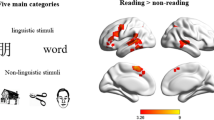
Abstract
Chinese, as a logographic language, fundamentally differs from alphabetic languages like English. Previous neuroimaging studies have mainly focused on alphabetic languages, while the exploration of Chinese reading is still an emerging and fast-growing research field. Recently, a growing number of neuroimaging studies have explored the neural circuit of Chinese reading. Here, we summarize previous research on Chinese reading from a connectomic perspective. Converging evidence indicates that the left middle frontal gyrus is a specialized hub region that connects the ventral with dorsal pathways for Chinese reading. Notably, the orthography-to-phonology and orthography-to-semantics mapping, mainly processed in the ventral pathway, are more specific during Chinese reading. Besides, in addition to the left-lateralized language-related regions, reading pathways in the right hemisphere also play an important role in Chinese reading. Throughout, we comprehensively review prior findings and emphasize several challenging issues to be explored in future work.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Reading is a complex skill used to obtain information by literate humans. Specifically, reading includes word and text reading. For word reading, mental procedures consist of early visual processing, orthographic processing, extracting phonological and semantic information from the mental lexicon, and accessing meaning [1,2,3,4]. Based on word recognition, text reading additionally requires individuals to make metalinguistic decisions about isolated words or characters, which not only necessitates a dynamic integration of vision, visual attention, eye movements, and linguistic processes of the visual stimuli presented but also involves attendant perceptual, memory, and reasoning processes [5,6,7]. Based on the long history of behavioral experiments with normal and neurologically impaired readers, some cognitive models have been proposed to simulate reading based on the dissociation of different cognitive processes, such as the dual-route model and the connectionist model [8,9,10,11]. Based on these models, neuroimaging methods and experimental paradigms have been applied to explore the underlying neurocognitive mechanisms for each cognitive process, especially visual word recognition [1, 12]. Consequently, brain regions centered on the left ventral occipitotemporal cortex, left inferior and middle frontal gyrus, and left superior and middle temporal cortices have been found to be consistently recruited by reading across different writing systems [13,14,15]. However, the depiction of the neural basis of the hierarchical coding and transformation across the different cognitive processes during reading remains a challenge [16]; the characterization of neural circuits for reading is mostly inferred empirically [15].
Recently, researchers found that for a brain region, the accomplishment of its specific functions depends on its embedding in the brain connectome [17,18,19,20]. The brain connectome refers to the characterization of structural or functional connectivity patterns through the combination of non-invasive neuroimaging techniques and graph theoretical approaches. These approaches have been widely applied to investigate the topological organization of interconnections between neural elements (i.e., nodes) [18, 21]. Nodes usually refer to imaging voxels, or regions of interest defined by functional or structural similarity [22, 23]. Structural connections can be constructed as diffusion magnetic resonance imaging (MRI)-traced white matter projections or by computing the covariance of morphological features (e.g., cortical thickness and gray matter volume) [24, 25]. Functional connections can be obtained by examining synchronous neural activity over distributed brain areas with signals recorded by functional MRI (fMRI), electroencephalography/magnetoencephalography (EEG/MEG), or functional near-infrared spectroscopy [26, 27]. After network construction, graph theory-based analysis methods are used to evaluate the topological organization [21, 28, 29]. Notably, exploring the brain connectome of reading could help us not only to better interpret the complex interacting cognitive processes but also reveal the underlying neural circuit for reading.
Writing systems exist for the sole purpose of representing spoken language and vary in how their visual forms represent units of spoken language [30, 31]. The world’s writing systems can be mainly divided into three types: alphabetic, such as English, syllabic, such as Japanese Kana, and logographic, such as Chinese [32]. The alphabetic writing system, whose grapheme is composed of a letter or digraph of letters, encodes spoken units at the level of the phoneme [33]. The syllabic system uses basic operational graphic units to encode speech syllables, and Japanese Kana is one of the purest examples [31]. The logographic system, whose grapheme consists of strokes and radicals that fit into a square space, represents spoken language in terms of syllable and morpheme, thus often called the morpheme-syllabic system [31]. Notably, logographic systems use symbols to represent meaning directly and have no and comparatively few cues to pronunciation, whereas alphabetic and syllabic systems use symbols to represent speech sounds [34]. Thus, the main discrepancies between these different systems are in the level of linguistic analysis to which their basic functional units relate [31, 35,36,37,38].
Each Chinese character is associated with a syllable and a meaning [31]. The stroke is the smallest visual unit of Chinese characters and the number and position of strokes within characters are critical for recognition [39]. Different from letters in alphabetic languages, there is no systematic mapping relation between strokes and segments; that is, strokes do not contain any phonological and semantic information nor incorporate orthography-to-phonology mapping clues. But radicals, which consist of strokes, have various orthographic patterns and can provide phonological or semantic cues for the whole character [40, 41]. Nevertheless, the pronunciation of a character cannot be computed sound-by-sound from its constituent parts [32]. Thus, the orthography-to-phonology conversion for Chinese character recognition is accessible only by recourse to direct retrieval of phonological information stored in the mental lexicon [42]. Notably, as a morphologically non-inflectional language, Chinese does not have syntactic markers or features, and neither does it have a rigid word order, so Chinese syntax is inherently dependent on semantics [43, 44].
Therefore, the exploration of neurocognitive mechanisms underlying Chinese reading provides evidence for both the universality and specificity of linguistic mechanisms. Currently, cognitive and neuroimaging studies are largely focused on alphabetic writing systems, while exploration of the Chinese system is relatively lacking. Until recently, many studies have been conducted to investigate both the cognitive models and the neural mechanisms of Chinese reading by applying various methods and experimental paradigms, while the findings remain inconclusive. In addition, although several meta-analytic studies have been conducted on the neuroimaging findings of Chinese reading, a review on Chinese reading from a connectomics perspective is still lacking.
In the connectomics framework, the brain can be modeled as a network comprising neural elements or brain regions (i.e., nodes) and their pairwise interactions (i.e., connections/edges) [45, 46]. The nodes are typically defined by using a prior anatomical/functional parcellation or regions of interest identified by functional imaging studies [47]. The connections can be defined by either structural connectivity, which is defined by structural or diffusion MRI, or functional interaction which is defined by functional MRI, between network nodes [47]. Based on the constructed brain networks, general organization principles like the modular, small-world, and heavy-tailed can be scaled with graph-theory analysis methods [48]. Here, we summarize and review the relevant findings and challenges concerning the brain networks for Chinese reading, especially its neural mechanisms with respect to the specific linguistic properties of Chinese characters. First, we introduce the important element, nodes, for the establishment of the specific brain reading connectome, by summarizing the functional identification of cortical areas involved in Chinese reading, including word recognition and reading comprehension. Then, we review the findings on connections, the other important element for brain language network construction. Given that structural connections constrain functional connections, we review the findings on the characterization of structural and functional connections for Chinese reading. After the network construction, we review the topological organization evaluations of the brain connectome for Chinese reading based on graph-theory analysis methods. Finally, we discuss future perspectives in this field.
Node Identification for the Chinese Reading Connectome
Valid node identification is critical for estimates of true network interactions [49]. The nodes in imaging connectomics should represent distinct, functionally homogeneous neural elements or brain regions, and are defined by the four most common methods: (1) voxel-based, (2) random, (3) anatomical, and (4) functional [47, 50]. The first is to treat each voxel as a separate node, which results in very large, high-resolution networks (>104), but this method may yield a noisy or underpowered estimate of brain network properties [47, 50]. The second is to randomly parcellate the brain into discrete nodes of similar size, at various resolutions, and with unclear validity [50]. The third is based on a prior anatomical parcellation, such as the Anatomical Automatic Labeling (AAL) template [50]. The last is based on prior functional information identified in functional imaging studies, such as the coordinates of peak activations or meta-analytic results [50]. Therefore, the definition of nodes in function-specific studies, such as decision making and language, are mostly based on regions of interest centered on stereotaxic coordinates identified in functional neuroimaging studies [47].
Over the past decades, functional neuroimaging and electromagnetic techniques such as positron emission tomography (PET), fMRI, MEG, and EEG have been widely used to explore brain activity during language processing, and these data can be used to identify nodes for the brain reading network [13]. Among these, fMRI is a very popular method that measures hemodynamic responses in the brain through the blood-oxygenation-level-dependent (BOLD) signal, a ratio of oxygenated versus deoxygenated hemoglobin in given regions. Using task- and resting-state fMRI, researchers have identified the specific brain activation patterns for Chinese word recognition as well as text reading.
Regions for the Orthographic Processing of Chinese Reading
Orthography refers to the information stored in memory or mental lexicon that informs us how to represent oral presentation in written form. Thus, orthographic processing is a dynamic practice of extracting the invariant, abstract structural representation from the surface structures of written words and is regarded as an antecedent stage of word recognition, providing information for subsequent processing steps such as phonological transformation [51].
Unlike the linear sequence of letters combined into words in alphabetic languages, Chinese characters consist of strokes and radicals in a square spatial configuration [12]. The visual complexity of Chinese characters places greater cognitive demands on visuospatial analytic skills than other orthographic systems and involves holistic processing [52]. Although many prior studies have revealed that the left middle fusiform gyrus (FG), named the visual word form area (VWFA), is involved in extracting the orthographic information of written words across different writing systems despite orthographic differences [13, 53,54,55,56], several recent studies have identified specific neural mechanisms for the orthographic processing of Chinese characters.
Specifically, although alphabetic writing systems mainly activate the left FG, Chinese character recognition tends to elicit activity in the bilateral FG [33, 42, 57,58,59,60]. Tan et al. found that activation in the right FG is stronger than in the left FG, revealing right lateralization [59]. Moreover, Guo and Burgund supposed that the left FG stores information in terms of parts and their relationships, while the right FG stores information holistically [61]. Comparing reading Chinese characters and English words, the right occipital gyrus is activated only in Chinese word reading, which is related to holistic visual-orthographic analysis [62].
In addition to the occipital cortex, recent studies have found that the left middle frontal gyrus (MFG) and left posterior temporal cortex are also associated with orthographic processing using lexical decision tasks during Chinese character recognition [58, 63]. Specifically, the left MFG in reading Chinese is associated with the unique square configuration of the logographeme. The function of the left posterior temporal cortex is involved in the active retrieval of the orthographic architecture of Chinese characters. In addition, a set of right hemisphere cortical areas, such as the right frontal pole, frontal operculum, dorsal lateral frontal gyrus, and superior and inferior parietal lobules, is involved in orthographic processing both in semantic and homophonic judgment tasks [59]. Among these brain areas, the right frontal and parietal regions are proposed to perceive the spatial locations of the strokes and the processes of stroke combinations. The right MFG is also thought to be relevant to orthographic processing and specifically involved in visual working memory and attentional load [64]. Nevertheless, some studies still indicated that the neural network underlying naming a single Chinese word is left-lateralized, and brain activations relevant to orthographic processing are noted in the left extrastriate areas (lateral inferior/middle occipital gyrus), left basal temporal cortex, and left superior temporal region [65, 66].
Notably, focusing on the development of Chinese reading, Cao et al. suggested that the right superior parietal lobule and right inferior temporal gyrus are associated with orthographic processing [67]. The right superior parietal lobule is involved in visual spatial analysis in mental rotation and the shifting of spatial attention, and the right inferior temporal gyrus is involved in nonlinguistic visual configuration processing. In addition, the authors indicated that with development, the involvement of the right superior parietal lobule increases, while the involvement of the right inferior temporal gyrus decreases.
To summarize, the left middle occipital gyrus, left ventral occipitotemporal cortex, left superior temporal gyrus, and left MFG are involved in orthographic processing (Fig. 1). Particularly, in line with the specialized linguistic properties of Chinese characters, the right middle occipital gyrus, right ventral occipitotemporal cortex, right inferior parietal lobule, right superior parietal lobule, dorsal part of the right inferior frontal gyrus (IFG), and right MFG are activated more in Chinese than in alphabetic languages during orthographic processing. However, whether the Chinese VWFA is left-lateralized, right-lateralized, or bilateral has not been conclusively established.
Identification of nodes for the Chinese reading connectome. The bilateral middle occipital gyrus (MOG) and ventral occipitotemporal cortex (vOT), right inferior parietal lobule (IPL), right superior parietal lobule (SPL), and right middle frontal gyrus (MFG) are involved in orthographic processing. The left angular gyrus (AG) and ventral part of the left inferior frontal gyrus (vIFG) are involved in semantic processing. The left middle temporal gyrus (MTG) and left anterior temporal lobe (ATL) are involved in syntactic processing. The left supramarginal gyrus (SMG), left superior temporal gyrus (STG), and dorsal part of the right inferior frontal gyrus are involved in orthographic and phonological processing. The dorsal part of the left inferior frontal gyrus (dIFG) is involved in phonological and syntactic processing. The left SPL and left IPL are involved in phonological and semantic processing. The left MFG is involved in orthographic, phonological, semantic, and syntactic processing. L, left hemisphere; R, right hemisphere.
Regions for the Phonological Processing of Chinese Reading
Phonological processing transforms the abstract structural representation from orthographic processing to its abstract phonological form and maps onto its phonology, deemed a subsequent stage of orthographic processing [51]. Although different cognitive models of word recognition have different views on how the three sets of processing units (orthography, phonology, and semantics) interact with each other, it has been consistently hypothesized that phonological processing can be dissociated into two stages: orthography-to-phonology mapping and general phonological access [68].
According to the orthographic differences between alphabetic and logographic scripts, cognitive models have hypothesized that these two writing systems have distinct depth and consistency in their phonological clues during visual word recognition. Specifically, the mapping between orthography and phonology is more transparent and consistent in alphabetic languages such as Italian and English but extremely opaque and less consistent in Chinese characters with weak phonological clues (morphemic reading) [36, 62, 69, 70]. Therefore, the orthography-to-phonology conversion of alphabetic scripts relies more on the sublexical route, which involves assembling the phonology associated with the whole word from its sublexical parts (sequential reading), whereas the phonological access of Chinese characters relies more on the lexical route involved in retrieving phonology directly from the orthography of the whole character [13].
Recent functional neuroimaging studies with various experimental paradigms, such as reading aloud [58], reading silently [65, 71, 72], homophone judgment [51, 59], and lexical decisions [57, 73], have been used to investigate the orthography-to-phonology conversion and phonological processing for Chinese reading. The former three tasks explicitly require the orthography-to-phonology transformation, while lexical decision tasks do not necessarily require such transformation [72]. Task studies that are theoretically involved in the orthography-to-phonology transformation proposed that the left (pre)motor cortex, left superior temporal lobe, bilateral inferior frontal cortices, left temporoparietal region (superior parietal gyrus and supramarginal gyrus), left temporal–occipital junction, left MFG, bilateral inferior prefrontal cortex, and left medial prefrontal lobe are involved in the orthography-to-phonology mapping in naming Chinese [58, 59, 72, 74, 75]. For instance, one fMRI study suggested that the posterior dorsal part of the mid-inferior frontal regions and the left inferior parietal lobule are associated with phonological processing, especially when involved in orthography-to-phonology mapping, by applying parallel rhyming and meaning association judgment tasks [75]. In addition, a lifespan fMRI study on reading Chinese words identified seven activation clusters in the homophone task, including the left mid-inferior frontal regions, the medial frontal cortex, supplementary motor area, and left FG, and the striate-extrastriate cortices, the left caudate nucleus and thalamus, right inferior frontal cortex and insula, left superior parietal lobule, and right caudate nucleus and putamen [74].
However, most neuroimaging studies of Chinese reading cannot dissociate the orthography-to-phonology mapping and general phonological processing due to limitations of the methodology and temporal resolution of fMRI. Three meta-analytic studies consistently proposed that the phonological processing of Chinese characters recruits the left inferior parietal lobule, the more posterior dorsal part of the left IFG, and the left MFG, which differ from the areas recruited by the phonological processing of alphabetic words [15, 33, 42]. Consistently, a Chinese reading study by Cao et al. focused on the phonological deficits of Chinese children with dyslexia, and they found that Chinese dyslexia was impaired in the dorsal part of the left IFG [76].
To summarize, the left supramarginal gyrus, left superior temporal gyrus, dorsal part of the left IFG, left superior parietal lobule, left inferior parietal lobule, and left MFG are involved in phonological processing (Fig. 1). Hence, consistent with the differences between Chinese and alphabetic languages on addressed and assembled phonology, neuroimaging studies have found that the left MFG is more involved in addressed phonology during Chinese character recognition, whereas the left temporoparietal areas are more relevant to assembled phonology during alphabetic word reading [33, 42]. In addition, in line with the assumptions of the cognitive models of Chinese word recognition [36, 77], especially the identification-with-phonology hypothesis that places phonology as a constituent of word recognition, these neuroimaging findings have indicated that phonological information is indeed processed during Chinese character recognition.
Regions for the Semantic Processing of Chinese Reading
Prior work has indicated that reading acquisition builds on spoken language, which is already well developed before learning to read [78, 79]. Once a written word is decoded phonologically, its meaning becomes accessible through the existing phonology-to-semantics correspondence in the spoken language system [67, 79, 80]. In this sense, the neural basis of semantic processing in reading should be more consistent than that of orthographic processing and phonological processing across different writing systems. In the literature on English and Chinese word reading, it has been consistently shown that the left frontal gyrus contributes to semantic processing [57, 81, 82].
Previous fMRI studies in Chinese reading suggested that the left MFG, bilateral inferior and middle prefrontal gyri, bilateral frontal pole, and precentral (motor) gyri are activated in both word generation and semantic decision tasks [57, 59]. In addition, in the semantic decision task, the left and right superior parietal lobules and inferior or postcentral parietal gyri are significantly activated [59]. By applying a semantic association task, Dong et al. also demonstrated that the ventral part of the left IFG and the right parietal and frontal networks are involved in semantic processing [63]. In addition, the results of this study supported the hypothesis that the anterior ventral part of the left IFG is associated with semantic processing, while the posterior dorsal part is related to phonological processing.
The left angular gyrus has also been considered a critical area for semantic processing in word reading, particularly for lexical-semantic processing [83]. In Chinese reading, the left angular gyrus indicates a function of integration between the orthographic input and the meaning [14]. It has been shown that the left angular gyrus is more active for Chinese characters than for Chinese pinyin scripts [84]. Both types of the script can convey meaning and sound, whereas more orthographic processing is engaged when reading characters than in alphabetic-like pinyin scripts. Therefore, additional activity in the left angular gyrus for Chinese characters compared with the activity noted for Chinese pinyin reflects the processing of mapping from orthography to meaning.
A point that needs explicit discussion is that these studies are largely task-dependent, and it is difficult to dissociate the neural substrates underlying these three language-processing components by applying current experimental paradigms and traditional methodologies. Therefore, it is necessary to introduce more elaborate experimental designs and methodologies to investigate the relationships between specific language-processing components and neural bases. For instance, although the left middle FG has been consistently regarded as the VWFA, two new fMRI studies using the representational similarity analysis (RSA) method have suggested that the left middle FG not only influences orthographic representations but also represents phonological and semantic processing [85, 86]. In addition, a recent study indicated that the VWFA is a key site for subserving general semantic processes linking words and meaning by spoken and written word comprehension and production tasks (i.e., listening, speaking, reading, and writing) [87].
Overall, the left angular gyrus, ventral part of the left IFG, left superior parietal lobule, left inferior parietal lobule, left MFG, and right inferior frontal gyrus are involved in semantic processing (Fig. 1). Although the linguistic features of Chinese characters differ significantly from those of alphabetic languages such as English, consistent neuroimaging findings have shown that the ventral part of the left IFG is involved in the semantic processing of both writing systems.
Regions for the Syntactic Processing of Chinese Reading
Given that syntax is a set of rules that guide the combination of words into phrases and sentences based on their syntactic category information, only reading comprehension comprising phrase and sentence reading involves syntactic processing [88]. Reading comprehension integrates information at different levels through an online cognitive process [89]. Specifically, sentence comprehension requires the engagement and coordination of word recognition, lexical access and retrieval, syntactic parsing based on word category and morphosyntactic information, semantic integration, and pragmatic inferencing among other processes. However, in the adult brain, the semantic and syntactic domains are dissociable both in terms of their neuroanatomical localization and in terms of the time windows at the last stage of language comprehension [90]. Hence, a critical question in the cognitive neuroscience of language is whether semantic and syntactic processing involve distinct cortical regions. In addition, unlike alphabetic languages, Chinese has limited morphosyntax and a lack of agreement between sentential arguments [91]. Thus, the syntactic status of a Chinese word can be more context-dependent derived from semantic information so that syntax and semantics are not always clearly demarcated [88]. In general, due to the difference between Chinese and English in syntax and semantics, research in Chinese will expand our knowledge of the universality and specificity of the neural mechanisms underlying syntactic and semantic processing.
To test whether semantic and syntactic processing in Chinese involve distinctive brain structures, Luke et al. conducted the first neuroimaging study comparing semantic and syntactic processing during Chinese phrase comprehension [88]. Their study found that the cortical regions involved in the syntactic processing of Chinese phrases are consistent with the cortical regions relevant to semantic processing, including the left mid-inferior frontal and mid-superior temporal cortices. In addition, they indicated that the left MFG is more involved in syntactic processing and that the left inferior prefrontal gyrus and mid-superior temporal gyrus are more relevant to semantic processing. Nevertheless, this study hypothesized that syntactic processing is less dependent on reading Chinese than on reading English and that it heavily interacts with semantic processing.
Following the study of Luke et al., Wang et al. examined the cortical activations of syntactic and semantic processing during Chinese sentence reading to test whether any brain region is specialized in syntactic processing in Chinese by applying a different experimental paradigm [92]. In contrast with the implications of Luke’s study, they suggested that the left IFG (BA44) is uniquely involved in syntactic processing in Chinese sentence reading, which is consistent with the literature on alphabetic languages. This implies that there are some common neural substrates underlying syntactic processing across different writing systems. By comparing brain activation patterns when reading sentences, unstructured word lists, and unstructured character lists in Chinese, Bulut et al. also indicated that reading Chinese sentences involves a common network including the left anterior temporal regions and left IFG, as when reading alphabetic languages [91]. In addition, the independent role of the left IFG on syntactic processing was also found during the syntactic processing of Chinese classifier phrases and sentences [93,94,95]. More recently, an fMRI study investigated the neural basis of Chinese syntactic representation by using a syntactic priming paradigm [96]. They revealed syntactic repetition suppression effects in the left temporal pole, left IFG, and left precentral gyrus.
In conclusion, the left middle temporal gyrus, left anterior temporal lobe, dorsal part of the left IFG, and left MFG are engaged in syntactic processing (Fig. 1). In addition, the left IFG may be a common region involved in syntactic processing across different languages. In this sense, there is a common distributed network of syntactic processing during reading comprehension, indicating that the language function at the higher level is more language-general. Although many EEG studies found that syntactic processing is independent of semantic processing during Chinese sentence comprehension (e.g., [97, 98]), given that research on the neural mechanisms underlying Chinese syntax is quite limited, whether and to what extent Chinese syntax is independent of semantics is still hotly debated.
Taken together, converging evidence indicates that the bilateral middle occipital gyrus, bilateral ventral occipitotemporal cortex, left angular gyrus, left supramarginal gyrus, bilateral superior and inferior parietal lobule, left middle and superior temporal gyrus, left anterior temporal regions, dorsal part of the left IFG, ventral part of the bilateral IFG, bilateral frontal pole, bilateral middle prefrontal gyri, and bilateral MFG construct the neural circuit for Chinese reading (Fig. 1). Notably, converging findings have shown that the left MFG, which is involved in the integration of orthography, phonology, semantics, and syntax, is an important region for Chinese character recognition. The functional identification of these brain regions allows us to define the nodes of the Chinese reading connectome and to further construct the reading networks and explore the relevant topological organization.
Characterization of the Connections for the Chinese Reading Connectome
For the harmonious functioning of segregated brain regions, efficient information exchange among regions through white matter fiber systems is vital. Notably, functional networks reflect the architecture of coupling connections, whereby structural connections may provide the neural basis for the accomplishment of functional connectivity [18]. Given that reading constitutes a complex multifaceted process involving visual and linguistic components, it is not surprising that it activates distributed and distant cortical regions. Thus, fluent reading not only requires adequate activation of the relevant cortical areas but also adequate communication between these areas [99]. The structural and functional connectivity of the brain connectome for reading reflecting interactions between language-related brain regions serves as the neural substrate for the phylogeny and ontogeny of reading.
Structural Connections for Chinese Reading
White matter fiber tracts connecting the language areas ensure efficient signal transmission between distant cortical regions, and anatomical connectivity also provides neurobiological bases for functional connectivity. Until recently, postmortem and lesion studies of white matter pathways have identified the anatomical basis of connections between reading-related cortical areas [99]. Currently, the application of diffusion tensor imaging (DTI) offers the possibility to reconstruct white matter pathways via tractography and examine white matter properties noninvasively in humans. The major white matter pathways that connect reading-related cortical areas are the arcuate fasciculus (AF, part of the superior longitudinal fasciculus), the inferior fronto-occipital fasciculus (IFOF), and the inferior longitudinal fasciculus (ILF) [99].
As discussed above, due to the vast differences in alphabetic languages (e.g., English) and logographic languages (e.g., Chinese character), in recent decades, neuroimaging studies have identified common cortical regions for reading across different writing systems as well as cortical regions that are specific for alphabetic or logographic languages. Previous studies found that the left ventral occipitotemporal cortex, anterior part of the superior temporal gyrus, and superior posterior part of the IFG are involved in orthographic processing, phonological identification of word form, and semantic processing across different writing systems [33, 100, 101]. However, Chinese character recognition might elicit additional involvement of the right occipitotemporal cortex and the left MFG [15, 33, 59, 102, 103]. It has been shown that the major hubs within the structural network are highly correlated with the major activation foci [18]. In addition, stimulus configurations and learning experiences might influence anatomical connections [18]. Therefore, investigation of the structural networks of Chinese reading can not only elucidate the basic neural mechanisms of logographic languages but also expand the findings on the structural networks for reading. Given that the relevant neural mechanisms underlying anatomical connections in the Chinese population are still largely unexplored, here, the systematic introduction of relevant studies as shown in Table 1, although very few, is necessary for future studies.
As the most important language-related pathway, the AF has been investigated for >200 years [99], and it has been consistently demonstrated that the AF connects Broca’s area and Wernicke’s area [104,105,106,107]. Based on the previous findings conducted on native readers of alphabetic languages, the AF has been suggested to subserve the dorsal phonological route (i.e., connecting the left IFG and the left temporoparietal cortex) [17, 99]. In alphabetic language populations, there is still confusion about the subcomponents of the AF and to which cortical regions these subcomponents are connected. Different DTI studies have differentiated the segments of the AF, such as twofold, threefold, or fourfold divisions (for a detailed review on the AF, see [17, 99]). For the Chinese population, until the past decade, several studies have investigated the relationship between white matter maturation and Chinese reading by using tract-based spatial statistics and probabilistic tractography of DTI [108,109,110]. With Chinese reading development, prior studies found prominent and asymmetrical changes in the left AF, which connects the frontal lobe to other brain regions [108, 109, 111]. Consistently, Shu et al. also revealed the leftward asymmetry of the AF in both micro- and macro-structural properties [112]. In addition, two recent studies by Su and colleagues have suggested that the AF-direct (connecting the IFG with the superior temporal gyrus) and AF-posterior (connecting the inferior parietal cortex with the superior temporal gyrus) have a significant correlation with reading development and developmental dyslexia by using the fiber tractography method [113, 114]. Specifically, Su and colleagues examined the effect of long-term vocabulary development on the structure of the AF, and reported a significant association between Chinese children’s vocabulary development and the left AF-direct and AF-posterior pathways [113]. Furthermore, Su et al. investigated whether Chinese children with dyslexia show a white matter disruption pattern similar to the alphabetic population with dyslexia [114]. They found reduced fractional anisotropy in the left AF-direct pathway for children with dyslexia, indicating that the disrupted integrity of the left AF-direct pathway is a universal characteristic of dyslexia. Basically, the left AF in the dorsal pathway is associated with phonological processing and may be a language-general pathway.
With regard to the ventral pathway of Chinese reading, the left ILF and IFOF are the main white matter tracts associated with orthographic and semantic processing, respectively [99, 110, 115, 116]. In children with dyslexia, Su and colleagues reported reduced fractional anisotropy in the left ILF, which is associated with morphological processing skills [114]. They also suggested that the disrupted integrity of the left ILF may be more specific to Chinese individuals. Furthermore, one lesion-behavior mapping analysis study, which investigated the correlations between the integrity of major tracts and orthographic ability, suggested a vital role of the left ILF in orthographic processing [117]. In addition, Qiu et al. found the asymmetrical changes in the right ILF during Chinese reading development [108]. However, it is still under debate whether the structure of the left IFOF is significantly correlated with developmental dyslexia or Chinese character recognition [113, 114, 118].
More recently, to establish a reading-related global subnetwork, Li and colleagues revealed that the left AF, ILF, and IFOF are the white matter pathways connecting reading-related brain regions such as the left inferior occipital gyrus, middle temporal gyrus, superior temporal gyrus, triangular part of IFG, middle occipital gyrus, supramarginal gyrus, and superior parietal lobe in Chinese brain-damaged patients by using a network-based lesion-symptom mapping method [119]. Moreover, the global reading subnetwork that they revealed is left-lateralized.
In summary, the AF, IFOF, and ILF have been suggested to be the dorsal and ventral pathways of Chinese reading in the left hemisphere (Fig. 2). The leftward asymmetry of the reading subnetwork may reflect the anatomical substrate of language-related functional lateralization of the human brain. In addition, the corpus callosum has been proposed to integrate visual information generated from the bilateral occipital cortex during Chinese character recognition [118, 120]. However, due to limited research, the picture of the neural substrate underlying the structural networks in the Chinese population is not very clear. Prior work has mainly examined the correlation between white matter changes and reading ability or development and put less attention on the details about which brain regions are connected by those white matter pathways, particularly the left ILF and IFOF.
Structural connections for Chinese reading. The left arcuate fasciculus (AF) in the dorsal pathway, which connects the frontal lobe to the temporoparietal cortex, is associated with phonological processing. The bilateral inferior longitudinal fasciculus (ILF) is associated with orthographic processing which is language-specific for Chinese reading (bold italic). The left inferior fronto-occipital fasciculus (IFOF) is associated with semantic processing. The corpus callosum (CC) is proposed to integrate visual information generated from the bilateral occipital cortex.
Functional Connections of Chinese Reading
Over the past decades, functional neuroimaging has become a predominant technique for investigating the brain’s functional architecture and operational principles [121]. As an effective method of revealing the temporal synchronization of functionally-related brain areas, resting-state functional connectivity (RSFC) analysis has been used to investigate the connection between the visual stream and reading-related areas and to analyze the correlation between the RSFC and reading competence. Although research on the interaction of distributed brain areas for reading is limited, more recently, several studies have investigated the neural pathway of Chinese reading by using fMRI data (Table 2).
Prior studies of alphabetic languages have shown that visual word recognition involves both ventral and dorsal neural pathways, corresponding to the ventral ‘what’ stream and the dorsal ‘where’ stream in the dual-route theory [122]. However, the role of the ventral and dorsal pathways in Chinese reading is still under debate. On the one hand, two studies have consistently revealed that the ventral pathway is more involved in Chinese word recognition [4, 123]. Specifically, one effective connectivity (EC) study investigated the multiple pathways model of reading Chinese and they indicated that it is the ventral pathway from the visual system to the left ventral occipitotemporal cortex but not the dorsal pathway from the visual system to the left parietal region that is engaged in Chinese character recognition [123]. Furthermore, another EC study on word recognition in Chinese indicated that while the dorsal visual pathway is mainly involved in eye-movement control, the ventral visual pathway is engaged in orthographic processing and orthography-phonology mapping [4].
On the other hand, some studies have suggested that the dorsal pathway is highly engaged in Chinese reading by analyzing the correlation between RSFC and reading skills [124,125,126]. Specifically, a recent resting-state fMRI study investigated the role of intrinsic interregional connections during Chinese reading by correlating RSFC maps containing nine Chinese reading-related seed regions with the reading competence of Chinese native speakers. The results revealed that Chinese reading competence is positively correlated with the connectivity between the left occipital gyrus and left superior parietal lobule, between the right posterior FG and right superior parietal lobule, and between the left inferior temporal gyrus and left inferior parietal lobule [125]. In addition, one study applied RSFC and Granger causality analysis to investigate the relationship between intrinsic couplings of the VWFA with the spoken language network and reading competence in Chinese children and adults and found that suppressed connections of the left supramarginal gyrus with the VWFA (i.e., the dorsal pathway) facilitate Chinese reading in developed readers [127].
More recently, several neuroimaging studies on Chinese text reading have adopted the dual-route theory to investigate the principles of information processing in the brain [5]. Zhou et al. showed that there is significant functional connectivity (FC) and EC among the left MFG, the left intraparietal sulcus, and the VWFA in both reading-driven fMRI and resting-state fMRI [5]. In addition, this study revealed dual top-down effects from the left MFG to both the dorsal region [e.g., the left intraparietal sulcus (IPS)] and the ventral region (e.g., the left VWFA). In other words, the left MFG is proposed to be a region of convergence of the dorsal and ventral visual systems during text reading. In addition, recent fMRI studies in Chinese text reading have indicated that brain areas in the dorsal pathway (e.g., the left MFG and IPS) are of great importance in visuospatial attention or eye movements when reading an entire sentence [128, 129]. Accordingly, the VWFA and the left IPS are central nodes for the ventral and dorsal reading pathways, respectively [130]. In addition, researchers have concluded that dorsal areas such as the left MFG and the IPS are responsible for eye movements and the coordination between subprocesses of text reading [5]. Therefore, recent neuroimaging studies of text reading have suggested that on the one hand, the dual-route visual systems play different roles in word reading and text reading; and on the other hand, the left MFG is a central region that connects the dorsal visual pathway with the ventral visual pathway. Notably, neuroimaging studies on Chinese text reading are still very limited, and more research is needed to investigate the neural mechanisms of text reading in detail.
Overall, converging studies have focused on the functional localization of Chinese reading; however, less attention has been focused on the neural pathways of Chinese character recognition. Particularly, due to the limitation of the temporal resolution of MRI and spatial resolution of EEG, combined neuroimaging and EEG methods, as well as MEG and intracranial methods that have a high spatiotemporal resolution, need to be applied to explore language processing, especially the functional networks of language. For example, an intracranial EEG study investigated whether high-level lexical information is involved in early visual word-form processing using a lexical decision task, and they identified directional connectivity from the high-level linguistic regions to the left ventral occipitotemporal and occipital cortices [131]. Hence, this study provided direct supportive evidence for the Chinese reading network with direction. In general, the functional connections of Chinese reading can be coarsely summarized, as shown in Fig. 3.
Topological Organization Estimation of the Chinese Reading Connectome
As described in the introduction, by using various graph-theoretic approaches, the global topological organization of a complex system can be quantified and hence provide important clues about the global and local information communication through an integrative network [21, 45]. However, to our knowledge, only a few studies have investigated the brain networks of Chinese reading from a connectomic perspective.
As discussed above, in the literature, the neural network underlying word reading has been determined through reading-relevant brain regions or white matter tracts, mainly based on MRI data. Specifically, a large-scale functional brain network study using graph theoretical analysis showed that healthy adults rely more on visual-orthographic processing, whereas children rely more on auditory phonological processing during word reading [132]. A similar shift from reliance on phonology-related networks to semantics- and vision-related networks with age has also been reported for text reading [133]. In addition, this study suggested that transmission to vision-related brain circuits enhance reading performance in adults, whereas phonology-related brain circuits play important roles in children's reading before they develop into fluent readers.
Based on cortical thickness and surface area, two MRI studies have investigated large-scale structural networks in Chinese children with dyslexia or poor reading ability. One study focused on the abnormal development of structural connectivity in dyslexic individuals, and they indicated a more bilateral, anteriorly-connected structural network in Chinese dyslexia [134]. Specifically, compared to typically developing children, in the cortical thickness network, reading disabled children showed reduced nodal network properties in the left hemisphere along with enhanced nodal properties in the right hemisphere, while in the surface area network, reading disabled children demonstrated lower nodal properties in the posterior brain regions and higher nodal properties in the anterior brain regions. Another study by Mao et al. investigated the neural topological organization of the language network in Chinese children with poor reading ability during an auditory rhyming task and a visual spelling task, and they found an aberrant topological organization in poor readers, as the opercular part of the left IFG forms deficient functional segregation and the bilateral superior temporal gyrus shows higher nodal efficiency [135].
Given that reading is a high-level cognitive ability, both regional brain activity and dynamic inter-regional communication within a large-scale brain connectome play a crucial role during reading. Over the past three decades, functional identification of Chinese reading, especially visual word recognition, has been studied based on various experimental stimuli and paradigms as well as distinct methodologies. However, research on the characterization of the Chinese reading connectome is still quite limited, thus the interaction of functional systems and neural systems remains unclear.
Concluding Remarks and Future Directions
We reviewed the brain connectome of Chinese reading based on evidence from previous studies on the functional identification, and the structural and functional networks that provide the neural basis for understanding Chinese reading. In addition, we comprehensively reviewed this important emerging field, and highlighted important implications for the language-universal and -specific neural basis across different writing systems. However, the study of Chinese reading is still in its infancy. Significant scientific questions remain to be addressed, including the complete characterization of reading-related brain regions as well as their connections with each other; and how the developmental language system changes as well as their interactions with genetic and environmental factors.
Language is a complex ability that requires the brain to process multiple heterogeneous types of information. Characterizing the complete function of related brain regions remains challenging. The most typical method applied in task fMRI studies is linearly fitting the experimental stimulus and the hemodynamic activities of brain voxels. However, shortcomings remain, such as the generalization of findings and the multifunctionality of a single region. New data-analytical frameworks have been developed to solve these problems [136,137,138]. The voxel-based encoding and decoding models have been complementarily used to investigate what sensory, cognitive, or motor information is represented in the brain. Specifically, the voxel-based encoding model can provide a complete functional description of a brain region, which is impossible for decoding models, whereas the decoding model can test direct relationships between patterns of brain activity and behavioral performance, which is also difficult for encoding models to achieve [136]. These novel models have already been used to expand the understanding of the neural basis for language processing, particularly for natural language comprehension [139, 140]. Gallant and colleagues created detailed semantic maps for the first time by using the combined encoding and decoding model and indicated that the semantic maps are independent of sensory modalities (i.e., listening and reading). Predictive models of brain activity are the gold standard of computational neuroscience and are thus critical for the long-term advancement of neuroscience and artificial intelligence. Therefore, it is important to decode brain activity, such as internal speech. Multivariate methods such as RSA, as one form of decoding, have been applied to investigate how our brain represents the cognitive processes of language [136]. By correlating neural representational similarity matrices with behavioral prediction matrices, RSA is able to better decode information represented by the pattern of activity in different cortical areas and detect fine-grained pattern differences without eliminating information by averaging [137]. Recently, RSA studies focused on word recognition have indicated that the VWFA (i.e., the left ventral occipitotemporal cortex) represents orthographic, phonological, and semantic information, which challenges the traditional VWFA hypothesis [85, 86, 141, 142].
Furthermore, language is a unique capacity of humans, which distinguishes humans from other animals in essence. Although the language system is complex with distinct linguistic processes, we use language quite effortlessly. The origin and genetic basis for language have aroused multidisciplinary discussion for centuries. Recently, modern genetic tools have been applied to explore this fascinating aspect of human beings, and the first identified language gene [forkhead box p2 (FOXP2)] was finally discovered in the field of molecular genetics [143, 144]. Specifically, mutation of FOXP2 has been identified in a family with speech and language impairment involving problems with controlling finely coordinated movement sequences of the mouth, tongue, lips, and soft palate [145]. However, FOXP2 is not a human-specific gene and is also found in nonhuman primates, birds, and fish [146,147,148]. Hence, it is still inconclusive what the species-specific differences in the regulation of expression are and how animal models can help elucidate the biological mechanisms involved in the evolution of language. In addition, the FOXP2 gene has been shown to be involved in susceptibility to other neurodevelopmental disorders with symptoms of language impairment, such as autism and schizophrenia [149, 150]. Nevertheless, the association between FOXP2 rs10447760 and schizophrenia has not always been reproduced in the Chinese Han population [151,152,153]. Thus, large-scale genetic replication studies with participants of different racial and geographic origins are required in the future. In addition to the FOXP2 gene, linkage studies on several heritable language-related disorders with various disabilities in speech or reading, such as stuttering and developmental dyslexia, have identified other language-related candidate genes [154,155,156,157]. For instance, dyslexia susceptibility 1 candidate 1 (DYX1C1) and roundabout, axon guidance receptor, homolog 1 (ROBO1) have been identified as candidates for dyslexia susceptibility by mapping translocation breakpoints to known regions of interest [158, 159]. However, linkage studies focused purely on candidate genes for language disabilities may not resolve key questions about the development of language. More recently, genome-wide association (GWAS) studies, which explore millions of common variants in the human genome, have been conducted to investigate not only language-related disorders but also language-related abilities in the general population (see review [154]). Notably, the GWAS method has been applied to investigate the genetic variations of language functional networks, using resting-state fMRI data from the UK biobank [160]. Although there has been an explosion of research on the genetic basis of human language, it is still inconclusive whether specific genes for language exist across all populations.
The intricate inner mechanisms of the brain remain one of the greatest mysteries defying resolution by contemporary scientific inquiry [48]. We need to model and characterize the interactions between scales and modalities in future research. Brain structural networks define the connectivity between neural elements based on physical measures of neural wiring, such as synapses between neurons and white matter tracts between brain regions, whereas brain functional networks define connectivity based on the similarity between the dynamics of two neural elements [161]. Although structural networks constrain functional networks, the diverse functionality of the brain derives from a fixed structure [162, 163]. Given that the relationship between structural and functional brain networks is non-linear and one-to-many, it is difficult to characterize the brain’s structure-function relationship, which is also one crucial topic of academic focus [162].
In general, the study of the brain connectome for Chinese reading remains in its infancy. Significant scientific challenges need to be addressed. Further elaboration of the organizational principles of the brain language system for Chinese reading and its development will significantly aid in uncovering the biological mechanisms of language processing.
References
Yang JF, Wang XJ, Shu H, Zevin JD. Brain networks associated with sublexical properties of Chinese characters. Brain Lang 2011, 119: 68–79.
Marshall JC, Newcombe F. Patterns of paralexia: A psycholinguistic approach. J Psycholinguist Res 1973, 2: 175–199.
Patterson K, Shewell C. Speak and spell: Dissociations and word-class effects. In Coltheart M, Sartori G, Job R (Eds.), The cognitive neuropsychology of language. Lawrence Erlbaum Associates, Inc. 1987: 273–294.
Zhou W, Xia ZC, Georgiou GK, Shu H. The distinct roles of dorsal and ventral visual systems in Naming of Chinese characters. Neuroscience 2018, 390: 256–264.
Zhou W, Wang XJ, Xia ZC, Bi YC, Li P, Shu H. Neural mechanisms of dorsal and ventral visual regions during text reading. Front Psychol 2016, 7: 1399.
Zhou W, Zhang LJ, Shu H, Li P. Neural mechanisms for the processing of Chinese. The Routledge Handbook of Chinese Applied Linguistics. Abingdon, Oxon; New York, NY: Routledge, 2019: 659–672.
Wang XJ, Yang JF, Yang J, Mencl WE, Shu H, Zevin JD. Language differences in the brain network for reading in naturalistic story reading and lexical decision. PLoS One 2015, 10: e0124388.
Seidenberg MS, McClelland JL. A distributed, developmental model of word recognition and Naming. Psychol Rev 1989, 96: 523–568.
Plaut DC, McClelland JL, Seidenberg MS, Patterson K. Understanding normal and impaired word reading: Computational principles in quasi-regular domains. Psychol Rev 1996, 103: 56–115.
Bjaalid IK, Høien T, Lundberg I. Dual-route and connectionist models: A step towards a combined model. Scand J Psychol 1997, 38: 73–82.
Coltheart M, Curtis B, Atkins P, Haller M. Models of reading aloud: Dual-route and parallel-distributed-processing approaches. Psychol Rev 1993, 100: 589–608.
Li P, Shu H, Liu YY. Neurocognitive approaches to the processing of Chinese. The Handbook of Chinese Linguistics. Hoboken, USA: John Wiley & Sons, Inc., 2014: 511–533.
Price CJ. A review and synthesis of the first 20 years of PET and fMRI studies of heard speech, spoken language and reading. Neuroimage 2012, 62: 816–847.
Zhao R, Fan R, Liu MX, Wang XJ, Yang JF. Rethinking the function of brain regions for reading Chinese characters in a meta-analysis of fMRI studies. J Neurolinguistics 2017, 44: 120–133.
Wu CY, Ho MHR, Chen SHA. A meta-analysis of fMRI studies on Chinese orthographic, phonological, and semantic processing. Neuroimage 2012, 63: 381–391.
Price CJ, Friston KJ. Functional ontologies for cognition: The systematic definition of structure and function. Cogn Neuropsychol 2005, 22: 262–275.
Friederici AD. Pathways to language: Fiber tracts in the human brain. Trends Cogn Sci 2009, 13: 175–181.
Friederici AD, Singer W. Grounding language processing on basic neurophysiological principles. Trends Cogn Sci 2015, 19: 329–338.
Li J, Osher DE, Hansen HA, Saygin ZM. Innate connectivity patterns drive the development of the visual word form area. Sci Rep 2020, 10: 18039.
Ekstrand C, Neudorf J, Kress S, Borowsky R. Structural connectivity predicts functional activation during lexical and sublexical reading. Neuroimage 2020, 218: 117008.
Cao M, Huang H, Peng Y, Dong Q, He Y. Toward developmental connectomics of the human brain. Front Neuroanat 2016, 10: 25.
Pugh KR, Shaywitz BA, Shaywitz SE, Constable RT, Skudlarski P, Fulbright RK. Cerebral organization of component processes in reading. Brain 1996, 119(Pt 4): 1221–1238.
Rumsey JM, Horwitz B, Donohue BC, Nace K, Maisog JM, Andreason P. Phonological and orthographic components of word recognition A PET-rCBF study. Brain 1997, 120(Pt 5): 739–759.
Mori S, van Zijl PCM. Fiber tracking: Principles and strategies - a technical review. NMR Biomed 2002, 15: 468–480.
Gong GL, He Y, Chen ZJ, Evans AC. Convergence and divergence of thickness correlations with diffusion connections across the human cerebral cortex. Neuroimage 2012, 59: 1239–1248.
Friston KJ, Frith CD, Liddle PF, Frackowiak RS. Functional connectivity: The principal-component analysis of large (PET) data sets. J Cereb Blood Flow Metab 1993, 13: 5–14.
Niu HJ, He Y. Resting-state functional brain connectivity: Lessons from functional near-infrared spectroscopy. Neuroscientist 2014, 20: 173–188.
Bullmore ET, Bassett DS. Brain graphs: Graphical models of the human brain connectome. Annu Rev Clin Psychol 2011, 7: 113–140.
Sporns O, Tononi G, Kötter R. The human connectome: A structural description of the human brain. PLoS Comput Biol 2005, 1: e42.
Saussure FD. Course in General Linguistics. The Philosophical Library, New York, 1959.
Florian C. Writing Systems an Introduction to Their Linguistic Analysis. Cambridge Textbooks in Linguistics, Cambridge, UK, 2003.
DeFrancis J. Visible Speech: The Diverse Oneness of Writing Systems. University of Hawaii Press, Honolulu, 1989.
Bolger DJ, Perfetti CA, Schneider W. Cross-cultural effect on the brain revisited: Universal structures plus writing system variation. Hum Brain Mapp 2005, 25: 92–104.
Ellis NC, Natsume M, Stavropoulou K, Hoxhallari L, van Daal VHP, Polyzoe N, et al. The effects of orthographic depth on learning to read alphabetic, syllabic, and logographic scripts. Read Res Q 2004, 39: 438–468.
Reichle ED, Yu LL. Models of Chinese reading: Review and analysis. Cogn Sci 2018, 42: 1154–1165.
Perfetti CA, Liu Y, Tan LH. The lexical constituency model: Some implications of research on Chinese for general theories of reading. Psychol Rev 2005, 112: 43–59.
Zhao JJ, Wang XY, Frost SJ, Sun W, Fang SY, Mencl WE, et al. Neural division of labor in reading is constrained by culture: A training study of reading Chinese characters. Cortex 2014, 53: 90–106.
Liu Y, Perfetti CA. The time course of brain activity in reading English and Chinese: An ERP study of Chinese bilinguals. Hum Brain Mapp 2003, 18: 167–175.
Shu H, Anderson RC. Learning to read Chinese: The development of metalinguistic awareness. Reading Chinese Script: A Cognitive Analysis 1999: 1–18.
Yu LL, Reichle ED. Chinese versus English: Insights on cognition during reading. Trends Cogn Sci 2017, 21: 721–724.
Tong XH, Wang Y, Tong SX. Neurocognitive correlates of statistical learning of orthographic-semantic connections in Chinese adult learners. Neurosci Bull 2020, 36: 895–906.
Tan LH, Laird AR, Li K, Fox PT. Neuroanatomical correlates of phonological processing of Chinese characters and alphabetic words: A meta-analysis. Hum Brain Mapp 2005, 25: 83–91.
Ma QZ. Categories in a Chinese semantic grammar (Hanyu Yuyi Yufa Fanchou Wenti). Beijing University of Language and Culture Press, Beijing, 1998.
Xu TQ. On language (Yuyan Lun). Northeast Normal Uiversity Press (Dongbei Shifan Daxue Chubanshe), Changchun, 2000.
Xu YW, He Y, Bi YC. A tri-network model of human semantic processing. Front Psychol 2017, 8: 1538.
Faskowitz J, Esfahlani FZ, Jo Y, Sporns O, Betzel RF. Edge-centric functional network representations of human cerebral cortex reveal overlapping system-level architecture. Nat Neurosci 2020, 23: 1644–1654.
Fornito A, Zalesky A, Pantelis C, Bullmore ET. Schizophrenia, neuroimaging and connectomics. Neuroimage 2012, 62: 2296–2314.
Lynn CW, Bassett DS. The physics of brain network structure, function and control. Nat Rev Physics 2019, 1: 318–332.
Smith SM, Miller KL, Salimi-Khorshidi G, Webster M, Beckmann CF, Nichols TE, et al. Network modelling methods for fMRI. Neuroimage 2011, 54: 875–891.
Fornito A, Zalesky A, Breakspear M. Graph analysis of the human connectome: Promise, progress, and pitfalls. Neuroimage 2013, 80: 426–444.
Kuo WJ, Yeh TC, Lee JR, Chen LF, Lee PL, Chen SS, et al. Orthographic and phonological processing of Chinese characters: An fMRI study. Neuroimage 2004, 21: 1721–1731.
Ren SW, Shao HY, He S. Interaction between conscious and unconscious information-processing of faces and words. Neurosci Bull 2021, 37: 1583–1594.
Cohen L, Lehéricy S, Chochon F, Lemer C, Rivaud S, Dehaene S. Language-specific tuning of visual cortex? Functional properties of the Visual Word Form Area. Brain 2002, 125: 1054–1069.
Cohen L, Dehaene S, Naccache L, Lehéricy S, Dehaene-Lambertz G, Hénaff MA, et al. The visual word form area: Spatial and temporal characterization of an initial stage of reading in normal subjects and posterior split-brain patients. Brain 2000, 123(Pt 2): 291–307.
Dehaene S, le Clec’H G, Poline JB, le Bihan D, Cohen L. The visual word form area: A prelexical representation of visual words in the fusiform gyrus. Neuroreport 2002, 13: 321–325.
Cohen L, Dehaene S. Specialization within the ventral stream: The case for the visual word form area. Neuroimage 2004, 22: 466–476.
Tan LH, Spinks JA, Gao JH, Liu HL, Perfetti CA, Xiong J, et al. Brain activation in the processing of Chinese characters and words: A functional MRI study. Hum Brain Mapp 2000, 10: 16–27.
Tan LH, Feng CM, Fox PT, Gao JH. An fMRI study with written Chinese. Neuroreport 2001, 12: 83–88.
Tan LH, Liu HL, Perfetti CA, Spinks JA, Fox PT, Gao JH. The neural system underlying Chinese logograph reading. Neuroimage 2001, 13: 836–846.
Tian MY, Li HH, Chu MY, Ding GS. Functional organization of the ventral occipitotemporal regions for Chinese orthographic processing. J Neurolinguistics 2020, 55: 100909.
Guo Y, Burgund ED. Task effects in the mid-fusiform gyrus: A comparison of orthographic, phonological, and semantic processing of Chinese characters. Brain Lang 2010, 115: 113–120.
Cao F, Brennan C, Booth JR. The brain adapts to orthography with experience: Evidence from English and Chinese. Dev Sci 2015, 18: 785–798.
Dong Y, Nakamura K, Okada T, Hanakawa T, Fukuyama H, Mazziotta JC, et al. Neural mechanisms underlying the processing of Chinese words: An fMRI study. Neurosci Res 2005, 52: 139–145.
Kuo WJ, Yeh TC, Lee CY, Wu YT, Chou CC, Ho LT, et al. Frequency effects of Chinese character processing in the brain: An event-related fMRI study. Neuroimage 2003, 18: 720–730.
Kuo WJ, Yeh TC, Duann JR, Wu YT, Ho LT, Hung D, et al. A left-lateralized network for reading Chinese words: A 3 T fMRI study. Neuroreport 2001, 12: 3997–4001.
Liu C, Zhang WT, Tang YY, Mai XQ, Chen HC, Tardif T, et al. The Visual Word Form Area: Evidence from an fMRI study of implicit processing of Chinese characters. Neuroimage 2008, 40: 1350–1361.
Cao F, Lee R, Shu H, Yang YH, Xu GQ, Li KC, et al. Cultural constraints on brain development: Evidence from a developmental study of visual word processing in mandarin Chinese. Cereb Cortex 2010, 20: 1223–1233.
Coltheart M. Dual route and connectionist models of reading: An overview. Lond Rev Educ 2006.
Xu YD, Pollatsek A, Potter MC. The activation of phonology during silent Chinese word reading. J Exp Psychol Learn Mem Cogn 1999, 25: 838–857.
Coltheart M, Rastle K, Perry C, Langdon R, Ziegler J. DRC: a dual route cascaded model of visual word recognition and reading aloud. Psychol Rev 2001, 108: 204–256.
Fu SM, Chen YP, Smith S, Iversen S, Matthews PM. Effects of word form on brain processing of written Chinese. Neuroimage 2002, 17: 1538–1548.
Lee CY, Tsai JL, Kuo WJ, Yeh TC, Wu YT, Ho LT, et al. Neuronal correlates of consistency and frequency effects on Chinese character Naming: An event-related fMRI study. Neuroimage 2004, 23: 1235–1245.
Sun YF, Yang YH, Desroches AS, Liu L, Peng DL. The role of the ventral and dorsal pathways in reading Chinese characters and English words. Brain Lang 2011, 119: 80–88.
Siok WT, Jia FL, Liu CY, Perfetti CA, Tan LH. A lifespan fMRI study of neurodevelopment associated with reading Chinese. Cereb Cortex 2020, 30: 4140–4157.
Booth JR, Lu D, Burman DD, Chou TL, Jin Z, Peng DL, et al. Specialization of phonological and semantic processing in Chinese word reading. Brain Res 2006, 1071: 197–207.
Cao F, Yan X, Wang Z, Liu YN, Wang J, Spray GJ, et al. Neural signatures of phonological deficits in Chinese developmental dyslexia. Neuroimage 2017, 146: 301–311.
Tan LH, Perfetti CA. Phonological codes as early sources of constraint in Chinese word identification: A review of current discoveries and theoretical accounts. Reading and Writing 1998, 10: 165–200.
Perfetti CA (1985) Reading Aability. Oxford University Press, New York.
Tan LH, Spinks JA, Eden GF, Perfetti CA, Siok WT. Reading depends on writing, in Chinese. Proc Natl Acad Sci USA 2005, 102: 8781–8785.
Rueckl JG, Paz-Alonso PM, Molfese PJ, Kuo WJ, Bick A, Frost SJ, et al. Universal brain signature of proficient reading: Evidence from four contrasting languages. Proc Natl Acad Sci USA 2015, 112: 15510–15515.
Petersen SE, Fox PT, Snyder AZ, Raichle ME. Activation of extrastriate and frontal cortical areas by visual words and word-like stimuli. Science 1990, 249: 1041–1044.
Fiez JA. Phonology, semantics, and the role of the left inferior prefrontal cortex. Hum Brain Mapp 1997, 5: 79–83.
Binder JR, Desai RH, Graves WW, Conant LL. Where is the semantic system? A critical review and meta-analysis of 120 functional neuroimaging studies. Cereb Cortex 2009, 19: 2767–2796.
Chen YP, Fu SM, Iversen SD, Smith SM, Matthews PM. Testing for dual brain processing routes in reading: A direct contrast of Chinese character and pinyin reading using fMRI. J Cogn Neurosci 2002, 14: 1088–1098.
Zhao LB, Chen CH, Shao LY, Wang YP, Xiao XQ, Chen CS, et al. Orthographic and phonological representations in the fusiform cortex. Cereb Cortex 2017, 27: 5197–5210.
Wang X, Xu Y, Wang Y, Zeng Y, Zhang J, Ling Z, et al. Representational similarity analysis reveals task-dependent semantic influence of the visual word form area. Sci Rep 2018, 8: 3047.
Qin L, Lyu BJ, Shu S, Yin YY, Wang XF, Ge JQ, et al. A heteromodal word-meaning binding site in the visual word form area under top-down frontoparietal control. J Neurosci 2021, 41: 3854–3869.
Luke KK, Liu HL, Wai YY, Wan YL, Tan LH. Functional anatomy of syntactic and semantic processing in language comprehension. Hum Brain Mapp 2002, 16: 133–145.
Yang JM, Wang SP, Chen HC, Rayner K. The time course of semantic and syntactic processing in Chinese sentence comprehension: Evidence from eye movements. Mem Cognit 2009, 37: 1164–1176.
Skeide MA, Friederici AD. The ontogeny of the cortical language network. Nat Rev Neurosci 2016, 17: 323–332.
Bulut T, Hung YH, Tzeng O, Wu DH. Neural correlates of processing sentences and compound words in Chinese. PLoS One 2017, 12: e0188526.
Wang SP, Zhu ZD, Zhang JX, Wang ZX, Xiao ZW, Xiang HD, et al. Broca’s area plays a role in syntactic processing during Chinese reading comprehension. Neuropsychologia 2008, 46: 1371–1378.
Chou TL, Lee SH, Hung SM, Chen HC. The role of inferior frontal gyrus in processing Chinese classifiers. Neuropsychologia 2012, 50: 1408–1415.
Feng SW, Legault J, Yang L, Zhu JW, Shao KQ, Yang YM. Differences in grammatical processing strategies for active and passive sentences: An fMRI study. J Neurolinguistics 2015, 33: 104–117.
Feng SW, Qi RN, Yang J, Yu AY, Yang YM. Neural correlates for nouns and verbs in phrases during syntactic and semantic processing: An fMRI study. J Neurolinguistics 2020, 53: 100860.
Sun ZH, Shi YJ, Guo P, Yang YM, Zhu ZD. Independent syntactic representation identified in left front-temporal cortex during Chinese sentence comprehension. Brain Lang 2021, 214: 104907.
Zhang YX, Yu J, Boland JE. Semantics does not need a processing license from syntax in reading Chinese. J Exp Psychol Learn Mem Cogn 2010, 36: 765–781.
Ye Z, Luo YJ, Friederici AD, Zhou XL. Semantic and syntactic processing in Chinese sentence comprehension: Evidence from event-related potentials. Brain Res 2006, 1071: 186–196.
Vandermosten M, Boets B, Wouters J, Ghesquière P. A qualitative and quantitative review of diffusion tensor imaging studies in reading and dyslexia. Neurosci Biobehav Rev 2012, 36: 1532–1552.
Thuy DHD, Matsuo K, Nakamura K, Toma K, Oga T, Nakai T, et al. Implicit and explicit processing of kanji and kana words and non-words studied with fMRI. Neuroimage 2004, 23: 878–889.
Zhang MX, Li J, Chen CS, Mei LL, Xue G, Lu ZL, et al. The contribution of the left mid-fusiform cortical thickness to Chinese and English reading in a large Chinese sample. Neuroimage 2013, 65: 250–256.
Siok WT, Niu ZD, Jin Z, Perfetti CA, Tan LH. A structural–functional basis for dyslexia in the cortex of Chinese readers. Proc Natl Acad Sci U S A 2008, 105: 5561–5566.
Zhu LL, Nie YX, Chang CQ, Gao JH, Niu ZD. Different patterns and development characteristics of processing written logographic characters and alphabetic words: An ALE meta-analysis. Hum Brain Mapp 2014, 35: 2607–2618.
Catani M, Jones DK, Ffytche DH. Perisylvian language networks of the human brain. Ann Neurol 2005, 57: 8–16.
Glasser MF, Rilling JK. DTI tractography of the human brain’s language pathways. Cereb Cortex 2008, 18: 2471–2482.
Wakana S, Caprihan A, Panzenboeck MM, Fallon JH, Perry M, Gollub RL, et al. Reproducibility of quantitative tractography methods applied to cerebral white matter. Neuroimage 2007, 36: 630–644.
Paus T, Zijdenbos A, Worsley K, Collins DL, Blumenthal J, Giedd JN, et al. Structural maturation of neural pathways in children and adolescents: in vivo study. Science 1999, 283: 1908–1911.
Qiu DQ, Tan LH, Zhou K, Khong PL. Diffusion tensor imaging of normal white matter maturation from late childhood to young adulthood: Voxel-wise evaluation of mean diffusivity, fractional anisotropy, radial and axial diffusivities, and correlation with reading development. Neuroimage 2008, 41: 223–232.
Qiu DQ, Tan LH, Siok WT, Zhou K, Khong PL. Lateralization of the arcuate fasciculus and its differential correlation with reading ability between young learners and experienced readers: A diffusion tensor tractography study in a Chinese cohort. Hum Brain Mapp 2011, 32: 2054–2063.
Zhang MX, Chen CS, Xue G, Lu ZL, Mei LL, Xue HL, et al. Language-general and-specific white matter microstructural bases for reading. Neuroimage 2014, 98: 435–441.
Cui ZX, Xia ZC, Su MM, Shu H, Gong GL. Disrupted white matter connectivity underlying developmental dyslexia: A machine learning approach. Hum Brain Mapp 2016, 37: 1443–1458.
Shu N, Liu YO, Duan YY, Li KC. Hemispheric asymmetry of human brain anatomical network revealed by diffusion tensor tractography. Biomed Res Int 2015, 2015: 908917.
Su MM, Thiebaut de Schotten M, Zhao JJ, Song S, Zhou W, Gong GL, et al. Vocabulary growth rate from preschool to school-age years is reflected in the connectivity of the arcuate fasciculus in 14-year-old children. Dev Sci 2018, 21: e12647.
Su MM, Zhao JJ, Thiebaut de Schotten M, Zhou W, Gong GL, Ramus F, et al. Alterations in white matter pathways underlying phonological and morphological processing in Chinese developmental dyslexia. Dev Cogn Neurosci 2018, 31: 11–19.
Zhao JJ, Thiebaut de Schotten M, Altarelli I, Dubois J, Ramus F. Altered hemispheric lateralization of white matter pathways in developmental dyslexia: Evidence from spherical deconvolution tractography. Cortex 2016, 76: 51–62.
Han ZZ, Ma YJ, Gong GL, He Y, Caramazza A, Bi YC. White matter structural connectivity underlying semantic processing: Evidence from brain damaged patients. Brain 2013, 136: 2952–2965.
Wang K, Li XN, Huang RW, Ding JH, Song LP, Han ZZ. The left inferior longitudinal fasciculus supports orthographic processing: Evidence from a lesion-behavior mapping analysis. Brain Lang 2020, 201: 104721.
Wang HLS, Wang NYH, Yeh FC. Specifying the diffusion MRI connectome in Chinese-speaking children with developmental dyslexia and auditory processing deficits. Pediatr Neonatol 2019, 60: 297–304.
Li MY, Song LP, Zhang YM, Han ZZ. White matter network of oral word reading identified by network-based lesion-symptom mapping. iScience 2021, 24: 102862.
Shan CL, Weng XC, Wang T, Yu MX, Cong F, Zhao XY, et al. The role of splenium of corpus callosum in words, objects and faces processing. Chinese Journal of Rehabilitation 2010, 25: 426–429.
Friston KJ. Functional and effective connectivity: A review. Brain Connect 2011, 1: 13–36.
Levy J, Pernet C, Treserras S, Boulanouar K, Aubry F, Démonet JF, et al. Testing for the dual-route cascade reading model in the brain: An fMRI effective connectivity account of an efficient reading style. PLoS One 2009, 4: e6675.
Xu M, Wang TF, Chen SP, Fox PT, Tan LH. Effective connectivity of brain regions related to visual word recognition: An fMRI study of Chinese reading. Hum Brain Mapp 2015, 36: 2580–2591.
Li L, Liu JG, Chen FY, Feng L, Li H, Tian J, et al. Resting state neural networks for visual Chinese word processing in Chinese adults and children. Neuropsychologia 2013, 51: 1571–1583.
Wang XS, Han ZZ, He Y, Liu L, Bi YC. Resting-state functional connectivity patterns predict Chinese word reading competency. PLoS One 2012, 7: e44848.
Zhang MX, Li J, Chen CS, Xue G, Lu ZL, Mei LL, et al. Resting-state functional connectivity and reading abilities in first and second languages. Neuroimage 2014, 84: 546–553.
Li Y, Zhang LJ, Xia ZC, Yang J, Shu H, Li P. The relationship between intrinsic couplings of the visual word form area with spoken language network and reading ability in children and adults. Front Hum Neurosci 2017, 11: 327.
Hillen R, Günther T, Kohlen C, Eckers C, van Ermingen-Marbach M, Sass K, et al. Identifying brain systems for gaze orienting during reading: fMRI investigation of the Landolt paradigm. Front Hum Neurosci 2013, 7: 384.
Choi W, Desai RH, Henderson JM. The neural substrates of natural reading: A comparison of normal and nonword text using eyetracking and fMRI. Front Hum Neurosci 2014, 8: 1024.
Zhou W, Xia ZC, Bi YC, Shu H. Altered connectivity of the dorsal and ventral visual regions in dyslexic children: A resting-state fMRI study. Front Hum Neurosci 2015, 9: 495.
Liu Y, Shi GF, Li MY, Xing HB, Song Y, Xiao LC, et al. Early top-down modulation in visual word form processing: Evidence from an intracranial SEEG study. J Neurosci 2021, 41: 6102–6115.
Liu X, Gao Y, Di QQ, Hu JL, Lu CM, Nan Y, et al. Differences between child and adult large-scale functional brain networks for reading tasks. Hum Brain Mapp 2018, 39: 662–679.
Zhou W, Cui XH, Shi BG, Su MM, Cao M. The development of brain functional connectome during text reading. Dev Cogn Neurosci 2021, 48: 100927.
Qi T, Gu B, Ding GS, Gong GL, Lu CM, Peng DL, et al. More bilateral, more anterior: Alterations of brain organization in the large-scale structural network in Chinese dyslexia. Neuroimage 2016, 124: 63–74.
Mao JQ, Liu LF, Perkins K, Cao F. Poor reading is characterized by a more connected network with wrong hubs. Brain Lang 2021, 220: 104983.
Naselaris T, Kay KN, Nishimoto S, Gallant JL. Encoding and decoding in fMRI. Neuroimage 2011, 56: 400–410.
Kriegeskorte N, Mur M, Bandettini P. Representational similarity analysis - connecting the branches of systems neuroscience. Front Syst Neurosci 2008, 2: 4.
Kriegeskorte N, Goebel R, Bandettini P. Information-based functional brain mapping. Proc Natl Acad Sci U S A 2006, 103: 3863–3868.
Huth AG, de Heer WA, Griffiths TL, Theunissen FE, Gallant JL. Natural speech reveals the semantic maps that tile human cerebral cortex. Nature 2016, 532: 453–458.
Deniz F, Nunez-Elizalde AO, Huth AG, Gallant JL. The representation of semantic information across human cerebral cortex during listening versus reading is invariant to stimulus modality. J Neurosci 2019, 39: 7722–7736.
Fischer-Baum S, Bruggemann D, Gallego IF, Li DSP, Tamez ER. Decoding levels of representation in reading: A representational similarity approach. Cortex 2017, 90: 88–102.
Dong J, Li AQ, Chen CS, Qu J, Jiang N, Sun Y, et al. Language distance in orthographic transparency affects cross-language pattern similarity between native and non-native languages. Hum Brain Mapp 2021, 42: 893–907.
Fisher SE, Vargha-Khadem F, Watkins KE, Monaco AP, Pembrey ME. Localisation of a gene implicated in a severe speech and language disorder. Nat Genet 1998, 18: 168–170.
Fisher SE, Scharff C. FOXP2 as a molecular window into speech and language. Trends Genet 2009, 25: 166–177.
Fisher SE, Lai CSL, Monaco AP. Deciphering the genetic basis of speech and language disorders. Annu Rev Neurosci 2003, 26: 57–80.
Takahashi K, Liu FC, Oishi T, Mori T, Higo N, Hayashi M, et al. Expression of FOXP2 in the developing monkey forebrain: Comparison with the expression of the genes FOXP1, PBX3, and MEIS2. J Comp Neurol 2008, 509: 180–189.
Haesler S, Wada K, Nshdejan A, Morrisey EE, Lints T, Jarvis ED, et al. FoxP2 expression in avian vocal learners and non-learners. J Neurosci 2004, 24: 3164–3175.
Bonkowsky JL, Chien CB. Molecular cloning and developmental expression of foxP2 in zebrafish. Dev Dyn 2005, 234: 740–746.
Tolosa A, Sanjuán J, Dagnall AM, Moltó MD, Herrero N, de Frutos R. FOXP2 gene and language impairment in schizophrenia: Association and epigenetic studies. BMC Med Genet 2010, 11: 114.
Alarcón M, Abrahams BS, Stone JL, Duvall JA, Perederiy JV, Bomar JM, et al. Linkage, association, and gene-expression analyses identify CNTNAP2 as an autism-susceptibility gene. Am J Hum Genet 2008, 82: 150–159.
Rao WW, Du XD, Zhang YY, Yu Q, Hui L, Yu YQ, et al. Association between forkhead-box P2 gene polymorphism and clinical symptoms in chronic schizophrenia in a Chinese population. J Neural Transm (Vienna) 2017, 124: 891–897.
Yin JJ, Jia NR, Liu YS, Jin CH, Zhang FQ, Yu S, et al. No association between FOXP2 rs10447760 and schizophrenia in a replication study of the Chinese Han population. Psychiatr Genet 2018, 28: 19–23.
Du JN, Palaniyappan L, Liu ZW, Cheng W, Gong WK, Zhu MM, et al. The genetic determinants of language network dysconnectivity in drug-naïve early stage schizophrenia. NPJ Schizophr 2021, 7: 18.
Graham SA, Fisher SE. Understanding language from a genomic perspective. Annu Rev Genet 2015, 49: 131–160.
Unger N, Heim S, Hilger DI, Bludau S, Pieperhoff P, Cichon S, et al. Identification of phonology-related genes and functional characterization of Broca’s and Wernicke’s regions in language and learning disorders. Front Neurosci 2021, 15: 680762.
Peterson RL, Pennington BF. Developmental dyslexia. Lancet 2012, 379: 1997–2007.
Peterson RL, Pennington BF. Developmental dyslexia. Annu Rev Clin Psychol 2015, 11: 283–307.
Taipale M, Kaminen N, Nopola-Hemmi J, Haltia T, Myllyluoma B, Lyytinen H, et al. A candidate gene for developmental dyslexia encodes a nuclear tetratricopeptide repeat domain protein dynamically regulated in brain. Proc Natl Acad Sci U S A 2003, 100: 11553–11558.
Hannula-Jouppi K, Kaminen-Ahola N, Taipale M, Eklund R, Nopola-Hemmi J, Kääriäinen H, et al. The axon guidance receptor gene ROBO1 is a candidate gene for developmental dyslexia. PLoS Genet 2005, 1: e50.
Mekki Y, Guillemot V, Lemaître H, Carrión-Castillo A, Forkel S, Frouin V, et al. The genetic architecture of language functional connectivity. Neuroimage 2022, 249: 118795.
Bullmore E, Sporns O. Complex brain networks: Graph theoretical analysis of structural and functional systems. Nat Rev Neurosci 2009, 10: 186–198.
Park HJ, Friston K. Structural and functional brain networks: From connections to cognition. Science 2013, 342: 1238411.
Medaglia JD, Huang WY, Karuza EA, Kelkar A, Thompson-Schill SL, Ribeiro A, et al. Functional alignment with anatomical networks is associated with cognitive flexibility. Nat Hum Behav 2018, 2: 156–164.
Acknowledgments
We would like to thank Drs Kunyu Xu and Wei Zhou for their insightful comments on the manuscript. This review was supported by the Natural Science Foundation of China (81901826 and 61932008), the Natural Science Foundation of Shanghai (19ZR1405600 and 20ZR1404900), the Shanghai Municipal Science and Technology Major Project (2018SHZDZX01), ZJLab and Shanghai Center for Brain Science and Brain-inspired Technology.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors claim that there are no conflicts of interest.
Rights and permissions
About this article
Cite this article
Guo, W., Geng, S., Cao, M. et al. The Brain Connectome for Chinese Reading. Neurosci. Bull. 38, 1097–1113 (2022). https://doi.org/10.1007/s12264-022-00864-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12264-022-00864-3