Abstract
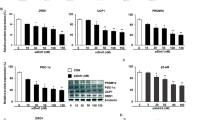
Due to its propensity to boost energy expenditure, browning of white fat is emerging as an intriguing and prospective target for therapeutic intervention in obesity. Here, we report that L-dihydroxyphenylalanine (L-Dopa), used as a gold standard therapy in Parkinson’s disease, induces browning in 3T3-L1 adipocytes by increasing the expression levels of beige-specific marker genes such as Cd137, Cited1, Cidea, Tbx1, Prdm16, and Ucp1. In addition, exposure to L-Dopa induces a remarkable increase in the expressions of proteins involved in thermogenesis in white adipocytes. L-Dopa treatment also regulates 3T3-L1 adipocytes by markedly increasing protein expressions of p-AMPK, p-HSL, CPT1, ACOX1, and PPARα while decreasing FAS, ACC, C/EBPα, and PPARγ, suggesting enhanced lipolysis and fatty acid oxidation as well as reduced lipogenesis and adipogenesis, respectively. Molecular docking studies elucidated that L-Dopa binds to dopamine receptor D1 (DRD1) and β3-AR, thereby predicting the potential receptor candidates that activate protein kinase A (PKA), the master regulator of lipid metabolism. Mechanistic studies indicate that the browning potential of L-Dopa in 3T3-L1 white adipocytes is mediated by DRD1 and β3-AR activation, which consequently stimulates the PKA/p38 MAPK/ERK signaling pathway. In conclusion, L-Dopa appears to be a promising therapeutic candidate in the fight against obesity due to its inherent role in the browning of 3T3-L1 adipocytes via both the dopaminergic and adrenergic pathways. To our knowledge, this is the first report that demonstrates the browning potential of L-Dopa in white adipocytes. Our results may assist to expand the understanding on the contradictory findings in literature, related to the association between L-Dopa and weight loss observed in Parkinson’s disease patients.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Change history
24 December 2022
An Erratum to this paper has been published: https://doi.org/10.1007/s12257-022-1111-8
References
Williams, E. P., M. Mesidor, K. Winters, P. M. Dubbert, and S. B. Wyatt (2015) Overweight and obesity: prevalence, consequences, and causes of a growing public health problem. Curr. Obes. Rep. 4: 363–370.
Barber, T. M., P. Hanson, M. O. Weickert, and S. Franks (2019) Obesity and polycystic ovary syndrome: implications for pathogenesis and novel management strategies. Clin. Med. Insights Reprod. Health. 13: 1179558119874042.
Jackson, S. E., C. H. Llewellyn, and L. Smith (2020) The obesity epidemic - nature via nurture: a narrative review of high-income countries. SAGE Open Med. 8: 2050312120918265.
Vernochet, C., A. Mourier, O. Bezy, Y. Macotela, J. Boucher, M. J. Rardin, D. An, K. Y. Lee, O. R. Ilkayeva, C. M. Zingaretti, B. Emanuelli, G. Smyth, S. Cinti, C. B. Newgard, B. W. Gibson, N. G. Larsson, and C. R. Kahn (2012) Adipose-specific deletion of TFAM increases mitochondrial oxidation and protects mice against obesity and insulin resistance. Cell Metab. 16: 765–776.
Whitehead, A., F. N. Krause, A. Moran, A. D. MacCannell, J. L. Scragg, B. D. McNally, E. Boateng, S. A. Murfitt, S. Virtue, J. Wright, J. Garnham, G. R. Davies, J. Dodgson, J. E. Schneider, A. J. Murray, C. Church, A. Vidal-Puig, K. K. Witte, J. L. Griffin, and L. D. Roberts (2021) Brown and beige adipose tissue regulate systemic metabolism through a metabolite interorgan signaling axis. Nat. Commun. 12: 1905.
Makki, K., P. Froguel, and I. Wolowczuk (2013) Adipose tissue in obesity-related inflammation and insulin resistance: cells, cytokines, and chemokines. ISRN Inflamm. 2013: 139239.
Zu, Y., H. Overby, G. Ren, Z. Fan, L. Zhao, and S. Wang (2018) Resveratrol liposomes and lipid nanocarriers: comparison of characteristics and inducing browning of white adipocytes. Colloids Surf. B Biointerfaces. 164: 414–423.
Chouchani, E. T., L. Kazak, M. P. Jedrychowski, G. Z. Lu, B. K. Erickson, J. Szpyt, K. A. Pierce, D. Laznik-Bogoslavski, R. Vetrivelan, C. B. Clish, A. J. Robinson, S. P. Gygi, and B. M. Spiegelman (2016) Mitochondrial ROS regulate thermogenic energy expenditure and sulfenylation of UCP1. Nature. 532: 112–116. (Erratum published 2016, Nature 536: 360)
Choi, M., S. Mukherjee, and J. W. Yun (2021) Trigonelline induces browning in 3T3-L1 white adipocytes. Phytother. Res. 35: 1113–1124.
Jang, M. H., S. Mukherjee, M. J. Choi, N. H. Kang, H. G. Pham, and J. W. Yun (2020) Theobromine alleviates diet-induced obesity in mice via phosphodiesterase-4 inhibition. Eur. J. Nutr. 59: 3503–3516.
Manigandan, S. and J. W. Yun (2020) Urolithin A induces brown-like phenotype in 3T3-L1 white adipocytes via β3-adrenergic receptor-p38 MAPK signaling pathway. Biotechnol. Bioprocess Eng. 25: 345–355.
Song, N. J., S. H. Chang, D. Y. Li, C. J. Villanueva, and K. W. Park (2017) Induction of thermogenic adipocytes: molecular targets and thermogenic small molecules. Exp. Mol. Med. 49: e353.
Duan, Y. N., X. Ge, H. W. Jiang, H. J. Zhang, Y. Zhao, J. L. Li, W. Zhang, and J. Y. Li (2020) Diphyllin improves high-fat diet-induced obesity in mice through brown and beige adipocytes. Front. Endocrinol. (Lausanne) 11: 592818.
Daubner, S. C., T. Le, and S. Wang (2011) Tyrosine hydroxylase and regulation of dopamine synthesis. Arch. Biochem. Biophys. 508: 1–12.
Mercuri, N. B. and G. Bernardi (2005) The ‘magic’ of L-dopa: why is it the gold standard Parkinson’s disease therapy? Trends Pharmacol. Sci. 26: 341–344.
Tambasco, N., M. Romoli, and P. Calabresi (2018) Levodopa in Parkinson’s disease: current status and future developments. Curr. Neuropharmacol. 16: 1239–1252.
Colamartino, M., L. Padua, C. Meneghini, S. Leone, T. Cornetta, A. Testa, and R. Cozzi (2012) Protective effects of L-dopa and carbidopa combined treatments on human catecholaminergic cells. DNA Cell Biol. 31: 1572–1579.
Bliwise, D. L., L. M. Trotti, A. G. Wilson, S. A. Greer, C. Wood-Siverio, J. J. Juncos, S. A. Factor, A. Freeman, and D. B. Rye (2012) Daytime alertness in Parkinson’s disease: potentially dose-dependent, divergent effects by drug class. Mov. Disord. 27: 1118–1124.
Sürücü, O., H. Baumann-Vogel, M. Uhl, L. L. Imbach, and C. R. Baumann (2013) Subthalamic deep brain stimulation versus best medical therapy for L-dopa responsive pain in Parkinson’s disease. Pain. 154: 1477–1479.
Okun, M. S., S. S. Wu, D. Jennings, K. Marek, R. L. Rodriguez, and H. H. Fernandez (2014) Testosterone level and the effect of levodopa and agonists in early Parkinson disease: results from the INSPECT cohort. J. Clin. Mov. Disord. 1: 8.
Li, Z., X. Li, X. He, X. Jia, X. Zhang, B. Lu, J. Zhao, J. Lu, L. Chen, Z. Dong, K. Liu, and Z. Dong (2020) Proteomics reveal the inhibitory mechanism of levodopa against esophageal squamous cell carcinoma. Front. Pharmacol. 11: 568459.
Rivera-Calimlim, L. and J. R. Bianchine (1972) Effect of L-dopa on plasma free fatty acids and plasma glucose. Metabolism. 21: 611–617.
Vardi, J., Z. Oberman, I. Rabey, M. Streifler, D. Ayalon, and M. Herzberg (1976) Weight loss in patients treated long-term with levodopa: Metabolic aspects. J. Neurol. Sci. 30: 33–40.
Bachmann, C. G., A. Zapf, E. Brunner, and C. Trenkwalder (2009) Dopaminergic treatment is associated with decreased body weight in patients with Parkinson’s disease and dyskinesias. Eur. J. Neurol. 16: 895–901.
Pålhagen, S., B. Lorefält, M. Carlsson, W. Ganowiak, G. Toss, M. Unosson, and A. K. Granérus (2005) Does L-dopa treatment contribute to reduction in body weight in elderly patients with Parkinson’s disease? Acta Neurol. Scand. 111: 12–20.
Barichella, M., A. Marczewska, A. Vairo, M. Canesi, and G. Pezzoli (2003) Is underweightness still a major problem in Parkinson’s disease patients? Eur. J. Clin. Nutr. 57: 543–547.
Levi, S., M. Cox, M. Lugon, M. Hodkinson, and A. Tomkins (1990) Increased energy expenditure in Parkinson’s disease. BMJ. 301: 1256–1257.
Markus, H. S., M. Cox, and A. M. Tomkins (1992) Raised resting energy expenditure in Parkinson’s disease and its relationship to muscle rigidity. Clin. Sci. (Lond.) 83: 199–204.
Kohlie, R., N. Perwitz, J. Resch, S. M. Schmid, H. Lehnert, J. Klein, and K. A. Iwen (2017) Dopamine directly increases mitochondrial mass and thermogenesis in brown adipocytes. J. Mol. Endocrinol. 58: 57–66.
Tiraby, C., G. Tavernier, C. Lefort, D. Larrouy, F. Bouillaud, D. Ricquier, and D. Langin (2003) Acquirement of brown fat cell features by human white adipocytes. J. Biol. Chem. 278: 33370–33376.
Sharp, L. Z., K. Shinoda, H. Ohno, D. W. Scheel, E. Tomoda, L. Ruiz, H. Hu, L. Wang, Z. Pavlova, V. Gilsanz, and S. Kajimura (2012) Human BAT possesses molecular signatures that resemble beige/brite cells. PLoS One. 7: e49452.
Seale, P., S. Kajimura, W. Yang, S. Chin, L. M. Rohas, M. Uldry, G. Tavernier, D. Langin, and B. M. Spiegelman (2007) Transcriptional control of brown fat determination by PRDM16. Cell Metab. 6: 38–54.
Ahmadian, M., M. J. Abbott, T. Tang, C. S. Hudak, Y. Kim, M. Bruss, M. K. Hellerstein, H. Y. Lee, V. T. Samuel, G. I. Shulman, Y. Wang, R. E. Duncan, C. Kang, and H. S. Sul (2011) Desnutrin/ATGL is regulated by AMPK and is required for a brown adipose phenotype. Cell Metab. 13: 739–748.
Hardie, D. G., F. A. Ross, and S. A. Hawley (2012) AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat. Rev. Mol. Cell Biol. 13: 251–262.
Rosso, P., M. Fioramonti, A. Fracassi, M. Marangoni, V. Taglietti, S. Siteni, and M. Segatto (2016) AMPK in the central nervous system: physiological roles and pathological implications. Res. Rep. Biol. 7: 1–13.
Watt, M. J., A. G. Holmes, S. K. Pinnamaneni, A. P. Garnham, G. R. Steinberg, B. E. Kemp, and M. A. Febbraio (2006) Regulation of HSL serine phosphorylation in skeletal muscle and adipose tissue. Am. J. Physiol. Endocrinol. Metab. 290: E500–E508. (Erratum published 2009, Am. J. Physiol. Endocrinol. Metab. 296: E401)
Kim, T. W., H. M. Cho, S. Y. Choi, Y. Suguira, T. Hayasaka, M. Setou, H. C. Koh, E. M. Hwang, J. Y. Park, S. J. Kang, H. S. Kim, H. Kim, and W. Sun (2013) (ADP-ribose) polymerase 1 and AMP-activated protein kinase mediate progressive dopaminergic neuronal degeneration in a mouse model of Parkinson’s disease. Cell Death Dis. 4: e919.
Collins, S., E. Yehuda-Shnaidman, and H. Wang (2010) Positive and negative control of Ucp1 gene transcription and the role of β-adrenergic signaling networks. Int. J. Obes. (Lond.) 34 Suppl 1: S28–S33.
Julien, C., L. Berthiaume, A. Hadj-Tahar, A. H. Rajput, P. J. Bédard, T. Di Paolo, P. Julien, and F. Calon (2006) Postmortem brain fatty acid profile of levodopa-treated Parkinson disease patients and parkinsonian monkeys. Neurochem. Int. 48: 404–414.
Adams, F., M. Boschmann, E. Lobsien, A. Kupsch, A. Lipp, G. Franke, M. C. Leisse, J. Janke, S. Gottschalk, J. Spranger, and J. Jordan (2008) Influences of levodopa on adipose tissue and skeletal muscle metabolism in patients with idiopathic Parkinson’s disease. Eur. J. Clin. Pharmacol. 64: 863–870.
Kim, K. S., Y. R. Yoon, H. J. Lee, S. Yoon, S. Y. Kim, S. W. Shin, J. J. An, M. S. Kim, S. Y. Choi, W. Sun, and J. H. Baik (2010) Enhanced hypothalamic leptin signaling in mice lacking dopamine D2 receptors. J. Biol. Chem. 285: 8905–8917.
Folgueira, C., D. Beiroa, B. Porteiro, M. Duquenne, E. Puighermanal, M. F. Fondevila, S. Barja-Fernández, R. Gallego, R. Hernández-Bautista, C. Castelao, A. Senra, P. Seoane, N. Gómez, P. Aguiar, D. Guallar, M. Fidalgo, A. Romero-Pico, R. Adan, C. Blouet, J. L. Labandeira-García, F. Jeanrenaud, I. Kallo, Z. Liposits, J. Salvador, V. Prevot, C. Dieguez, M. Lopez, E. Valjent, G. Frühbeck, L. M. Seoane, and R. Nogueiras (2019) Hypothalamic dopamine signaling regulates brown fat thermogenesis. Nat. Metab. 1: 811–829.
Borcherding, D. C., E. R. Hugo, G. Idelman, A. De Silva, N. W. Richtand, J. Loftus, and N. Ben-Jonathan (2011) Dopamine receptors in human adipocytes: expression and functions. PLoS One. 6: e25537.
Beeler, J. A., R. P. Faust, S. Turkson, H. Ye, and X. Zhuang (2016) Low dopamine D2 receptor increases vulnerability to obesity via reduced physical activity, not increased appetitive motivation. Biol. Psychiatry. 79: 887–897.
de Leeuw van Weenen, J. E., E. T. Parlevliet, J. P. Schröder-van der Elst, S. A. van den Berg, K. Willems van Dijk, J. A. Romijn, and H. Pijl (2011) Pharmacological modulation of dopamine receptor D2-mediated transmission alters the metabolic phenotype of diet induced obese and diet resistant C57Bl6 mice. Exp. Diabetes Res. 2011: 928523.
Brizuela, M., A. Antipov, W. W. Blessing, and Y. Ootsuka (2019) Activating dopamine D2 receptors reduces brown adipose tissue thermogenesis induced by psychological stress and by activation of the lateral habenula. Sci. Rep. 9: 19512.
Ootsuka, Y., C. A. Heidbreder, J. J. Hagan, and W. W. Blessing (2007) Dopamine D2 receptor stimulation inhibits cold-initiated thermogenesis in brown adipose tissue in conscious rats. Neuroscience. 147: 127–135.
Raffaelli, F. M., J. Resch, R. Oelkrug, K. A. Iwen, and J. Mittag (2020) Dopamine receptor D1- and D2-agonists do not spark brown adipose tissue thermogenesis in mice. Sci. Rep. 10: 20203.
Chen, J., M. Rusnak, R. R. Luedtke, and A. Sidhu (2004) D1 dopamine receptor mediates dopamine-induced cytotoxicity via the ERK signal cascade. J. Biol. Chem. 279: 39317–39330.
Hondares, E., R. Iglesias, A. Giralt, F. J. Gonzalez, M. Giralt, T. Mampel, and F. Villarroya (2011) Thermogenic activation induces FGF21 expression and release in brown adipose tissue. J. Biol. Chem. 286: 12983–12990.
Wang, H., J. Xu, P. Lazarovici, R. Quirion, and W. Zheng (2018) cAMP response element-binding protein (CREB): a possible signaling molecule link in the pathophysiology of schizophrenia. Front. Mol. Neurosci. 11: 255.
Pham, H. G., J. P. Park, and J. W. Yun (2020) BMP11 negatively regulates lipid metabolism in C2C12 muscle cells. Biotechnol. Bioprocess Eng. 25: 670–680.
Choi, M. J., S. Mukherjee, and J. W. Yun (2021) Loss of ADAMTS15 promotes browning in 3T3-L1 white adipocytes via activation of β3-adrenergic receptor. Biotechnol. Bioprocess Eng. 26: 188–200.
Lee, T. L., C. T. Hsu, S. T. Yen, C. W. Lai, and J. T. Cheng (1998) Activation of beta3-adrenoceptors by exogenous dopamine to lower glucose uptake into rat adipocytes. J. Auton. Nerv. Syst. 74: 86–90.
Lei, S. (2014) Cross interaction of dopaminergic and adrenergic systems in neural modulation. Int. J. Physiol. Pathophysiol. Pharmacol. 6: 137–142.
Acknowledgement
This study was supported by a Daegu University Research Grant 2022.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Neither ethical approval nor informed consent was required for this study.
Additional information
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Haddish, K., Yun, J.W. L-Dihydroxyphenylalanine (L-Dopa) Induces Brown-like Phenotype in 3T3-L1 White Adipocytes via Activation of Dopaminergic and β3-adrenergic Receptors. Biotechnol Bioproc E 27, 818–832 (2022). https://doi.org/10.1007/s12257-021-0361-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-021-0361-1