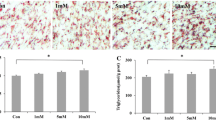
Abstract
Muscle tissue influences energy and protein metabolism throughout the body. Earlier reports demonstrated that bone morphogenetic protein 11 (BMP11) could inhibit skeleton muscle proliferation and development. However, the role of BMP11 in the fatty acid metabolism of muscle has not been explored. In this study, we investigated the physiological functions of exogenous BMP11 on lipid metabolism in C2C12 cells using recombinant BMP11 (rBMP11). Treatment by rBMP11 inhibits myogenesis while inducing lipid accumulation in C2C12 cells. Moreover, induction of rBMP11 inhibits not only fatty acid uptake by downregulation of the fatty acid transport proteins CD36, FATP1, FATP4, and FABP3 but also suppresses fatty acid oxidation by decreasing the levels of p-HSL, ATGL, ACSL, and CGI-58 via Smad 2/3 pathway. Taken together, BMP11 negatively regulates lipid metabolism in muscle cells, which is an opposite result to that in adipocytes where BMP11 improves metabolic homeostasis. Considering the contrasting roles of BMP11 in adipocytes and muscle cells, BMP11 could be a promising target for pharmacological intervention in the treatment of metabolic diseases.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- Abhd5 :
-
gene encoding CGI-58
- Acvrl1 :
-
gene encoding activin receptor-like kinase 1 (ALK1)
- Acvr1 :
-
gene encoding activin receptor-like kinase-2 (ALK2)
- Acvr1b :
-
gene encoding activin receptor type-1B (ALK4)
- Acvr2a :
-
gene encoding activin receptor type-2A (ACTRIIA)
- Acvr2b :
-
gene encoding activin receptor type-2B (ACTRIIB)
- ACOX1 :
-
acyl-coenzyme A oxidase 1
- ACSL :
-
long-chainfatty-acid-CoA ligase
- ATGL :
-
adipose triglyceride lipase
- BMP :
-
bone morphogenetic protein
- Bmpr1a :
-
gene encoding bone morphogenetic protein receptor type-1A (ALK3)
- Bmpr1b :
-
gene encoding bone morphogenetic protein receptor type-1B (ALK6)
- Bmpr2 :
-
gene encoding bone morphogenetic protein receptor type 2 (BMPRII)
- CD36 :
-
cluster of differentiation 36
- CGI-58 :
-
comparative gene identification-58
- CPT1 :
-
carnitine palmitoyltransferase 1
- FFA :
-
free fatty acid
- FAS :
-
fatty acid synthase
- FATP4 :
-
long-chain fatty acid transport protein 4
- GDF11 :
-
growth differentiation factor-11
- MYH/Myh :
-
myosin heavy chain/encoding gene
- MYOG/Myog :
-
myogenin/encoding gene
- p-HSL :
-
phosphorylated hormone-sensitive lipase
- Plin :
-
gene encoding perilipin
- Slc27a :
-
gene encoding FATP
- Tbx1 :
-
gene encoding T-box protein 1
- Tgfbr-1 :
-
transforming growth factor beta receptor type 1 (ALK5)
- TG :
-
triglyceride
- UCP/Ucp :
-
uncoupling protein/encoding gene.
References
Morales, P. E., J. L. Bucarey, and A. Espinosa (2017) Muscle lipid metabolism: role of lipid droplets and perilipins. J. Diabetes Res. 2017: 1789395.
Cahova, M., H. Vavrinkova, and L. Kazdova (2007) Glucosefatty acid interaction in skeletal muscle and adipose tissue in insulin resistance. Physiol. Res. 56: 1–15.
Schenk, S. and J. F. Horowitz (2007) Acute exercise increases triglyceride synthesis in skeletal muscle and prevents fatty acid-induced insulin resistance. J. Clin. Invest. 117: 1690–1698.
Watt, M. J. and A. J. Hoy (2012) Lipid metabolism in skeletal muscle: generation of adaptive and maladaptive intracellular signals for cellular function. Am. J. Physiol. Endocrinol. Metab. 302: E1315–E1328.
Amati, F., J. J. Dubé, E. Alvarez-Carnero, M. M. Edreira, P. Chomentowski, P. M. Coen, G. E. Switzer, P. E. Bickel, M. Stefanovic-Racic, F. G. S. Toledo, and B. H. Goodpaster (2011) Skeletal muscle triglycerides, diacylglycerols, and ceramides in insulin resistance: another paradox in endurance-trained athletes? Diabetes. 60: 2588–2597.
Li, L. J., J. Ma, S. B. Li, X. F. Chen, and J. Zhang (2019) Electric pulse stimulation inhibited lipid accumulation on C2C12 myotubes incubated with oleic acid and palmitic acid. Arch. Physiol. Biochem.https://doi.org/10.1080/13813455.2019.1639763.
Guitart, M., O. Osorio-Conles, T. Pentinat, J. Cebrià, J. García-Villoria, D. Sala, D. Sebastián, A. Zorzano, A. Ribes, J. C. Jiménez-Chillarón, C. García-Martínez, and A. M. Gómez-Foix (2014) Fatty acid transport protein 1 (FATP1) localizes in mitochondria in mouse skeletal muscle and regulates lipid and ketone body disposal. PLoS One. 9: e98109.
Kawaguchi, M., Y. Tamura, S. Kakehi, K. Takeno, Y. Sakurai, T. Watanabe, T. Funayama, F. Sato, S. Ikeda, Y. Ogura, N. Saga, H. Naito, Y. Fujitani, A. Kanazawa, R. Kawamori, and H. Watada (2014) Association between expression of FABPpm in skeletal muscle and insulin sensitivity in intramyocellular lipid-accumulated nonobese men. J. Clin. Endocrinol. Metab. 99: 3343–3352.
Bruce, C. R., A. J. Hoy, N. Turner, M. J. Watt, T. L. Allen, K. Carpenter, G. J. Cooney, M. A. Febbraio, and E. W. Kraegen (2009) Overexpression of carnitine palmitoyltransferase-1 in skeletal muscle is sufficient to enhance fatty acid oxidation and improve high-fat diet-induced insulin resistance. Diabetes. 58: 550–558.
Shi, Y. and P. Burn (2004) Lipid metabolic enzymes: emerging drug targets for the treatment of obesity. Nat. Rev. Drug Discov. 3: 695–710.
Luquet, S., J. Lopez-Soriano, D. Holst, C. Gaudel, C. Jehl-Pietri, A. Fredenrich, and P. A. Grimaldi (2004) Roles of peroxisome proliferator-activated receptor delta (PPARdelta) in the control of fatty acid catabolism. A new target for the treatment of metabolic syndrome. Biochimie. 86: 833–837.
Chen, D., M. Zhao, and G. R. Mundy (2004) Bone morphogenetic proteins. Growth Factors. 22: 233–241.
Wozney, J. M., V. Rosen, A. J. Celeste, L. M. Mitsock, M. J. Whitters, R. W. Kriz, R. M. Hewick, and E. A. Wang (1988) Novel regulators of bone formation: molecular clones and activities. Science. 242: 1528–1534.
Urist, M. R. (1965) Bone: formation by autoinduction. Science. 150: 893–899.
Cobourne, M. T. and P. T. Sharpe (2003) Tooth and jaw: molecular mechanisms of patterning in the first branchial arch. Arch. Oral Biol. 48: 1–14.
Walker, R. G., T. Poggioli, L. Katsimpardi, S. M. Buchanan, J. Oh, S. Wattrus, B. Heidecker, Y. W. Fong, L. L. Rubin, P. Ganz, T. B. Thompson, A. J. Wagers, and R. T. Lee (2016) Biochemistry and biology of GDF11 and myostatin: similarities, differences, and questions for future investigation. Circ. Res. 118: 1125–1142.
Zhang, Y., Y. Wei, D. Liu, F. Liu, X. Li, L. Pan, Y. Pang, and D. Chen (2017) Role of growth differentiation factor 11 in development, physiology and disease. Oncotarget. 8: 81604–81616.
Liu, Y., L. Shao, K. Chen, Z. Wang, J. Wang, W. Jing, and M. Hu (2018) GDF11 restrains tumor growth by promoting apoptosis in pancreatic cancer. Onco Targets Ther. 11: 8371–8379.
Bajikar, S. S., C. C. Wang, M. A. Borten, E. J. Pereira, K. A. Atkins, and K. A. Janes (2017) Tumor-Suppressor Inactivation of GDF11 occurs by precursor sequestration in triple-negative breast cancer. Dev. Cell. 43: 418–435.
Loffredo, F. S., M. L. Steinhauser, S. M. Jay, J. Gannon, J. R. Pancoast, P. Yalamanchi, M. Sinha, C. Dall’Osso, D. Khong, J. L. Shadrach, C. M. Miller, B. S. Singer, A. Stewart, N. Psychogios, R. E. Gerszten, A. J. Hartigan, M. J. Kim, T. Serwold, A. J. Wagers, and R. T. Lee (2013) Growth differentiation factor 11 is a circulating factor that reverses age-related cardiac hypertrophy. Cell. 153: 828–839.
Añón-Hidalgo, J., V. Catalán, A. Rodríguez, B. Ramírez, C. Silva, J. C. Galofré, J. Salvador, G. Frühbeck, and J. Gómez-Ambrosi (2019) Circulating GDF11 levels are decreased with age but are unchanged with obesity and type 2 diabetes. Aging (Albany NY). 11: 1733–1744.
Harmon, E. B., A. A. Apelqvist, N. G. Smart, X. Gu, D. H. Osborne, and S. K. Kim (2004) GDF11 modulates NGN3+ islet progenitor cell number and promotes beta-cell differentiation in pancreas development. Development. 131: 6163–6174.
Freitas-Rodríguez, S., F. Rodríguez, and A. R. Folgueras (2016) GDF11 administration does not extend lifespan in a mouse model of premature aging. Oncotarget. 7: 55951–55956.
Kim, T. N., M. S. Park, S. J. Yang, H. J. Yoo, H. J. Kang, W. Song, J. A. Seo, S. G. Kim, N. H. Kim, S. H. Baik, D. S. Choi, and K. M. Choi (2010) Prevalence and determinant factors of sarcopenia in patients with type 2 diabetes: the Korean Sarcopenic Obesity Study (KSOS). Diabetes Care. 33: 1497–1499.
Krssak, M., K. F. Petersen, A. Dresner, L. DiPietro, S. M. Vogel, D. L. Rothman, M. Roden and G. I. Shulman (1999) Intra-myocellular lipid concentrations are correlated with insulin sensitivity in humans: a 1H NMR spectroscopy study. Diabetologia. 42: 113–116.
Lu, B., J. Zhong, J. Pan, X. Yuan, M. Ren, L. Jiang, Y. Yang, G. Zhang, D. Liu, C. Zhang (2019) Gdf11 gene transfer prevents high fat diet-induced obesity and improves metabolic homeostasis in obese and STZ-induced diabetic mice. J Transl Med. 17: 422.
Luo, H., Y. Guo, Y. Liu, Y. Wang, R. Zheng, Y. Ban, L. Peng, Q. Yuan, and W. Liu (2019) Growth differentiation factor 11 inhibits adipogenic differentiation by activating TGF-beta/Smad signalling pathway. Cell Prolif. 52: e12631.
Lukaszuk, B., I. Bialuk, J. Górski, M. Zajączkiewicz, M. M. Winnicka, and A. Chabowski (2012) A single bout of exercise increases the expression of glucose but not fatty acid transporters in skeletal muscle of IL-6 KO mice. Lipids. 47: 763–772.
Pedersen, B. K. and M. A. Febbraio (2012) Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat. Rev. Endocrinol. 8: 457–465.
Mukherjee, S., K. R. Aseer, and J. W. Yun (2020) Roles of macrophage colony stimulating factor in white and brown adipocytes. Biotechnol. Bioprocess Eng. 25: 29–38.
Jackson, V. M., D. M. Breen, J. P. Fortin, A. Liou, J. B. Kuzmiski, A. K. Loomis, M. L. Rives, B. Shah, and P. A. Carpino (2015) Latest approaches for the treatment of obesity. Expert. Opin. Drug Discov. 10: 825–839.
Jang, M. H., N. H. Kang, S. Mukherjee, and J. W. Yun (2018) Theobromine, a methylxanthine in cocoa bean, stimulates thermogenesis by inducing white fat browning and activating brown adipocytes. Biotechnol. Bioprocess Eng. 23: 617–626.
Manigandan, S. and J. W. Yun (2020) Urolithin A induces brown-like phenotype in 3T3-L1 white adipocytes via β3-adrenergic receptor-p38 MAPK signaling pathway. Biotechnol. Bioprocess Eng. 25: 345–355.
Egerman, M. A., S. M. Cadena, J. A. Gilbert, A. Meyer, H. N. Nelson, S. E. Swalley, C. Mallozzi, C. Jacobi, L. L. Jennings, I. Clay, G. Laurent, S. Ma, S. Brachat, E. Lach-Trifilieff, T. Shavlakadze, A. U. Trendelenburg, A. S. Brack, and D. J. Glass (2015) GDF11 increases with age and inhibits skeletal muscle regeneration. Cell Metab. 22: 164–174.
Jeanplong, F., S. J. Falconer, M. Thomas, K. G. Matthews, J. M. Oldham, T. Watson, and C. D. McMahon (2012) Growth and differentiation factor-11 is developmentally regulated in skeletal muscle and inhibits myoblast differentiation. Open J. Mol. Integr. Physiol. 2:127–138.
Fujita, T., S. Furukawa, K. Morita, T. Ishihara, M. Shiotani, Y. Matsushita, M. Matsuda, and I. Shimomura (2005) Glucosamine induces lipid accumulation and adipogenic change in C2C12 myoblasts. Biochem. Biophys. Res. Commun. 328: 369–374.
Yin, C., Q. Long, T. Lei, X. Chen, H. Long, B. Feng, Y. Peng, Y. Wu, and Z. Yang (2009) Lipid accumulation mediated by adiponectin in C2C12 myogenesis. BMB Rep. 42: 667–672.
McCormack, S. E., M. A. McCarthy, S. G. Harrington, L. Farilla, M. I. Hrovat, D. M. Systrom, B. J. Thomas, M. Torriani, K. McInnis, S. K. Grinspoon, and A. Fleischman (2014) Effects of exercise and lifestyle modification on fitness, insulin resistance, skeletal muscle oxidative phosphorylation and intramyocellular lipid content in obese children and adolescents. Pediatric Obesity. 9: 281–291
Watt, M. J., G. J. F. Heigenhauser, D. J. Dyck, and L. L. Spriet (2002) Intramuscular triacylglycerol, glycogen and acetyl group metabolism during 4 h of moderate exercise in man. J. Physiol. 541: 969–978.
Pan, D. A., S. Lillioj, A. D. Kriketos, M. R. Milner, L. A. Baur, C. Bogardus, A. B. Jenkins, and L. H. Storlien (1997) Skeletal muscle triglyceride levels are inversely related to insulin action. Diabetes. 46: 983–988.
Goodpaster, B. H., R. Teriault, S. C. Watkins, and D. E. Kelley (2000) Intramuscular lipid content is increased in obesity and decreased by weight loss. Metabolism. 49: 467–472.
Cortright, R. N., D. M. Muoio, and G. L. Dohm (1997) Skeletal muscle lipid metabolism: A frontier for new insights into fuel homeostasis. J. Nutr. Biochem. 8: 228–245.
Newsom, S. A., S. Schenk, M. Li, A. C. Everett, and J. F. Horowitz (2011) High fatty acid availability after exercise alters the regulation of muscle lipid metabolism. Metabolism. 60: 852–859.
Van Hall, G. (2015) The physiological regulation of skeletal muscle fatty acid supply and oxidation during moderate-intensity exercise. Sports Med. 45: 23–32.
Acknowledgments
This work was supported by a National Research Foundation of Korea grant funded by the Korean government (MSIT) (No. 2019R1A2C2002163).
The authors declare no conflict of interest.
Neither ethical approval nor informed consent was required for this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pham, H.G., Park, J.P. & Yun, J.W. BMP11 Negatively Regulates Lipid Metabolism in C2C12 Muscle Cells. Biotechnol Bioproc E 25, 670–680 (2020). https://doi.org/10.1007/s12257-020-0254-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-020-0254-8