N-bridged Co, Ni dual-atom catalyst. J. Am. Chem. Soc. 2023, 145, 16218–16227.
Qin, Y. C.; Wang, F. Q.; Liu, P.; Ye, J. Y.; Wang, Q.; Wang, Y.; Jiang, G. C.; Liu, L. J.; Zhang, P. F.; Liu, X. B. et al. Enhancement of CH3CO* adsorption by editing d-orbital states of Pd to boost C–C bond cleavage of ethanol eletrooxidation. Sci China Chem, in press, https://doi.org/10.1007/s11426-023-1756-8.
Li, W. H.; Yang, J.; Wang, D. Long-range interactions in diatomic catalysts boosting electrocatalysis. Angew. Chem., Int. Ed. 2022, 61, e202213318.
Gao, Y.; Liu, B. Z.; Wang, D. S. Microenvironment engineering of single/dual-atom catalysts for electrocatalytic application. Adv. Mater. 2023, 35, 2209654.
Hu, Y. M.; Chao, T. T.; Li, Y. P.; Liu, P. G.; Zhao, T. H.; Yu, G.; Chen, C.; Liang, X.; Jin, H. L.; Niu, S. W. et al. Cooperative Ni(Co)-Ru-P sites activate dehydrogenation for hydrazine oxidation assisting self-powered H2 pooduttion. Angew. Chem., Int. Ed. 2023, 22, e202308800.
Zheng, X. B.; Yang, J. R,; Li, P.; Wang, Q. S.; Wu, J. B.; Zhang, E. H.; Chen, S. H.; Zhuang, Z. C.; Lai, W. H.; Dou, S. X.; Sun, W. P.; Wang, D. S.; Li, Y. D. Ir-Sn pair-site triggers key oxygen radical intermediate for efficient acidic water oxidation. Sci. Adv, 2023, 9, eadi8025.
Wang, Y.; Zhuo, H. Y.; Sun, H.; Zhang, X.; Dai, X. P.; Luan, C. L.; Qin, C. L.; Zhao, H. H.; Li, J.; Wang, M. L. et al. Implanting Mo atoms into surface lattice of Pt3Mn alloys enclosed by high-indexed facets: Promoting highly active sites for ethylene glycol oxidation. ACS Catal. 2019, 9, 442–455.
Wang, P. T.; Shao, Q.; Cui, X. N.; Zhu, X.; Huang, X. Q. Hydroxide-membrane-coated Pt3Ni nanowires as highly efficient catalysts for selective hydrogenation reaction. Adv. Funct. Mater. 2018, 28, 1705918.
Cheng, R. Q.; Min, Y. L.; Li, H. X.; Fu, C. P. Electronic structure regulation in the design of low-cost efficient electrocatalysts: From theory to applications. Nano Energy 2023, 115, 108718.
Huang, W. J.; Wang, H. T.; Zhou, J. G.; Wang, J.; Duchesne, P. N.; Muir, D.; Zhang, P.; Han, N.; Zhao, F. P.; Zeng, M. et al. Highly active and durable methanol oxidation electrocatalyst based on the synergy of platinum-nickel hydroxide-graphene. Nat. Commun. 2015, 6, 10035.
Hunt, S. T.; Milina, M.; Wang, Z. S.; Román-Leshkov, Y. Activating earth-abundant electrocatalysts for efficient, low-cost hydrogen evolution/oxidation: Sub-monolayer platinum coatings on titanium tungsten carbide nanoparticles. Energy Environ. Sci. 2016, 9, 3290–3301.
Kye, J.; Shin, M.; Lim, B.; Jang, J. W.; Oh, I.; Hwang, S. Platinum monolayer electrocatalyst on gold nanostructures on silicon for photoelectrochemical hydrogen evolution. ACS Nano 2013, 7, 6017–6023.
Zhang, B.; Zhu, H.; Zou, M. L.; Liu, X. R.; Yang, H.; Zhang, M.; Wu, W. W.; Yao, J. M.; Du, M. L. Design and fabrication of size-controlled Pt-Au bimetallic alloy nanostructure in carbon nanofibers: A bifunctional material for biosensors and the hydrogen evolution reaction. J. Mater. Sci. 2017, 52, 8207–8218.
Li, L. G.; Wang, P. T.; Shao, Q.; Huang, X. Q. Metallic nanostructures with low dimensionality for electrochemical water splitting. Chem. Soc. Red. 2020, 49, 3072–3106.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 22305101), the Natural Science Foundation of Jiangsu Province (No. BK20231032), the Fundamental Research Funds for the Central Universities (No. JUSRP123020), Doctoral Science Research Foundation of Zhengzhou University of Light Industry (No. 2021BSJJ008), the Scientific and Technological Project of Henan Province (Nos. 222102240079, 232102230139, and 212102210209), and Henan Province College Students Innovation Project (No. 202310462017).
Author information
Authors and Affiliations
Corresponding authors
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Liu, C., Zhang, P., Liu, B. et al. Long-range Pt-Ni dual sites boost hydrogen evolution through optimizing the adsorption configuration. Nano Res. 17, 3700–3706 (2024). https://doi.org/10.1007/s12274-023-6271-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-023-6271-0