Abstract
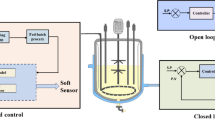
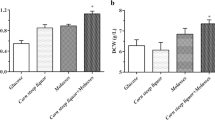
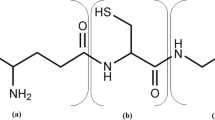
Reduced glutathione (GSH), the abundant bioactive tripeptide in most living cells, is widely used in pharmaceutical, food, and cosmetic industries. Specific growth rate (μ) is a key physiological parameter for GSH high-cell-density cultivation using microbial cell factories. Here, based on a biomass probe, an on-line μ feedback control was developed to regulate glucose feeding rate during the fed-batch phase for overproducing GSH in Saccharomyces cerevisiae. Compared with real-time μ controlled at 0.15/h, μ controlled at 0.2/h achieved yeast dry weight (120 g/L), GSH concentration (1.5 g/L), and intracellular GSH content (1.25%), which improved by 9, 150, and 129.1%, respectively. To our knowledge, this is the first report about on-line μ feedback control for GSH production. On-line μ control led to 59.38 mg/L/h of GSH productivity and 3.52 mg/g of GSH yield on glucose, which improved by 107.6 and 7.2%, respectively, in comparison with those of traditional ethanol feedback control (maintaining ethanol concentration at 1%). Taken together, the on-line μ feedback control is a promising method as an efficient alternative to conventional feed control techniques presently practiced in the GSH industry, and has the potential for the production of other valuable chemicals.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Penninckx, M. (2000) A short review on the role of glutathione in the response of yeasts to nutritional, environmental, and oxidative stresses. Enz. Microb. Technol. 26: 737–742.
Li, Y., G. Y. Wei, and J. Chen (2004) Glutathione: A review on biotechnological production. Appl. Microbiol. Biotechnol. 66: 233–242.
Korz, D. J., U. Rinas, K. Hellmuth, E. A. Sanders, and W. D. Deckwer (1995) Simple fed-batch technique for high cell density cultivation of Escherichia coli. J. Biotechnol. 39: 59–65.
Navarro, J., E. Obrador, J. Carretero, I. Petschen, J. Avino, P. Perez, and J. M. Estrela (1999) Changes in glutathione status and the antioxidant system in blood and in cancer cells associate with tumour growth in vivo. Free Radic. Biol. Med. 26: 410–418.
Cha, J. Y., J. C. Park, B. S. Jeon, Y. C. Lee, and Y. S. Cho (2004) Optimal fermentation conditions for enhanced glutathione production by Saccharomyces cerevisiae FF-8. J. Microbiol. 42: 51–55.
Xiong, Z. Q., M. J. Guo, Y. X. Guo, J. Chu, Y. P. Zhuang, N. S. Wang, and S. L. Zhang (2010) RQ feedback control for simultaneous improvement of GSH yield and GSH content in Saccharomyces cerevisiae T65. Enz. Microb. Technol. 46: 598–602.
Sakato, K. and H. Tanaka (1992) Advanced control of glutathione fermentation process. Biotechnol. Bioeng. 40: 904–912.
Alfafara, C. G., K. Miura, H. Shimizu, S. Shioya, K. I. Suga, and K. Suzuki (1993) Fuzzy control of ethanol concentration its application to maximum glutathione production in yeast fed-batch culture. Biotechnol. Bioeng. 41: 493–501.
Shimizu, H., K. Araki, S. Shioya, and K. I. Suga (1991) Optimal production of glutathione by controlling the specific growth rate of yeast in fed-batch culture. Biotechnol. Bioeng. 38: 196–205.
Dabros, M., M. M. Schuler, and I. W. Marison (2010) Simple control of specific growth rate in biotechnological fed-batch processes based on enhanced online measurements of biomass. Bioproc. Biosyst. Eng. 33: 1109–1118.
Schuler, M. M. and I. W. Marison (2012) Real-time monitoring and control of microbial bioprocesses with focus on the specific growth rate: current state and perspectives. Appl. Microbiol. Biotechnol. 94: 1469–1482.
Xiong, Z. Q., M. J. Guo, Y. X. Guo, J. Chu, Y. P. Zhuang, and S. L. Zhang (2008) Real-time viable-cell mass monitoring in high-cell-density fed-batch glutathione fermentation by Saccharomyces cerevisiae T65 in industrial complex medium. J. Biosci. Bioeng. 105: 409–413.
Wang, Y., D. Wang, G. Wei, and C. Wang (2013) Improved co-production of S-adenosylmethionine and glutathione using citrate as an auxiliary energy substrate. Bioresour. Technol. 131: 28–32.
Ge, S., T. Zhu, and Y. Li (2012) Expression of bacterial GshF in Pichia pastoris for glutathione production. Appl. Environ. Microbiol. 78: 5435–5439.
Shang, F., Z. Wang, and T. Tan (2008) High-cell-density cultivation for co-production of ergosterol and reduced glutathione by Saccharomyces cerevisiae. Appl. Microbiol. Biotechnol. 77: 1233–1240.
Wen, S. H., T. Zhang, and T. W. Tan (2005) Optimization of the amino acid composition in glutathione fermentation. Proc. Biochem. 40: 3474–3479.
Gao, M. J., Z. Y. Zheng, J. R. Wu, S. J. Dong, Z. Li, H. Jin, X. B. Zhan, and C. C. Lin (2012) Improvement of specific growth rate of Pichia pastoris for effective porcine interferon-alpha production with an on-line model-based glycerol feeding strategy. Appl. Microbiol. Biotechnol. 93: 1437–1445.
Li, Y., J. Chen, Y. Y. Mao, S. Y. Lun, and Y. M. Koo (1998) Effect of additives and fed-batch culture strategies on the production of glutathione by recombinant Escherichia coli. Proc. Biochem. 33: 709–714.
Lin, H. P., J. Tian, J. F. You, Z. H. Jin, Z. N. Xu, and P. L. Cen (2004) An effective strategy for the co-production of S-adenosyl-L-methionine and glutathione by fed-batch fermentation. Biochem. Eng. J. 21: 19–25.
Vemuri, G. N., M. A. Eiteman, J. E. McEwen, L. Olsson, and J. Nielsen (2007) Increasing NADH oxidation reduces overflow metabolism in Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. U. S. A. 104: 2402–2407.
Nisamedtinov, I., K. Kevvai, K. Orumets, J. J. Rautio, and T. Paalme (2010) Glutathione accumulation in ethanol-stat fed-batch culture of Saccharomyces cerevisiae with a switch to cysteine feeding. Appl. Microbiol. Biotechnol. 87: 175–183.
Liu, H., J. P. Lin, P. L. Cen, and Y. J. Pan (2004) Co-production of S-adenosyl—methionine and glutathione from spent brewer’s yeast cells. Proc. Biochem. 39: 1993–1997.
Fei, L., Y. Wang, and S. Chen (2009) Improved glutathione production by gene expression in Pichia pastoris. Bioproc. Biosyst. Eng. 32: 729–735.
Liang, G., G. Du, and J. Chen (2008) A novel strategy of enhanced glutathione production in high cell density cultivation of Candida utilis-Cysteine addition combined with dissolved oxyen controlling. Enz. Microb. Technol. 42: 284–289.
Nie, M., G. Y. Wei, N. Shao, and X. G. Ge (2010) A novel strategy on the high-cell-density cultivation of Candida utilis for the enhanced production of glutathione. Kor. J. Chem. Eng. 27: 1246–1251.
Ubiyvovk, V. M., V. M. Ananin, A. Y. Malyshev, H. A. Kang, and A. A. Sibirny (2011) Optimization of glutathione production in batch and fed-batch cultures by the wild-type and recombinant strains of the methylotrophic yeast Hansenula polymorpha DL-1. BMC Biotechnol. 11:8.
Min, C. K., J. W. Lee, K. H. Chung, and H. W. Park (2010) Control of specific growth rate to enhance the production of a novel disintegrin, saxatilin, in recombinant Pichia pastoris. J. Biosci. Bioeng. 110: 314–319.
Wang, Y., D. Wang, G. Wei, and N. Shao (2012) Enhanced co-production of S-adenosylmethionine and glutathione by an ATP-oriented amino acid addition strategy. Bioresour. Technol. 107: 19–24.
Suzuki, T., A. Yokoyama, T. Tsuji, E. Ikeshima, K. Nakashima, S. Ikushima, C. Kobayashi, and S. Yoshida (2011) Identification and characterization of genes involved in glutathione production in yeast. J. Biosci. Bioeng. 112: 107–113.
Hara, K. Y., K. Kiriyama, A. Inagaki, H. Nakayama, and A. Kondo (2012) Improvement of glutathione production by metabolic engineering the sulfate assimilation pathway of Saccharomyces cerevisiae. Appl. Microbiol. Biotechnol. 94: 1313–1319.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xiong, ZQ., Guo, MJ., Chu, J. et al. On-line specific growth rate control for improving reduced glutathione production in Saccharomyces cerevisiae . Biotechnol Bioproc E 20, 887–893 (2015). https://doi.org/10.1007/s12257-015-0018-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-015-0018-z