Abstract
Hernia is a ubiquitous patho-anatomical surgical entity that a surgeon encounters and treats employing various methods and following certain principles to affect a durable repair. In recent years, the increasing understanding of the bio-physiology and mechanics of the abdominal wall coupled with innovations in surgical technique and evolution of novel procedures has created a sphere of reconstructive surgery wherein large complex hernias can be treated. There are various technical factors pertaining to a hernia repair including the surgical approach, extent and plane of dissection, the use of component separation, and placement of the mesh which needs to be addressed to bring about an optimum result. Complex hernias with loss of domain pose a challenge wherein an abdominal wall repair surgeon is tasked to bring about a tissue-based reconstruction with prosthetic reinforcement utilizing the now available various pre-operative tissue expansion techniques followed by a myofascial advancement procedure. With the advent and adoption of minimal invasive surgery, such procedures can be effectively undertaken in centers with available expertise. Similarly, groin hernias are now treated using a laparo-endoscopic approach. The modern abdominal wall repair surgeon should have in his armamentarium the knowledge and skill to effectively carry out a hernia repair with minimum morbidity but without affecting the durability or compromising the basic underlying principles. In this review, we endeavor to address these relevant issues so that an informed decision making for the accomplishment of a safe hernia surgery can be taken.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The evolving techniques and innovations in the surgical field combined with the increasing understanding of the bio-physiology and mechanics of the abdominal wall have created a sphere of reconstructive surgery wherein complex hernias can be treated with good results. Incisional hernias after laparotomy occur in 9–20% of patients at the end of 1 year [1]. Recurrence rates following primary repair range from 24 to 54%. Higher recurrence rates have been reported after suture repair (~ 43%) and significantly less after mesh repair (24%) [2, 3]. During the past two decades, the treatment of hernias has evolved noticeably and expectedly along the lines of minimal invasive surgery (Fig. 1). The complex hernias with loss of domain call for a durable management that now utilizes one of the component separation techniques. Similarly, groin hernias are commonly encountered in surgical practice and their management has refined itself along with the evolution of newer and better techniques. Contemporary surgical practice is now defined by incorporating improved and standardized recovery programs in conjunction with the use of minimal invasive approach with a resultant superior outcome in terms of surgical results and expeditious patient convalescence. The present scenario dictates that the surgeon be aware of the expanding scope of different available techniques and apply his skills in the most judicious manner to achieve results with minimal recurrence and morbidity.
The spectrum of recent surgical innovations in the field of minimal invasive surgery. SCOLA: subcutaneous onlay aponeuroplasty, LIRA: laparoscopic intracorporeal rectus aponeuroplasty, E-MILOS: endoscopic minimally invasive Mini or Less Open Sublay Repair, BARS: bony anchoring reinforcement system, e-TEP: extended or enhanced view total extra peritoneal, TAR: transversus abdominis release, ELAR: endoscopic linea alba reconstruction, IPOM: intra peritoneal onlay meshplasty
In this review, we seek to address the following pertinent topics of conflict to provide an informed decision making for the accomplishment of a safe hernia surgery-open vs laparoscopic approach, sublay vs onlay mesh placement, anterior component separation vs posterior component separation, loss of domain, recent innovations, tissue-based reconstruction with prosthetic reinforcement—the concept of eTEP and its application, closure of fascial defects, mesh fixation techniques, and role of imaging in hernia.
Methods
A comprehensive literature search was performed through the following databases: PubMed, Embase, Scopus, Cochrane database of systematic reviews, and Google scholar with the search terms: “hernia,” “abdominal wall hernia,” “ventral,” “laparoscopy,” “abdominal wall reconstruction,” “component separation,” “mesh fixation,” “loss of domain,” “closure of fascial defects,” “transversus abdominis release,” “enhanced view totally extraperitoneal technique,” and “imaging.” A further search of the bibliography of each article for additional articles that may have been missed during the original search was also done. Inclusion criteria included systematic reviews, narrative reviews, meta-analysis, and original articles. Duplicates, letter to editor, and case reports were excluded from the study.
Results
The systematic reviews, meta-analysis, and randomized controlled trials have shown that there is an advantage of minimal invasive approach for the treatment of abdominal wall and inguinal hernia in terms of less post-operative pain, return to work, occurrence of hematoma, and wound infection; the sublay repair has lower risk for recurrence and surgical site infections when compared to an onlay repair; when compared to open anterior approach, the technique of posterior component separation with transversus abdominis release has a lower incidence of recurrence and wound complications; botulinum toxin as a method of abdominal wall expansion provides a primary fascial closure in almost all the patients. Closure of fascial defects during open and laparoscopic repair has the advantage of lower seroma formation and length of stay; and finally, the manner of mesh fixation has no effect on the immediate or long-term postoperative pain, quality of life, rate of seroma formation, and recurrence rate. When there is an indication to obtain imaging in a case of abdominal wall hernia, a CT scan remains the preferred approach as it provides the necessary information to undertake a complex reconstruction. In case of a groin pathology when there is diagnostic dilemma, a step wise approach, utilizing ultrasound initially and proceeding to CT scan or MRI subsequently, should be employed.
Discussion
Open Versus Laparoscopic Approach
What was previously endorsed has now been ratified by various randomized controlled trials (RCTs) [4,5,6] and meta-analysis [7,8,9] including a Cochrane systematic review [10] regarding the advantages of a minimal invasive approach with level-1 evidence now proving its advantage in terms of less wound infections (Table 1). A laparoscopic approach is associated with less postoperative pain and earlier return to work while matching the open approach in terms of recurrence. A large prospective study has shown that the quality of life is also similar after 6 months of surgery [11]. The role of minimally invasive approach is also validated for the component separation technique in terms of less postoperative wound complications [12].
A recent systematic review and network meta-analyses of randomized controlled trials comparing the open Lichtenstein tension-free repair, laparoscopic transabdominal preperitoneal (TAPP) repair, and totally extraperitoneal repair (TEP) showed that the minimally invasive techniques were associated with significantly reduced early postoperative pain, return to work/activities, chronic pain, hematoma, and wound infection [13].
Sublay Versus Onlay Mesh Placement
The mesh can be positioned in different planes across the anterior abdominal wall. Vital to successful mesh placement is the presence of well-vascularized flaps with good tissue coverage to allow mesh-tissue integration. This can be optimally achieved when the sublay plane is used as the vascularized bilaminar coverage allows for good tissue integration and furthermore, avoids exposure from superficial wound complications, intra-abdominal adhesions, and contamination. Animal studies have shown improved fibrous capsule formation and collagen deposition when a mesh is placed in the sublay plane which correlates with better cellular penetration and potentially superior clearance of infection. A systematic review and meta-analysis has shown that sublay repair has a lower risk for recurrence and surgical site infections compared to onlay repair [14]. Sublay was ranked the best mesh placement option with a 94.2% probability of having the lowest odds of recurrence and 77.3% probability of having the lowest odds for surgical site infections [14]. The sublay repair is offset by its challenging technique and difficult to realize in cases of previous abdominal surgery. In such circumstances, the preference in order of surgical prudence would be underlay followed by onlay followed by inlay.
Anterior Component Separation Versus Posterior Component Separation
Local myofascial advancement for abdominal wall reconstruction is rapidly becoming the accepted technique by abdominal wall reconstruction surgeons. Component separation technique (CST) is based on the concept of re-establishing a functional abdominal wall with autologous tissue repair [12]. After Albanese and Ramirez demonstrated that that the external oblique muscle can be separated from the internal oblique muscle in a relatively avascular plane and midline can be advanced by 10 cm on either side [15, 16], various iterations [17,18,19,20,21] of component separation have been developed (Table 2). Anterior component separation has a disadvantage in that a large area of subcutaneous undermining is required which predisposes to seroma formation, skin flap necrosis, and mesh exposure if wound complications occur. The high wound morbidity associated with anterior component separation ranges from 25 to 57% precluding its use as a preliminary procedure [12]. Though there are no proper randomized controlled trials addressing this issue, a systemic review [22] shows that posterior component separation with transversus abdominis release is advantageous in terms of recurrence. Expert consensus guided by systematic review states that posterior component separation technique is associated with a lower wound complication rate compared to the standard anterior component separation technique [23].
Loss of Domain
Patients undergoing damage control laparotomies, laparotomy for hemorrhage and patients with bowel edema secondary to sepsis who have been managed by a laparostomy eventually require a planned ventral hernia repair with abdominal wall reconstruction. Defects more than 10 cm in size qualify as giant ventral hernia [24]. Restoring the hernia sac contents back into the abdominal cavity may lead to compartment syndrome with the resultant respiratory and circulatory disturbances. Expansion of the abdominal wall can be achieved by the use of progressive preoperative pneumoperitoneum (PPP), tissue expanders, and Botulinum toxin. PPP, introduced by Goñi Moreno [25], involves the controlled induction of pneumoperitoneum by an endo-peritoneal catheter which is maintained over a period of days or weeks. The introduction of pneumoperitoneum can be done by various methods, surgically using implantable subcutaneous port or dialysis catheter, or under ultrasound guidance. The volume ratio calculated as HSV/ACV (hernia sac volume/ abdominal cavity volume) is a guide to the use of PPP, with values > 20 to 25% serving as an indication [26, 27].
Botulinum Toxin A (BTA) with an ever-increasing range of clinical applications is now used as a method of “chemical component separation.” First reported by Cakmak et al. [28], it acts by causing relaxation of the lateral abdominal wall muscles which results in a significant decrease in thickness and increase in length of these muscles. Maximum effect is reached in 2 weeks with a prolonged plateau of approximately two and half months, with duration of effect lasting 6 to 9 months [29].
Tissue expanders are placed in a specially created pocket between the external oblique and the superficial aspect of the internal oblique fascia. The expanders have remote ports which allow the injection of saline on a weekly or biweekly basis over a period of months depending on the size of the defect, until there has been an adequate lengthening of musculature to allow primary fascial closure. The reported primary fascial closure rate with PPP has been 84%, with Botox 100% and with tissue expanders 93% [30].
Recent Innovations
Laparoscopic intraperitoneal onlay mesh (IPOM) repair and open sublay mesh repair remain the most practiced procedural modalities [9, 10, 31] with established benefits but the former is offset by its difficult realization in presence of large defects (> 10–15 cm). Cadaveric dissections and clinical studies have shown that the abdominal wall is not only an anatomical entity but a physiological and functional segment with biomechanical dynamics. Utilizing the ability to advance the constituent myofascial segments with intervening avascular planes, various innovations have come up as part of abdominal wall reconstruction. These minimal invasive techniques gain access to the extraperitoneal space offering the dual advantage of avoiding an open approach and the associated complications along with placing the mesh in the sublay plane thus avoiding contact with intra-peritoneal organs. The added benefit in all these procedures is the ability to use uncoated synthetic mesh with minimal need for fixation offering a more economical approach than the more widely practiced IPOM.
The surgical approach may be open, laparoscopic, endoscopic, or hybrid. The minimally invasive Mini or Less Open Sublay Repair (MILOS) and its endoscopic variant (EMILOS) access the retro-rectus plane through a mini (≤ 5 cm) or less open (6–12 cm) incision and laparo-endoscopic trans-hernial approach respectively. The authors further showed in a prospective study that this technique is associated with significantly fewer complications, recurrences, and less chronic pain than IPOM and less infection when compared to open sublay repair [32]. The Enhanced or Extended-view Total Extraperitoneal Preperitoneal (eTEP) approach can access any part of the abdominal wall including the groin region. Both of these novel techniques can be used to perform a posterior component separation, re-approximation of the linea alba, and a wide mesh coverage required for treating large complex hernias.
Tissue-Based Reconstruction with Prosthetic Reinforcement–the Concept of e-TEP and Its Application
Myofascial advancements with concurrent mesh placement after primary closure of the sheath can be achieved through a minimal access approach facilitated by the concept of enhanced or extended view total extraperitoneal technique (e-TEP). e-TEP finds application in accomplishment of various procedures including incisional, inguinal, recurrent, and complex hernia providing an expansive view and a field which helps in placement of a wide mesh covering the entire visceral sac.
Inguinal hernia is now approached endoscopically as a first-line procedure. The TEP technique was further modified by Jorge Daes who contrived the enhanced view concept and applied it successfully for the repair of inguinal hernia [33]. The e-TEP technique offers the advantages of an expeditious creation of the extraperitoneal space, a large surgical field, a flexible port setup adaptable to many clinical situations, an unencumbered parietalization of the cord structures, easier management of the distal sac in cases of large inguinoscrotal hernias, and an easier to master technique for a novice surgeon. It is particularly helpful in obese patients, patients with a short distance between the umbilicus and the pubic tubercle and patients with prior pelvic surgery. Jorge Daes further described the critical view of the myopectineal orifice [34] for the appropriate exposure of the anatomical area that must be attained before placing mesh during laparoscopic and robotic repair to accomplish a safe and durable endoscopic inguinal hernia repair.
e-TEP has rapidly gained acceptance with satisfactory results in the procedure of laparoscopic retro muscular hernia repair [35] and the technique has further extended its application to carry out a transversus abdominis release (TAR) [21]. This procedure offers a bilaminar tissue closure with prosthetic re-enforcement. There is a biomechanical basis for performing the TAR. The tranversus abdominis (TA) muscle physiologically functions in the maintenance of the intra-abdominal pressure and together with the internal oblique provides good tension throughout the entire thoracolumbar fascia forming a “corset” of the abdomen. The horizontal orientation of TA muscle fibers facilitates the advancement of rectus muscle complex and coupled with the possibility to mobilize it off the underlying fascia facilitates a successful medialization of all abdominal components during TAR of upto 8 to 12 cm on each side. The TAR is actually an extension of the stoppa philosophy of providing “giant prosthetic reinforcement of the visceral sac” providing a bilaminar fascial coverage with restoration of the linea alba ventral to the mesh (Fig. 2). Candidates for TAR include patients with large abdominal wall incisional or ventral hernia (including hernias with loss of domain), large non-midline hernias, para-stomal hernias, and recurrent incisional hernias after IPOM or anterior component separation.
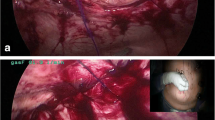
A pictorial representation of the transversus abdominis release procedure. a The posterior rectus sheath (highlighted in red color) is divided medial to the neurovascular bundles to gain access to the transversus abdominis muscle (blue arrow). b The transversus abdominis muscle is divided (yellow flash-arrowhead) to gain access to the TAR plane (grey arrows). E.O.: external oblique muscle, I.O.: internal oblique muscle, T.A.: transversus abdominis muscle, LA: linea alba, ARS: anterior rectus sheath, R: rectus abdominis muscle, PRS: posterior rectus sheath
Advantages of TAR include a tissue-based reconstruction with concurrent prosthetic reinforcement with subsequent ability to provide more durable repairs, preservation of neurovascular bundles, significant myofascial advancement with subsequent creation of a well-vascularized plane ideal for sublay mesh placement. The sublay plane provides a barrier to exclude the mesh from contact with the viscera and also a bacteriologic benefit by allowing the use of uncoated synthetic meshes placed in a sublay manner. Another important advantage lies in the intraoperative discovery and concomitant treatment of subtle defects not identified on clinical and radiological examination. A recent systematic review of 646 patients showed that TAR is associated with a low wound morbidity (15% versus 20%) and also a lower recurrence rate (4% versus 13%) [36].
Closure of Fascial Defects
Closure of the fascial defect (CFD) aims to restore the integrity, and hence function, of the abdominal wall by better distribution of tension across the repaired abdominal wall and the mesh. CFD increases the size of the overlap ratio of mesh to defect and increases the contact surface area of the mesh with the abdominal wall, which is likely to promote improved tissue ingrowth and strength of the repair. The technique of primary fascial closure with mesh reinforcement also reduces recurrence after open repairs of abdominal wall defects [37].
The advantages of CFD include decreased seroma formation as the dead space is closed, improvement in cosmesis and decreased recurrence as the abdominal wall muscle layer is intact with the rectus muscles restored to the midline. A meta-analysis which included 3638 patients of which 2963 underwent fascial defect closure, a significantly fewer adverse events including a lower seroma rate formation and shorter length of hospital stay were noted following fascial closure [38].
Mesh Fixation Technique
Placing a prosthesis is an integral part of the modern surgical repair. Minimally invasive procedure requires placing a mesh intraperitoneally or, with the newer techniques, extraperitoneally, with the former method requiring fixing the mesh to the anterior abdominal wall to facilitate a wide overlap and a firm anchorage. Fixation techniques lead to post-operative pain which affects the quality of life [11]. Pain can be attributed to various factors including implantation of foreign material on the abdominal wall, inflammation of the parietal peritoneum, trauma caused by insertion of trocars, adhesionolysis, use of various fixation devices, and nerve entrapment during trans-fascial fixation.
A wide array of selection is available for fixation material which can be divided into four categories: nonabsorbable tacks, absorbable tacks, sutures, and glues. Controversy exists regarding the choice, amount, strength, and type (absorbable or permanent) of fixation required. A systematic review and meta-analysis showed that tack fixation and suture showed comparable results with regard to postoperative chronic pain incidence and hernia recurrence with a shorter operative time when tackers are used [39]. The tackers are applied in a “double crown” manner (an outer row 0.5 cm from the mesh edge, and an inner row around the fascial defect) and should not be spaced more than 1–2 cm apart. Systematic review comparing absorbable and non-absorbable tacks found no difference between the two mesh fixation techniques in terms of recurrence, chronic pain, seroma, and hematoma [40]. A recent randomized controlled trial comparing permanent tacks, absorbable tacks, and synthetic glue for mesh fixation showed that the type of fixation device did not affect the immediate or long-term postoperative pain, quality of life, or recurrence rate [41]. The manner of fixation, whether tacks alone or sutures alone or a combination of both, has a divided opinion. Benefits of tacks-only fixation include shorter operating time, fewer skin incisions, improved cosmesis, and less acute and chronic pain. Advantages of suture-only fixation include the lower cost and stronger attachment of the prosthetic to the abdominal wall. Regardless of the manner of fixation, pain is inevitable and the results of these modalities are equivalent. The choice of fixation depends largely on surgeon’s preference and the institutional practice ensuring an overlap of at least 5 cm with proper fixation especially to the bony points, if needed.
Role of Imaging in Hernia
While the diagnosis of hernia is clinically evident in a significant proportion of cases, there are instances when occult hernias may be present or a recurrence is in question. Imaging is useful to diagnose and characterize the hernia, and aid in preoperative planning as well, when a complex abdominal wall reconstruction is to be undertaken.
Imaging in Ventral Hernia
In specific circumstances, imaging in the form of an ultrasound or a cross-sectional imaging is required. Imaging is useful to aid in diagnosis of a suspected ventral hernia in certain instances such as a scarred abdomen where a recurrence is suspected or in an obese patient precluding a clinical diagnosis. Imaging also helps to discern relevant anatomical details when a complex hernia requires an abdominal wall reconstruction and in non-midline hernias. The current gold standard, CT scan accurately characterizes most incisional hernias [42]. However, the supine, static nature of the study can miss some clinically apparent hernias, and CT scan also incurs exposure to ionizing radiation, which may have a long-term cumulative effect in patients undergoing multiple studies [43]. The additional cost of MRI limits its use as a routine imaging modality for ventral hernia. An innovative technique using a 12 MHz linear ultrasound probe has been described, called the Dynamic Abdominal Sonography for Hernia (DASH) [42]. The ventral abdominal wall is scanned in five sequential cranio-caudal passes to detect even small defects. The added advantage of DASH apart from non-invasive nature and low cost is the high sensitivity (98%) and specificity (88%). Nonetheless, CT scan remains an irreplaceable tool when the global management of incisional hernia is considered. Particularly, when managing complex hernias, CT scan helps in determining the hernia metrics, envisage the need for component separation, and subsequently predict the likelihood of abdominal wall closure and wound complications. Certain features are sought by the surgeon for optimal planning such as mesh location, whether a previously done component separation is unilateral or bilateral, presence of adhesions or fistulas, location of tacks which are hyper attenuating which point out the margins of the mesh, the status of muscles (which group is atrophic—to plan a component separation) and to quantify the loss of domain.
Imaging in Inguinal Hernia
Only groin pain that is obscure or groin swelling of unclear origin is required to have a further diagnostic evaluation. No consensus exists presently on the best imaging modality for these diagnostic dilemmas. A meta-analysis has shown that herniography is the most accurate imaging modality for occult inguinal hernias [44]. This analysis also showed that overall sensitivity and specificity of CT is approximately 80% and 65%, respectively. A study comparing CT with herniography showed CT to have a sensitivity of 67% and a specificity of 95% compared to a sensitivity of 63% and a specificity of 100% for herniography [45]. Since herniography is invasive and involves the use of a contrast medium, it is not widely available and has a potential for complications such as injection site hematoma, visceral puncture, and a vasovagal reaction. Its practical application is limited.
Ultrasound can be considered the first-line diagnostic test for the occult inguinal hernia. It is quick, inexpensive, and non-invasive, but there is an operator variability and it is limited by an obese patient’s body habitus. A meta-analysis by Robinson et al. [44] demonstrated ultrasound to have a sensitivity of 96.6%, specificity of 84.8%, and a positive predictive value of 92.6%. It is reasonable to proceed next, to a CT scan in the setting of a compelling history for inguinal hernia but a negative clinical exam or equivocal ultrasound study. CT may help identify other sources of pain such as soft tissue or skeletal abnormalities.
A report by Miller et al. showed that MRI has a sensitivity of 91%, and appeared superior to both ultrasound and CT scan in the diagnosis of occult inguinal hernias [46]. When a groin pathology is suspected but not diagnosed, MRI should be the preferred modality, as it can not only rule out an occult hernia but also elucidate other causes of groin pain. In terms of inguinal hernia detection, the initial use of ultrasound, followed by a CT scan, represents a sensitive and cost-effective progression for the evaluation of inguinal pathology. However, MRI is an expensive diagnostic modality, and surgeons are typically more comfortable with image interpretation of CT compared to MRI. An MRI can be considered the penultimate choice in the ladder of workup preceding surgical exploration via open or laparoscopic approach.
Conclusion
To manage inguinal and ventral hernia, a minimal invasive approach should be the preferred modality. The concept of e-TEP has initiated a domain of laparo-endoscopic techniques which allow a tissue-based prosthetic reinforcement of the defects. Abdominal wall reconstruction is evolving as a divergent practice wherein protocol-based management for loss of domain followed by a bilaminar closure with a prosthetic reinforcement is undertaken. A posterior component separation with sublay mesh placement should be the goal of hernia management. Ultrasound should be the initial choice of imaging followed by a CT scan for evaluation of inguinal or ventral hernia. Incorporation into surgical practice and standardization of these innovative techniques should result in a more durable repair and rapid convalescence. Further research will help ascertain these advantages.
References
Diener MK, Voss S, Jensen K, Büchler MW, Seiler CM (2010) Elective midline laparotomy closure: the INLINE systematic review and meta-analysis. Ann Surg 251(5):843–856. https://doi.org/10.1097/SLA.0b013e3181d973e4
Anthony T, Bergen PC, Kim LT, Henderson M, Fahey T, Rege RV, Turnage RH (2000) Factors affecting recurrence following incisional herniorrhaphy. World J Surg 24(1):95–100. discussion 101. https://doi.org/10.1007/s002689910018
Burger JW, Luijendijk RW, Hop WC, Halm JA, Verdaasdonk EG, Jeekel J (2004) Long-term follow-up of a randomized controlled trial of suture versus mesh repair of incisional hernia. Ann Surg 240(4):578–83. discussion 583–5. https://doi.org/10.1097/01.sla.0000141193.08524.e7
Itani KM, Hur K, Kim LT, Anthony T, Berger DH, Reda D, Neumayer L, Veterans Affairs Ventral Incisional Hernia Investigators (2010) Comparison of laparoscopic and open repair with mesh for the treatment of ventral incisional hernia: a randomized trial. Arch Surg 145(4):322–8. discussion 328. https://doi.org/10.1001/archsurg.2010.18
Rogmark P, Petersson U, Bringman S, Eklund A, Ezra E, Sevonius D, Smedberg S, Osterberg J, Montgomery A (2013) Short-term outcomes for open and laparoscopic midline incisional hernia repair: a randomized multicenter controlled trial: the ProLOVE (prospective randomized trial on open versus laparoscopic operation of ventral eventrations) trial. Ann Surg 258(1):37–45. https://doi.org/10.1097/SLA.0b013e31828fe1b2
Eker HH, Hansson BM, Buunen M, Janssen IM, Pierik RE, Hop WC, Bonjer HJ, Jeekel J, Lange JF (2013) Laparoscopic vs. open incisional hernia repair: a randomized clinical trial. JAMA Surg 148(3):259–63. https://doi.org/10.1001/jamasurg.2013.1466
Forbes SS, Eskicioglu C, McLeod RS, Okrainec A (2009) Meta-analysis of randomized controlled trials comparing open and laparoscopic ventral and incisional hernia repair with mesh. Br J Surg 96(8):851–858. https://doi.org/10.1002/bjs.6668
Sajid MS, Bokhari SA, Mallick AS, Cheek E, Baig MK (2009) Laparoscopic versus open repair of incisional/ventral hernia: a meta-analysis. Am J Surg 197(1):64–72. https://doi.org/10.1016/j.amjsurg.2007.12.051
Al Chalabi H, Larkin J, Mehigan B, McCormick P (2015) A systematic review of laparoscopic versus open abdominal incisional hernia repair, with meta-analysis of randomized controlled trials. Int J Surg 20:65–74. https://doi.org/10.1016/j.ijsu.2015.05.050
Sauerland S, Walgenbach M, Habermalz B, Seiler CM, & Miserez M (2011) Laparoscopic versus open surgical techniques for ventral or incisional hernia repair. Cochrane Database Syst Rev (3):CD007781. https://doi.org/10.1002/14651858.CD007781.pub2
Colavita PD, Tsirline VB, Belyansky I, Walters AL, Lincourt AE, Sing RF, Heniford BT (2012) Prospective, long-term comparison of quality of life in laparoscopic versus open ventral hernia repair. Ann Surg 256(5):714–723. https://doi.org/10.1097/SLA.0b013e3182734130
Switzer NJ, Dykstra MA, Gill RS, Lim S, Lester E, de Gara C, Shi X, Birch DW, Karmali S (2015) Endoscopic versus open component separation: systematic review and meta-analysis. Surg Endosc 29(4):787–795. https://doi.org/10.1007/s00464-014-3741-1
Aiolfi A, Cavalli M, Del Ferraro S, Manfredini L, Bonitta G, Bruni PG, Bona D, & Campanelli G (2021) Treatment of inguinal hernia: systematic review and updated network meta-analysis of randomized controlled trials. Ann Surg. Publish Ahead of Print. Advance online publication. https://doi.org/10.1097/SLA.0000000000004735
Holihan JL, Nguyen DH, Nguyen MT, Mo J, Kao LS, Liang MK (2016) Mesh location in open ventral hernia repair: a systematic review and network meta-analysis. World J Surg 40(1):89–99. https://doi.org/10.1007/s00268-015-3252-9
Albanese AR (1951) Eventración mediana xifoumbilical gigante; metodo para su tratamiento [Gigantic median xipho-umbilical eventration; method for treatment]. Rev Asoc Med Argent 65(709–710):376–378
Ramirez OM, Ruas E, Dellon AL (1990) “Components separation” method for closure of abdominal-wall defects: an anatomic and clinical study. Plast Reconstr Surg 86(3):519–526. https://doi.org/10.1097/00006534-199009000-00023
Carbonell AM, Cobb WS, Chen SM (2008) Posterior components separation during retromuscular hernia repair. Hernia 12(4):359–362. https://doi.org/10.1007/s10029-008-0356-2
Saulis AS, Dumanian GA (2002) Periumbilical rectus abdominis perforator preservation significantly reduces superficial wound complications in “separation of parts” hernia repairs. Plast Reconstr Surg 109(7):2275–2282. https://doi.org/10.1097/00006534-200206000-00016
Rosen MJ, Jin J, McGee MF, Williams C, Marks J, Ponsky JL (2007) Laparoscopic component separation in the single-stage treatment of infected abdominal wall prosthetic removal. Hernia 11(5):435–440. https://doi.org/10.1007/s10029-007-0255-y
Novitsky YW, Elliott HL, Orenstein SB, Rosen MJ (2012) Transversus abdominis muscle release: a novel approach to posterior component separation during complex abdominal wall reconstruction. Am J Surg 204(5):709–716. https://doi.org/10.1016/j.amjsurg.2012.02.008
Belyansky I, Zahiri HR, Park A (2016) Laparoscopic transversus abdominis release, a novel minimally invasive approach to complex abdominal wall reconstruction. Surg Innov 23(2):134–141. https://doi.org/10.1177/1553350615618290
Cornette B, De Bacquer D, Berrevoet F (2018) Component separation technique for giant incisional hernia: a systematic review. Am J Surg 215(4):719–726. https://doi.org/10.1016/j.amjsurg.2017.07.032
Liang MK, Holihan JL, Itani K, Alawadi ZM, Gonzalez JR, Askenasy EP, Ballecer C, Chong HS, Goldblatt MI, Greenberg JA, Harvin JA, Keith JN, Martindale RG, Orenstein S, Richmond B, Roth JS, Szotek P, Towfigh S, Tsuda S, Vaziri K, …, Berger DH (2017) Ventral hernia management: expert consensus guided by systematic review. Ann Surg 265(1):80–89. https://doi.org/10.1097/SLA.0000000000001701
Passot G, Villeneuve L, Sabbagh C, Renard Y, Regimbeau JM, Verhaeghe P, Kianmanesh R, Palot JP, Vaudoyer D, Glehen O, Cotte E, Fédération de RechercheenChirurgie (FRENCH) (2016) Definition of giant ventral hernias: development of standardization through a practice survey. Int J Surg (London, England) 28:136–140. https://doi.org/10.1016/j.ijsu.2016.01.097
Moreno IG (1947) Chronic eventrations and large hernias; preoperative treatment by progressive pneumoperitoneum; original procedure. Surgery 22(6):945–953
Tanaka EY, Yoo JH, Rodrigues AJ Jr, Utiyama EM, Birolini D, Rasslan S (2010) A computerized tomography scan method for calculating the hernia sac and abdominal cavity volume in complex large incisional hernia with loss of domain. Hernia 14(1):63–69. https://doi.org/10.1007/s10029-009-0560-8
Sabbagh C, Dumont F, Robert B, Badaoui R, Verhaeghe P, Regimbeau JM (2011) Peritoneal volume is predictive of tension-free fascia closure of large incisional hernias with loss of domain: a prospective study. Hernia 15(5):559–565. https://doi.org/10.1007/s10029-011-0832-y
Cakmak M, Caglayan F, Somuncu S, Leventoglu A, Ulusoy S, Akman H, Kaya M (2006) Effect of paralysis of the abdominal wall muscles by botulinum A toxin to intraabdominal pressure: an experimental study. J Pediatr Surg 41(4):821–825. https://doi.org/10.1016/j.jpedsurg.2005.12.023
Dressler D (2012) Clinical applications of botulinum toxin. Curr Opin Microbiol 15(3):325–336. https://doi.org/10.1016/j.mib.2012.05.012
Alam NN, Narang SK, Pathak S, Daniels IR, Smart NJ (2016) Methods of abdominal wall expansion for repair of incisional herniae: a systematic review. Hernia 20(2):191–199. https://doi.org/10.1007/s10029-016-1463-0
Awaiz A, Rahman F, Hossain MB, Yunus RM, Khan S, Memon B, Memon MA (2015) Meta-analysis and systematic review of laparoscopic versus open mesh repair for elective incisional hernia. Hernia 19(3):449–463. https://doi.org/10.1007/s10029-015-1351-z
Reinpold W, Schröder M, Berger C, Nehls J, Schröder A, Hukauf M, Köckerling F, Bittner R (2019) Mini- or Less-open Sublay Operation (MILOS): a new minimally invasive technique for the extraperitoneal mesh repair of incisional hernias. Ann Surg 269(4):748–755. https://doi.org/10.1097/SLA.0000000000002661
Daes J (2012) The enhanced view-totally extraperitoneal technique for repair of inguinal hernia. Surg Endosc 26(4):1187–1189. https://doi.org/10.1007/s00464-011-1993-6
Daes J, Felix E (2017) Critical view of the myopectineal orifice. Ann Surg 266(1):e1–e2. https://doi.org/10.1097/SLA.0000000000002104
Belyansky I, Daes J, Radu VG, Balasubramanian R, Reza Zahiri H, Weltz AS, Sibia US, Park A, Novitsky Y (2018) A novel approach using the enhanced-view totally extraperitoneal (eTEP) technique for laparoscopic retromuscular hernia repair. Surg Endosc 32(3):1525–1532. https://doi.org/10.1007/s00464-017-5840-2
Wegdam JA, Thoolen J, Nienhuijs SW, de Bouvy N, de Vries Reilingh TS (2019) Systematic review of transversus abdominis release in complex abdominal wall reconstruction. Hernia 23(1):5–15. https://doi.org/10.1007/s10029-018-1870-5
Booth JH, Garvey PB, Baumann DP, Selber JC, Nguyen AT, Clemens MW, Liu J, Butler CE (2013) Primary fascial closure with mesh reinforcement is superior to bridged mesh repair for abdominal wall reconstruction. J Am Coll Surg 217(6):999–1009. https://doi.org/10.1016/j.jamcollsurg.2013.08.015
Tandon A, Pathak S, Lyons NJ, Nunes QM, Daniels IR, Smart NJ (2016) Meta-analysis of closure of the fascial defect during laparoscopic incisional and ventral hernia repair. Br J Surg 103(12):1598–1607. https://doi.org/10.1002/bjs.10268
Ahmed MA, Tawfic QA, Schlachta CM, Alkhamesi NA (2018) Pain and surgical outcomes reporting after laparoscopic ventral hernia repair in relation to mesh fixation technique: a systematic review and meta-analysis of randomized clinical trials. J Laparoendosc Adv Surg Tech A 28(11):1298–1315. https://doi.org/10.1089/lap.2017.0609
Khan R, Bughio M, Ali B, Hajibandeh S, Hajibandeh S (2018) Absorbable versus non-absorbable tacks for mesh fixation in laparoscopic ventral hernia repair: a systematic review and meta-analysis. Int J Surg (London, England) 53:184–192. https://doi.org/10.1016/j.ijsu.2018.03.042
Harsløf S, Krum-Møller P, Sommer T, Zinther N, Wara P, Friis-Andersen H (2018) Effect of fixation devices on postoperative pain after laparoscopic ventral hernia repair: a randomized clinical trial of permanent tacks, absorbable tacks, and synthetic glue. Langenbeck’s Arch Surg 403(4):529–537. https://doi.org/10.1007/s00423-018-1676-z
Beck WC, Holzman MD, Sharp KW, Nealon WH, Dupont WD, Poulose BK (2013) Comparative effectiveness of dynamic abdominal sonography for hernia vs computed tomography in the diagnosis of incisional hernia. J Am Coll Surg 216(3):447–511. https://doi.org/10.1016/j.jamcollsurg.2012.11.012
Berrington de González A, Darby S (2004) Risk of cancer from diagnostic X-rays: estimates for the UK and 14 other countries. Lancet (London, England) 363(9406):345–351. https://doi.org/10.1016/S0140-6736(04)15433-0
Robinson A, Light D, Kasim A, Nice C (2013) A systematic review and meta-analysis of the role of radiology in the diagnosis of occult inguinal hernia. Surg Endosc 27(1):11–18. https://doi.org/10.1007/s00464-012-2412-3
Markos V, Brown EF (2005) CT herniography in the diagnosis of occult groin hernias. Clin Radiol 60(2):251–256. https://doi.org/10.1016/j.crad.2004.02.013
Miller J, Cho J, Michael MJ, Saouaf R, Towfigh S (2014) Role of imaging in the diagnosis of occult hernias. JAMA Surg 149(10):1077–1080. https://doi.org/10.1001/jamasurg.2014.484
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jain, N., Upadhyay, Y. & Bhojwani, R. Emerging Concepts in the Minimal Access Repair of Abdominal Wall Hernia—a Narrative Review. Indian J Surg (2021). https://doi.org/10.1007/s12262-021-03018-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12262-021-03018-3