Abstract
Low back pain is a leading cause of disability worldwide and the second most common cause of physician visits. There are many causes of back pain, and among them, disc herniation and intervertebral disc degeneration are the most common diagnoses and targets for intervention. Currently, clinical treatment outcomes are not strongly correlated with diagnoses, emphasizing the importance for characterizing more completely the mechanisms of degeneration and their relationships with symptoms. This review covers recent studies elucidating cellular and molecular changes associated with disc mechanobiology, as it relates to degeneration and regeneration. Specifically, we review findings on the biochemical changes in disc diseases, including cytokines, chemokines, and proteases; advancements in disc disease diagnostics using imaging modalities; updates on studies examining the response of the intervertebral disc to injury; and recent developments in repair strategies, including cell-based repair, biomaterials, and tissue engineering. Findings on the effects of the omega-6 fatty acid, linoleic acid, on nucleus pulposus tissue engineering are presented. Studies described in this review provide greater insights into the pathogenesis of disc degeneration and may define new paradigms for early or differential diagnostics of degeneration using new techniques such as systemic biomarkers. In addition, research on the mechanobiology of disease enriches the development of therapeutics for disc repair, with potential to diminish pain and disability associated with disc degeneration.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Low back pain (LBP) is a leading cause of disability worldwide, with a lifetime incidence of 80 % [1] and an estimated 11.9 % of the worldwide population affected at any time [2]. In the USA, the population incidence of LBP is even greater with CDC surveillance demonstrating that 30.1 % of American women and 26 % of men report LBP within a 3-month period [3, 4]. As the second most common cause of doctor visits, LBP contributes $20–100 billion in direct health care spending [5], and between $100 and 200 billion annually in total economic burden [6, 7]. There are many causes of LBP, and among them, disc herniation and intervertebral disc (IVD) degeneration are the most common diagnoses and targets for intervention [8]. Disc degeneration (DD) is a multifactorial process characterized by cellular and biochemical changes in disc tissue that result in structural failure [9]. While DD is a part of normal aging, a significant number of people with indications of DD on MRI are actually asymptomatic, with no history of pain or disability [10, 11]. Consequently, routine radiological diagnostics do not strongly correlate with symptoms including pain, except in more severe and persistent conditions [12]. In addition, diagnostic criteria such as changes in disc height, bulging, or herniation do not provide strong correlations with treatment outcomes in patients who are in pain [13, 14]. This discrepancy between the asymptomatic state despite physiologic degeneration and the symptomatic state emphasizes the importance of characterizing more completely the mechanisms of degeneration and their relationships with symptoms.
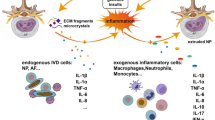
Multiple changes are observed in DD, each of which provides insights into the disease process. A normal disc is a composite of two tissue types—the nucleus pulposus and the annulus fibrosus, which have different functions in load bearing. The nucleus pulposus (NP) is gelatinous, composed predominantly of type II collagen and proteoglycans, with high water content that contributes to its hydrostatic pressurization and resistance to compressive forces [15, 16]. In degeneration, loss of proteoglycans and water content are observed by changes in disc height and MRI signal [17], which results in redistribution of load onto the fibrochondrocyte-like cells in the annulus fibrosus (AF) [18]. The AF consists of concentric lamellae rich in collagen and elastin, whose content varies with distance from inner to outer portions. This review covers recent studies elucidating cellular and molecular changes in DD, which provide greater insights into pathogenesis, may define new paradigms for early and/or differential diagnostics of DD using new techniques such as systemic biomarkers, and enriches the development of therapeutics for IVD repair (Fig. 1).
Biomarkers of disc degeneration and back pain. Disc degeneration process begins years before disc height narrowing is observable by current diagnostic methods (structural, radiological). There is an urgent need for earlier methods of detection of disc degeneration, which can lead to novel and earlier, less invasive therapies
Updates on currently available treatments
Current standard of care for LBP due to degenerative disc changes includes non-operative (pain management) and operative approaches. Non-operative management focuses on symptomatic pain relief, while permitting possible endogenous recovery such as resolution of herniation [19, 20] or repair of structural damage [21, 22]. Analgesia is the primary target of non-operative management and is accomplished by a combination of NSAIDs and physical therapy to strengthen core muscles among other programs. Surgical management may begin with epidural injections of local anesthetic, steroids, or a combination of both prior to more invasive surgical approach. The evidence of efficacy is variable among different epidural injection techniques, such as interlaminar, caudal, and transforaminal injections, and for various conditions including intervertebral disc herniation (DH), spinal stenosis (SS), and discogenic pain without disc herniation or radiculitis. The debate continues with regard to the effectiveness, indications, and medical necessity of epidural injections [23, 24]. For radicular pain secondary to DH, treatment with epidural injections of steroids in conjunction with local anesthetic resulted in greater pain relief compared to anesthetic alone, reinforcing the contribution of neuro or disc inflammation in LBP [23, 24]. The indications for use of lumbar epidural injections for discogenic pain without DH or radicular pain due to SS are fair, and a recent study found that injection with anesthetic alone is comparable to use of anesthetic and steroids for pain relief [23–25]. While significant complications can arise from needle placement, including epidural hematomas, disc puncture, and infections in the epidural space, meninges, IVD, and vertebrae, the use of fluoroscopic guidance mitigates some risk of adverse events involved with this invasive therapy [26].
Operative management includes in situ repairs using minimally invasive microdiscectomy or open procedures for removal of herniated tissue, impingement relief, and spinal stabilization [8, 27]. In cases where laminectomy and spinal fusion are performed, achieving stability is crucial. Recombinant human bone morphogenetic protein-2 (rhBMP-2), a popular adjuvant for spinal fusion procedures, has recently garnered attention due to safety concerns. Though previous studies have indicated that rhBMP-2 achieves higher fusion rates in spinal surgery compared to iliac crest bone graft [28, 29], recent studies have focused on potential side effects and complications. The recent analyses were performed by independent groups under the auspices of the Yale Open Data Access project, where complete patient data generated from clinical trials sponsored by the manufacturer, Medtronic, were made publically available for review. Two independent teams performed meta-analyses on studies using BMP-2 and found similar, but not identical, results. Fu et al. found no improvement in outcomes with rhBMP-2 compared with iliac crest bone graft and increased rate of cancer with rhBMP-2 use [30]. In contrast, the analysis by Simmonds et al. found better rates of fusion at 24 months with rhBMP-2, but increased postoperative pain and no significant improvements in clinical outcomes. They also found a significant increased risk of heterotopic bone formation and the possibility of increased cancer risk [31]. These analyses provide the most comprehensive review of side effects and complications of rhBMP-2 in spinal fusion and form the basis for the current consensus statement regarding the use of rhBMP-2 in spine surgery for clinically indicated or off-label procedures [32]. However, their nuanced messages support continued controversy surrounding the use of rhBMP-2 for spinal fusion following operative management of DD.
Biochemical changes in disc disease: cytokines, chemokines, and proteases
Inflammation is implicated in both the triggers and effects of DD, which can both involve cells and molecules that participate in immune responses (reviewed in [33••]). Inflammation is a process that the body engages in response to harmful stimuli, such as damaged cells or infectious material. While inflammation is a necessary step in wound healing, persistent inflammation can damage tissue and organs and may contribute to pain [34]. Factors associated with adaptive immunity, and more recently innate immunity, have been implicated in IVD disease. In healthy conditions, NP tissue is considered to be immunologically privileged or naïve because of lack of direct vasculature into the tissue or interaction with the systemic immune system once formed. In DH, NP tissue and cells protrude beyond the interspace and interact with the systemic immune system, leading to accumulation of immune cells on the herniated NP and within the IVD, including macrophages, T cells, and neutrophils (reviewed in [33••]). Thus, DH triggers an immune response mediated by pro-inflammatory signaling by disc or immune cells and generates pain by physically touching, impinging, or chemically stimulating nerve roots or dorsal root ganglion.
To investigate the role of sterile inflammation in IVD, Rajan et al. modeled IVD degeneration by stimulating Toll-like receptor 4 (TLR-4), a pattern recognition receptor of the innate immune system, with one of its ligands, lipopolysaccharide (LPS) [35]. TLR-4 activation stimulated increases in inflammatory mediators (TNF-α, IL-1β, and IL-6) in vivo and in vitro and identified production of novel mediators (HGMB1 and MIF) in vivo. Activation of inflammatory mediators was also achieved by exposure to individual cytokines (TNF-α, IL-1β); however, the inflammatory response was significantly higher with LPS stimulation [35]. Klawitter et al. also demonstrated that expression of TLR-1, 2, 3, and 4 is enhanced in the presence of TNF-α in human IVD cells, and TLR-2 expression is elevated by IL-1β stimulation. Interestingly, expression levels of TLR-1, 2, 4, and 6 were also found to increase with increasing degeneration severity in human IVD [36]. Activation of TLR-4 in the IVD induced moderate disc degeneration in vivo, in the absence of traumatic injury, confirming that inflammation alone is able to trigger DD [35]. Studies on pattern recognition receptors and the interaction of cytokines in IVD tissue highlight the importance in further defining the role of innate immunity in DD. In addition, the presence of autoimmune antibodies in degenerating discs obtained from human subjects supports continued examination of the adaptive immune system’s involvement in disc disease [37].
RANTES (CCL5), a chemoattractant for monocytes, macrophages, and T cells, also changes significantly with DD and was elevated in discs from patients with more severe degeneration, independent of herniation. This expression was increased in response to TNF-α, and both baseline and induced RANTES expression were particularly localized to AF cells [38]. AF cells also demonstrated constitutive expression of growth differentiation factor-5 (GDF-5), a growth factor that belongs to the BMP gene family and is implicated in mitigating DD [39, 40]. The expression of GDF-5 in the AF was significantly upregulated in herniated versus non-herniated discs but was independent of degeneration, suggesting a response induced explicitly by herniation. Exposure of AF cells in vitro to inflammatory cytokines TNF-α and IL-1β induced a significant downregulation of GDF-5 expression [41]. These findings support the concept that an endogenous repair mechanism may be activated in response to herniation and impaired by inflammation. While this repair mechanism may be amenable to therapeutic treatments, one important gap in knowledge is the efficacy of anti-inflammatory drugs at promoting recovery of GDF-5 expression and activity in AF cells.
Understanding the development of DD has increasingly implicated the presence and dynamics of inflammatory mediators which act to stimulate chemokine, protease activity, and calcification in and by disc cells [33••]. Recent efforts to identify differences between pathologic processes and normal aging suggest that matrix metalloproteinase (MMP)-3 is associated with pathological processes in diseased discs [42, 43]. MMPs contribute to extracellular matrix turnover and disruption, and recently MMP-3 and to a lesser extent MMP-8 have been shown to be strongly upregulated in human samples with histological evidence for DD [42]. MMP-3 expression levels were correlated with histomorphologic age-related alterations, such as cell proliferation, tears and cleft formation, and mucoid degeneration. MMP-8 expression levels were correlated with pain duration and histologic degeneration score [42]. Interestingly, an upregulation in expression of MMP inhibitors (TIMP-1 and TIMP-2) paralleled that of MMPs [42]. More recently, Canbay et al. examined relationship between Pfirrmann grade, immunohistochemical expression of MMP-3, and the histopathologic signs of IVD degeneration [44]. This investigation found that histopathological score was significantly correlated with DD severity on MRI (Pfirrmann grade) and MMP-3 expression, although MMP-3 expression was not significantly correlated with age. Thus, the absence of correlation of MMP-3 with age suggests that mechanisms of IVD may be distinct from normal aging [44]. Increases in MMP-2 were also recently shown to occur in experimentally induced preclinical models of degeneration. AF cells appear to use MMP-2 in a directed fashion for local matrix degradation and collagen remodeling [45]. These findings strengthen the association between catabolic breakdown associated with protease activity and DD. For example, Furtwangler et al. observed that injecting disc explants with known proteases associated with DD (MMP-3 or ADAMTS-4) and a less studied protease in disc (HTRA1) decreases cellular activity and disc height; however, the histopathologic features of degeneration were not observed on the time scale tested [46].
Increasingly, recent research has focused on associations between disc disease and comorbidities that may induce inflammatory signaling. Albert et al. found high prevalence of asymptomatic infection in surgically removed herniated IVD tissue (46 % of patients had positive bacterial culture) [47]. Patients with positive cultures were significantly more likely to have evidence of bone edema (Modic changes [48]) 1 year after surgery, in the absence of symptomatic or persistent infection [47]. These findings may represent a nidus of infection leading to persistent inflammation with associated changes in the adjacent bony tissues. Modic changes are associated with pain [49], so these findings of chronic inflammation secondary to bacterial infection may contribute to the frustrating diagnosis of chronic LBP.
There is a common relationship among LBP and DD, obesity, and metabolic syndrome including diabetes, in that they are associated with increased cytokine activity. The oxidative stress resulting from diabetes mellitus [50, 51] poses a risk to IVD tissues by potentially inducing or accelerating inflammation. A recent animal model used by Tsai et al. demonstrated that a rat model of type I diabetes mellitus had increased expression of MMP-2 when exposed to advanced glycation end products (AGEs), as compared to similar exposure in non-diabetic rats [52]. Considering the role of MMPs in degeneration and painful symptoms of disc disease, the possibility that diabetes further accelerates inflammation and degeneration may offer recommendations for the prevention of disc disease in diabetics, as well as an explanation for its increasing incidence [53]. A similar model by Illien-Junger et al. examined disc height, glycosaminoglycan (GAG) content, and inflammatory factors in a murine model of type I diabetes treated with broad spectrum anti-inflammatory and AGE-inhibiting medications. Untreated diabetic mice showed decreased IVD height, decreased GAG content, and increased catabolic factors (including AGEs and TNF-α) compared to both control mice and diabetic mice in the treatment group. Treatment also decreased, but did not entirely protect, against expression of catabolic factors (ADAMTS-5, MMP-13) in diabetic mice [54]. The response to treatment adds a dimension of specificity to the proposed pathogenesis of disc disease in diabetes and may help focus clinical management in diabetics. These studies suggest multiple sources of inflammation in disc disease, including infection and diabetes, which may represent important future therapeutic targets.
Select drugs that are used in the treatment of rheumatoid arthritis or osteoarthritis have been examined for treatment of DD. Drugs targeting inflammatory cytokines implicated in inflammation, such as TNF-α and IL-1, are being widely used clinically in the treatment of rheumatologic diseases through systemic administration. Clinical trials have also explored the use of these same anti-cytokine therapies for the treatment of LBP and radicular pain, using epidural injection delivery. The TNF-α inhibitor etanercept has demonstrated varied results for treatment of sciatica or spinal stenosis, with some studies showing significant improvement in pain over the placebo groups, while others indicating no benefit over placebo and found it to be less effective than steroids [55–59]. Another TNF-α inhibitor, adalimumab, demonstrated improvement in leg pain and reduced the proportion of patients requiring follow-up back surgery, while another inhibitor, infliximab, did not demonstrate significant effect on pain [60–62]. A study on an inhibitor of IL-6, tocilizumab, had significant improvement in leg pain, leg numbness, and back pain compared with demographically matched patients who received steroid treatment. Currently, the evidence supporting these therapies in back pain and related disc etiologies remains limited [63]. The use of glucosamine supplements for osteoarthritis has been widely adopted; however, concerns have been raised regarding the use of glucosamine supplementation for LBP [64, 65]. Studies have explored the effects of glucosamine on disc matrix homeostasis in an animal model of DD and demonstrate a net negative effect on the matrix [64]. In vitro studies revealed a complex mechanism of action, where glucosamine alleviated some inflammatory effects but increased deleterious effects of mechanical loading on AF cells [65]. These results may partially explain the conflicting reports of efficacy, though future research is warranted to establish clinical relevance [64, 65].
Advancement in disc disease diagnostics by imaging modalities
The measurement of biomechanical properties of the IVD provides the opportunity for quantitative analyses of tissue integrity and improved opportunities in the diagnosis of DD and LBP. The establishment of benchmarks for the biomechanical properties of IVD tissues will further allow for improved clinical diagnosis of DD. IVD biomechanical properties, including elastic moduli, water content, permeability, GAG content, and Poisson’s ratio have all been proposed [66] as a reference for comparative analysis in clinical diagnostic settings. Additionally, the use of finite element modeling has been used to predict biomechanical behavior and properties of the IVD and simulate changes in those properties during DD [67]. Therefore, biomechanical properties predicted for DD may be useful in validation of benchmarks for the clinical diagnosis of DD.
Conventional MRI used clinically for diagnosis of DD is largely qualitative and dependent on subjective assessment of anatomical changes associated with disease. Quantitative MRI (qMRI) measures, specifically T1 and T2 relaxation times, represent inherent properties of tissues which may change with structural alterations due to disease. In DD, T1 and T2 have been shown to significantly decrease in both the NP and AF with increasing degeneration grades [68]. QMRI measures have also been shown to be correlated with regional or overall changes in disc tissue biomechanical properties [68]. Other specialized sequences, such as T2* MRI mapping was shown to be significantly correlated with changes in biomechanical properties that are indicative of the IVD degenerative processes, specifically range of motion and stiffness changes [69]. Similarly, magnetic resonance elastography (MRE) is an imaging technique that allows for non-invasive measurement of the shear modulus of soft tissues [70•]. Shear modulus measured with MRE provides a new diagnostic measure that could potentially be compared to known values of healthy and degenerative IVD tissue. Shear modulus measured with MRE was also shown to correlate significantly with clinical DD severity measures on MRI (Pfirrmann grade). The use of quantitative computed tomography (QCT) and micro-QCT (μQCT) has also been used to investigate relationships between the microstructure of the IVD in degenerative conditions. In a study by Jackman et al., increased deflection of the IVD end plate was found to be a significant indicator of vertebral fracture [71]. The ability to measure IVD microstructure using QCT and μQCT can potentially allow for the measurement of biomechanical properties that can be used in the diagnostics. The development and use of these tools for DD diagnosis represent opportunities for potential quantification of degeneration severity, which is largely a qualitative assessment in disc degeneration. In addition, the development of μCT-based 3D kinematic analysis can provide a quantitative assessment of repair in preclinical models to complement current gold standard methods used for efficacy assessment of new therapeutic approaches [72].
Response of IVD to injury
Structural changes and misalignment in the spine may induce injurious changes in the IVD. For example, the facet joints transfer loads, guide, and constrain motions in the spine. Injury, degeneration, or anatomical perturbations due to surgery may alter the facet joints, leading to misalignment and biomechanical dysfunction (summarized in [73]). Fractures within the vertebral bodies also affect the alignment of adjacent facet joints that can cause increased wear and altered loading and can subsequently lead to IVD degeneration. Degeneration of the IVD and adjacent facet joints have been observed to occur coordinately, suggesting the involvement of an interactive stimuli (such as mechanical tissue insults) or shared mechanisms (such as inflammation or chemical irritation) [73, 74]. Indeed, we have recently observed that inflammatory and degradative expression in spinal ligaments (the ligamentum flavum) of subjects with DD or HD increases with disc disease severity [75].
Fractures of the vertebral body and adjacent cartilaginous end plate (CEP) of the IVD have been implicated in the progression of degenerative changes within the disc. In a study by Dudli et al., burst fracture of the CEP, in which a highly energetic impact causes fracture of the CEP and expulsion of NP from the IVD in organ culture studies, leads to significant increases in DD markers and cell death. Non-fracturing impact as well as non-energetic NP depressurization did not lead to the same patterns of degenerative markers, as identified by primary component and clustering analyses [76, 77]. Degeneration in a preclinical rabbit model was also found to increase the potential for delamination between AF layers. Substantial change to the integrity of the AF after degeneration may facilitate disc herniation and represents an additional therapeutic target for strengthening the AF [78]. These findings further support the notion that localized mechanical disruption of a distinct tissue (CEP, AF, or NP) dysregulates interactions among them and facilitates fracture or delamination that may dispose the IVD to subsequent degeneration or herniation [76–79].
Dynamic mechanical stimulation of IVD affects the metabolic activity of IVD cells and may consequently alter their ability to maintain the integrity of disc extracellular matrix (ECM). Dynamic loading has been shown to increase the metabolic activity of both NP and AF cells by increased glycolysis [80, 81]. Increased levels of ATP and ATP consumption due to these changes in metabolic activity can alter the production of GAGs within the disc, an ATP-demanding process, leading to possible changes in disc ECM integrity. Increased ATP levels have also been suggested to cause CEP calcification and consequently reduced delivery of nutrients to the inner IVD, leading to cell death and subsequent degeneration. Differences between the responses of AF and NP cells to dynamic loading have also been observed in cell cytoskeleton [82], viability, and gene expression [83], all of which can contribute to compromised IVD structure and degenerative effects. Dynamic loading experiments have also shown cells isolated from older subjects are even more susceptible to degenerative changes following tensile loading, versus cells of younger subjects [84]. Complex loading of disc explants, which combines compression and torsion, induced a stronger degree of disc degeneration compared with compression or torsion loading alone [82, 83].
The behavior of IVD cells, especially in their response to mechanical loading, is dependent upon their morphology, mechanical properties, the properties of the extracellular matrix (ECM), and cell-matrix interactions. NP cells respond to mechanical stimuli such as compression, tension, and shear, through cell-ECM interactions which control the deformation and stress propagated from the tissue down to the cell and cytoskeleton. Inflammatory stimulation of IVD cells has been shown to alter the mechanobiology of single cells, by altering morphology, the cytoskeleton, and hydraulic permeability of the cell [85]. Significant changes in ECM composition with degeneration can also alter the mechanobiology of disc cells. Manipulation of the cell-ECM interaction, such as substrate stiffness and content of the ECM constituents, can be used to control and modify cell responses, promoting NP cell phenotype and morphology [86•]. Changes in the mechanobiological properties of these cells can affect the properties and integrity of the IVD as a whole potentially making it susceptible to further degeneration. The control of cell-ECM interactions can be used for altering the response of cells to loading in disease or for the development of tissue engineering or cell delivery strategies [86•].
Inflammatory response of IVD cells to mechanical stimulation or injury has also been suggested to play a role in the presentation of discogenic and lower back pain. Exposure of dorsal root ganglion to NP tissue following disc herniation is thought to induce nerve hyperalgesia and associated lower back pain, due to interaction with cytokines released from NP cells [87]. Increases in the expression of brain-derived neurotrophic factor (BDNF) and nerve growth factor (NGF) within the IVD have been observed in response to whole body vibration [88]. Interactions between AF cells and endothelial cells (ECs) that line the microvasculature were also investigated [89]. AF cells from DD secreted factors which stimulated EC production of factors known to induce matrix degradation, angiogenesis, and innervations, including IL-8 and VEGF [89]. Increases in such factors may promote ingrowth of blood vessels and nerves into the IVD and may be associated with increased discogenic pain.
Advancement in repair strategies
Management of IVD degeneration currently focuses on resolution of symptoms and, to a lesser degree, prevention. With more information regarding the pathogenesis and mechanobiological changes, there is an increased therapeutic focus on biological treatments with growth factors, cell transplantation, and biomaterial-based repair.
Growth factors
The premise for growth factors in disc repair is to encourage endogenous repair processes and maximize the efficacy of other interventions, but utilization depends on understanding other interactions in the cellular environment and limiting unintended effects. To address the change in anabolic factors in response to inflammation, Tran et al. performed a study on connective tissue growth factor (CCN2), a protein found in the extracellular matrix with important implications in responding to growth factors and promoting wound healing [90, 91]. They found reduced expression of CCN2 following inflammatory stimulation, and this reduction was mediated by nuclear factor-κB (NF-κB). The protective effects of CCN2 resulted in decreased expression of catabolic genes in IVD cells, and this protection persisted with NF-κB inhibition. However, severely degenerated cells were unable to generate the same response to CCN2 [92] independent of NF-κB exposure, suggesting that inflamed and degenerated cells lack a robust response to CCN2. Kim et al. found anti-inflammatory and protective effects of autologous platelet-rich plasma (PRP) on IVD tissue in a small study on patients (N = 3) with intermediate level of degeneration. While this study did not address the mechanism, PRP is also able to inhibit NF-κB [93, 94]. Application of PRP significantly restored gene expression of collagen type II and aggrecan and significantly reduced COX-2 and MMP-3 gene expression following exposure to pro-inflammatory stimuli [95]. The intersection of many anti- and pro-inflammatory pathways onto the NF-κB pathway makes it an important target for further elucidation and potential therapeutic targeting.
The TGF-β superfamily and its constituent members of the BMP family are also implicated in DD. In culture, stimulation with bone mineralizing protein-2 (BMP-2) or transforming growth factor-β3 (TGF-β3) increases expression of matrix proteins, such as collagen and aggrecan. However, a rabbit tissue explant model has also shown pronounced ossification in the AF following application of BMP-2 and TGF-β3 [96]. Similarly, insulin-like growth factor-1 (IGF-1) in IVD tissue increases production of proteoglycans in a dose-dependent fashion [97]. Huang et al. explored a clinically relevant mechanism of introducing IGF-1 into the disc via injection, for evaluating competition between growth factor diffusion and binding kinetics required to produce anabolic stimulation. IGF-1 demonstrated a significant binding reaction to IGF-1 binding protein, thus lowering the availability of freely soluble IGF [98]. The resulting increased metabolic demand of this anabolic stimulation was found to also decrease pH, decrease glucose, and decrease oxygen tension, all biophysical shifts thought to hinder anabolic benefits of growth factor stimulation [98]. The therapeutic use of IGF-1 and other growth factors at effective dosages needs to balance the metabolic demands in the avascular disc and changes in cellularity with increasing disease severity. This class of treatment may be more effective before severe DD onset, further highlighting the need for augmented diagnostics in DD for better identification of subjects who may find benefit from potential treatments (Fig. 1). Several growth factors have overlapping signaling pathways, including CCN-2 and IGF-1 [99], emphasizing the need for a greater understanding of growth factor activation mechanisms and physical interactions. One feature of mid- to late-stage DD is the onset of calcification in the disc. Suppression of calcification potential has been hypothesized as a possible therapeutic target. A study by Madiraju et al. demonstrated that human parathyroid hormone (PTH) can potentially retard IVD degeneration by stimulating matrix synthesis and suppressing markers of calcification potential in degenerated disc cells [100]. Tissue engineering provides a promising approach to recover the functionality of the degenerative intervertebral disc. Many studies are directed toward nucleus pulposus (NP) tissue engineering because DD is associated with weakening and matrix loss in the NP. Tissue-engineered IVDs are designed to be utilized as a living tissue replacement in IVD repair, promoting healing and recovery of mechanical properties. Previous studies of engineered NP have borrowed techniques from cartilage tissue engineering, including culture media composition, hydrogel-based scaffolds, and biophysical conditioning. However, important phenotypic differences exist between NP and articular cartilage, including higher water content and GAG to collagen ratio in NP tissue [101, 102], supporting disc-focused investigations. Understanding how biological factors regulate in vitro NP matrix development represents an important step toward generating a physiologically relevant model for NP tissue engineering.
We are investigating effects of the fatty acid linoleic acid (LA) and the morphogen transforming growth factor (TGF-β) on engineered NP tissues. LA is an omega-6 polyunsaturated fatty acid that has been shown to decrease production of inflammatory cytokines in osteoarthritic chondrocytes [103]. TGF-β is a potent morphogen used for induction of a chondrogenic phenotype in undifferentiated mesenchymal stem cells (MSCs) and chondrocytes. Transient supplementation of TGF-β for the first 14 days in culture improves chondrogenic growth and mechanical properties of articular chondrocytes in tissue-engineered constructs, compared to continuous growth factor supplementation [104, 105]. However, the effect of transient TGF-β supplementation on NP cells in 3D culture has not been widely investigated. We explored the temporal effects of TGF-β (10 ng/ml) and LA (470 μg/ml) exposure on the biomechanical and biochemical development on NP cells grown in 3D hydrogel scaffolds (2 % agarose), mimicking an engineered NP tissue replacement (Fig. 2). While transient TGF-β is more potent in inducing chondrogenic tissue growth of articular chondrocytes compared to continuous culture [104, 105], our findings suggest that NP cells respond less robustly to transient TGF-β exposure. Short-term TGF-β exposure results in comparable GAG content and mechanical properties as compared to continuous exposure for 6 weeks in vitro, confirming the pro-anabolic effects of TGF-β on NP cell tissue engineering [106, 107]. The presence of LA synergized with short-term TGF-β to yield the greatest increase in GAG content and mechanical properties. Consequently, LA appears to be a critical supplement for engineered NP tissue. LA inhibits MSC proliferation and alters MSC expression and secretion of known mediators of angiogenesis [108]. In addition, LA decreases production of inflammatory cytokines in osteoarthritic chondrocytes [103], suggesting potential mechanisms for the observed effects of LA on NP cells.
Temporal effects of TGF-β and LA on NP Cells. a NP cells were cultured in 2 % agarose hydrogels for up to 42 days in culture. Bars indicate duration of exposure. b Percent increase in equilibrium modulus from day 14 values measured by unconfined compression tests. *p < 0.001 versus all other groups. c Percent increase in GAG content per wet weight of NP constructs from day 14 values. *p < 0.001 versus respective LA− group. + p < 0.002 versus LA−/TGF+
A recent preclinical study described the evaluation of an electrosurgical technique for ablation or tissue removal in the IVD using plasma-mediated radiofrequency-based ablation. This technique appears to have an anabolic effect on disc cells, stimulating proteoglycan and IL-8 production and maintaining annulus architecture [109]. The increasing IL-8 may promote maturation and remodeling of the disc matrix and may restore overall disc architecture [109].
Cell-based repair
Stem cell therapies are gaining attention in many diseases, and IVD degeneration is no exception. However, the avascular environment of the disc and concerns over disruption of the matrix integrity during cell delivery via needle injection present unique obstacles to the successful introduction of regenerative cells. Chan et al. developed a model for evaluating efficacy of cell-based repair, using papain to enzymatically degrade the proteoglycans of the NP, and found that discs with moderate to severe loss of extracellular matrix were better able to accommodate cell delivery via intradiscal injection [110]. While this model simulates structural and biomechanical changes of degeneration, the ability to understand the inflammatory environment and its role in intracellular interactions are limited.
Multipotent stem cells (MPSCs) derived from umbilical cord blood show promise at protecting disc height and increase aggrecan expression in IVD repair. The availability of umbilical cord blood MPSCs through cord blood banks may also be less invasive than use of autologous stem cells collected from bone marrow or adipose tissue. However, the avascular nature of IVD and concern for disrupting the integrity of the AF complicate delivery of cells for disc repair. A mouse model by Tam et al. evaluated the therapeutic tradeoff between disc puncture for administration of MPSC directly to the IVD versus delivery via intravenous injection. Discs that were punctured and did not receive stem cells showed significant loss of disc height, whereas discs that were injected with the MPSCs preserved their height. Both intradisc and intravenous administration of stem cells resulted in an improvement in aggrecan expression and appeared to stimulate endogenous repair, though the delivered stem cells did not persist in the disc [111]. Another study in a rabbit model showed similar trends for repair with MPSC, but was unable to establish superior outcomes in stem cell-treated groups versus rabbits treated with hydrogel carrier alone [112].
Notochordal cells are the developmental origin of the nucleus pulposus; however, they are not expressed in the adult human IVD. Induced pluripotent stem cells (iPSCs) have demonstrated their capacity to differentiate into various cell types. In IVD applications, mouse and human iPSCs have been shown to differentiate into NP-like cells expressing notochordal markers and suggest the possibility that they may be used as a novel cell source for cellular therapy [113, 114]. Notochordal cells have been observed to substantially stimulate biosynthetic activity of NP cells through factors secreted into conditioned medium [115, 116]. These findings support the notion that molecular agents secreted by NCs constitute a promising alternative for disc repair. In another approach, preconditioning of stem cells prior to injection was explored. Improved proteoglycan content and expression of IVD-specific markers were achieved by non-viral overexpression of GDF-5. The model did not demonstrate an increase in type II collagen but introduced a novel way of utilizing stem cells optimized for IVD repair [117]. Controlled interactions between stem cells and NP cells using bilaminar coculture pellets were developed as a cell-based therapy, adapting inductive mechanisms from development to trigger differentiation [118]. This 3D coculture system was found to stimulate greater MSC proliferation and differentiation, compared to single cell type or randomly mixed coculture controls [119]. Results of stem cell studies in IVD are developing and, if delivery obstacles can be overcome, may offer alternative future treatment strategies.
Hydrogels/biomaterials
Material science is developing scaffold materials, such as hydrogels and other biomaterials, for support of and delivery of cells for restoration of tissue function. Hydrogels are polymers with high water content and the ability to serve as a scaffold for cells, making them a promising choice for NP prostheses as they perform a similar biomechanical function, and can metabolically support endogenous NP cells. In contrast, repairing the AF necessitates restoration of circumferential integrity, preventing NP extrusion and interfacing with the cartilaginous end plates of the vertebrae. The duality of these tissues within the IVD necessitates development of prostheses that combine different biomechanical properties, and the application of appropriate mechanical influences allows for the optimization of function-specific tissue engineering [120].
Damage to the NP with loss of water content and disc height has been shown to compromise the IVD mechanical function. Range of motion, axial rotation, and compressive stiffness are all vulnerable to deterioration when the NP has been compromised. In fact, Reitmaier et al. demonstrated an AF injury without disruption of the NP does not exhibit these mechanical signs of degeneration [121]. Injection of hydrogels and biomimetic composites into the NP restores axial compressive stiffness when delivered directly into the disc via needle injection or when used to fill a defect created with a balloon catheter (modified kyphoplasty) [122]. However, the selection of the optimal prosthesis relies on both mechanical properties of the hydrogel and its ability to assimilate biologically [123]. A study of a hydrogel prosthesis that swells in situ upon delivery in a canine model showed improvement in flexion and extension and in disc height following replacement of injured IVDs with prostheses. In this work, Kranenburg et al. describes a model for designing a prosthetic NP with subsequent restoration of the biomechanical neutral zone, which has been clinically posited to correlate with reduction in pain [124]. An injectable prosthesis studied by Malhotra et al. was also able to correct changes in range of motion following replacement with an NP injection to an ovine model, but could not restore compressive stiffness without addressing the AF defect [125].
Most commonly, AF repair is performed by sutures or an increasing array of devices that insert through the defect and seal the AF to prevent NP herniation. Though sutures and implants are capable of preventing extrusion of the central tissue in some studies, Reitmaier et al. found repair strategy neither held the NP in place [121] nor restored biomechanical function of the AF [126]. The concept of an adhesive for repair injected to the IVD following restoration of the NP by stem cell or growth factor treatment is attractive because it may maximize therapeutic benefit and minimizing iatrogenic damage. One such adhesive, a fibrin-genipin hydrogel optimized for AF repair showed excellent success at sealing AF and preventing loss of disc height in response to experimental compression [127]. The biomechanical function of the repaired disc approached that of uninjured tissue and was not significantly different from intact tissue with regard to compressive stiffness. However, these findings were tempered by the histological observation of inflammatory cells and fibrosis, and better understanding of the immune response to such materials is required for clinical application [127]. Another approach to AF repair also utilizes hydrogel scaffolds with fiber reinforcement to improve cellular differentiation and fusion with cartilaginous end plate. The mechanisms behind these improvements are uncertain, and inflammatory response of these composites was not explored [128].
Summary
Low back pain occurs with many diseases and represents a tremendous burden to the health care system and economy. Understanding more about these underlying pathologies helps delineate differences between these disease processes and describe opportunities that may represent critical therapeutic targets. Inflammation is increasingly critical as both a cause and effect of IVD disease. Treatments that utilize inherent growth potential, through growth factors or stem cells, can stimulate tissue repair but may also provide benefit by mitigating inflammation. Understanding of IVD mechanics contributes an essential piece of the repair puzzle, as much of the damage in DD is associated with mechanical dysfunction of the IVD, and appropriate loading affects the growth and metabolism of IVD cells. Optimum management of LBP will continue to integrate new and refined concepts in pathophysiology, earlier detection of disease, and improved developments in tissue engineering.
With regard to the authors’ research cited in this paper, all institutional and national guidelines for the care and use of laboratory animals were followed.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of outstanding importance
ConnellyL.B, Woolf A, and Brooks P. Cost-effectiveness of interventions for musculoskeletal conditions. 2006.
Hoy D, Bain C, Williams G, March L, Brooks P, Blyth F, et al. A systematic review of the global prevalence of low back pain. Arthritis Rheum. 2012;64(6):2028–37.
Pleis JR, Ward BW, Lucas JW. Summary health statistics for U.S. adults: national health interview survey. Vital Health Stat. 2009;10(249):1–207.
Smitherman TA, Burch R, Sheikh H, Loder E. The prevalence, impact, and treatment of migraine and severe headaches in the United States: a review of statistics from national surveillance studies. Headache. 2013;53(3):427–36.
Dagenais S, Caro J, Haldeman S. A systematic review of low back pain cost of illness studies in the United States and internationally. Spine J. 2008;8(1):8–20.
Initiative, U.S.B.a.J. Health care utilization and economic cost of musculoskeletal disease, In The burden of musculoskeletal diseases in the United States, A.A.o.O. Surgeons, Editor. 2011.
Katz JN. Lumbar disc disorders and low-back pain: socioeconomic factors and consequences. J Bone Joint Surg Am. 2006;88 Suppl 2:21–4.
Deyo RA, Weinstein JN. Low back pain. N Engl J Med. 2001;344(5):363–70.
Adams MA, Roughley PJ. What is intervertebral disc degeneration, and what causes it? Spine (Phila Pa 1976). 2006;31(18):2151–61.
Boden SD, McCowin PR, Davis DO, Dina TS, Mark AS, Wiesel S. Abnormal magnetic-resonance scans of the cervical spine in asymptomatic subjects. A prospective investigation. J Bone Joint Surg Am. 1990;72(8):1178–84.
Jensen MC, Brant-Zawadzki MN, Obuchowski N, Modic MT, Malkasian D, Ross JS. Magnetic resonance imaging of the lumbar spine in people without back pain. N Engl J Med. 1994;331(2):69–73.
Takatalo J, Karppinen J, Niinimaki J, Taimela S, Nayha S, Mutanen P, et al. Does lumbar disc degeneration on magnetic resonance imaging associate with low back symptom severity in young Finnish adults? Spine (Phila Pa 1976). 2011;36(25):2180–9.
Mirza SK, Deyo RA. Systematic review of randomized trials comparing lumbar fusion surgery to nonoperative care for treatment of chronic back pain. Spine (Phila Pa 1976). 2007;32(7):816–23.
DeVine J, Norvell DC, Ecker E, Fourney DR, Vaccaro A, Wang J, et al. Evaluating the correlation and responsiveness of patient-reported pain with function and quality-of-life outcomes after spine surgery. Spine (Phila Pa 1976). 2011;36(21 Suppl):S69–74.
Antoniou J, Steffen T, Nelson F, Winterbottom N, Hollander AP, Poole RA, et al. The human lumbar intervertebral disc: evidence for changes in the biosynthesis and denaturation of the extracellular matrix with growth, maturation, ageing, and degeneration. J Clin Invest. 1996;98(4):996–1003.
Ohshima H, Tsuji H, Hirano N, Ishihara H, Katoh Y, Yamada H. Water diffusion pathway, swelling pressure, and biomechanical properties of the intervertebral disc during compression load. Spine (Phila Pa 1976). 1989;14(11):1234–44.
Mwale F, Iatridis JC, Antoniou J. Quantitative MRI as a diagnostic tool of intervertebral disc matrix composition and integrity. Eur Spine J. 2008;17 Suppl 4:432–40.
Iatridis JC, Nicoll SB, Michalek AJ, Walter BA, Gupta MS. Role of biomechanics in intervertebral disc degeneration and regenerative therapies: what needs repairing in the disc and what are promising biomaterials for its repair? Spine J. 2013;13(3):243–62.
Guinto Jr FC, Hashim H, Stumer M. CT demonstration of disk regression after conservative therapy. AJNR Am J Neuroradiol. 1984;5(5):632–3.
Keskil S, Ayberk G, Evliyaoglu C, Kizartici T, Yucel E, Anbarci H. Spontaneous resolution of “protruded” lumbar discs. Minim Invasive Neurosurg. 2004;47(4):226–9.
Hasue M, Fujiwara M. Epidemiologic and clinical studies of long-term prognosis of low-back pain and sciatica. Spine (Phila Pa 1976). 1979;4(2):150–5.
Komori H, Shinomiya K, Nakai O, Yamaura I, Takeda S, Furuya K. The natural history of herniated nucleus pulposus with radiculopathy. Spine (Phila Pa 1976). 1996;21(2):225–9.
Benyamin RM, Manchikanti L, Parr AT, Diwan S, Singh V, Falco FJ, et al. The effectiveness of lumbar interlaminar epidural injections in managing chronic low back and lower extremity pain. Pain Physician. 2012;15(4):E363–404.
Manchikanti L, Buenaventura RM, Manchikanti KN, Ruan X, Gupta S, Smith HS, et al. Effectiveness of therapeutic lumbar transforaminal epidural steroid injections in managing lumbar spinal pain. Pain Physician. 2012;15(3):E199–245.
Friedly JL, Comstock BA, Turner JA, Heagerty PJ, Deyo RA, Sullivan SD, et al. A randomized trial of epidural glucocorticoid injections for spinal stenosis. N Engl J Med. 2014;371(1):11–21.
Benyamin RM, Manchikanti L, Parr AT, Diwan S, Singh V, Falco FJ, et al. The effectiveness of lumbar interlaminar epidural injections in managing chronic low back and lower extremity pain. Pain Physician. 2012;15(4):E363–404.
Gibson JN, Waddell G. Surgical interventions for lumbar disc prolapse. Cochrane Database Syst Rev. 2007;2:CD001350.
Baskin DS, Ryan P, Sonntag V, Westmark R, Widmayer MA. A prospective, randomized, controlled cervical fusion study using recombinant human bone morphogenetic protein-2 with the CORNERSTONE-SR allograft ring and the ATLANTIS anterior cervical plate. Spine (Phila Pa 1976). 2003;28(12):1219–24. discussion 1225.
Burkus JK, Transfeldt EE, Kitchel SH, Watkins RG, Balderston RA. Clinical and radiographic outcomes of anterior lumbar interbody fusion using recombinant human bone morphogenetic protein-2. Spine (Phila Pa 1976). 2002;27(21):2396–408.
Fu R, Selph S, McDonagh M, Peterson K, Tiwari A, Chou R, et al. Effectiveness and harms of recombinant human bone morphogenetic protein-2 in spine fusion: a systematic review and meta-analysis. Ann Intern Med. 2013;158(12):890–902.
Simmonds MC, Brown JV, Heirs MK, Higgins JP, Mannion RJ, Rodgers MA, et al. Safety and effectiveness of recombinant human bone morphogenetic protein-2 for spinal fusion: a meta-analysis of individual-participant data. Ann Intern Med. 2013;158(12):877–89.
Walker B, Koerner J, Sankarayanaryanan S, and Radcliff K, A consensus statement regarding the utilization of BMP in spine surgery. Curr Rev Musculoskelet Med.
Risbud MV, Shapiro IM. Role of cytokines in intervertebral disc degeneration: pain and disc content. Nat Rev Rheumatol. 2013;10(1):44–56. This paper reviews the contribution of cytokines and immune cells to catabolic, angiogenic and nociceptive processes in disc diseases.
Tracey KJ. Physiology and immunology of the cholinergic antiinflammatory pathway. J Clin Invest. 2007;117(2):289–96.
Rajan NE, Bloom O, Maidhof R, Stetson N, Sherry B, Levine M, et al. Toll-like receptor 4 (TLR4) expression and stimulation in a model of intervertebral disc inflammation and degeneration. Spine (Phila Pa 1976). 2012;38(16):1343–51.
Klawitter M, Hakozaki M, Kobayashi H, Krupkova O, Quero L, Ospelt C, Gay S, Hausmann O, Liebscher T, Meier U, Sekiguchi M, Konno SI, Boos N, Ferguson SJ, and Wuertz . Expression and regulation of Toll-like receptors (TLRs) in human intervertebral disc cells. Eur Spine J.
Capossela S, Schlafli P, Bertolo A, Janner T, Stadler BM, Potzel T, et al. Degenerated human intervertebral discs contain autoantibodies against extracellular matrix proteins. Eur Cell Mater. 2014;27:251–63. discussion 263.
Gruber HE, Hoelscher GL, Ingram JA, Bethea S, Norton HJ, Hanley Jr EN. Production and expression of RANTES (CCL5) by human disc cells and modulation by IL-1-beta and TNF-alpha in 3D culture. Exp Mol Pathol. 2014;96(2):133–8.
Le Maitre CL, Freemont AJ, Hoyland JA. Expression of cartilage-derived morphogenetic protein in human intervertebral discs and its effect on matrix synthesis in degenerate human nucleus pulposus cells. Arthritis Res Ther. 2009;11(5):R137.
Walsh AJ, Bradford DS, Lotz JC. In vivo growth factor treatment of degenerated intervertebral discs. Spine (Phila Pa 1976). 2004;29(2):156–63.
Gruber HE, Hoelscher GL, Ingram JA, Bethea S, Hanley Jr EN. Growth and differentiation factor-5 (GDF-5) in the human intervertebral annulus cells and its modulation by IL-1ss and TNF-alpha in vitro. Exp Mol Pathol. 2014;96(2):225–9.
Bachmeier BE, Nerlich A, Mittermaier N, Weiler C, Lumenta C, Wuertz K, et al. Matrix metalloproteinase expression levels suggest distinct enzyme roles during lumbar disc herniation and degeneration. Eur Spine J. 2009;18(11):1573–86.
Zigouris A, Batistatou A, Alexiou GA, Pachatouridis D, Mihos E, Drosos D, et al. Correlation of matrix metalloproteinases-1 and -3 with patient age and grade of lumbar disc herniation. J Neurosurg Spine. 2011;14(2):268–72.
Canbay S, Turhan N, Bozkurt M, Arda K, Caglar S. Correlation of matrix metalloproteinase-3 expression with patient age, magnetic resonance imaging and histopathological grade in lumbar disc degeneration. Turk Neurosurg. 2013;23(4):427–33.
Rastogi A, Kim H, Twomey JD, Hsieh AH. MMP-2 mediates local degradation and remodeling of collagen by annulus fibrosus cells of the intervertebral disc. Arthritis Res Ther. 2013;15(2):R57.
Furtwangler T, Chan SC, Bahrenberg G, Richards PJ, Gantenbein-Ritter B. Assessment of the matrix degenerative effects of MMP-3, ADAMTS-4, and HTRA1, injected into a bovine intervertebral disc organ culture model. Spine (Phila Pa 1976). 2013;38(22):E1377–87.
Albert HB, Lambert P, Rollason J, Sorensen JS, Worthington T, Pedersen MB, et al. Does nuclear tissue infected with bacteria following disc herniations lead to Modic changes in the adjacent vertebrae? Eur Spine J. 2013;22(4):690–6.
Modic MT, Steinberg PM, Ross JS, Masaryk TJ, Carter JR. Degenerative disk disease: assessment of changes in vertebral body marrow with MR imaging. Radiology. 1988;166(1 Pt 1):193–9.
Jensen TS, Karppinen J, Sorensen JS, Niinimaki J, Leboeuf-Yde C. Vertebral endplate signal changes (Modic change): a systematic literature review of prevalence and association with non-specific low back pain. Eur Spine J. 2008;17(11):1407–22.
Bekyarova GY, Ivanova DG, Madjova VH. Molecular mechanisms associating oxidative stress with endothelial dysfunction in the development of various vascular complications in diabetes mellitus. Folia Med (Plovdiv). 2007;49(3–4):13–9.
Matsuda M, Shimomura I. Increased oxidative stress in obesity: implications for metabolic syndrome, diabetes, hypertension, dyslipidemia, atherosclerosis, and cancer. Obes Res Clin Pract. 2013;7(5):e330–41.
Tsai TT, Ho NY, Lin YT, Lai PL, Fu TS, Niu CC, et al. Advanced glycation end products in degenerative nucleus pulposus with diabetes. J Orthop Res. 2013;32(2):238–44.
Garg A, Hegmann KT, Moore JS, Kapellusch J, Thiese MS, Boda S, et al. Study protocol title: a prospective cohort study of low back pain. BMC Musculoskelet Disord. 2013;14:84.
Illien-Junger S, Grosjean F, Laudier DM, Vlassara H, Striker GE, Iatridis JC. Combined anti-inflammatory and anti-AGE drug treatments have a protective effect on intervertebral discs in mice with diabetes. PLoS One. 2013;8(5):e64302.
Ohtori S, Miyagi M, Eguchi Y, Inoue G, Orita S, Ochiai N, et al. Epidural administration of spinal nerves with the tumor necrosis factor-alpha inhibitor, etanercept, compared with dexamethasone for treatment of sciatica in patients with lumbar spinal stenosis: a prospective randomized study. Spine (Phila Pa 1976). 2012;37(6):439–44.
Cohen SP, Bogduk N, Dragovich A, Buckenmaier 3rd CC, Griffith S, Kurihara C, et al. Randomized, double-blind, placebo-controlled, dose-response, and preclinical safety study of transforaminal epidural etanercept for the treatment of sciatica. Anesthesiology. 2009;110(5):1116–26.
Okoro T, Tafazal SI, Longworth S, Sell PJ. Tumor necrosis alpha-blocking agent (etanercept): a triple blind randomized controlled trial of its use in treatment of sciatica. J Spinal Disord Tech. 2010;23(1):74–7.
Cohen SP, White RL, Kurihara C, Larkin TM, Chang A, Griffith SR, et al. Epidural steroids, etanercept, or saline in subacute sciatica: a multicenter, randomized trial. Ann Intern Med. 2012;156(8):551–9.
Genevay S, Stingelin S, Gabay C. Efficacy of etanercept in the treatment of acute, severe sciatica: a pilot study. Ann Rheum Dis. 2004;63(9):1120–3.
Genevay S, Finckh A, Zufferey P, Viatte S, Balague F, Gabay C. Adalimumab in acute sciatica reduces the long-term need for surgery: a 3-year follow-up of a randomised double-blind placebo-controlled trial. Ann Rheum Dis. 2012;71(4):560–2.
Genevay S, Viatte S, Finckh A, Zufferey P, Balague F, Gabay C. Adalimumab in severe and acute sciatica: a multicenter, randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 2010;62(8):2339–46.
Korhonen T, Karppinen J, Paimela L, Malmivaara A, Lindgren KA, Bowman C, et al. The treatment of disc-herniation-induced sciatica with infliximab: one-year follow-up results of FIRST II, a randomized controlled trial. Spine (Phila Pa 1976). 2006;31(24):2759–66.
Williams NH, Lewis R, Din NU, Matar HE, Fitzsimmons D, Phillips CJ, et al. A systematic review and meta-analysis of biological treatments targeting tumour necrosis factor alpha for sciatica. Eur Spine J. 2013;22(9):1921–35.
Jacobs L, Vo N, Coehlo JP, Dong Q, Bechara B, Woods B, Hempen E, Hartman R, Preuss H, Balk J, Kang J, and Sowa G. Glucosamine supplementation demonstrates a negative effect on intervertebral disc matrix in an animal model of disc degeneration. Spine (Phila Pa 1976), 2013.
Sowa GA, Coelho JP, Jacobs LJ, Komperda K, Sherry N, Vo NV, Preuss HG, Balk JL, and Kang JD. The effects of glucosamine sulfate on intervertebral disc annulus fibrosus cells in vitro. Spine J. 2013
Cortes DH, Jacobs NT, DeLucca JF, Elliott DM. Elastic, permeability and swelling properties of human intervertebral disc tissues: a benchmark for tissue engineering. J Biomech. 2014;47(9):2088–94.
Huang J, Yan H, Jian F, Wang X, and Li H. Numerical analysis of the influence of nucleus pulposus removal on the biomechanical behavior of a lumbar motion segment. Comput Methods Biomech Biomed Engin: p. 1–9.
Antoniou J, Epure LM, Michalek AJ, Grant MP, Iatridis JC, Mwale F. Analysis of quantitative magnetic resonance imaging and biomechanical parameters on human discs with different grades of degeneration. J Magn Reson Imaging. 2013;38(6):1402–14.
Ellingson AM, Mehta H, Polly DW, Ellermann J, Nuckley DJ. Disc degeneration assessed by quantitative T2* (T2 star) correlated with functional lumbar mechanics. Spine (Phila Pa 1976). 2013;38(24):E1533–40.
Cortes DH, Magland JF, Wright AC, Elliott DM. The shear modulus of the nucleus pulposus measured using magnetic resonance elastography: a potential biomarker for intervertebral disc degeneration. Magn Reson Med. 2014;72(1):211–9. This paper demonstrates a new imaging tool for disc diagnostics using magnetic resonance elastography.
Jackman TM, Hussein AI, Adams AM, Makhnejia KK, Morgan EF. Endplate deflection is a defining feature of vertebral fracture and is associated with properties of the underlying trabecular bone. J Orthop Res. 2014;32(7):880–6.
Yamaguchi T, Inoue N, Sah RL, Lee YP, Taborek AP, Williams GM, et al. Micro-computed tomography-based three-dimensional kinematic analysis during lateral bending for spinal fusion assessment in a rat posterolateral lumbar fusion model. Tissue Eng Part C Methods. 2014;20(7):578–87.
Jaumard NV, Welch WC, Winkelstein BA. Spinal facet joint biomechanics and mechanotransduction in normal, injury and degenerative conditions. J Biomech Eng. 2011;133(7):071010.
Igarashi A, Kikuchi S, Konno S. Correlation between inflammatory cytokines released from the lumbar facet joint tissue and symptoms in degenerative lumbar spinal disorders. J Orthop Sci. 2007;12(2):154–60.
Alipui DO, Chen D, Houseman C, Stetson N, Black K, Overby C, Levine M, and Chahine N. Molecular Profiles of Degenerative Biomarkers in the ligamentum flavum and their relationships with disc disease severity. In Transactions of the Orthopedics Research Society. 2014. New Orleans, LA.
Dudli S, Ferguson SJ, Haschtmann D. Severity and pattern of post-traumatic intervertebral disc degeneration depend on the type of injury. Spine J. 2014;14(7):1256–64.
Dudli S, Haschtmann D, Ferguson SJ. Fracture of the vertebral endplates, but not equienergetic impact load, promotes disc degeneration in vitro. J Orthop Res. 2012;30(5):809–16.
Gregory DE, Bae WC, Sah RL, Masuda K. Disc degeneration reduces the delamination strength of the annulus fibrosus in the rabbit annular disc puncture model. Spine J. 2014;14(7):1265–71.
Neidlinger-Wilke C, Boldt A, Brochhausen C, Galbusera F, Carstens C, Copf F, et al. Molecular interactions between human cartilaginous endplates and nucleus pulposus cells: a preliminary investigation. Spine (Phila Pa 1976). 2014;39(17):1355–64.
Fernando HN, Czamanski J, Yuan TY, Gu W, Salahadin A, Huang CY. Mechanical loading affects the energy metabolism of intervertebral disc cells. J Orthop Res. 2011;29(11):1634–41.
Salvatierra JC, Yuan TY, Fernando H, Castillo A, Gu WY, Cheung HS, et al. Difference in energy metabolism of annulus fibrosus and nucleus pulposus cells of the intervertebral disc. Cell Mol Bioeng. 2011;4(2):302–10.
Li S, Jia X, Duance VC, Blain EJ. The effects of cyclic tensile strain on the organisation and expression of cytoskeletal elements in bovine intervertebral disc cells: an in vitro study. Eur Cell Mater. 2011;21:508–22.
Chan SC, Walser J, Kappeli P, Shamsollahi MJ, Ferguson SJ, Gantenbein-Ritter B. Region specific response of intervertebral disc cells to complex dynamic loading: an organ culture study using a dynamic torsion-compression bioreactor. PLoS One. 2013;8(8):e72489.
Cho H, Seth A, Warmbold J, Robertson JT, Hasty KA. Aging affects response to cyclic tensile stretch: paradigm for intervertebral disc degeneration. Eur Cell Mater. 2011;22:137–45. discussion 145–6.
Maidhof R, Jacobsen T, Papatheodorou A, Chahine NO. Inflammation induces irreversible biophysical changes in isolated nucleus pulposus cells. PLoS One. 2014;9(6):e99621.
Hwang PY, Chen J, Jing L, Hoffman BD, Setton LA. The role of extracellular matrix elasticity and composition in regulating the nucleus pulposus cell phenotype in the intervertebral disc: a narrative review. J Biomech Eng. 2014;136(2):021010. This paper reviews the complex mechanobiology of the disc, highlighting the effects of cell-matrix interactions and susbtrate stiffness in the cellular microenvironment.
de Souza Grava AL, Ferrari LF, Defino HL. Cytokine inhibition and time-related influence of inflammatory stimuli on the hyperalgesia induced by the nucleus pulposus. Eur Spine J. 2012;21(3):537–45.
Kartha S, Zeeman ME, Baig HA, Guarino BB, and Winkelstein BA. Upregulation of BDNF & NGF in cervical intervertebral discs exposed to painful whole body vibration. Spine (Phila Pa 1976).
Moon HJ, Yurube T, Lozito TP, Pohl P, Hartman RA, Sowa GA, et al. Effects of secreted factors in culture medium of annulus fibrosus cells on microvascular endothelial cells: elucidating the possible pathomechanisms of matrix degradation and nerve in-growth in disc degeneration. Osteoarthritis Cartilage. 2014;22(2):344–54.
Gao R, Brigstock DR. Connective tissue growth factor (CCN2) induces adhesion of rat activated hepatic stellate cells by binding of its C-terminal domain to integrin alpha (v) beta (3) and heparan sulfate proteoglycan. J Biol Chem. 2004;279(10):8848–55.
Tran CM, Markova D, Smith HE, Susarla B, Ponnappan RK, Anderson DG, et al. Regulation of CCN2/connective tissue growth factor expression in the nucleus pulposus of the intervertebral disc: role of Smad and activator protein 1 signaling. Arthritis Rheum. 2010;62(7):1983–92.
Tran CM, Schoepflin ZR, Markova DZ, Kepler CK, Anderson DG, Shapiro IM, et al. CCN2 suppresses catabolic effects of interleukin-1beta through alpha5beta1 and alphaVbeta3 integrins in nucleus pulposus cells: implications in intervertebral disc degeneration. J Biol Chem. 2014;289(11):7374–87.
Bendinelli P, Matteucci E, Dogliotti G, Corsi MM, Banfi G, Maroni P, et al. Molecular basis of anti-inflammatory action of platelet-rich plasma on human chondrocytes: mechanisms of NF-kappaB inhibition via HGF. J Cell Physiol. 2010;225(3):757–66.
van Buul GM, Koevoet WL, Kops N, Bos PK, Verhaar JA, Weinans H, et al. Platelet-rich plasma releasate inhibits inflammatory processes in osteoarthritic chondrocytes. Am J Sports Med. 2011;39(11):2362–70.
Kim HJ, Yeom JS, Koh YG, Yeo JE, Kang KT, Kang YM, et al. Anti-inflammatory effect of platelet-rich plasma on nucleus pulposus cells with response of TNF-alpha and IL-1. J Orthop Res. 2014;32(4):551–6.
Haschtmann D, Ferguson SJ, Stoyanov JV. BMP-2 and TGF-beta3 do not prevent spontaneous degeneration in rabbit disc explants but induce ossification of the annulus fibrosus. Eur Spine J. 2012;21(9):1724–33.
Osada R, Ohshima H, Ishihara H, Yudoh K, Sakai K, Matsui H, et al. Autocrine/paracrine mechanism of insulin-like growth factor-1 secretion, and the effect of insulin-like growth factor-1 on proteoglycan synthesis in bovine intervertebral discs. J Orthop Res. 1996;14(5):690–9.
Huang CY, Travascio F, Gu WY. Quantitative analysis of exogenous IGF-1 administration of intervertebral disc through intradiscal injection. J Biomech. 2012;45(7):1149–55.
Aoyama E, Hattori T, Hoshijima M, Araki D, Nishida T, Kubota S, et al. N-terminal domains of CCN family 2/connective tissue growth factor bind to aggrecan. Biochem J. 2009;420(3):413–20.
Madiraju P, Gawri R, Wang H, Antoniou J, Mwale F. Mechanism of parathyroid hormone-mediated suppression of calcification markers in human intervertebral disc cells. Eur Cell Mater. 2013;25:268–83.
Lee CS, Szczesny SE, Soslowsky LJ. Remodeling and repair of orthopedic tissue: role of mechanical loading and biologics: part II: cartilage and bone. Am J Orthop (Belle Mead NJ). 2011;40(3):122–8.
Mwale F, Roughley P, Antoniou J. Distinction between the extracellular matrix of the nucleus pulposus and hyaline cartilage: a requisite for tissue engineering of intervertebral disc. Eur Cell Mater. 2004;8:58–63. discussion 63–4.
Shen CL, Dunn DM, Henry JH, Li Y, Watkins BA. Decreased production of inflammatory mediators in human osteoarthritic chondrocytes by conjugated linoleic acids. Lipids. 2004;39(2):161–6.
Byers BA, Mauck RL, Chiang IE, Tuan RS. Transient exposure to transforming growth factor beta 3 under serum-free conditions enhances the biomechanical and biochemical maturation of tissue-engineered cartilage. Tissue Eng Part A. 2008;14(11):1821–34.
Lima EG, Bian L, Ng KW, Mauck RL, Byers BA, Tuan RS, et al. The beneficial effect of delayed compressive loading on tissue-engineered cartilage constructs cultured with TGF-beta3. Osteoarthritis Cartilage. 2007;15(9):1025–33.
Reza AT, Nicoll SB. Serum-free, chemically defined medium with TGF-beta (3) enhances functional properties of nucleus pulposus cell-laden carboxymethylcellulose hydrogel constructs. Biotechnol Bioeng. 2010;105(2):384–95.
Smith LJ, Chiaro JA, Nerurkar NL, Cortes DH, Horava SD, Hebela NM, et al. Nucleus pulposus cells synthesize a functional extracellular matrix and respond to inflammatory cytokine challenge following long-term agarose culture. Eur Cell Mater. 2011;22:291–301.
Smith AN, Muffley LA, Bell AN, Numhom S, Hocking AM. Unsaturated fatty acids induce mesenchymal stem cells to increase secretion of angiogenic mediators. J Cell Physiol. 2012;227(9):3225–33.
Kuelling FA, Foley KT, Liu JJ, Liebenberg E, Sin AH, Matsukawa A, et al. The anabolic effect of plasma-mediated ablation on the intervertebral disc: stimulation of proteoglycan and interleukin-8 production. Spine J. 2014;14(10):2479–87.
Chan SC, Burki A, Bonel HM, Benneker LM, Gantenbein-Ritter B. Papain-induced in vitro disc degeneration model for the study of injectable nucleus pulposus therapy. Spine J. 2013;13(3):273–83.
Tam V, Rogers I, Chan D, Leung VY, Cheung KM. A comparison of intravenous and intradiscal delivery of multipotential stem cells on the healing of injured intervertebral disk. J Orthop Res. 2014;32(6):819–25.
Leckie SK, Sowa GA, Bechara BP, Hartman RA, Coelho JP, Witt WT, et al. Injection of human umbilical tissue-derived cells into the nucleus pulposus alters the course of intervertebral disc degeneration in vivo. Spine J. 2013;13(3):263–72.
Chen J, Lee EJ, Jing L, Christoforou N, Leong KW, Setton LA. Differentiation of mouse induced pluripotent stem cells (iPSCs) into nucleus pulposus-like cells in vitro. PLoS One. 2013;8(9):e75548.
Liu Y, Rahaman MN, Bal BS. Modulating notochordal differentiation of human induced pluripotent stem cells using natural nucleus pulposus tissue matrix. PLoS One. 2014;9(7):e100885.
Potier EK. Ito, Can notochordal cells promote bone marrow stromal cell potential for nucleus pulposus enrichment? A simplified in vitro system. Tissue Eng Part A.
Purmessur D, Cornejo MC, Cho SK, Hecht AC, Iatridis JC. Notochordal cell-derived therapeutic strategies for discogenic back pain. Glob Spine J. 2013;3(3):201–18.
Bucher C, Gazdhar A, Benneker LM, Geiser T, and Gantenbein-Ritter B. Nonviral gene delivery of growth and differentiation factor 5 to human mesenchymal stem cells injected into a 3D bovine intervertebral disc organ culture system. Stem Cells Int. 2013: p. 326828.
Allon AA, Butcher K, Schneider RA, Lotz JC. Structured bilaminar coculture outperforms stem cells and disc cells in a simulated degenerate disc environment. Spine (Phila Pa 1976). 2012;37(10):813–8.
Allon AA, Butcher K, Schneider RA, Lotz JC. Structured coculture of mesenchymal stem cells and disc cells enhances differentiation and proliferation. Cells Tissues Organs. 2012;196(2):99–106.
MacBarb RF, Chen AL, Hu JC, Athanasiou KA. Engineering functional anisotropy in fibrocartilage neotissues. Biomaterials. 2013;34(38):9980–9.
Reitmaier S, Shirazi-Adl A, Bashkuev M, Wilke HJ, Gloria A, Schmidt H. In vitro and in silico investigations of disc nucleus replacement. J R Soc Interface. 2012;9(73):1869–79.
Leckie AE, Akens MK, Woodhouse KA, Yee AJ, Whyne CM. Evaluation of thiol-modified hyaluronan and elastin-like polypeptide composite augmentation in early-stage disc degeneration: comparing 2 minimally invasive techniques. Spine (Phila Pa 1976). 2012;37(20):E1296–303.
Reitmaier S, Wolfram U, Ignatius A, Wilke HJ, Gloria A, Martin-Martinez JM, et al. Hydrogels for nucleus replacement—facing the biomechanical challenge. J Mech Behav Biomed Mater. 2012;14:67–77.
Kranenburg HJ, Meij BP, Onis D, van der Veen AJ, Saralidze K, Smolders LA, et al. Design, synthesis, imaging, and biomechanics of a softness-gradient hydrogel nucleus pulposus prosthesis in a canine lumbar spine model. J Biomed Mater Res B Appl Biomater. 2012;100(8):2148–55.
Malhotra NR, Han WM, Beckstein J, Cloyd J, Chen W, Elliott DM. An injectable nucleus pulposus implant restores compressive range of motion in the ovine disc. Spine (Phila Pa 1976). 2012;37(18):E1099–105.
Ahlgren BD, Lui W, Herkowitz HN, Panjabi MM, Guiboux JP. Effect of anular repair on the healing strength of the intervertebral disc: a sheep model. Spine (Phila Pa 1976). 2000;25(17):2165–70.
Likhitpanichkul M, Dreischarf M, Illien-Junger S, Walter BA, Nukaga T, Long RG, et al. Fibrin-genipin adhesive hydrogel for annulus fibrosus repair: performance evaluation with large animal organ culture, in situ biomechanics, and in vivo degradation tests. Eur Cell Mater. 2014;28:25–38.
Pei BQ, Li H, Zhu G, Li DY, Fan YB, and Wu SQ. The application of fiber-reinforced materials in disc repair. Biomed Res Int. 2013: p. 714103.
Compliance with Ethics Guidelines
Conflict of Interest
Kathryn T. Weber, Timothy D. Jacobsen, Robert Maidhof, Justin Virojanapa, Chris Overby, Ona Bloom, Shaheda Quraishi, and Mitchell Levine declare that they have no conflict of interest.
Nadeen Chahine reports grants from National Science Foundation (NSF CAREER 1151605), grants from New York State Department of Health (Empire Clinical Research Investigator Program), grants from National Institute of Health (R41AG050021), and grants from American Orthopedic Society for Sports Medicine, during the conduct of the study.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Biological Adjuvants in Orthopedic Surgery
Rights and permissions
About this article
Cite this article
Weber, K.T., Jacobsen, T.D., Maidhof, R. et al. Developments in intervertebral disc disease research: pathophysiology, mechanobiology, and therapeutics. Curr Rev Musculoskelet Med 8, 18–31 (2015). https://doi.org/10.1007/s12178-014-9253-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12178-014-9253-8