Summary
Onuris hauthalii is a species distributed in Southern Argentina and Chile. Its generic assignment has been controversial, being included in Eudema, Brayopsis and Onuris. In order to clarify its generic affinities, we studied its morphology and phylogenetic relationships using multilocus/multispecies coalescent species-tree analyses based on four DNA regions (ITS, trnL-F, trnH-psbA, and rps16 intron). Results show that while O. hauthalii is related to Onuris s.s., it represents a well-differentiated lineage characterised primarily by having branched rhizomes, rosette flowering with ebracteate flowers, and latiseptate silicles. Based on the morphological and molecular evidence, we place this species in the new monospecific genus Alshehbazia. A description, morphological key, updated geographical distributions, map, and illustration are provided.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Onuris hauthalii (Gilg & Muschl.) Al-Shehbaz (Brassicaceae) is distributed in southern Argentina (Santa Cruz Province) and Chile (Magallanes and Antártica Chilena Region). Generic assignation of this species has been controversial. Gilg & Muschler (1909) originally placed it in Eudema Humb. & Bonpl., based on the presence of a scapose perennial habit with linear entire basal leaves, much reduced inflorescence rachis with the flowers borne on long pedicels originating from the centre of rosettes, and pyriform silicles. Skottsberg (1916) treated it under Brayopsis Gilg & Muschl. but Schulz (1924) reassigned it to Eudema based on fruit shape. Al-Shehbaz (2012a, b) transferred the species to the Patagonian Onuris Phil. (hereafter Onuris s.s.), differing from Eudema basically by having latiseptate (vs angustiseptate) silicles, and smooth (vs conspicuously reticulate) seeds. In addition, the distribution of Onuris s.s. overlaps with that of O. hauthalii, while species of Eudema grow in Ecuador and northern Peru. Therefore, Al-Shehbaz’s (2012a) transfer of the species to Onuris (hereafter Onuris s.l.) may be justified, but it clearly differs from species of Onuris s.s. by its habit and inflorescence.
Salariato et al. (2015) published a molecular phylogeny of tribe Eudemeae, which included Onuris s.s., O. hauthalii, Aschersoniodoxa Gilg & Muschl., Brayopsis, Eudema, Dactylocardamum Al-Shehbaz, and Xerodraba Skottsb. Their results showed O. hauthalii in a clade (“Southern Andean clade”) with Onuris s.s. and Xerodraba, although their relationships were poorly supported.
The aim of this work is to analyse the generic placement of Onuris hauthalii using morphological data and estimating the species tree for Onuris s.l. and related taxa with multilocus DNA sequence data.
Materials & Methods
Morphological studies
Morphological studies of Onuris hauthalii and species of Onuris s.s. were based on herbarium specimens from B, BA, BAA, BAB, CONC, HIP, SGO, SI, and UPS (herbarium acronyms follow Thiers 2014). All cited specimens have been seen by the authors. Leaves, flowers and fruits from herbarium specimens were re-hydrated prior to observations using stereoscopic microscope.
Molecular studies
Sampling and DNA sequence selection. For the estimation of the species tree of Onuris s.l., we used the multilocus DNA sequence data of Salariato et al. (2015). Sequence data from the nuclear ribosomal ITS region and chloroplast regions trnL intron + trnL-F spacer, trnH-psbA spacer, and rps16 intron, were included for O. hauthalii (four specimens) and all species of Onuris s.s.: O. alismatifolia Gilg in Skottsb. (1), O. graminifolia Phil. (2), O. hatcheriana (Gilg ex Macloskie) Gilg & Muschl. (3), O. papillosa O. E. Schulz (2), and O. spegazziniana Gilg & Muschl. (2). We also included species of the related genus Xerodraba: X. patagonica (Speg.) Skottsb. (2), X. pectinata (Speg.) Skottsb. (3), and X. pycnophylloides (Speg.) Skottsb. (1), as well as representatives of the “Northern-Central Andean clade”: Aschersoniodoxa, Brayopsis, Eudema, and Dactylocardamum (one specimen each). All analyses were rooted with Menonvillea chilensis (Turcz.) B. D. Jacks. Vouchers and GenBank accession numbers are provided in Appendix 1.
Phylogenetic analyses. Alignments were generated with Muscle v.3.6 (Edgar 2004) using a first round of multiple alignments and posterior rounds of refinement under the default settings. The alignments obtained were then checked and improved manually where necessary by visual refinement using the program Bioedit v.7.0.9.0 (Hall 1999). All aligned matrices of each locus are available from TreeBase (www.treebase.org) (http://purl.org/phylo/treebase/phylows/study/TB2:S16488).
The coalescent-based method implemented in *BEAST extension (Heled & Drummond 2010) of software BEAST v2.1.3 (Bouckaert et al. 2014) was used to obtain the Bayesian species trees based on all four sampled sequences (ITS, trnL-F, trnH-psbA, rps16 intron). Best nucleotide substitution models from each region were determined using jModeltest v2.1.6 (Darriba et al. 2012) under the AIC as follows: ITS: TVMef+G, trnL-F: TVM+I, trnH-psbA spacer: TPM1uf+G, rps16 intron: TIM1+G. All molecular evolutionary models were unlinked across loci and with an uncorrelated log-normal model (UCLN) of molecular evolutionary heterogeneity assigned to each sampled locus. We linked the tree model for the three chloroplast regions (trnL-F, trnH-psbA, rps16 intron), since they are genetically linked, and set separate tree models for the chloroplast dataset and the nuclear ribosomal ITS region. A Yule process was used for the species tree prior, and the piecewise linear and constant root was used for the population size model. The program BEAUti was used to generate input files for the analyses, in which substitution models were edited manually on the xml file to fit the models selected using jModeltest. All remaining priors were set to the defaults. Two independent runs of 5 × 108, sampling every 2.5 × 104 were executed, discarding as burn-in the first 25% of the samples. Convergence of the chains was checked using Tracer v1.6.0 (Rambaut et al. 2013) ensuring that ESS values for parameters were > 200. Trees from the two independent runs were combined using LogCombiner v1.2.3, and the maximum-clade credibility tree from 3000 trees was generated with TreeAnnotator v1.8 using the posterior probability limit set to 0.5 and the median node heights, visualising it in Figtree v1.4. Finally, results from the species-tree analyses were compared with the concatenated analyses conducted in Salariato et al. (2015).
Results
Morphological Studies
Morphological analysis of Onuris hauthalii agrees with previous observations of Boelcke (1984), Boelcke & Romanczuk (1984), and Al-Shehbaz (2012a, b), and confirm that this species has a unique combination of characters lacking in both Eudema and Onuris s.s. O. hauthalii is characterised by forming scapose perennial herbs with slender rhizomes, the branches of which end in rosettes (Fig. 1A, B). In contrast, both Eudema and Onuris s.s. have a woody caudex. Also, the inflorescences in O. hauthalii have a shortened rachis, generally less than 1.5 mm, with ebracteate flowers on long pedicels originating from the centre of the rosette (Fig. 1C). This flowering type is also present in Eudema, but absent in species of Onuris s.s., which have corymbose racemes bracteate throughout and elongated in fruit. Fruits in O. hauthalii are latiseptate, pyriform silicles (Fig. 1G), while Eudema has angustiseptate silicles. Species in Onuris s.s. also have latiseptate silicles, but they are oblong to ovoid or ellipsoid. Finally, the seed coat in O. hauthalii and Onuris s.s. is smooth, but it is reticulate in Eudema.
Molecular studies
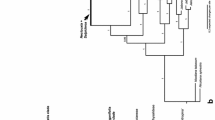
The inferred species tree obtained (Fig 2.) is similar to the phylogenetic tree of Eudemeae presented by Salariato et al. (2015) using concatenated phylogenetic analyses. The tribe Eudemeae is grouped in two main clades, the northern-central Andes clade (NCA clade) that includes Aschersoniodoxa, Brayopsis, Dactylocardamum and Eudema (posterior probability “PP”=1); and the southern Andes clade, including Xerodraba, Onuris s.s., and O. hauthalii (PP=0.96). Monophyly was strongly recovered for Onuris s.s. (PP=1), but not for Onuris s.l. O. hauthalii was sister to Xerodraba in a weekly supported relationship (PP: 0.78).
Maximum clade credibility species tree estimated from one nuclear ribosomal (ITS) and three chloroplast (trnL-F, trnH-psbA, rps16 intron) DNA regions using the multispecies coalescent method implemented in *BEAST. Bayesian posterior support values are given at each node. The arrow shows the position of Alshehbazia hauthalii. Abbreviations: NCA north-central Andes; SA, southern Andes.
Discussion
Morphological and molecular studies show that Onuris hauthalii is a taxon differentiated from both Onuris s.s. and Eudema. Its morphology exhibits a unique combination of habit, inflorescence, and fruits characters that distinguish it from species of Eudema and the closely related Onuris s.s. Species trees obtained in this work, as well as the phylogenetic trees obtained with the concatenated phylogenetic analyses of Salariato et al. (2015), show that O. hauthalii represents a morphologically unique lineage, which is sufficiently phylogenetically isolated from related genera to warrant its generic status. Regarding the weakly supported sister-group relationship between O. hauthalii and Xerodraba, it should be mentioned that the former is easily distinguished from the latter by not forming cushions with imbricate leaves and solitary flowers or occasionally 2- or 3-flowered racemes. The molecular results do not fully support the phylogenetic position of O. hauthalii as sister to Xerodraba; it is also possible that additional data would establish that this species is sister to Onuris s.s. Nevertheless, our analysis indicates that O. hauthalii is a well-differentiated lineage both from Xerodraba and from Onuris s.s. Therefore, based on the morphological and molecular results obtained, we place O. hauthalii into the new monospecific genus Alshehbazia.
Key to Alshehbazia and related genera
-
1. Plants producing loose to compact woody cushions with tiny imbricate leaves (shorter than 1 cm) and solitary flowers, rarely 2 or 3- flowered racemes ………………………………………………………..................Xerodraba
-
1. Plant producing rosettes of non-imbricate leaves, inflorescences several- to many- flowered racemes..................2
-
2 (1). Inflorescences elongated, rachis elongated at least in fruiting, flowers bracteate ...................................Onuris
-
2. Inflorescences not elongated, rachis of inflorescences reduced, flowers ebracteate, with long pedicels arising from the centre of the rosette.........................................................................................................................................3
-
3 (2). Silicles angustiseptate; seed coat coarsely reticulate....................................................................................Eudema
-
3. Silicles latiseptate, pyriform; seed coat smooth ........................................................................................Alshehbazia
Taxonomic treatment
Alshehbazia Salariato & Zuloaga gen. nov. Type: Alshehbazia hauthalii (Gilg & Muschl.) Salariato & Zuloaga (Eudema hauthalii Gilg & Muschl.).
http://www.ipni.org/urn:lsid:ipni.org:names:77150415-1
Herbs perennial, scapose, with slender rhizomes the branches of which terminated by rosettes with petiolar remains of previous years. Basal leaves rosulate, petiolate; petioles persistent, as long as or longer than blade, strongly flattened at base; leaf blade oblanceolate-spatulate to narrowly oblanceolate or linear, glabrous. Inflorescences not elongated, with the rachis obsolete, generally shorter than 1.5 mm; flowers borne on long pedicels originating from the centre of rosette, maturing centripetally; fruiting pedicels ascending to divaricate or recurved, persistent. Sepals ovate, free, deciduous, erect, equal, base of inner pair not saccate. Petals creamy white, erect at base, longer than sepals; blade oblanceolate, apex rounded; claw not differentiated from blade. Stamens 6, exserted or slightly so, erect, slightly tetradynamous; filaments filiform, wingless, unappendaged, not dilated at base, glabrous; anthers ovate, rounded at base, not apiculate at apex. Nectar glands 4, distinct; median nectaries absent; lateral nectaries 1 on each side of lateral stamen. Ovules 4 − 10 per ovary; placentation parietal. Fruits dehiscent, capsular silicles, obovoid to pyriform, rarely ovoid, latiseptate, unsegmented, dehiscing from apex downward; valves thin, obscurely veined, smooth, wingless, unappendaged; gynophore absent; replum rounded; septum reduced to a narrow rim, hyaline, not veined; style slender; stigma capitate, entire, slightly 2-lobed, unappendaged. Seeds biseriate, wingless, oblong, plump, light brown; seed coat smooth, not mucilaginous when wetted; cotyledons incumbent, entire.
recognition. Alshehbazia is easily distinguished from other genera of tribe Eudemeae by having slender and branched rhizomes, not elongated inflorescences with an obsolete rachis, ebracteate flowers on long pedicels originating from the centre of rosette, and obovoid to pyriform latiseptate silicles with smooth seeds.
distribution. The genus is restricted to Southern Argentina (Santa Cruz Province) and Chile (Magallanes and Antartica Chilena region).
etymology . Alshehbazia is named after Dr Ihsan A. Al-Shehbaz, Senior Curator at Missouri Botanical Garden (MO) and an expert on the Brassicaceae (Cruciferae). His contributions to the worldwide knowledge of the family have been invaluable.
Alshehbazia hauthalii (Gilg & Muschl.) Salariato & Zuloaga comb. nov.
http://www.ipni.org/urn:lsid:ipni.org:names:77150416-1
Eudema hauthalii Gilg & Muschl. (Gilg & Muschler, Bot. Jahrb. Syst. 42 (5): 471 (1909)). Brayopsis hauthalii (Gilg & Muschl.) Skottsb. (Skottsberg 1916: 236). Onuris hauthalii (Gilg & Muschl.) Al-Shehbaz (2012b: 949). Type: Argentina, Prov. Santa Cruz, Río Gallegos, al frente del Cerro Buitres (Basalto), 15 leguas de la costa, 1899, R. Hauthal 10618 (holotype B!; isotypes BAA! fragm. ex B, CORD!).
Brayopsis skottsbergii Gilg in Skottsb. (Skottsberg 1916: 236). Type: Argentina, Prov. Santa Cruz, Sierra de los Baguales, Paso Centinela, 5 Feb. 1909, C. Skottsberg s.n. (holotype, UPS!).
Plants glabrous throughout, prostrate. Leaves (6 –) 9 – 15 (– 22) mm long, (0.7 –) 1 – 1.5 (– 2) mm wide, base attenuate, margin entire, apex rounded. Fruiting pedicels (3 –) 5 – 10 mm long. Sepals 2.5 – 3.5 mm long, (1.2 – ) 1.5 – 2 mm wide; petals (4.5 –) 5.5 – 6.5 mm long, (1.2 –) 1.5 – 1.8 mm wide; filaments erect, slender, 2.5 – 3.5 mm long; anthers 0.5 – 0.6 mm long; ovules 4 – 10 per ovary. Fruits (4 –) 5 – 11 (– 12) mm long, 4 – 7 mm wide, style 0.5 – 1 mm long. Seeds light brown 2 – 2.5 (– 3) mm long, 1 – 1.3 (–1.8) mm wide; funicles thick. Fig. 1.
distribution . Alshehbazia hauthalii is restricted to Southern Argentina in Santa Cruz province (Depts Güer Aike and Lago Argentino) and Chile in XII Region (Prov. Última Esperanza). Map 1.
specimens examined. argentina. Prov. Santa Cruz: Dept. Güer Aike, Estancia Las Vizcachas, Cerro Pto. la Piedra, faldeos SE, W de Arroyo Bueno, 26 Jan. 1977, TBPA 2542 (SI); Cerro sin Nombre, ladera E, 29 Jan. 1977, TBPA 2674 (SI); ensenada de Rique, al pié del cerro sin nombre, 3 Jan. 1977, TBPA 2679 (BAA); Laguna las Vizcachas, ladera SSE de la laguna, 28 Jan. 1977, TBPA 2600 (BAB, SI); Dept. Lago Argentino: Cerro Argentino, 8 Jan. 1964, Zöllner s.n. (CONC-94665, SI-167056); Lago Argentino, Cerro Buenos Aires, 14 Jan. 1941, Spegazzini 482 (BAA, BAB); margen de la Meseta Quemada, arriba de la estancia Pérez (Río de las Vueltas), 28 Dec. 1950, Sleumer 1386 (BAA, LIL). chile. Magallanes y de la Antártica Chilena Region (XII): Prov. Última Esperanza, Torres del Paine, Cerro Donoso, Sector Río de las Chinas, 9 − 11 Feb. 1987, Arroyo et al. 870226 (CONC); Cordillera Señoret, 1 Jan. 1931, Donat 436½ (BA); Parque Nacional Torres del Paine, Cerro Agudo, 15 − 17 Jan. 1987, Arroyo & Squeo 870015 (CONC); Cerro Diente, 16 Dec. 1985, Arroyo & Squeo 850848 (CONC); 25 Dec. 1985, Arroyo & Squeo 850953 (CONC); Cerro Daudet, 18 Jan. 1987, Arroyo 870141 (CONC); Sierra Baguales, Estancia La Cumbre, Cerro Cono, 19 Dec. 1975, TBPA 757 (CONC, SI); Estancia La Cumbre, campo Laguna Grande, 9 Dec. 1986, Landero 651 (CONC); Sierra del Toro, 10 Feb. 1992, Arroyo et al. 92115 (CONC); Arroyo et al. 92191A (CONC); Arroyo et al. 92293 (CONC).
habitat. It grows on fine gravel, along mountain wetlands, peat bogs, and moist seepage areas; between 700 − 1400 m.
conservation status. Alshehbazia hauthalii has an apparently restricted distribution in southern Argentina and Chile, but most certainly its populations are under-collected: DD (Data Deficient) following IUCN Categories and Criteria.
phenology. The species flowers from December to January, and produces fruits until February.
notes. Al-Shehbaz (2012b) cited this species for San Juan and La Rioja Provinces in Argentina, based on three specimens, Krapovickas & Hunziker 5996 (BAA, LP), Nicora et al. 8508 (LP, P) and Múlgura & Deginani 169 (SI), but a closer examination of the leaves show branched trichomes characteristic of Petroravenia werdermannii (O. E. Schulz) Al-Shehbaz. Therefore, the distribution of Alshehbazia hauthalii is restricted to southern Argentina and Chile.
References
Al-Shehbaz, I. A. (2012a). A generic and tribal synopsis of the Brassicaceae (Cruciferae). Taxon 61: 931 – 954.
____ (2012b). Eudemeae. In: A. M. Anton & F. O. Zuloaga (eds), Brassicaceae, Flora Argentina, Vol. 8: 135 – 150. Editorial Sigma, Buenos Aires.
Boelcke, O. (1984). El género Onuris (Cruciferae), endémico de la Patagonia. Parodiana 3(1): 53 – 65.
____ & Romanczuk, M. C. (1984). Cruciferae. In: M. N. Correa (ed.), Flora Patagonica 8 (4a): 373 – 544. Instituto Nacional de Tecnología Agropecuaria, Buenos Aires.
Bouckaert, R., Heled, J., Kühnert, D., Vaughan, T., Wu, C. H., Xie, D., Suchard, M. A., Rambaut, A. & Drummond, A. J. (2014). BEAST 2: a software platform for Bayesian evolutionary analysis. PLoS computational biology 10: e1003537. doi:10.1371/journal.pcbi.1003537.
Darriba, D., Taboada, G. L., Doallo, R. & Posada, D. (2012). jModelTest 2: more models, new heuristics and parallel computing. Nat. Methods 9: 772 – 772. doi:10.1038/nmeth.2109.
Edgar, R. C. (2004). MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucl. Acids Res. 32: 1792 – 1797.
Gilg, E. F. & Muschler, R. C. (1909). Aufzählung aller zur Zeit bekannten Südamerikanischen Cruciferen. Bot. Jahrb. Syst. 42(5): 437 – 487.
Hall, T. A. (1999). Bioedit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl. Acids Symp. Ser. 41: 95 – 98.
Heled, J. & Drummond, A. J. (2010). Bayesian inference of species trees from multilocus data. Molec. Biol. Evol. 27: 570 – 580. doi:10.1093/molbev/msp274.
Rambaut, A., Suchard, M. A., Xie, D. & Drummond, A. J. (2013). Tracer v1.6, Available from <http://beast.bio.ed.ac.uk/Tracer>
Salariato, D. L., Zuloaga, F. O., Cano, A. & Al-Shehbaz, I. A. (2015). Molecular phylogeny of the tribe Eudemeae (Brassicaceae) and implications for its distribution and morphology. Molec. Phylogenet. Evol. 82: 43 – 59. doi:10.1016/j.ympev.2014.09.030.
Schulz, O. E. (1924). Cruciferae-Sisymbrieae. Pflanzenr. (Engler) IV 105(Heft 86): 1 – 388.
Skottsberg, C. J. F. (1916). Botanische Ergebnisse der Schwedischen Expedition Nach Patagonie und dem Feuerlande 1907 – 1909, V. Die Vegetationsverhältnisse Längs der Cordillera de los Andes S. von 41° S Br. Ein Beitrag Zur Kenntnis der Vegetation on Chiloé, West-Patagonien, dem Andinen Patagonien und Feurland. Kongl. Svenska Vetensk. Acad. Handl. 56(5): 1 – 366.
Thiers, B. (2014). Index Herbariorum: A global directory of public herbaria and associated staff. New York Botanical Garden’s Virtual Herbarium. [April 2014]. http://sweetgum.nybg.org/ih
Acknowledgements
Funding of this work was provided by ANPCyT (Agencia Nacional de Promoción Científica y Tecnológica) grant PICT-2013-1042, CONICET (Consejo Nacional de Investigaciones Científicas y Técnicas) grants D4541-12 and PIP-112-201301-00124CO, and the National Geographic Society grant #9398-13, for which we are profoundly grateful. Fieldwork and visits to herbaria were also supported by the Myndel Botanical Foundation grants in 2011 and 2012. Our deep gratitude goes to Dr Ihsan A. Al-Shehbaz for his valuable support, guidance, and suggestions for this work and for the study of South American Brassicaceae over the years. We especially thank Dr David Simpson and the two anonymous reviewers for suggestions to improve an early version of this paper. We are grateful to the directors and curators of the herbaria listed and to Francisco Rojas for preparing the illustration.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Salariato, D.L., Zuloaga, F.O. Taxonomic placement of Onuris hauthalii (Brassicaceae: Eudemeae), based on morphology and multilocus species tree analyses, and the recognition of the new genus Alshehbazia . Kew Bull 70, 49 (2015). https://doi.org/10.1007/s12225-015-9602-9
Accepted:
Published:
DOI: https://doi.org/10.1007/s12225-015-9602-9