Abstract
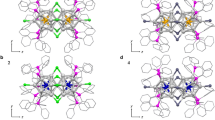
Bottom-up constructing all-metal functional materials is challenging, because the metal clusters are prone to lose their original structures during coalensence. In this work, we report that closed-shell coinage metal superatoms can achieve direct chemical bonding without losing their electronic properties. The reason is that the supermolecule formed by two superatoms has the same number of bonding and anti-bonding supermolecular orbitals, in which the bonding orbitals contribute to bonding and the anti-bonding orbitals with anti-phase orbitals delocalized over each monomer to maintain the individual geometric and electronic structural properties. Further analysis indicates the interactions between two superatoms are too weak to break the structure of monomers, which is confirmed by the first-principles molecular dynamics simulations. With these superatoms as the basic units, a series of robust one-dimensional and two-dimensional nanostructures are fabricated. Our findings provide a general strategy to take advantage of superatoms in regulating bonding compared to natural atoms, which paves the way for the bottom-up design of materials with collective properties.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Doud, E. A.; Voevodin, A.; Hochuli, T. J.; Champsaur, A. M.; Nuckolls, C.; Roy, X. Superatoms in materials science. Nat. Rev. Mater. 2020, 5, 371–387.
Li, J.; Li, X.; Zhai, H. J.; Wang, L. S. Au20: A tetrahedral cluster. Science 2003, 299, 864–867.
Desireddy, A.; Conn, B. E.; Guo, J. S.; Yoon, B.; Barnett, R. N.; Monahan, B. M.; Kirschbaum, K.; Griffith, W. P.; Whetten, R. L.; Landman, U. et al. Ultrastable silver nanoparticles. Nature 2013, 501, 399–402.
Joshi, C. P.; Bootharaju, M. S.; Alhilaly, M. J.; Bakr, O. M. [Ag25(SR)18]−:The “golden” silver nanoparticle. J. Am. Chem. Soc. 2015, 137, 11578–11581.
Wang, Z. Y.; Wang, M. Q.; Li, Y. L.; Luo, P.; Jia, T. T.; Huang, R. W.; Zang, S. Q.; Mak, T. C. W. Atomically precise site-specific tailoring and directional assembly of superatomic silver nanoclusters. J. Am. Chem. Soc. 2018, 140, 1069–1076.
Kwak, K.; Lee, D. Electrochemistry of atomically precise metal nanoclusters. Acc. Chem. Res. 2019, 52, 12–22.
Xu, W. W.; Zhu, B. E.; Zeng, X. C.; Gao, Y. A grand unified model for liganded gold clusters. Nat. Commun. 2016, 7, 13574.
Liu, X.; Xu, W. W.; Huang, X. Y.; Wang, E. D.; Cai, X.; Zhao, Y.; Li, J.; Xiao, M.; Zhang, C. F.; Gao, Y. et al. De novo design of Au36(SR)24 nanoclusters. Nat. Commun. 2020, 11, 3349.
Lei, Z.; Wan, X. K.; Yuan, S. F.; Guan, Z. J.; Wang, Q. M. Alkynyl approach toward the protection of metal nanoclusters. Acc. Chem. Res. 2018, 51, 2465–2474.
Jadzinsky, P. D.; Calero, G.; Ackerson, C. J.; Bushnell, D. A.; Kornberg, R. D. Structure of a thiol monolayer-protected gold nanoparticle at 1. 1 Å resolution. Science 2007, 318, 430–433.
Klajn, R.; Bishop, K. J. M.; Fialkowski, M.; Paszewski, M.; Campbell, C. J.; Gray, T. P.; Grzybowski, B. A. Plastic and moldable metals by self-assembly of sticky nanoparticle aggregates. Science 2007, 316, 261–264.
Wu, X.; Lu, X. Dimetalloendofullerene U2@C60 has a U−U multiple bond consisting of sixfold one-electron-two-center bonds. J. Am. Chem. Soc. 2007, 129, 2171–2177.
Li, J.; Huang, H. C.; Chen, J.; Bu, Y. X.; Cheng, S. B. Organic ligand mediated evolution from aluminum-based superalkalis to superatomic molecules and one-dimensional nanowires. Nano Res, 2022, 15, 1162–1170.
Guo, P.; Gao, Y. Coalescence of Au nanoparticles without ligand detachment. Phys. Rev. Lett. 2020, 124, 066101.
Yin, B. Q.; Luo, Z. X. Coinage metal clusters: From superatom chemistry to genetic materials. Coord. Chem. Rev. 2021, 429, 213643.
Williamson, M. J.; Tromp, R. M.; Vereecken, P. M.; Hull, R.; Ross, F. M. Dynamic microscopy of nanoscale cluster growth at the solid-liquid interface. Nat. Mater. 2003, 2, 532–536.
Fiorio, J. L.; Barbosa, E. C. M.; Kikuchi, D. K.; Camargo, P. H. C.; Rudolph, M.; Hashmi, A. S. K.; Rossi, L. M. Piperazine-promoted gold-catalyzed hydrogenation: The influence of capping ligands. Catal. Sci. Technol. 2020, 10, 1996–2003.
Ma, Y. Y.; Zhang, S.; Chang, C. R.; Huang, Z. Q.; Ho, J. C.; Qu, Y. Semi-solid and solid frustrated Lewis pair catalysts. Chem. Soc. Rev. 2018, 47, 5541–5553.
Fiorio, J. L.; López, N.; Rossi, L. M. Gold-ligand-catalyzed selective hydrogenation of alkynes into cis-alkenes via H2 heeroolycic activation by frustrated Lewis pairs. ACS Catal. 2017, 7, 2973–2980.
Izquierdo, F.; Manzini, S.; Nolan, S. P. The use of the sterically demanding IPr* and related ligands in catalysis. Chem. Commun. 2014, 50, 14926–14937.
Chen, S. P.; Li, M. F.; Yu, S.; Louisia, S.; Chuang, W.; Gao, M. Y.; Chen, C. B.; Jin, J. B.; Salmeron, M. B.; Yang, P. D. Ligand removal of Au25 nanoclusters by thermal and electrochemical treatments for selective CO2 electroreduction to CO. J. Chem. Phys. 2021, 155, 051101.
Xie, W. Y.; Zhu, Y.; Wang, J. P.; Cheng, A. H.; Wang, Z. G. Magnetic coupling induced self-assembly at atomic level. Chin. Phys. Lett. 2019, 36, 116401.
Iwasa, T.; Nakajima, A. Geometric, electronic, and optical properties of monomer and assembly of endohedral aluminum superatomic clusters. J. Phys. Chem. C 2013, 117, 21551–21557.
Robles, R.; Khanna, S. N. Magnetism in assembled and supported silicon endohedral cages: First-principles electronic structure calculations. Phys. Rev. B 2009, 80, 115414.
Singh, A.; Sen, P. Finding the right substrate support for magnetic superatom assembly from density functional calculations. Phys. Rev. B 2015, 91, 035438.
Ohta, T.; Shibuta, M.; Tsunoyama, H.; Eguchi, T.; Nakajima, A. Charge transfer complexation of Ta-encapsulating Ta@Si16 superatom with C60. J. Phys. Chem. C 2016, 120, 15265–15271.
Du, Q. Y.; Wang, Z.; Zhou, S.; Zhao, J. J.; Kumar, V. Searching for cluster Lego blocks for three-dimensional and two-dimensional assemblies. Phys. Rev. Mater. 2021, 5, 066001.
Khanna, S. N.; Jena, P. Atomic clusters: Building blocks for a class of solids. Phys. Rev. B 1995, 51, 13705–13716.
Bergeron, D. E.; Castleman, A. W. Jr.; Morisato, T.; Khanna, S. N. Formation of Al13I−: Evidence for the superhalogen character of Al13. Science 2004, 304, 84–87.
Yoon, B.; Hakkinen, H.; Landman, U.; Worz, A. S.; Antonietti, J. M.; Abbet, S.; Judai, K.; Heiz, U. Charging effects on bonding and catalyzed oxidation of CO on Au8 clusters on MgO. Science 2005, 307, 403–407.
Reber, A. C.; Khanna, S. N.; Castleman, A. W. Superatom compounds, clusters, and assemblies: Ultra alkali motifs and architectures. J. Am. Chem. Soc. 2007, 129, 10189–10194.
Luo, Z. X.; Castleman, A. W. Special and general superatoms. Acc. Chem. Res. 2014, 47, 2931–2940.
Liu, G. X.; Pinkard, A.; Ciborowski, S. M.; Chauhan, V.; Zhu, Z. G.; Aydt, A. P.; Khanna, S. N.; Roy, X.; Bowen, K. H. Tuning the electronic properties of hexanuclear cobalt sulfide superatoms via ligand substitution. Chem. Sci. 2019, 10, 1760–1766.
Lee, J.; Yang, J.; Kwon, S. G.; Hyeon, T. Nonclassical nucleation and growth of inorganic nanoparticles. Nat. Rev. Mater. 2016, 1, 16034.
Zhou, T. W.; Wang, M.; Zang, Z. G.; Tang, X. S.; Fang, L. Two-dimensional lead-free hybrid halide perovskite using superatom anions with tunable electronic properties. Sol. Energy Mater. Sol. Cells 2019, 191, 33–38.
Gao, Y.; Wang, B.; Lei, Y. Y.; Teo, B. K.; Wang, Z. G. Actinide-embedded gold superatom models: Electronic structure, spectroscopic properties, and applications in surface-enhanced Raman scattering. Nano Res. 2016, 2, 622–632.
Shen, H.; Xiang, S. J.; Xu, Z.; Liu, C.; Li, X. H.; Sun, C. F.; Lin, S. C.; Teo, B. K.; Zheng, N. F. Superatomic Au13 clusters ligated by different N-heterocyclic carbenes and their ligand-dependent catalysis, photoluminescence, and proton sensitivity. Nano Res. 2020, 13, 1908–1911.
Liu, J.; Guo, P.; Zheng, J. M.; Zhao, P. J.; Jiang, Z. Y.; Shen, L. Self-assembly of a two-dimensional sheet with Ta@Si16 superatoms and its magnetic and photocatalytic properties. J. Phys. Chem. C 2020, 124, 6861–6870.
Liu, L. R.; Cheng, L. J.; Yang, J. L. The superatomic molecule theory of metal clusters. Sci. Sin.: Chim. 2018, 48, 143–153.
Mingos, D. M. P. Bonding in molecular clusters and their relationship to bulk metals. Chem. Soc. Rev. 1986, 15, 31–61.
Rodriguez, J. A.; Goodman, D. W. The nature of the metal-metal bond in bimetallic surfaces. Science 1992, 257, 897–903.
Perdew, J. P. Density-functional approximation for the correlation energy of the inhomogeneous electron gas. Phys. Rev. B 1986, 33, 8822–8824.
Becke, A. D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 1988, 38, 3098–3100.
Van Lenthe, E.; Ehlers, A.; Baerends, E. J. Geometry optimizations in the zero order regular approximation for relativistic effects. J. Chem. Phys. 1999, 110, 8943–8953.
Morokuma, K. Molecular orbital studies of hydrogen bonds. III. C=O⋯H−O hydrogen bond in H2CO⋯H2O and H2CO⋯2H2O. J. Chem. Phys. 1971, 55, 1236–1244.
Ziegler, T.; Rauk, A. On the calculation of bonding energies by the Hartree Fock Slater method: I. The transition state method. Theor. Chim. Acta 1977, 46, 1–10.
Te Velde, G.; Bickelhaupt, F. M.; Baerends, E. J.; Guerra, C. F.; Van Gisbergen, S. J. A.; Snijders, J. G.; Ziegler, T. Chemistry with ADF. J. Comput. Chem. 2001, 22, 931–967.
Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G. A. et al. Gaussian 09; Gaussian, Inc.: Wallingford, CT, USA, 2013.
Lu, T.; Chen, F. W. Multiwfn: A multifunctional wavefunction analyzer. J. Comput. Chem. 2012, 33, 580–592.
Car, R.; Parrinello, M. Unified approach for molecular dynamics and density-functional theory. Phys. Rev. Lett. 1985, 55, 2471–2474.
Hutter, J.; Iannuzzi, M.; Schiffmann, F.; VandeVondele, J. CP2K:Atomistic simulations of condensed matter systems. WIREs Comput. Mol. Sci. 2014, 4, 15–25.
Pham, H. T.; Cuong, N. T.; Tam, N. M.; Tung, N. T. A systematic investigation on CrCun clusters with n = 9–16: Noble gas and tunable magnetic property. J. Phys. Chem. A 2016, 120, 7335–7343.
Chen, L.; Wang, Z. G.; Li, Z. Q.; Zhang, R. Q. Chemical coupling SERS properties of pyridine on silver-caged metal clusters M@Ag12 (M = V−, Nb−, Ta−, Cr, Mo, W, Mn+, Tc+, Re+). J. Electron. Mater. 2017, 46, 3904–3909.
Nijamudheen, A.; Jose, D.; Datta, A. Metal encapsulation mediated planar to three dimensional structural transformation in Au-clusters: The venus flytrap effect. Comput. Theor. Chem. 2011, 966, 133–136.
Li, X.; Kiran, B.; Li, J.; Zhai, H. J.; Wang, L. S. Experimental observation and confirmation of icosahedral W@Au12 and Mo@Au12 molecules. Angew. Chem., Int. Ed. 2002, 41, 4786–4789.
Pyykkö, P.; Runeberg, N. Icosahedral WAu12: A predicted closed-shell species, stabilized by aurophilic attraction and relativity and in accord with the 18-electron rule. Angew. Chem., Int. Ed. 2002, 41, 2174–2176.
Pyykkö, P. Understanding the eighteen-electron rule. J. Organomet. Chem. 2006, 691, 4336–4340.
Park, S.; Kim, G.; Kwon, Y. K. First-principles investigation on dimerization of metal-encapsulated gold nanoclusters. RSC Adv. 2014, 4, 192–198.
Bader, R. F. W. Atoms in molecules. Acc. Chem. Res. 1985, 15, 9–15.
Bianchi, R.; Gervasio, G.; Marabello, D. Experimental electron density analysis of Mn2(CO)10: Metal-metal and metal-ligand bond characterization. Inorg. Chem. 2000, 39, 2360–2366.
Lu, T.; Chen, Q. X. Independent gradient model based on Hirshfeld partition: A new method for visual study of interactions in chemical systems. J. Comput. Chem. 2022, 43, 539–555.
Cheng, L. J.; Yang, J. L. Communication: New insight into electronic shells of metal clusters: Analogues of simple molecules. J. Chem. Phys. 2013, 138, 141101.
Cheng, L. J.; Zhang, X. Z.; Jin, B. K.; Yang, J. L. Superatom-atom super-bonding in metallic clusters: A new look to the mystery of an Au20 pyramid. Nanoscale 2014, 6, 12440–12444.
Wan, Q. Y.; Yang, J.; To, W. P.; Che, C. M. Strong metal-metal Pauli repulsion leads to repulsive metallophilicity in closed-shell d8 and d10 organometallic complexes. Proc. Natl. Acad. Sci. USA 2021, 118, e2019265118.
Luo, Z. X.; Reber, A. C.; Jia, M. Y.; Blades, W. H.; Khanna, S. N.; Castleman, A. W. Jr. What determines if a ligand activates or passivates a superatom cluster? Chem. Sci. 2016, 7, 3067–3074.
Cheng, L. J.; Ren, C. D.; Zhang, X. Z.; Yang, J. L. New insight into the electronic shell of Au38(SR)24: A superatomic molecule. Nanoscale 2013, 5, 1475–1478.
Acknowledgements
We would like to thank Ms. Zheng Liu and Ms. Aihua Cheng for the stimulating discussion. This work was supported by the National Science Foundation of China (Nos. 11974136 and 11674123). Z. G. W. also acknowledges the High-Performance Computing Center of Jilin University and National Supercomputing Center in Shanghai.
Author information
Authors and Affiliations
Corresponding authors
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Yu, F., Zhu, Y., Gao, Y. et al. Direct assembly between closed-shell coinage metal superatoms. Nano Res. 15, 8665–8672 (2022). https://doi.org/10.1007/s12274-022-4478-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-022-4478-0