Abstract
Foodborne illnesses caused by Escherichia coli (E. coli) are causing worldwide morbidity and mortality, threating human health. One way to limit and eliminate E. coli infections is developing fast and sensitive methods for detection. Biosensors have emerged to complement traditional strategies for E. coli detection. This review aims at providing an updated summary of developed biosensors for specific detection of E. coli in the last 5 years. Since biosensor development first initiated with immobilizing biorecognition element on the transducer, this review starts with a brief discussion on the immobilization methods of biorecognition elements on the biosensor surface, followed by a detailed discussion focused on the progresses made in principles and applications of various biorecognition element-based biosensors for E. coli detection. Especially, the limitations of each method using various biorecognition elements are discussed. Furthermore, the advancements of paper-based biosensors are generalized. In addition, a few suggestions and challenges for prospective development of biosensor are also provided.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Food safety has attracted increasing attention globally. Foodborne illnesses caused by microorganisms are causing worldwide morbidity and mortality, threating human health. Escherichia coli (E. coli) is one type of normal resident bacteria in the intestines of animals, some special serotypes of which cause disease under certain conditions via specific pili antigen, pathogenic toxin, and other infections. These diseases include human body or animal gastrointestinal infection, urinary tract infection, arthritis, meningitis, and septic infection (Zhao et al. 2018). The Centers for Disease Control and Prevention (CDC) have listed the multistate foodborne outbreak caused by E. coli group, e.g., as of April 22, 2020, 51 people were infected with E. coli O103 (CDC, in 2020). Therefore, reducing the harm of E. coli to human health is extremely important and urgent. One way to limit E. coli infections is developing fast and sensitive sensing methods for their detection. The majority of traditional methods for E. coli detection mainly focused on culture-based method, enzyme-linked immune-sorbent assay (ELISA), and polymerase chain reaction (PCR). However, the obvious drawbacks of time-consuming culture-dependent methods, requirement of complicated DNA extraction procedure for PCR, and insensitivity for ELISA are the barriers hampering the application of these methods (Zheng et al. 2019). Therefore, the development of a simple, sensitive, rapid, and portable detection strategy for E. coli is crucial to ensure food safety.
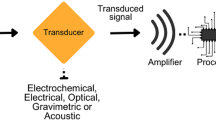
Over the past few years, biosensors have emerged to complement traditional methods for E. coli detection. A biosensor is an independent and integrated device that can provide specific quantitative or semi-quantitative analytical information (Maas et al. 2017). Biosensors are generally made up of three components: a biorecognition element grafted on the biosensor surface for specific recognition of targets, a transducer for transformation of recognition events to physically quantifiable signal, and a detector for signal amplification and collection (Templier et al. 2016). A biosensor definitely contains a biorecognition element directly fixed on a transduction element (Thévenot et al. 2001), which can be incorporated into portable device system, making it as an alternative platform to PCR and ELISA to identify and quantify E. coli. In most cases, biosensors should be able to detect pathogens with high sensitivity and selectivity without sample preparation in real time at various environments including food samples, body fluids, water, and object surfaces (Cesewski and Johnson 2020). Attributing to the use of selected biorecognition elements, biosensors are often highly specific and are combined with advanced technologies (such as nanotechnology and biotechnology) to produce highly robust and portable devices.
Biorecognition elements of E. coli usually include DNA, antibodies, aptamers, antimicrobial peptides, and phages. Reasonable DNA sequences can be designed as biorecognition elements to specifically identify genes extracted from E. coli. Nucleic acid amplification technologies (Miao et al. 2015; Zhao et al. 2015) have led to extremely sensitive gene sensor; however, the sample processing procedure is time-consuming and laborious. Antibodies, divided into monoclonal and polyclonal antibodies, are a class of immunoglobulin that can bind specifically to antigens (Templier et al. 2016; Banerjee and Jaiswal 2018). What is noteworthy is that polyclonal antibodies are less specific than monoclonal antibodies (Amiri et al. 2018). Artificially selected aptamers, which can selectively bind with a variety of target substances with high specificity, granting the extensive use in the field of biosensors, is an outstanding alternative to antibodies (Davydova et al. 2016). Antimicrobial peptides, which can also be synthesized artificially, are less specific in recognition than the aforementioned recognition elements due to their broad-spectrum antimicrobial properties (Pardoux et al. 2020). Phages can tolerate extreme environmental conditions, endowing the important advantage for developing fast and fairly inexpensive biosensors for E. coli detection (Amiri et al. 2018).
E. coli biosensors have been designed with various types of transducers including electrochemical, mechanical, magnetic, and optical transducers (Cesewski and Johnson 2020). Among them, electrochemical biosensors are the most widely used for E. coli detection. The powerful frequency function for measuring dielectric properties by electrochemical impedance spectroscopy (EIS) was well received for possible eliminating target labeling procedures and reducing costs (Siavash Moakhar et al. 2020). Other biosensors basically need to be labeled, and the labeling molecules (or reporters) are usually organic molecules, enzymes, or nanomaterials (Burlage and Tillmann 2017). Some of these labeling molecules especially enzymes (including deoxyribozymes) and nanoparticles are often amplifiers with the power of signal amplification, and their application can improve the sensitivity of biosensors (Sharifi et al. 2020; Rajapaksha et al. 2019).
The possible construction of flexible, inexpensive, and disposable devices has made biosensors very attractive for E. coli detection. Benefiting from the development of nanomaterials and composite materials with excellent features, the improvements in specificity and sensitivity of biosensors for E. coli detection have been extensively reported. This review aims at providing an updated summary of developed biosensors for E. coli detection in the last 5 years. Since biorecognition element fixed on the transducer is the first procedure for the biosensor development, this review starts with a brief discussion on the immobilization methods of biorecognition elements on the biosensor surface. Then, a detailed discussion focuses on the progress made in principles and applications of various biorecognition element-based biosensors for E. coli detection. In addition, the limitations of each method are discussed at the end. Furthermore, as paper-based biosensor is suitable for point-of-care testing (POCT) system without the need for sophisticated lab facility, the advancements of paper-based biosensors will be generalized. In addition, this review puts forth a few suggestions and challenges for prospective development.
Immobilization of Biorecognition Elements
The development of biosensor started with fixing biorecognition element on the biosensor’s surface. Therefore, summarizing the ways in which recognition elements are immobilized on the surface is necessary. Several ways to immobilize biorecognition elements onto biosensor’s surface are outlined as follows.
Nonspecific Adsorption
Adsorption in a completely random way is the easiest manner to immobilize E. coli biorecognition element to the biosensor surface. Recognition molecules, especially antibodies, favor direct nonspecific adsorption. Nonspecific adsorption disorients the recognition molecules at the biosensor surface, disrupting the contact of active centers with the E. coli surface. Disorientation also partially denatures and loses their function, compromising the sensitivity of biosensors (Luo et al. 2019).
Specific Interaction
Specific interaction is mainly based on three models: biotin-avidin, protein A/G-immunoglobulin G (IgG), and DNA hybridization. Biotin is usually modified to the terminals or side-chain groups of the recognition molecules (such as protein, peptide, and DNA), and each avidin can combine with four molecules of biotin, which can be used to construct a multi-level signal amplification system (Choiniere et al. 2019). Protein A/G can specifically bind the Fc region of IgG. Each protein A or G indeed binds four or two IgG (Amiri et al. 2018). Therefore, the orientational immobilization of the antibodies can be realized by pre-encapsulating protein A/G to improve the detection sensitivity. In addition, DNA molecules with complementary sequences can form stable DNA duplexes by forming hydrogen bonds between the base pairs.
Covalent Linking
Covalent linking of biorecognition elements onto the surface is the extensively utilized approach to fasten biorecognition element to the biosensor surface. The most popular covalent bonding methods include the conjugation of amino and carboxyl groups using 1-ethyl-3-[3-dimethylaminopropyl]carbodiimide hydrochloride (EDC)/N-hydroxysuccinimide (NHS) peptide coupling reagent and activated ester (Zhang et al. 2016). The covalent interaction between azide group and acetylene group forming triazole is also an alternative. Recognition element functionalized with thiol can be assembled on gold electrode surface by forming via SH-Au bond (Castillo-Torres et al. 2019).
Imprinting
Molecular imprinting technology (MIT) is a method that uses molecular imprinting polymers (MIPs) to simulate the interaction between enzyme/substrate and antibody/antigen and specifically recognize template molecules. MIPs possess abundant tailor-made binding sites, which are complementary to the corresponding template targets in functional groups, size, and shape (Ding et al. 2020).
Integrated in Composites
Monoclonal antibodies against E. coli O157:H7, enzymes, and other inorganic substances are directly formed into immune invertase-nanoclusters (Huang et al. 2018). However, this practice results in many unknown properties of the antibodies such as the orientation, activity, and depth.
Biosensors for Escherichia coli
Various biorecognition elements of E. coli such as antibodies, aptamers, antimicrobial peptides (AMPs), and genes have been used to develop biosensors. In addition, there are only two types of targets for E. coli biosensors: whole cells and cellular components (or metabolites). Researchers are more likely to develop biosensors that detect whole cells, because preprocessing is relatively uncomplicated. Whole cells biosensors include imprinted polymer-based biosensors, AMP-based biosensors, most immuno-biosensor, and aptamer-based biosensors. In addition, the primary target of cellular components is the gene of E. coli. Preprocessing such as bacterial isolation, gene extraction, and amplification is prerequisite (Chen et al. 2020). The principles, applications, and limitations of various biorecognition element-based biosensors are discussed in detail in the following sections.
Immuno-biosensor
Immuno-biosensor is an antibodies-based biosensor that can specifically recognize the target. Most sensing approaches are currently based on immunological detection principles. Antibodies, or immunoglobulins, are protective of “Y shape” proteins, which are produced as a result of the stimulation of an antigen. Antibodies possess two distinct regions, the antigen-binding region (Fab fragment) and the crystallizable region (Fc fragment), which can recognize/binding to antigenic agents by epitope and activate other immune system partners, respectively (Templier et al. 2016). Therefore, antibodies were taken for granted as the key affinity ligand for the detection of pathogens in food and clinical samples to meet requirement for modern biosensors (Helali et al. 2018). Widely used polyclonal antibodies often lack the mandatory specificity, and this can be remedied by monoclonal antibodies or recombinant antibodies produced by hybridoma technology or phage display technology, respectively (Amiri et al. 2018). For the biosensors, a detectable signal can be induced by the interaction of immobilized antibodies with the corresponding antigens on the bacterial surface.
Because of cellular properties, e.g., large volume and heavy weight, a small number of bacteria on the surface of biosensors can also have a significant effect on the conduction. Therefore, impedance is the most common method to develop biosensors for detecting E. coli, as it can directly obtain detectable signals that it does not necessarily need further treatment or labeling signaling substances, after the immobilization of E. coli (Gupta et al. 2019a; Wan et al. 2016). Based on the immunomagnetic separation using streptavidin-coated magnetic beads to self-assemble biotinylated antibodies, an impedimetric immunosensor using surficial undecorated screen-printed interdigitated microelectrode was developed to rapidly detect E. coli O157:H7 and other bacteria (Salmonella typhimurium (S. typhimurium)) in chicken rinse water and ground beef (Xu et al. 2016a). In order to improve the performance of the biosensor, most impedance detection methods are modified with nanomaterials. Commonly used nanomaterials are carbon nanomaterials (Pandey et al. 2017; Guner et al. 2017), precious metal nanomaterials (Guner et al. 2017), transition-metal oxide (Dhull et al. 2019), and metal–organic frameworks (MOFs) (Gupta et al. 2019b). Guner and coworkers developed a novel impedimetric immunosensor based on a type of complex nanocomposites modified pencil graphite electrode (Guner et al. 2017). Such hybrid bio-nanocomposites are composed of polypyrrole, gold nanoparticles (AuNPs), chitosan, and multiwalled carbon nanotubes (Fig. 1). Once this nanocomposite platform was modified with anti-E. coli O157:H7 monoclonal antibodies, it could detect E. coli O157:H7 ranging from 30 to 3 × 107 CFU/mL with a LOD of 30 CFU/mL. Discarding the tedious pre-composition, in situ synthesis of nanomaterials can simplify the experimental procedure. An impedimetric biosensor was prepared by integrating noble metal nanoparticle induced by one-step laser induction method to three-dimensional graphene array electrode (You et al. 2020a) for E. coli O157:H7 quantification. It is worth noting that not all impedance methods need label of signaling substance. Sometimes, the use of labeled signal molecules such as urease on composite material surface (Yao et al. 2018) can improve detection sensitivity. Field-effect transistor (FET) and electrochromism similar to most impedance biosensors do not require any extra signaling molecules. Thakur and collaborators developed a graphene-based FET immunosensor to detect E. coli in river water (Thakur et al. 2018). The FET channel conductivity was modulated by the negatively charged molecules on the E. coli surface against the passivation with ultrathin Al2O3. Such immunosensor could real time detect 1 cell in 1 µL sample within less than 1 min (the linear range: 1 × 103 to 1 × 105 CFU/mL). In addition, using cytochrome C oxidase expressed by bacteria themselves as signal “label,” such nonselective chemical principle-based biosensor was found suitable to detect a large number of different bacteria including E. coli by just substituting target-specific antibodies (Kuss et al. 2019). Electrochromic polyaniline electropolymerized on indium tin oxide electrode was used to develop a visual method for bacteria detection. Polyaniline oxidation state was affected by the increased resistance in the circuit caused by E. coli binding to change the electrochromic response (Ranjbar et al. 2019).
Schematic of the fabrication of a multicomponent immunosensor. This novel impedimetric immunosensor based on a kind of complex nanocomposites modified pencil graphite electrode. Such hybrid bio-nanocomposites are composed of polypyrrole, gold nanoparticles, multiwalled carbon nanotubes, and chitosan. Reproduce from (Guner et al. 2017) with permission
Sandwich-based strategies that require labeled signaling molecules are commonly used because the outer extra signaling layer can generally increase specificity and sensitivity. For sandwich immune-biosensors, differential pulse voltammetry (DPV) is a commonly used electrochemical method to record the signal (Zou et al. 2019; Nguyen et al. 2018). For example, DPV was used to measure the polyaniline catalytically deposited by the labels of HRP on the surface of carbon nanotubes surface, and this sensitive sandwich immunosensor was developed to detect E. coli in dairy products including milk and yogurt (Fig. 2) (Zhang et al. 2016). By optimizing the dendrimer-encapsulated AuNPs and reasonable designing enhanced nanoprobes, this proposed strategy displayed a wide linear relationship (1 × 102–1 × 106 CFU/mL) and a low LOD (50 CFU/mL). Amperometry is another widely used electrochemical method (Altintas et al. 2018). Based on giant magnetoresistance (GMR), Zhou’s research group developed novel separable detection systems using magnetic nanoparticle as labels to detect E. coli O157:H7 (Yang et al. 2016a; Sun et al. 2016). E. coli captured on the GMR biosensor further captures magnetic beads, therefore changing the resistance of the GMR biosensor. This newly proposed detection method possesses the advantage of reusability without cleaning (Sun et al. 2016). Moreover, Tang et al. developed a novel strategy based on the blockage effect on nanopore array to rapidly detect of E. coli (Tang et al. 2016). Antibodies can be bound to the surface of nanopore array, and the immunobinding E. coli with its antibodies blocks silicon nanopore array to decrease the pore accessibility.
Schematic of the synthetic procedures of A PAMAM(Au) nanocomposites and B dAb-CNT-horseradish peroxidase (HRP) nanoprobes. C is sandwich strategy of E. coli immunoassay. Catalytically deposited polyaniline by HRP enzyme loaded on CNTs was measured by DPV. Reproduce from (Zhang et al. 2016) with permission
Improvement of the sensitivity of a biosensor is significantly important to achieve the detection of target with low abundance. In recent years, significant efforts have focused on developing more sensitive detection technologies through exploring intrinsic chemical or physical properties of elements of biosensors and have initiated an uplifting opportunity for practical applications. Sensitivity of biosensors can be most commonly improved by optimization. For example, Zhang and collaborators improved the immunosensor sensitivity by orders of magnitude through optimizing the proportion of graphene oxide (GO) to AuNPs (Zhang et al. 2017). For the biosensors, the immobilization of antibodies on the surface of biosensors is critical for the performance efficiency of biosensors, such as limit of detection (LOD) and linear dynamic range (LDR). Although the operation of antibody immobilization is simple by non-covalent strategy, the antibody tends to be randomly oriented on the surface, which is a foreseen disadvantage. To overcome this potential disadvantage, different immobilization techniques of anti-E. coli antibodies on surface were investigated, leading to a high sensitivity detection method by oriented immobilization using a dendrimer (Malvano et al. 2018). In addition, immobilization of antibodies by synthetic materials (Huang et al. 2018) or glutaraldehyde covalent cross-linking (Zhong et al. 2019) can also improve the load of antibodies and stability, thus improving the performance of the immunosensor. A biosensor was developed using MOFs encapsulated with quantum dots (QDs) (CdS@ZIF-8) as signal-amplifying tags, exhibiting a LOD of 3 CFU/mL by measuring Cd2+ leached from core–shell CdS@ZIF-8 nanoparticles (Zhong et al. 2019). The methods using individual CdS or ZIF-8 MOFs as amplifying tags show a 10–16 times higher LOD than that of the proposed nanocomposite method. The usage of enzymes (such as HRP) (Eltzov and Marks 2016; Li et al. 2017a), enzyme-contained composite nanomaterials (e.g., immune invertase-nanoclusters (Huang et al. 2018)), or nanozymes (e.g., platinum nanoparticles (Mo et al. 2019)) as signal enhancer improved the sensitivity of immune-biosensors for E. coli in standard solution or food samples. Using enzyme-free chain reactions activated by nucleic acid, such as hybridization chain reaction (HCR, analyte-triggered isothermal HCR stimulates formation of DNA polymeric nanowires (Wang et al. 2014)) to structure multifunctional nanoconjugates, this signal amplification approach was demonstrated to detect and quantify E. coli with a LOD of 39 CFU/mL and a linear range from 50 to 1 × 106 CFU/mL (Li et al. 2017b).
Limitations of Immune-biosensor
Attributing to the higher affinity toward targets than other ligands, antibodies have been extensively engaged to structure biosensors for E. coli detection (Table 1). However, a few drawbacks limit their applications. Antibodies are 100 times more expensive than aptamers in general. The generation and application of antibodies are restricted by pH, temperature, and background environments as well. Antibodies especially polyclonal antibodies are potentially affected to some degree by the batch, indicating that transvaluation is probably required before using a new batch of antibody (Chen and Yang 2015). Furthermore, the shelf life of antibodies is short, and the preservation conditions are quite harsh because they require a continuous cold chain. In most cases, antibodies bind to the substrate by non-covalent adsorption (Mathelie-Guinlet et al. 2019), which is inefficient and instability yet. The antibodies in random orientation caused by non-covalent adsorption could decrease the capture efficiency toward E. coli (Luo et al. 2019), thereby reducing the performance of immunosensor. Although there are approaches to get uniform orientation of antibodies (Malvano et al. 2018; Crivianu-Gaita and Thompson 2016), these techniques are difficult and expensive to be modified. On the other hand, the instability of the antibody, subsequent treatment, such as the synthesis of antibodies-containing compound materials (Huang et al. 2018; Xu et al. 2016b), can probably inactivate the antibodies, causing potential problem.
Aptamer-Based Biosensors
Aptamer, also known as chemical antibodies, is a structured single-stranded oligonucleotide sequence (RNA or DNA) obtained by systematic evolution of ligands by exponential enrichment (SELEX) in vitro (Saad and Faucher 2021). Aptamers possess a strict recognition ability and a high affinity with their corresponding targets ranging from small molecules to whole cells in size (including proteins, bacteria, viruses, cells, small biological molecules, and heavy metal ions) (Li et al. 2019; Song et al. 2019). They exhibit several advantages, such as a low immunogenic antigen, short screening period, stability, small-batch difference, ease of chemical modification, low cost, and easy to preserve (Paniel and Noguer 2019). These advantages led to the discovery of attractive aptamers, which have received immense interest in biosensors replacing antibodies for food safety, medical diagnosis, and environment monitoring (McConnell et al. 2020). In recent years, a large number of reviews have summarized the application of aptamers in bacteria including E. coli (Zhao et al. 2018; Davydova et al. 2016; Sharifi et al. 2020; Majdinasab et al. 2018).
In general, impedance methods are the easiest to use in the construction of sensor or biosensor methods, as they do not necessarily need further labeling signaling materials. Using E. coli O157:H7 specific aptamer, Bratov’s research group developed a three-dimensional interdigitated impedimetric transducer with enhanced sensitivity (Brosel-Oliu et al. 2018). The chemical reactions between the outer membrane proteins of E. coli O157:H7 and its aptamers at the electrode surface provoke detectable changes in the surface conductivity as a result of the electrical charge redistribution. Such novel aptamer-based label-free impedimetric biosensor shows a low LOD (2.9 × 102 CFU/mL) and short detection time (30 min) with a wide linear response (10–1 × 105 CFU/mL). In addition, no response emerges for other bacterial strains such as E. coli K12, Staphylococcus aureus (S. aureus), and S. typhimurium. In addition, other techniques have also been used to detect E. coli in various samples. An electrochemical biosensor based on a turn-off strategy was used to rapidly and effectively detect E. coli in traditional Chinese medicine of licorice extract with a satisfying LOD (80 CFU/mL) (Wang et al. 2019a). Zhang et al. developed a conductometric aptasensor (aptamer biosensor) using magnetic analyte separation by aptamers, and its minimum detectable concentration of E. coli in spiked tap water is 2.3 × 104 CFU/mL (Zhang et al. 2020). Zhang et al. developed an electrokinetics-based capacitive biosensor using a commercialized functional microelectrode with a LOD of 1 × 102 cells/mL for E. coli (Zhang et al. 2018a). Wang et al. designed a general surface plasmon resonance (SPR) spectroscopy kinetic measurement for detecting bacteria of E. coli (LOD: 1 × 105 CFU/mL) or Staphylococcus aureus (S. aureus) (LOD: 1 × 106 CFU/mL) in ideal samples (Wang et al. 2019b) (in this study, aptamers were immobilized on gold substrate via the tails of polyadenine as anchoring groups (Pei et al. 2012)). In recent years, microchip capillary electrophoresis (Zhang et al. 2019a) and microfluidic chips (Hao et al. 2019; Jiang et al. 2017; Li et al. 2020) also recruited to explore novel aptamer strategies. For microfluidic strategy, 4-polyamidoamine dendrimers immobilized onto channel surfaces exhibit two effects: diminution of background noise and increase in multiple binding sites for bacteria. Rolling circle amplification (RCA), a type of isothermal amplification method, can further enhance detection sensitivity and lower the LOD (1 × 102 cells/mL) based on a sandwich strategy (Jiang et al. 2017).
With the development of nanomaterials science and technology, various nanomaterials were used in the development of aptasensor to improve the performance of detection methods. One of the most commonly used nanomaterials is AuNPs. Dong and collaborators constructed a photoelectrochemical platform to assay bacteria with an ultralow LOD of 1.125 CFU/mL E. coli O157:H7 (Dong et al. 2020). This aptamer-based electrode structured by AuNPs and aptamer-CdS QDs decorated ZnO nanowire array absorbed the light from UV to visible region. The ultralow LOD attributed to the incremental surface area of used nanoparticles (AuNPs and CdS QDs) and the synergistic effect of above-mentioned multiple enhancement factors. Moreover, aptamer conjugated AuNPs as the signal probes can be explored to develop lateral flow test strip to simultaneously detect E. coli O157:H7, S. typhimurium, and S. aureus (Lu et al. 2020), or surface-enhanced Raman spectroscopy analysis for E. coli O157:H7 assay in ground beef (Diaz-Amaya et al. 2019). Another type of common nanomaterials is carbon nanomaterials, including carbon nanotubes (CNTs), carbon dots, graphene QDs, or carbon material derivatives (e.g., carbon nitride nanosheet) (Hao et al. 2017). Owing to their various excellent features, carbon materials are more widely used than AuNPs in the fields of electrochemiluminescence, photoelectrochemical biosensor (Hua et al. 2018), chemiluminescence (Khang et al. 2016), and field-effect transistor (Wu et al. 2017). Hua and collaborators integrated a variety of carbon materials to develop ratiometric photoelectrochemical aptasensor (Hua et al. 2018). The clearly distinguished cathodic current and anodic current were generated by aptamer modified carbon quantum dots/graphene hydrogel and graphene-like carbon nitride (g-C3N4), respectively. Changes in steric hindrance from the binding E. coli with aptamer decreased the cathodic current and immutability of anodic current serving as the stable reference. Remarkably, a new type of functional materials, MOFs, was also used in the development of aptasensor (Shahrokhian and Ranjbar 2018; Zhang et al. 2019b). An electrochemical aptasensor was developed by Shahrokhian based on organic–inorganic hybrid nanocomposites comprising amino-functionalized MOFs and polyaniline (Shahrokhian and Ranjbar 2018). The amine-modified aptamer was attached to the hybrid nanocomposites by a cross-linking agent of glutaraldehyde. Using methylene blue as the electrochemical redox indicator, the data monitored by DPV show that this biosensor possessed a dynamic response ranging from 21 to 2.1 × 107 CFU/mL and a LOD of 2 CFU/mL for E. coli O157:H7. Furthermore, other nanomaterials, such as upconversion nanoparticles (Jin et al. 2017), metallic oxide nanowire (Dong et al. 2020), and iron nanocomposites (Khang et al. 2016), could also improve the performance of the aptasensors especially reducing the LOD.
A vast majority of studies used the anti-E. coli aptamers. However, some studies used aptamers selected by the researchers themselves. Kaur and collaborators used phenylboronic acid on microtiter plate and screened a specific anti-E. coli aptamer by a novel in situ developed SELEX approach (Kaur et al. 2017). The affinity constant (Kd) of such DNA aptamer was estimated as 14 nM. Then, they developed a novel functionalized Bridged Rebar Graphene-based method to design nanostructured aptamer biosensor to detect bacteria of E. coli O78:K80:H11 (Fig. 3). This label-free impedimetric biosensor demonstrated a low LOD of 10 CFU/mL. These excellent performances attributed to the synergistic effect of enhanced electrical properties between MWCNT and bridged terephthalaldehyde in a 3D-hierarchical nanostructure.
Schematic representation of the impedimetric aptamer platform fabricated by functionalized Bridged Rebar Graphene (BRG). Using phenylboronic acid (PBA) as capturing agent, microtiter plate-based SELEX was developed for anti-E. coli O78:K80:H11 aptamer screening. DNA library was exposed to E. coli O78:K80:H11 over PBA modified wells. Then, eluting and amplifying the binders were performed for iterative SELEX rounds. The selected aptamers were subsequently immobilized onto aptasensor with BRG nanostructure to detect E. coli O78:K80:H11. EIS was engaged to record the signal change in [Fe(CN)6]3−/4− solution. Reproduce from (Kaur et al. 2017) with permission
Attributing to many recognition sites on bacteria, multi-recognition elements is one of the strategies for making biosensors. Guo and coworkers developed a sensitive and label-free electrochemical method for E. coli detection using both aptamer and antibody to recognize target bacterium E. coli (Guo et al. 2016), in which, anti-E. coli polyclonal antibody was applied as the first recognition element to capture E. coli onto electrode surface via specific recognition, and anti-E. coli aptamer as the second recognition element containing the primer sequence complementary to a G-quadruplex DNAzyme units included circular probe, which trigger RCA-based polymerase elongation (Fig. 4). Under the optimal condition, this proposed biosensor exhibited an ultralow LOD of 8 CFU/mL with a good linear range (9.4 to 9.4 × 105 CFU/mL). Regrettably, no explanation was provided for the combined use of antibodies and aptamers.
Illustration of the dual recognition elements (anti-E. coli aptamer and antibody)-based electrochemical assay of highly sensitive E. coli detection using dual amplification from RCA and DNAzyme. The polyclonal antibody was used as first recognition element to capture E. coli onto electrode surface, and aptamer as second recognition element containing the primer sequence complementary to a G-quadruplex DNAzyme units included circular probe which were used to trigger RCA-based polymerase elongation. Reproduce from (Guo et al. 2016) with permission
Limitations of Aptasensors
Aptamers possess various excellent features such as cost effectiveness, easy to synthesize and modify, and high stability compared to that of antibodies; therefore, aptasensor field has been thoroughly investigated in recent years (Table 2). However, aptamers also have some intrinsic disadvantages because of the properties of nucleic acids, such as they are easy to be degraded by nucleases (Soutschek et al. 2004). In fact, aptamers have a shorter lifetime in vitro than non-biological molecules. To further enhance their stability, the oligonucleotide skeleton can be chemically modified using, e.g., locked nucleic acid (Hagedorn et al. 2018), peptide nucleic acid (Zhao et al. 2020a), or spiegelmers (enantiomeric nucleic acids) (Eulberg and Klussmann 2003), before their practical application. Furthermore, SELEX process is performed in an anthropogenic environment, which is different from the physiological environment; therefore, the affinity of the resulting aptamer may not be as high as the antibody, further restricting the development and application of aptasensors. More importantly, the lack of studies on binding sites does not provide a theoretical basis for subsequent studies on aptamer. Therefore, the study of aptamer also needs to analyze the three-dimensional structure such as proteins, laying a theoretical foundation for research of aptamer on biosensor.
Imprinted Polymer-Based Biosensors
In addition to antibodies and aptamers, MIPs are another recognition element to design biomimetic selectivity (Golabi et al. 2017). The synthesis processes include two main steps: polymerization of the monomers in the presence of the template (analyte) and the removal of the template to leave analyte matched cavities on supporting materials. The binding types of recognition include non-covalent, electrostatic/ionic, metal center coordination, or covalent types (Chen et al. 2016). The literature about imprinted polymers-based biosensors is rather rich (Table 3), and some examples are highlighted below.
The chemical properties and surface topographies of bacterial outer surfaces are important identities for exploring molecularly imprinted polymers. Various polymeric materials have been utilized to construct surface imprinted polymers. A polydopamine surface imprinted polymer was used to fabricate electrochemiluminescence (ECL) biosensor for the quantitative detection of E. coli O157:H7 (Chen et al. 2017). Dopamine and template E. coli cell were directly electropolymerized on the electrode surface. Polyclonal antibody labeled nitrogen-doped graphene QDs with a high quantum yield were synthesized as signal molecules to generate intensive ECL irradiation in the presence of coreactant. LOD as low as 8 CFU/mL was obtained under the optimal conditions. Wagner and his coworkers developed two label-free biomimetic sensors, aluminum chips with surface imprinting of polyurethane (Grinsven et al. 2016) and stainless-steel chips deposited with surface-imprinted polyurethane layers (Cornelis et al. 2019). Atomic force microscope image showed an empty cavity on the support material (Fig. 5). The selective binding of the target E. coli can increase the thermal resistance between the detecting solution and chip surface to sensitively and specifically detect bacteria in a broad concentration. They constructed a heat transfer method (HTM) device to measure the change in thermal resistance between the liquid and the chip. Such biosensor can distinguish living E. coli cells from dead cells, or discriminate Gram-negative from Gram-positive bacteria. In addition, histidine derivative under UV-polymerization can also be used to construct imprinting film to detect E. coli even in the concentration of 70 CFU/mL (Idil et al. 2017). Uropathogenic E. coli can be imprinted in ultrathin sol–gel matrices coatings on surface of gold electrodes to form impedimetric sensor to detect target E. coli below a detection limit of 1 CFU/mL (Jafari et al. 2019).
Photo of the heat transfer method (HTM)-sensor device (left), schematic drawing of the meander structure of the heat source (upper right), and AFM image of an empty cavity on steel. Reproduce from (Cornelis et al. 2019) with permission
Though topographies of outer surfaces of bacteria are crucial, the chemical properties of outer surfaces are more critical in order to recognize bacteria with similar size and shape. Chemical property commonly used is based on outer functional groups; however, the mechanisms of bacterial recognition and affinity remain unclear. Other chemical properties such as bacterial surface charge heterogeneity may avoid such disadvantage. Recently, a novel bacteria-imprinted polymer based on charge distribution on bacterial outer surfaces for highly specific E. coli and other bacteria recognition was developed (Bao et al. 2017). The self-assembly and polymerization of permanent cationic monomer and zwitterionic monomer encoded the charge distribution of bacterial outer surface, to inhibit nonspecific bacterial attachment. Such universal strategy can be extended to recognize other biologic molecules/cells associated with distinctive charge distribution.
Furthermore, advanced materials such as microsphere and nanomaterials are commonly used to improve the performance of imprinted biosensor. Shan and Shiigi reported a water-dispersible microsphere with the imprinting shape of whose surface complementary to E. coli O157:H7. Through surface area effects, this Nafion and overoxidized polypyrrole coated microsphere rebind E. coli spontaneously and specifically (Shan et al. 2017). Afterwards, to achieve high-throughput bacterial detection, they engineered an E. coli-imprinted 96-well gold nanoparticle-coated microplate using two same materials mentioned above (Shan et al. 2018). Furthermore, the imprinted microplates possess the ability to shorten bacterial detection time compared to conventional methods and discriminate target E. coli O157:H7 from bacterial mixtures.
Limitations of the Imprinted Polymers-Based Biosensors
Molecular imprinting shows strong specificity attributing to its large volume, large surface area, large number of surface molecules, and many interaction sites with the MIPs. Therefore, it has a strong advantage in distinguishing E. coli from other types of bacteria and even distinguishing living cells form dead cells (Cornelis et al. 2019). However, the little interest in molecular imprinting technology, related researches are quite behind other technologies such as antibody and nucleic acid-related techniques. One reason is the bacterial heterogeneity. Unlike small molecules, the morphology of bacteria varies greatly because of different growth time and growth conditions, and the distribution of constituent molecules on its surface is also uneven. Hardly any study examined the selectivity of the biosensor to bacteria growing in different environments. Most of the studies did not clearly identify specific patterns. The other reason is that the variety of functional monomers to structure molecular imprinting is limited, restraining the diversification and the further applications of molecular imprinting to some extent.
Antimicrobial Peptide-Based Biosensors
AMPs are short synthetic peptides (generally 10–50 amino acid residues) against bacteria and are generally positively charged and amphipathic (Amiri et al. 2018). The actions of AMPs possess several modes including carpet model, barrel-starve model, toroidal model, or detergent model (Nguyen et al. 2011). Though mechanisms of antibacterial action and structure are diverse, these peptides commonly work by interacting with bacterial cell membranes via electrostatic interactions with negatively charged molecules such as lipopolysaccharides (LPS), followed by disrupting membrane integrity through hydrophobic contacts (Templier et al. 2016). Because of their intrinsic stability and ease of synthesis, they must endow desirable properties as potential recognition elements for bacteria sensing (Hoyos-Nogues et al. 2018; Qiao et al. 2020).
As current methods of E. coli detection have drawbacks such as dissatisfactory sensitivity and stability, AMPs provide an alternative approach to explore excellent biosensor for E. coli assay (Table 4). The simplest biosensor is the self-assembly of terminal cysteine residue modified AMPs onto the electrode surface. Magainin I (MI), the most used AMP for structuring biosensors, is oriented assembly to efficiently capture bacterial pathogens. The approach based on site-specifically attached AMPs could distinguish between living and dead bacteria. These impedimetric detection methods showed a high sensitivity with a LOD of 1 × 102 CFU/mL for four bacterial strains including E. coli (Liu et al. 2016). Surface modification with nanomaterials or nanocomposites is a common and effective method to improve the performance of biosensors, for example, AMPs functionalized magnetic nanoparticles used by Wilson (Wilson et al. 2019) and silver nanoparticles-reduced GO nanocomposites synthesized by Zhou and collaborators (Zhou et al. 2018a). Andrade’s research group developed a simple nanostructured electrochemical biosensor for bacteria detection (Miranda et al. 2017). Using the EDC and NHS coupling strategy, cysteine capped AuNPs were first modified on gold electrode, followed by linking clavanin A (Clav A) AMP on AuNPs to structure the biosensor. They found that E. coli and S. typhimurium captured on biosensor generated observed response in higher level than other Gram-negative bacteria. This proposed sensitive biosensor effectively differentiated bacteria in the concentrations between 10 and 1 × 104 CFU, which is about 10 times lower than the unmodified method previously mentioned. Like antibodies and aptamers, the use of enzymes (such as HRP and invertase) as a signal enhancer improved the sensing property of AMPs-based biosensors and reduced LODs for E. coli detection (Bai et al. 2020; Qiao et al. 2017). Because of semi-selective binding of AMPs to target microbial cells, to improve the selectivity of AMPs based biosensors, aptamers assembled on biosensors were used to highly specifically capture microorganisms, MI in methylene blue@MI organic–inorganic nanocomposite were used only to capture electrochemical signaling molecules to yield amplified signals (Bu et al. 2020).
Limitation of Antimicrobial Peptide-Based Biosensors
Though the APMs-based biosensors are impressive owing to their high stability in harsh environmental conditions over other ligands such as antibody, they have apparent drawbacks. Bacterial detection with AMPs is not always capable especially for complex samples. The specific of AMPs is of less satisfactory, as their binding to the surface of Gram-negative species cells is semi-selective (Miranda et al. 2017; Singh et al. 2019). Achieving selectivity is challenging and requires further excavation. Moreover, because existing peptide synthesis techniques are not as sophisticated as DNA, AMPs are fairly less expensive than antibodies but far more expensive than aptamers. Achieving low costs is also another challenge. Various chemical groups in the side chain of AMPs possess active chemical components and thus are difficult to manipulate.
Nucleic Acid-Based Biosensors
The genetic material of various living things is nucleic acid, and the diversity of genes determines the diversity of living entities. Thus, by detecting the genes of a species, it is possible to specifically detect or identify that species without being affected by other species. Owing to the excellent characteristics such as stability, specificity, and low cost, DNA biosensors became a revolutionized modern analysis to fabricate newfangled devices in various fields including the detection and identification of pathogens (Trotter et al. 2020; Umesha and Manukumar 2018; Abi et al. 2018). The principle of the DNA biosensor-based detection depends on the fragment gene from the pathogen hybridized with specific DNA probes immobilized on biosensors (Wen et al. 2016). The changes generated by the hybridization of E. coli gene and specific DNA probe are measured using various readouts such as impedimetric (Sharif et al. 2019), voltammetric (Shoaie et al. 2018), and capacitive methods (Deshmukh et al. 2020). Last few years have witnessed an abundant literature on imprinted nucleic acid-based biosensors (Table 5), and some examples are highlighted below.
The direct methods for biosensors directly utilize the properties of the target DNA bases or the electrode itself, rather than to introduce additional signaling elements. The direct method is the most commonly used, because no subsequent processing step is required after the target DNA interacts with the probe DNA. Only the surface of the substrate needs to be modified before the hybridization. DNA comprises various bases, which are electrically active molecules, especially guanine. For example, Ozkan-Ariksoysal and collaborators, based on intrinsic guanine oxidation signal, developed a DNA-wrapped CNTs modified electrochemical biosensor for detecting 16S rDNA from E. coli ATCC 25922 in real samples (Ozkan-Ariksoysal et al. 2017). Although direct use of ordinary DNA probe DNA containing G leads to a high background signal since they explored an ingenious method to cross this barrier. Replacing guanine with inosine base (analogue guanine) significantly reduced the background signal as inosine can hybridize with cytosine like guanine but is inactive at the oxidation potential of guanine of about 1.0 V. Another type of direct method is alterations in the interfacial properties, e.g., impedance (Yuhana Ariffin et al. 2020), capacitance (Deshmukh et al. 2020), surface acoustic wave (Tsougeni et al. 2020), or light-addressable potentiometry of electrode (Tian et al. 2019). Among them, impedance analysis is the most commonly used method for E. coli detection using various surface modifications, such as ZnO nanorods with carboxylated graphene nanoflakes (Jaiswal et al. 2020, 2018), the gold/silver core/shell nanoparticles combined nanoporous alumina (Ye et al. 2017), and aminopropyltriethoxysilane functionalized hollow silica microspheres (Yuhana Ariffin et al. 2020). Furthermore, detecting RNA of E. coli can simplify the detection procedure, such as not requiring PCR amplification. Koo et al. developed an amplification-free platform for sequence-specific detection of E. coli 16S rRNA at 1 aM against a million-fold (1 pM) interferent RNA (Koo et al. 2018). Sequence-specific binding of negatively charged 16S rRNA with uncharged PNA on polystyrene bead makes it electrophoretically mobile to block the glass nanopore with small diameter, and significantly reduces current.
The indirect methods for biosensors introduce additional signaling elements, such as electroactive molecules and enzymes. Generally speaking, electroactive molecules were used as DNA redox intercalators (e.g., methylene blue and anthraquinone (Ariffin et al. 2018; Narang et al. 2018)) or labels (methylene blue and ferrocene (Li et al. 2020; Guo et al. 2017)). Enzymes including protein enzymes, nanozymes (Kim et al. 2019) (nanomaterials with catalytic activity, e.g., oxidase-mimicking activity of cerium oxide nanoparticles), and ribozyme (Zaouri et al. 2019) (e.g., HRP-mimicking DNAzyme) were used as labels to amplify the readout. Guo and colleagues used methylene blue and ferrocene as redox labels modified at hairpin probe DNA terminals and implemented voltammetric detection of E. coli and S. typhimurium simultaneously (Guo et al. 2017). This bacteria DNA biosensor shows a turn-off model, and with the hybridization of target DNA with hairpin probes, the DPV signals of ferrocene and methylene blue (+ 0.36 V and − 0.27 V vs. Ag/AgCl) decrease in the presence of bacterial DNA. The estimated LODs are 0.32 or 0.67 fM for E. coli DNA or S. typhimurium DNA with the assistance of polymerase, respectively. An electroactive molecule provides a finite signal; however, enzymes are commonly used as labels to amplify the readout attributing to their high catalytic capacity. The most used enzyme is HRP. This type of DNA biosensors is generally based on “sandwich-type” strategy. The first layer (modified layer) is the capture probes with various materials decorated on the substrate surface, the middle layer (target layer) is the target layer, and the top layer (signal layer) is the signal enzyme layer. Recently, studies of biosensors used self-assembly monolayer consecutive adenine (polyA) DNA electrochemical biosensor, three-dimensional DNA tetrahedral probe modified gold electrode and polyaniline, and AuNPs modified screen-printed carbon electrode (SPCE) to develop enzyme amplification-based strategy to detect E. coli DNA (Wen et al. 2016; Shoaie et al. 2018; Li et al. 2018a). Because of the enzyme amplification, the LOD of SPCE biosensor modified by polyaniline/AuNPs (Fig. 6) was found as 0.5 fM with a sensitivity of 398 μA pM−1 cm−2. Enzymes can be immobilized in two ways: (i) to form enzyme-antibody complex to be fixed through specific antigen–antibody binding (Li et al. 2018a) and (ii) to form enzyme–DNA to be immobilized through DNA hybridization (Zhang et al. 2018b).
Illustration of the DNA electrochemical HRP-amplification strategy to detect E. coli DNA. Aniline monomers were electropolymerized to form polyaniline and HAuCl4 were electrodeposited to form AuNPs on the surface of SPCE successively. Subsequently, biotinylated probe was immobilized on electrode surface. After the hybridization of target DNA in bacterial lysis, digoxin-labeled detector DNA and antibody-HRP were added in succession. E. coli DNA was detected based on HRP catalyzed redox reactions of H2O2/TMB using cyclic voltammetry measurement. Reproduce from (Shoaie et al. 2018) with permission
Nucleic acid-related amplifications prevail in DNA biosensor field. Although the genes are amplified by PCR (amplification of the target gene using polymerase) during the sample processing step in most studies, many other amplification methods (amplification of the target or process) have been also used during the biosensor construction to further improve the performance of the biosensor. PCR is a typical non-isothermal amplification technique, indicating that the reaction temperature periodically changes during nucleic acid amplification (Gibbs 1991). Besides PCR, various other polymerase-based in vitro isothermal amplification techniques, e.g., RCA (Li et al. 2020), terminal deoxynucleotidyl transferase (TdT)-based amplification (Zhou et al. 2019), and loop-mediated isothermal amplification (LAMP) (Sharif et al. 2019), have been utilized to amplify the target gene or probe DNA (Zhao et al. 2015; Zhong and Zhao 2018) and ultimately to increase the signal on the electrode surface. A typical example is the substituting LAMP for PCR for the amplification of the target gene to simplify the detecting process (Sharif et al. 2019). In this study, pathogens genomic DNA was first extracted using commercial bacterial DNA Kit to be amplified by LAMP. Then, LAMP amplicons of pathogens E. coli O157:H7 genomic DNA or other bacterial DNA (Vibrio parahaemolyticus, S. aureus, or Listeria monocytogenes) were collected by magnetic beads via electrostatic adsorption, subsequent injected amplicons-magnetic bead composite materials in the microfluidic impedance system as abundant negative charges of amplicons will obviously decrease the impedance signal. The LOD is about 10 copies with an assay time of less than 1 h. Besides polymerase-based amplification, enzyme-free entropy driven amplification is also a powerful strategy for DNA biosensors (Bi et al. 2017). Li and colleagues developed a HCR (entropy driven amplification)-based multiple amplification strategy combining RCA and 3D-DNA walker (Fig. 7) (Li et al. 2020). The extracted E. coli O157:H7 target DNA sequence was amplified by DNA walker assisted by Nb. BbvC I endonuclease on magnetic beads to first generate many DNA sequences. Subsequently, those RCA and HCR reactions were successively performed on the electrode surface to produce a long DNA sequence to hybridize and immobilize plentiful electrochemical indicator modified DNA probes; therefore, electrochemical signal enhanced significantly. This electrochemical biosensor exhibited an LOD of 7 CFU/mL.
Schematic representation of HCR-based multiple amplification strategy combining with RCA and 3D-DNA walker for E. coli O157:H7 detection. A Homogeneous reaction of 3D-DNA walker-based amplification reaction assisted by Nb. BbvC I endonuclease to produce many fragments for the following amplification. B Heterogeneous reaction of HCR and RCA reactions on GC electrode surface. The purpose of these two reactions is to produce a long DNA strand to immobilize plentiful ferrocene modified DNA probes. Reproduce from (Li et al. 2020) with permission
Limitation of the DNA-Based Biosensors
Though the DNA-based technique is not affected by environmental parameters showing the advantage over phenotypic approaches (Emerson et al. 2008), it still has apparent flaws. One of the obvious defects of DNA-based biosensor lies in its inability to distinguish living E. coli cells form dead cells, because DNA always exists whether the E. coli cells are dead or alive. Furthermore, the extraction of gene fragments requires skilled personnel, requiring a high level of skill on the part of the operators to avoid erroneous results (false positives or false negatives). Moreover, because the PCR of extracted gene fragments is required (Ozkan-Ariksoysal et al. 2017; Kim et al. 2019), the drawbacks in PCR method will eventually affect the testing result. For example, PCR technique is cumbersome and requires specific and expensive instruments; some complex components of real samples may have contamination or PCR inhibitors (e.g., the lipid content (Schrader et al. 2012)), which can affect primer hybridization or amplification efficiency and give misleading results (Rajapaksha et al. 2019).
Other Biosensors
In addition to the above-mentioned sensors, several other types of biosensors are also known (Table 6). Lectin can strongly bind to bacteria depending on the specific cognate carbohydrate on their surface (Amiri et al. 2018); therefore, it became an acclaimed candidate as recognition ligand used to develop newfangled biosensors for E. coli quantification application. Concanavalin A has mainly used lectin (Saucedo et al. 2018; Wang et al. 2019c). Zheng et al. prepared concanavalin, a functionalized ZnO nanorod array, to fabricate a 3D nano-bio-interface, with a higher binding ability than two-dimensional flat substrate (Zheng et al. 2017). This multivalent binding strategy-based biosensor efficiently captured and detected E. coli, allowing a low LOD of 90 CFU/mL.
Bacteriophages, owing to their specificity, accuracy, and reduced measurement time, have been taken into account as a recognition element of bacteria for the novel sensing method. In addition, compared to antibodies, phages are easy to produce and can tolerate extreme temperature, pH, and organic solvents. So far, several bacteriophage-based biosensors have been developed involving quartz crystal microbalance and electrochemical methods as reliable and cost-effective tools for bacterial detection (Farooq et al. 2018). Using charge-directed immobilization technology, T2 bacteriophage as the recognition element is orientated on the surface of positively charged CNTs modified glassy carbon (GC) electrode (Zhou et al. 2017). The impedance detection is selective to detect B strain of E. coli rather than nonhost K strain with 1 × 103 CFU/mL LOD using ESI. A T4 phage-based biosensor was developed using GOx&HRP-Cu3(PO4)2 organic–inorganic nanocomposite nanoflowers as signal amplifier to discriminate and quantify live E. coli in urine, reaching an ultralow LOD (1 CFU/mL) with a wide linearity range (15–1.5 × 108 CFU/mL) (Li et al. 2018b). For this sandwich strategy, AMPs and phages were used as the two layers of recognition elements. More recently, Sedki and coworkers used non-lytic M13 phage based on the mechanism of exiting cell by extrusion rather than cell lysis (such as for T4 & T7 phages) and introduced a selective biosensor for ultra-sensitive detection of E. coli within 14 cells/mL in synthetic river water (Sedki et al. 2020). The M13 phage-based biosensor is stable for 2 weeks in a wide pH range (3.0–10.0) and high temperature of 45 °C.
An adhesion peptide elutriated by the phage display biopanning method was conjugated to AuNPs to fabricate a dual-functional nanoconjugate (Au@P937 NRs), which specifically bind bacteria (Chen et al. 2018). Such biocompatible Au@P937 nanoconjugate has exhibited a low LOD of 46 CFU/mL and 89 CFU/mL for E. coli and S. aureus, respectively. Moreover, nanoconjugate could kill both bacteria in minutes under laser irradiation.
Antibacterial materials were engaged as recognition element of E. coli. Wu et al. developed a prickly antibacterial nanocomposite anchored on the surface of porous Ni electrode (Wu et al. 2018). They used nanocomposite of prickly Zn-CuO nanoparticles by in situ deposition and GO nanosheets (Fig. 8). Owing to the prick surface of nanostructures, the impedance sensor toward E. coli emerged with an excellent capture efficiency of more than 80% of 50 CFU/mL in 20 min. More importantly, this proposed nanocomposite-decorated sensor could quantify E. coli in a spiked rat blood sample. Cationic poly (phenylene vinylene) derivative as receptor was applied for binding with negatively charged microorganisms (Zhou et al. 2018b).
Upper figure: schematic presentation of the procedures for one-pot synthesizing prickly Zn-CuO@GO to decorate porous Ni electrode. Below figure: schematic presentation of the bacteria capture, killing and detection principles by prickly antibacterial nanocomposite. The procedure includes bacteria capture through strong binding affinity of nanocomposite toward E. coli, piercing on the bacterial surface, and acquisition of sensing signals. Reproduce from (Wu et al. 2018) with permission
Because of the abundance of nucleases in bacterial lysates, nucleases are used as alternative recognition elements to detect bacteria. In Gu’s work, hemin was encapsulated by DNA in the pores of mesoporous silicon nanoparticles, and the present nuclease of bacteria can hydrolyze DNA, thus releasing catalytic hemin from the pores and catalyzing the substrates to produce detectable signals (Gu et al. 2019). This cell enrichment-free strategy achieved a low LOD of 3 CFU/mL over a detection concentration ranging from 10 to 1 × 109 CFU/mL.
Paper-Based Biosensors
The demands for rapidly and accurately on-site bacteria detection progressively increase to limit the infectious incidence from contaminated water and food. Traditional analysis methods (such as culture-based assays, PCR, and ELISA) require time-consuming and labor intensive process of cultivation and identification, or need bacteria enrichment or sample preparation process to remove substance for inhibiting the sample background, greatly limiting these technologies in the field detection (Luo et al. 2019, 2020). To develop on-site, cost-efficient, disposable, and easy-to-use portable devices for meeting increasing market demands, paper-based portable biosensors for E. coli detection without the need of sophisticated lab facility were developed accordingly, adhering to the Affordable, Sensitive, Specific, User-friendly, Rapid and robust, Equipment-free, Delivered (ASSURED) criteria issued by World Health Organization (WHO) (Nguyen et al. 2020).
In recent years, paper-based colorimetric biosensors (e.g., LFA) can instantly identify the presence of bacteria in various real samples, and thus is a potential diagnostics platform (Table 7). The paper used is a filter paper, a nitrocellulose membrane, or even a waste newspaper (Ilhan et al. 2019; Yang et al. 2016b; Lin et al. 2020; Wang et al. 2020a). Lin developed an economical colorimetric strategy to fabricate microfluidic paper-based analytical devices for assaying E. coli BL21 in seawater and tap water (Lin et al. 2020). The surface structure of this environment-friendly paper device was manufactured by UV light curing water-based polyurethane acrylate, showing an excellent power against organic solvents and surfactant solutions. It revealed a linear range from 1 × 104 to 1 × 109 CFU/mL and a LOD of 3.7 × 103 CFU/mL. Fluorescence and photothermal methods are other alternative signal generation modes of paper-based biosensors. Song et al. presented a simple and time-saving method to fabricate label-free fluorescent strip immunosensor for detecting E. coli O157:H7, with a LOD of 1 cell/mL via target pre-incubation in food samples (Song et al. 2016a). Recently, a smartphone-integrated dual-readout (fluorescence and colorimetry) paper sensing system was developed depending on o-phenylenediamine-Cu2+ reaction system to quantitatively detect E. coli (Wang et al. 2020b). Relying on the photothermal effect of AuNPs to achieve the amplification, the immunofiltration strip method could detect a low concentration of 1.95 × 104 CFU/mL (Jia et al. 2019).
A majority of paper-based methods use antibodies as capture agents, other recognition elements such as aptamer and phage-based protein reporter are also used to avoid the disadvantages of antibodies (Alcaine et al. 2016; Wang et al. 2019d). Despite the fast pace growth of portable biosensors field and rapid development of breakthrough technologies (Song et al. 2016b; Zhu et al. 2018; Zhao et al. 2020b), majority of related lab-scale works cannot meet the market demands at all. In order to provide a repeatable and cost-efficient approach for pathogen E. coli detection in a real sample, Díaz-Amaya and collaborators developed an inkjet printing technique as a vigorous approach to fabricate paper-based biosensors for E. coli detection (Diaz-Amaya et al. 2019). Owing to nanomaterials can be enhanced the signal of bacteria detection, they have shown great application on paper-based biosensors (Nguyen and Kim 2020). A novel synthesized nanomaterial called aptamer conjugation (carboxyl-aptamer covalently conjugated on the polyetherimide-Au-polystyrene particles) as the optically signal enhancer was printed to nitrocellulose substrate by inkjet (Fig. 9). Utilizing a carefully controlled aptameric ink nanopatterning, this biosensor efficiently detected minimal 233 CFU/mL of E. coli O157:H7 in ground beef and 25 CFU/mL in pure culture.
Illustration of the mechanism using aptamer conjugation for improved optical detection of E. coli. Aptameric ink nanopatterning for inkjet printing platforms: image acquisition (a), data analysis (b), and test interpretation (c). Reproduce from (Diaz-Amaya et al. 2019) with permission
Most paper-based detection do not require nucleic acid extraction and amplification to detect whole pathogen cells (usually by sandwich strategy); however, their sensitivity does not meet (usually above 1 × 105 CFU/mL (Luo et al. 2020)) the demands of most food safety regulatory standards. The most common method to solve this problem is to improve the sensitivity through signal amplification technology, such as silver enhancement technology, nanozyme-based technology, and enzymatic catalytic amplification technology (Nguyen et al. 2020). The astatic arrangement of the antibodies on the carrier surface is also one of the reasons for the mediocre sensitivity; therefore, Luo increased the sensitivity of paper device by orienting antibodies to the surface of chromatic agents (Luo et al. 2019) using AuNPs as the chromatic agents. Gold binding peptide (GBP)-fused streptococcal protein G (SPG) possessed two functions: specific affinity of AuNPs by gold binding peptide and immobilization of Fc portion of the antibody by SPG (Fig. 10). In this case, antibodies on AuNPs surface present a great oriented arrangement (You et al. 2020b). This nanocomposite is colloidal stable even in high salt environment, granting the application to biological and food samples. This paper-based colorimetric biosensor successfully detected minimal E. coli of 1 × 103 CFU/mL, lowering an order of magnitude against conventional adsorption-based strategy (antibodies are unoriented on AuNPs). In addition, based on a Cas9 nickase-triggered isothermal DNA amplification, an affordable and convenient method for dual food-borne pathogen detection was achieved, allowing instrument-free and dual target detection (Wang et al. 2020c). The genomic DNAs of S. typhimurium and E. coli were simultaneously amplified using specific sgRNAs and primers by a one-pot reaction. The FITC/biotin and digoxin/biotin tags double-labeled amplicons were directly visualized on a lateral flow strip (Fig. 11). The LODs of such CRISPR-cas9 based method are 1 × 102 copies for genomic DNAs and 1 × 102 CFU/mL for bacteria.
Schematic illustration of paper-based radial flow chromatographic immunoassay. Streptococcal protein G (SPG) fused with gold binding peptide (GBP) tail has possessed two functions: specific affinity of AuNPs by GBP and immobilization of Fc portion of the antibody by SPG. In this case, antibodies are directly fixed on AuNPs surface to form immuno-AuNPs as chromatic agent. The size-dependent difference in mobility between E. coli and E. coli/immuno-AuNPs is vital for target analyte detection. A red inner ring arises because of the low mobility of E. coli/immuno-AuNPs, and its intensity is proportional to analyte concentration. Absence of E. coli results in the even diffusion of chromatic AuNPs in the radial direction without forming an inner ring. Reproduce from (Luo et al. 2019) with permission
Detection of dual Cas9 nickase-based amplification reaction products by the lateral flow strip. The genomic DNAs of S. typhimurium and E. coli were simultaneously amplified using specific sgRNAs and primers by a one-pot reaction. The FITC/biotin and digoxin/biotin tags double-labeled amplicons were directly visualized on a lateral flow strip. Reproduce from (Wang et al. 2020c) with permission
Conclusions and Perspectives
The identification of E. coli from food and environmental samples remains significant for ensuring food safety, preventing disease outbreaks, and protecting human health. The plenitudinous accessible literature suggests that various biorecognition element-based biosensors have been successfully used to detect E. coli in multifarious samples, especially in food samples. In this mini review, the literature with substantial effect on the current progress or significant potential for future applications was given prime importance. The principles, applications, and limitations of various biorecognition element-based biosensors are discussed in detail. A large majority of proposed biosensors, such as imprinted polymer-based, AMP-based, most immuno- and aptamer-based biosensors, have put their enthusiasm into determining whole E. coli cells, because of the relatively uncomplicated preprocessing. In addition, indisputably, antibodies are the most preferred and extensively used recognition molecules for many biosensors, particularly for paper-based biosensors. Other analyses are less mature than the immunoassays.
Advances in nanomaterials have boosted the development of biosensors for E. coli detection owing to their unique characteristics including distinct light, electricity, magnetism, and thermal properties. Nanomaterials ranging from zero dimension to three dimension are extensively used to construct biosensors surface and signal amplification complex, as the large specific surface area can remarkably improve the interaction between analytes and the surface. Among all nanomaterials, carbon materials, magnetic nanoparticles, and AuNPs have been used more frequently. Especially, AuNPs are the most commonly prescribed materials as chromogenic agent and carrier to manufacture paper-based lateral flow strip (Luo et al. 2020). The application of nanomaterials in biosensors possesses several crucial advantages such as the improved identification, augmented detectable signal, the increased sensitivity, and the decreased analysis time (Yang et al. 2016c). However, finding the traces of nanomaterials in the molecular imprinting-based method is difficult, and thus faces a great deal of challenge and future opportunity. Furthermore, some particular nanomaterials are enthusiastically selected, and some other emerging nanomaterials (such as MOFs and DNA nanostructures) are often overlooked unintentionally.
Biosensors have the potential to provide high selectivity, sensitivity, and methods for real-time analysis without time-consuming pretreatment steps and thus are of vital importance for field identification of E. coli. However, the development of these biosensors is still in the early stages, preventing them from entering market to meet realistic demands, food safety, and supervision are still based on traditional methods. Although a number of studies have stated that the required LOD or even lower than 1 CFU/mL of developed biosensors for E. coli quantitative detection has been achieved (Hao et al. 2017; Hua et al. 2018; Jafari et al. 2019), this situation is mainly suitable for detection in ideal solutions. However, in reality, the ingredients of real samples including food are of extreme complexity and are much more complicated than the ideal solution. Most of the biosensors developed cannot be used for such complex samples, because of interreference caused by other components, resulting in a decreased level sensitivity at least two orders of magnitude lower than the desired sensitivity. The development of in situ simple sample purification and separation method can further advance the development of biosensors.
Portable devices are favored in research studies as a consequence of their easy operation, compact size, and fast detection speed, and portability to different places for real-time detection. For example, paper-based LFA can instantly identify the presence of bacteria in various real samples, and is definitely a potential diagnostics platform. Unfortunately, their sensitivity just above 1 × 105 CFU/mL is insufficient (Luo et al. 2020) for meeting the demands of numerous food safety regulatory standards. The most common method of solving this problem is to improve the sensitivity by signal amplification technology; however, these methods undoubtedly increase the degree of labor, complexity, and sacrifice the portability. Therefore, the sensitivity of biosensors should be improved in order to achieve better recognition ability of biorecognition molecules. Therefore, the following measures may meet the LOD. (1) Improving the stability of biorecognition molecules, especially for antibodies. (2) Orienting biorecognition molecules on the surface. Directional fixation of the recognition molecules makes the specific binding sites completely exposed to E. coli cells and improves the ability of capture. (3) Integrating multiple recognition methods. For example, for sandwich mode biosensors, antibodies are immobilized on substrate surface (e.g., electrode, paper) to strongly capture target owing to the excellent specificity, as well as aptamers are fixed on signal particle surface to improve signal, because of malleable property. Especially in this study, the use of multiple recognition molecules can reduce the competition between nitrocellulose membrane and chromogenic nanoparticles for the target and thus is beneficial to improve the sensitivity.
The biosensors must be simple, fast, sensitive, stable, reproducible, and simple in operation without need of specialized personnel. With progress in technology (nanotechnology and biotechnology in particular) and advancement of commercialization, novel and high-impact biosensors for E. coli detection can be envisioned. In addition, as new laws and regulations are enacted, the requirements for biosensors have increased doubtlessly, leaving future researchers to explore these opportunities in order to meet the stringent demand.
References
Abi A, Mohammadpour Z, Zuo XL, Safavi A (2018) Nucleic acid-based electrochemical nanobiosensors. Biosens Bioelectron 102:479–489. https://doi.org/10.1016/j.bios.2017.11.019
Alcaine SD, Law K, Ho S, Kinchla AJ, Sela DA, Nugen SR (2016) Bioengineering bacteriophages to enhance the sensitivity of phage amplification-based paper fluidic detection of bacteria. Biosens Bioelectron 82:14–19. https://doi.org/10.1016/j.bios.2016.03.047
Altintas Z, Akgun M, Kokturk G, Uludag Y (2018) A fully automated microfluidic-based electrochemical sensor for real-time bacteria detection. Biosens Bioelectron 100:541–548. https://doi.org/10.1016/j.bios.2017.09.046
Amiri M, Bezaatpour A, Jafari H, Boukherroub R, Szunerits S (2018) Electrochemical methodologies for the detection of pathogens. ACS Sens 3:1069–1086. https://doi.org/10.1021/acssensors.8b00239
Ariffin EY, Lee YH, Futra D, Tan LL, Abd Karim NH, Ibrahim NNN, Ahmad A (2018) An ultrasensitive hollow-silica-based biosensor for pathogenic Escherichia coli DNA detection. Anal Bioanal Chem 410:2363–2375. https://doi.org/10.1007/s00216-018-0893-1
Bai HS, Bu SJ, Wang CY, Ma CY, Li ZY, Hao Z, Wan JY, Han Y (2020) Sandwich immunoassay based on antimicrobial peptide-mediated nanocomposite pair for determination of Escherichia coli O157:H7 using personal glucose meter as readout. Microchim Acta 187:220. https://doi.org/10.1007/s00604-020-4200-4
Banerjee R, Jaiswal A (2018) Recent advances in nanoparticle-based lateral flow immunoassay as a point-of-care diagnostic tool for infectious agents and diseases. Analyst 143:1970–1996. https://doi.org/10.1039/c8an00307f
Bao H, Yang B, Zhang X, Lei L, Li Z (2017) Bacteria-templated fabrication of a charge heterogeneous polymeric interface for highly specific bacterial recognition. Chem Commun 53:2319–2322. https://doi.org/10.1039/C6CC09242J
Bi S, Yue SZ, Zhang SS (2017) Hybridization chain reaction: a versatile molecular tool for biosensing, bioimaging, and biomedicine. Chem Soc Rev 46:4281–4298. https://doi.org/10.1039/c7cs00055c
Brosel-Oliu S, Ferreira R, Uria N, Abramova N, Gargallo R, Munoz-Pascual FX, Bratov A (2018) Novel impedimetric aptasensor for label-free detection of Escherichia coil O157:H7. Sens Actuator B-Chem 255:2988–2995. https://doi.org/10.1016/j.snb.2017.09.121
Bu S, Wang K, Li Z, Wang C, Hao Z, Liu W, Wan J (2020) An electrochemical biosensor based on methylene blue-loaded nanocomposites as signal-amplifying tags to detect pathogenic bacteria. Analyst 145:4328–4334. https://doi.org/10.1039/d0an00470g
Burlage RS, Tillmann J (2017) Biosensors of bacterial cells. J Microbiol Methods 138:2–11. https://doi.org/10.1016/j.mimet.2016.12.023
Castillo-Torres KY, Arnold DP, McLamore ES (2019) Rapid isolation of Escherichia coli from water samples using magnetic microdiscs. Sens Actuator B-Chem 291:58–66. https://doi.org/10.1016/j.snb.2019.04.043
CDC, in: https://www.cdc.gov/ecoli/outbreaks.html, , 2020.
Cesewski E, Johnson BN (2020) Electrochemical biosensors for pathogen detection. Biosens Bioelectron 159:112214. https://doi.org/10.1016/j.bios.2020.112214
Chen A, Yang S (2015) Replacing antibodies with aptamers in lateral flow immunoassay. Biosens Bioelectron 71:230–242. https://doi.org/10.1016/j.bios.2015.04.041
Chen L, Wang X, Lu W, Wu X, Li J (2016) Molecular imprinting: perspectives and applications. Chem Soc Rev 45:2137–2211. https://doi.org/10.1039/C6CS00061D
Chen S, Chen X, Zhang L, Gao J, Ma Q (2017) Electrochemiluminescence detection of Escherichia coli O157:H7 based on a novel polydopamine surface imprinted polymer biosensor. ACS Appl Mater Interfaces 9:5430–5436. https://doi.org/10.1021/acsami.6b12455
Chen QY, Zhang LW, Feng YH, Shi F, Wang YB, Wang P, Liu L (2018) Dual-functional peptide conjugated gold nanorods for the detection and photothermal ablation of pathogenic bacteria. J Mat Chem B 6:7643–7651. https://doi.org/10.1039/c8tb01835a
Chen YJ, Qian C, Liu CZ, Shen H, Wang ZJ, Ping JF, Wu J, Chen H (2020) Nucleic acid amplification free biosensors for pathogen detection. Biosens Bioelectron 153:17. https://doi.org/10.1016/j.bios.2020.112049
Choiniere S, Frost EH, Dubowski JJ (2019) Binding strategies for capturing and growing Escherichia coli on surfaces of biosensing devices. Talanta 192:270–277. https://doi.org/10.1016/j.talanta.2018.09.043
Cornelis P, Givanoudi S, Yongabi D, Iken H, Duwé S, Deschaume O, Robbens J, Dedecker P, Bartic C, Wübbenhorst M, Schöning MJ, Heyndrickx M, Wagner P (2019) Sensitive and specific detection of E. coli using biomimetic receptors in combination with a modified heat-transfer method. Biosens Bioelectron 136:97–105. https://doi.org/10.1016/j.bios.2019.04.026
Crivianu-Gaita V, Thompson M (2016) Aptamers, antibody scFv, and antibody Fab’ fragments: an overview and comparison of three of the most versatile biosensor biorecognition elements. Biosens Bioelectron 85:32–45. https://doi.org/10.1016/j.bios.2016.04.091
Davydova A, Vorobjeva M, Pyshnyi D, Altman S, Vlassov V, Venyaminova A (2016) Aptamers against pathogenic microorganisms. Crit Rev Microbiol 42:847–865. https://doi.org/10.3109/1040841X.2015.1070115
de Miranda JL, Oliveira MDL, Oliveira IS, Frias IAM, Franco OL, Andrade CAS (2017) A simple nanostructured biosensor based on clavanin A antimicrobial peptide for gram-negative bacteria detection. Biochem Eng J 124:108–114. https://doi.org/10.1016/j.bej.2017.04.013
Deshmukh R, Prusty AK, Roy U, Bhand S (2020) A capacitive DNA sensor for sensitive detection of Escherichia coli O157:H7 in potable water based on the z3276 genetic marker: fabrication and analytical performance. Analyst 145:2267–2278. https://doi.org/10.1039/c9an02291k
Dhull N, Kaur G, Jain P, Mishra P, Singh D, Ganju L, Gupta V, Tomar M (2019) Label-free amperometric biosensor for Escherichia coli O157:H7 detection. Appl Surf Sci 495:143548. https://doi.org/10.1016/j.apsusc.2019.143548
Diaz-Amaya S, Lin LK, Deering AJ, Stanciu LA (2019) Aptamer-based SERS biosensor for whole cell analytical detection of E. coli O157:H7. Anal Chim Acta 1081:146–156. https://doi.org/10.1016/j.aca.2019.07.028
Diaz-Amaya S, Zhao M, Lin LK, Ostos C, Allebach JP, Chiu GTC, Deering AJ, Stanciu LA (2019) Inkjet printed nanopatterned aptamer-based sensors for improved optical detection of foodborne pathogens. Small 15:1805342. https://doi.org/10.1002/smll.201805342
Ding S, Lyu Z, Niu X, Zhou Y, Liu D, Falahati M, Du D, Lin Y (2020) Integrating ionic liquids with molecular imprinting technology for biorecognition and biosensing: a review. Biosens Bioelectron 149:111830. https://doi.org/10.1016/j.bios.2019.111830
Dong XX, Shi ZL, Xu CX, Yang C, Chen F, Lei ML, Wang JJ, Cui QN (2020) CdS quantum dots/Au nanoparticles/ZnO nanowire array for self-powered photoelectrochemical detection of Escherichia coli O157:H7. Biosens Bioelectron 149:111843. https://doi.org/10.1016/j.bios.2019.111843
Eltzov E, Marks RS (2016) Miniaturized flow stacked immunoassay for detecting Escherichia coli in a single step. Anal Chem 88:6441–6449. https://doi.org/10.1021/acs.analchem.6b01034
Emerson D, Agulto L, Liu H, Liu L (2008) Identifying and characterizing bacteria in an era of genomics and proteomics. Bioscience 58:925–936. https://doi.org/10.1641/b581006
Eulberg D, Klussmann S (2003) Spiegelmers: biostable aptamers. ChemBioChem 4:979–983. https://doi.org/10.1002/cbic.200300663
Farooq U, Yang QL, Ullah MW, Wang SQ (2018) Bacterial biosensing: recent advances in phage-based bioassays and biosensors. Biosens Bioelectron 118:204–216. https://doi.org/10.1016/j.bios.2018.07.058
Gibbs RA (1991) Polymerase chain reaction techniques. Curr Opin Biotechnol 2:69–75. https://doi.org/10.1016/0958-1669(91)90063-B
Golabi M, Kuralay F, Jager EWH, Beni V, Turner APF (2017) Electrochemical bacterial detection using poly(3-aminophenylboronic acid)-based imprinted polymer. Biosens Bioelectron 93:87–93. https://doi.org/10.1016/j.bios.2016.09.088
Grinsven BV, Eersels K, Akkermans O, Ellermann S, Kordek A, Peeters M, Deschaume O, Bartic C, Diliën H, Steen Redeker E, Wagner P, Cleij TJ (2016) Label-free detection of Escherichia coli based on thermal transport through surface imprinted polymers. ACS Sens 1:1140–1147. https://doi.org/10.1021/acssensors.6b00435
Gu ZF, Fu AC, Ye L, Kuerban K, Wang Y, Cao ZJ (2019) Ultrasensitive chemiluminescence biosensor for nuclease and bacterial determination based on hemin-encapsulated mesoporous silica nanoparticles. ACS Sens 4:2922–2929. https://doi.org/10.1021/acssensors.9b01303
Guner A, Cevik E, Senel M, Alpsoy L (2017) An electrochemical immunosensor for sensitive detection of Escherichia coli O157:H7 by using chitosan, MWCNT, Polypyrrole with Gold Nanoparticles Hybrid Sensing Platform. Food Chem 229:358–365. https://doi.org/10.1016/j.foodchem.2017.02.083
Guo Y, Wang Y, Liu S, Yu J, Wang H, Wang Y, Huang J (2016) Label-free and highly sensitive electrochemical detection of E. coli based on rolling circle amplifications coupled peroxidase-mimicking DNAzyme amplification. Biosens Bioelectron 75:315–319. https://doi.org/10.1016/j.bios.2015.08.031
Guo YN, Wang Y, Liu S, Yu JH, Wang HZ, Liu XK, Huang JD (2017) Simultaneous voltammetric determination of E-coli and S-typhimurium based on target recycling amplification using self-assembled hairpin probes on a gold electrode. Microchim Acta 184:745–752. https://doi.org/10.1007/s00604-016-2017-y
Gupta A, Bhardwaj SK, Sharma AL, Deep A (2019a) A graphene electrode functionalized with aminoterephthalic acid for impedimetric immunosensing of Escherichia coli. Microchim Acta 186:800. https://doi.org/10.1007/s00604-019-3952-1
Gupta A, Bhardwaj SK, Sharma AL, Kim KH, Deep A (2019b) Development of an advanced electrochemical biosensing platform for E. coli using hybrid metal-organic framework/polyaniline composite. Environ Res 171:395–402. https://doi.org/10.1016/j.envres.2019.01.049
Hagedorn PH, Persson R, Funder ED, Albaek N, Diemer SL, Hansen DJ, Moller MR, Papargyri N, Christiansen H, Hansen BR, Hansen HF, Jensen MA, Koch T (2018) Locked nucleic acid: modality, diversity, and drug discovery. Drug Discov Today 23:101–114. https://doi.org/10.1016/j.drudis.2017.09.018
Hao N, Zhang X, Zhou Z, Hua R, Zhang Y, Liu Q, Qian J, Li H, Wang K (2017) AgBr nanoparticles/3D nitrogen-doped graphene hydrogel for fabricating all-solid-state luminol-electrochemiluminescence Escherichia coli aptasensors. Biosens Bioelectron 97:377–383. https://doi.org/10.1016/j.bios.2017.06.025
Hao XK, Yeh PY, Qin YB, Jiang YQ, Qiu ZY, Li SY, Le T, Cao XD (2019) Aptamer surface functionalization of microfluidic devices using dendrimers as multi-handled templates and its application in sensitive detections of foodborne pathogenic bacteria. Anal Chim Acta 1056:96–107. https://doi.org/10.1016/j.aca.2019.01.035
Helali S, Alatawi ASE, Abdelghani A (2018) Pathogenic Escherichia coli biosensor detection on chicken food samples. J Food Saf 38:e12510. https://doi.org/10.1111/jfs.12510
Hoyos-Nogues M, Gil FJ, Mas-Moruno C (2018) Antimicrobial peptides: powerful biorecognition elements to detect bacteria in biosensing technologies. Molecules 23:1683. https://doi.org/10.3390/molecules23071683
Hua R, Hao N, Lu JW, Qian J, Liu Q, Li HN, Wang K (2018) A sensitive potentiometric resolved ratiometric photoelectrochemical aptasensor for Escherichia coli detection fabricated with non-metallic nanomaterials. Biosens Bioelectron 106:57–63. https://doi.org/10.1016/j.bios.2018.01.053
Huang FC, Zhang HL, Wang L, Lai WH, Lin JH (2018) A sensitive biosensor using double-layer capillary based immunomagnetic separation and invertase-nanocluster based signal amplification for rapid detection of foodborne pathogen. Biosens Bioelectron 100:583–590. https://doi.org/10.1016/j.bios.2017.10.005
Idil N, Hedström M, Denizli A, Mattiasson B (2017) Whole cell based microcontact imprinted capacitive biosensor for the detection of Escherichia coli. Biosens Bioelectron 87:807–815. https://doi.org/10.1016/j.bios.2016.08.096
Ilhan H, Guven B, Dogan U, Torul H, Evran S, Cetin D, Suludere Z, Saglam N, Boyaci IH, Tamer U (2019) The coupling of immunomagnetic enrichment of bacteria with paper-based platform. Talanta 201:245–252. https://doi.org/10.1016/j.talanta.2019.04.017
Jaiswal N, Pandey CM, Soni A, Tiwari I, Rosillo-Lopez M, Salzmann CG, Malhotra BD, Sumana G (2018) Electrochemical genosensor based on carboxylated graphene for detection of water-borne pathogen. Sens Actuator B-Chem 275:312–321. https://doi.org/10.1016/j.snb.2018.07.055
Jaiswal N, Pandey CM, Solanki S, Tiwari I, Malhotra BD (2020) An impedimetric biosensor based on electrophoretically assembled ZnO nanorods and carboxylated graphene nanoflakes on an indium tin oxide electrode for detection of the DNA of Escherichia coli O157:H7. Microchim Acta 187:1. https://doi.org/10.1007/s00604-019-3921-8
Jafari H, Amiri M, Abdi E, Navid SL, Bouckaert J, Jijie R, Boukherroub R, Szunerits S (2019) Entrapment of uropathogenic E. coli cells into ultra-thin sol-gel matrices on gold thin films: a low cost alternative for impedimetric bacteria sensing, Biosens. Bioelectron 124(125):161–166. https://doi.org/10.1016/j.bios.2018.10.029.
Jia M, Liu JJ, Zhang JH, Zhang HY (2019) An immunofiltration strip method based on the photothermal effect of gold nanoparticles for the detection of Escherichia coli O157:H7. Analyst 144:573–578. https://doi.org/10.1039/c8an01004h
Jiang Y, Zou S, Cao X (2017) A simple dendrimer-aptamer based microfluidic platform for E. coli O157:H7 detection and signal intensification by rolling circle amplification. Sensors Actuators B: Chem 251:976–984. https://doi.org/10.1016/j.snb.2017.05.146
Jin BR, Wang SR, Lin M, Jin Y, Zhang SJ, Cui XY, Gong Y, Li A, Xu F, Lu TJ (2017) Upconversion nanoparticles based FRET aptasensor for rapid and ultrasenstive bacteria detection. Biosens Bioelectron 90:525–533. https://doi.org/10.1016/j.bios.2016.10.029
Kaur H, Shorie M, Sharma M, Ganguli AK, Sabherwal P (2017) Bridged Rebar Graphene functionalized aptasensor for pathogenic E. coli O78:K80:H11 detection. Biosens Bioelectron 98:486–493. https://doi.org/10.1016/j.bios.2017.07.004
Khang J, Kim D, Chung KW, Lee JH (2016) Chemiluminescent aptasensor capable of rapidly quantifying Escherichia coli O157:H7. Talanta 147:177–183. https://doi.org/10.1016/j.talanta.2015.09.055
Kim HY, Ahn JK, Kim MI, Park KS, Park HG (2019) Rapid and label-free, electrochemical DNA detection utilizing the oxidase-mimicking activity of cerium oxide nanoparticles. Electrochem Commun 99:5–10. https://doi.org/10.1016/j.elecom.2018.12.008
Koo B, Yorita AM, Schmidt JJ, Monbouquette HG (2018) Amplification-free, sequence-specific 16S rRNA detection at 1 aM. Lab Chip 18:2291–2299. https://doi.org/10.1039/c8lc00452h
Kuss S, Couto RAS, Evans RM, Lavender H, Tang CC, Compton RG (2019) Versatile electrochemical sensing platform for bacteria. Anal Chem 91:4317–4322. https://doi.org/10.1021/acs.analchem.9b00326
Li TC, Zhu FJ, Guo W, Gu HX, Zhao J, Yan M, Liu SQ (2017a) Selective capture and rapid identification of E. coli O157: H7 by carbon nanotube multilayer biosensors and microfluidic chip-based LAMP. RSC Adv 7:30446–30452. https://doi.org/10.1039/c7ra04583b
Li Y, Xiong Y, Fang LC, Jiang LL, Huang H, Deng J, Liang WB, Zheng JS (2017b) An Electrochemical strategy using multifunctional nanoconjugates for efficient simultaneous detection of Escherichia coli O157: H7 and Vibrio cholerae O1. Theranostics 7:935–944. https://doi.org/10.7150/thno.17544
Li LY, Wang LL, Xu Q, Xu L, Liang W, Li Y, Ding M, Aldalbahi A, Ge ZL, Wang LH, Yan J, Lu N, Li J, Wen YL, Liu G (2018a) Bacterial analysis using an electrochemical DNA biosensor with poly-adenine-mediated DNA self-assembly. ACS Appl Mater Interfaces 10:6895–6903. https://doi.org/10.1021/acsami.7b17327
Li YX, Xie GM, Qiu JH, Zhou DD, Gou D, Tao YY, Li Y, Chen H (2018b) A new biosensor based on the recognition of phages and the signal amplification of organic-inorganic hybrid nanoflowers for discriminating and quantitating live pathogenic bacteria in urine. Sens Actuator B-Chem 258:803–812. https://doi.org/10.1016/j.snb.2017.11.155
Li F, Yu Z, Han X, Lai RY (2019) Electrochemical aptamer-based sensors for food and water analysis: a review. Anal Chim Acta 1051:1–23. https://doi.org/10.1016/j.aca.2018.10.058
Li T, Ou G, Chen X, Li Z, Hu R, Li Y, Yang Y, Liu M (2020) Naked-eye based point-of-care detection of E.coli O157: H7 by a signal-amplified microfluidic aptasensor. Anal Chim Acta 1130:20–28. https://doi.org/10.1016/j.aca.2020.07.031
Li Y, Liu HM, Huang H, Deng J, Fang LC, Luo J, Zhang S, Huang J, Liang WB, Zheng JS (2020) A sensitive electrochemical strategy via multiple amplification reactions for the detection of E. coli O157: H7. Biosens Bioelectron 147:111752. https://doi.org/10.1016/j.bios.2019.111752
Lin D, Li BW, Qi J, Ji XF, Yang SX, Wang WH, Chen LX (2020) Low cost fabrication of microfluidic paper-based analytical devices with water-based polyurethane acrylate and their application for bacterial detection. Sens Actuator B-Chem 303:127213. https://doi.org/10.1016/j.snb.2019.127213
Liu XB, Marrakchi M, Xu DW, Dong H, Andreescu S (2016) Biosensors based on modularly designed synthetic peptides for recognition, detection and live/dead differentiation of pathogenic bacteria. Biosens Bioelectron 80:9–16. https://doi.org/10.1016/j.bios.2016.01.041
Lu CX, Gao XX, Chen Y, Ren JT, Liu CB (2020) Aptamer-based lateral flow test strip for the simultaneous detection of Salmonella typhimurium, Escherichia coli O157:H7 and Staphylococcus aureus. Anal Lett 53:646–659. https://doi.org/10.1080/00032719.2019.1663528
Luo K, Ryu J, Seol I-H, Jeong K-B, You S-M, Kim Y-R (2019) Paper-based radial chromatographic immunoassay for the detection of pathogenic bacteria in milk. ACS Appl Mater Interfaces 11:46472–46478. https://doi.org/10.1021/acsami.9b16075
Luo K, Kim HY, Oh MH, Kim YR (2020) Paper-based lateral flow strip assay for the detection of foodborne pathogens: principles, applications, technological challenges and opportunities. Crit Rev Food Sci Nutr 60:157–170. https://doi.org/10.1080/10408398.2018.1516623
Maas M, Perold W, Dicks L (2017) Biosensors for the detection of Escherichia coli. Water SA 43:707–721. https://doi.org/10.4314/wsa.v43i4.17
Majdinasab M, Hayat A, Marty JL (2018) Aptamer-based assays and aptasensors for detection of pathogenic bacteria in food samples, TrAC. Trends Anal Chem 107:60–77. https://doi.org/10.1016/j.trac.2018.07.016
Malvano F, Pilloton R, Albanese D (2018) Sensitive detection of Escherichia coli O157:H7 in food products by impedimetric immunosensors. Sensors 18:2168. https://doi.org/10.3390/s18072168
Mathelie-Guinlet M, Cohen-Bouhacina T, Gammoudi I, Martin A, Beven L, Delville MH, Grauby-Heywang C (2019) Silica nanoparticles-assisted electrochemical biosensor for the rapid, sensitive and specific detection of Escherichia coli. Sens Actuator B-Chem 292:314–320. https://doi.org/10.1016/j.snb.2019.03.144
McConnell EM, Nguyen J, Li Y (2020) Aptamer-based biosensors for environmental monitoring. Front Chem 8:434. https://doi.org/10.3389/fchem.2020.00434
Miao P, Tang YG, Wang BD, Yin J, Ning LM (2015) Signal amplification by enzymatic tools for nucleic acids. Trac-Trends Anal Chem 67:1–15. https://doi.org/10.1016/j.trac.2014.12.006
Mo XY, Wu ZY, Huang JF, Zhao GY, Dou WC (2019) A sensitive and regenerative electrochemical immunosensor for quantitative detection of Escherichia coli O157:H7 based on stable polyaniline coated screen-printed carbon electrode and rGO-NR-Au@Pt. Anal Methods 11:1475–1482. https://doi.org/10.1039/c8ay02594k
Narang J, Mishra A, Pilloton R, Alekhya VV, Wadhwa S, Pundir CS, Khanuja M (2018) Development of MoSe2 nano-urchins as a sensing platform for a selective bio-capturing of Escherichia coli shiga toxin DNA. Biosensors-Basel 8:77. https://doi.org/10.3390/bios8030077
Nguyen QH, Kim MI (2020) Nanomaterial-mediated paper-based biosensors for colorimetric pathogen detection. Trends Anal Chem 132:116038. https://doi.org/10.1016/j.trac.2020.116038
Nguyen LT, Haney EF, Vogel HJ (2011) The expanding scope of antimicrobial peptide structures and their modes of action. Trends Biotechnol 29:464–472. https://doi.org/10.1016/j.tibtech.2011.05.001
Nguyen DQ, Ishiki K, Shiigi H (2018) Single cell immunodetection of Escherichia coli O157:H7 on an indium-tin-oxide electrode by using an electrochemical label with an organic-inorganic nanostructure. Microchim Acta 185:465. https://doi.org/10.1007/s00604-018-3001-5
Nguyen V-T, Song S, Park S, Joo C (2020) Recent advances in high-sensitivity detection methods for paper-based lateral-flow assay. Biosens Bioelectron 152:112015. https://doi.org/10.1016/j.bios.2020.112015
Ozkan-Ariksoysal D, Kayran YU, Yilmaz FF, Ciucu AA, David IG, David V, Hosgor-Limoncu M, Ozsoz M (2017) DNA-wrapped multi-walled carbon nanotube modified electrochemical biosensor for the detection of Escherichia coli from real samples. Talanta 166:27–35. https://doi.org/10.1016/j.talanta.2017.01.005
Pandey A, Gurbuz Y, Ozguz V, Niazi JH, Qureshi A (2017) Graphene-interfaced electrical biosensor for label-free and sensitive detection of foodborne pathogenic E. coli O157:H7. Biosens Bioelectron 91:225–231. https://doi.org/10.1016/j.bios.2016.12.041
Paniel N, Noguer T (2019) Detection of Salmonella in food matrices, from conventional methods to recent aptamer-sensing technologies. Foods 8:371. https://doi.org/10.3390/foods8090371
Pardoux E, Boturyn D, Roupioz Y (2020) Antimicrobial peptides as probes in biosensors detecting whole bacteria: a review. Molecules (basel, Switzerland) 25:1998. https://doi.org/10.3390/molecules25081998
Pei H, Li F, Wan Y, Wei M, Liu H, Su Y, Chen N, Huang Q, Fan C (2012) Designed diblock oligonucleotide for the synthesis of spatially isolated and highly hybridizable functionalization of DNA–gold nanoparticle nanoconjugates. J Am Chem Soc 134:11876–11879. https://doi.org/10.1021/ja304118z
Qiao ZH, Lei CY, Fu YC, Li YB (2017) An antimicrobial peptide-based colorimetric bioassay for rapid and sensitive detection of E-coli O157:H7. RSC Adv 7:15769–15775. https://doi.org/10.1039/c6ra28362d
Qiao ZH, Fu YC, Lei CY, Li YB (2020) Advances in antimicrobial peptides-based biosensing methods for detection of foodborne pathogens: a review. Food Control 112:107116. https://doi.org/10.1016/j.foodcont.2020.107116
Rajapaksha P, Elbourne A, Gangadoo S, Brown R, Cozzolino D, Chapman J (2019) A review of methods for the detection of pathogenic microorganisms. Analyst 144:396–411. https://doi.org/10.1039/C8AN01488D
Ranjbar S, Nejad MAF, Parolo C, Shahrokhian S, Merkoçi A (2019) Smart chip for visual detection of bacteria using the electrochromic properties of polyaniline. Anal Chem 91:14960–14966. https://doi.org/10.1021/acs.analchem.9b03407
Saad M, Faucher SP (2021) Aptamers and aptamer-coupled biosensors to detect water-borne pathogens. Front Microbiol 12:643797. https://doi.org/10.3389/fmicb.2021.643797
Saucedo NM, Gao YN, Pham T, Mulchandani A (2018) Lectin- and saccharide-functionalized nano-chemiresistor arrays for detection and identification of pathogenic bacteria infection. Biosensors-Basel 8:63. https://doi.org/10.3390/bios8030063
Schrader C, Schielke A, Ellerbroek L, Johne R (2012) PCR inhibitors—occurrence, properties and removal. J Appl Microbiol 113:1014–1026. https://doi.org/10.1111/j.1365-2672.2012.05384.x
Sedki M, Chen XY, Chen C, Ge X, Mulchandani A (2020) Non-lytic M13 phage-based highly sensitive impedimetric cytosensor for detection of coliforms. Biosens Bioelectron 148:111794. https://doi.org/10.1016/j.bios.2019.111794
Shahrokhian S, Ranjbar S (2018) Aptamer immobilization on amino-functionalized metal-organic frameworks: an ultrasensitive platform for the electrochemical diagnostic of Escherichia coli O157:H7. Analyst 143:3191–3201. https://doi.org/10.1039/c8an00725j
Shan X, Yamauchi T, Yamamoto Y, Niyomdecha S, Ishiki K, Le DQ, Shiigi H, Nagaoka T (2017) Spontaneous and specific binding of enterohemorrhagic Escherichia coli to overoxidized polypyrrole-coated microspheres. Chem Commun 53:3890–3893. https://doi.org/10.1039/C7CC00244K
Shan X, Yamauchi T, Yamamoto Y, Shiigi H, Nagaoka T (2018) A rapid and specific bacterial detection method based on cell-imprinted microplates. Analyst 143:1568–1574. https://doi.org/10.1039/C7AN02057K
Sharif S, Wang YX, Ye ZZ, Wang Z, Qiu QM, Ying SN, Ying YB (2019) A novel impedimetric sensor for detecting LAMP amplicons of pathogenic DNA based on magnetic separation. Sens Actuator B-Chem 301:127051. https://doi.org/10.1016/j.snb.2019.127051
Sharifi S, Vahed SZ, Ahmadian E, Dizaj SM, Eftekhari A, Khalilov R, Ahmadi M, Hamidi-Asl E, Labib M (2020) Detection of pathogenic bacteria via nanomaterials-modified aptasensors. Biosens Bioelectron 150:111933. https://doi.org/10.1016/j.bios.2019.111933
Shoaie N, Forouzandeh M, Omidfar K (2018) Voltammetric determination of the Escherichia coli DNA using a screen-printed carbon electrode modified with polyaniline and gold nanoparticles. Microchim Acta 185:217. https://doi.org/10.1007/s00604-018-2749-y
SiavashMoakhar R, AbdelFatah T, Sanati A, Jalali M, Flynn SE, Mahshid SS, Mahshid S (2020) A nanostructured gold/graphene microfluidic device for direct and plasmonic-assisted impedimetric detection of bacteria. ACS Appl Mater Interfaces 12:23298–23310. https://doi.org/10.1021/acsami.0c02654
Singh S, Moudgil A, Mishra N, Das S, Mishra P (2019) Vancomycin functionalized WO3 thin film-based impedance sensor for efficient capture and highly selective detection of Gram-positive bacteria. Biosens Bioelectron 136:23–30. https://doi.org/10.1016/j.bios.2019.04.029
Song CM, Li JW, Liu JX, Liu Q (2016a) Simple sensitive rapid detection of Escherichia coli O157:H7 in food samples by label-free immunofluorescence strip sensor. Talanta 156:42–47. https://doi.org/10.1016/j.talanta.2016.04.054
Song CM, Liu C, Wu SY, Li HL, Guo HQ, Yang B, Qiu S, Li JW, Liu L, Zeng HJ, Zhai XZ, Liu Q (2016b) Development of a lateral flow colloidal gold immunoassay strip for the simultaneous detection of Shigella boydii and Escherichia coli O157:H7 in bread, milk and jelly samples. Food Control 59:345–351. https://doi.org/10.1016/j.foodcont.2015.06.012
Song S-H, Gao Z-F, Guo X, Chen G-H (2019) Aptamer-based detection methodology studies in food safety. Food Anal Methods 12:966–990. https://doi.org/10.1007/s12161-019-01437-3
Soutschek J, Akinc A, Bramlage B, Charisse K, Constien R, Donoghue M, Elbashir S, Geick A, Hadwiger P, Harborth J, John M, Kesavan V, Lavine G, Pandey RK, Racie T, Rajeev KG, Röhl I, Toudjarska I, Wang G, Wuschko S, Bumcrot D, Koteliansky V, Limmer S, Manoharan M, Vornlocher H-P (2004) Therapeutic silencing of an endogenous gene by systemic administration of modified siRNAs. Nature 432:173–178. https://doi.org/10.1038/nature03121
Sun XC, Lei C, Guo L, Zhou Y (2016) Separable detecting of Escherichia coli O157H:H7 by a giant magneto-resistance-based bio-sensing system. Sens Actuator B-Chem 234:485–492. https://doi.org/10.1016/j.snb.2016.04.183
Tang YY, Li Z, Luo QH, Liu JQ, Wu JM (2016) Bacteria detection based on its blockage effect on silicon nanopore array. Biosens Bioelectron 79:715–720. https://doi.org/10.1016/j.bios.2015.12.109
Templier V, Roux A, Roupioz Y, Livache T (2016) Ligands for label-free detection of whole bacteria on biosensors: a review. Trac-Trends Anal Chem 79:71–79. https://doi.org/10.1016/j.trac.2015.10.015
Thakur B, Zhou GH, Chang JB, Pu HH, Jin B, Sui XY, Yuan XC, Yang CH, Magruder M, Chen JH (2018) Rapid detection of single E. coli bacteria using a graphene-based field-effect transistor device. Biosens Bioelectron 110:16–22. https://doi.org/10.1016/j.bios.2018.03.014
Thévenot DR, Toth K, Durst RA, Wilson GS (2001) Electrochemical biosensors: recommended definitions and classification. Biosens Bioelectron 16:121–131. https://doi.org/10.1016/S0956-5663(01)00115-4
Tian Y, Liang T, Zhu P, Chen Y, Chen W, Du L, Wu C, Wang P (2019) Label-free detection of E. coli O157:H7 DNA using light-addressable potentiometric sensors with highly oriented ZnO nanorod arrays. Sensors (Basel, Switzerland) 19:5473. https://doi.org/10.3390/s19245473
Trotter M, Borst N, Thewes R, von Stetten F (2020) Review: electrochemical DNA sensing—principles, commercial systems, and applications. Biosens Bioelectron 154:112069. https://doi.org/10.1016/j.bios.2020.112069
Tsougeni K, Kaprou G, Loukas CM, Papadakis G, Hamiot A, Eck M, Rabus D, Kokkoris G, Chatzandroulis S, Papadopoulos V, Dupuy B, Jobst G, Gizeli E, Tserepi A, Gogolides E (2020) Lab-on-Chip platform and protocol for rapid foodborne pathogen detection comprising on-chip cell capture, lysis, DNA amplification and surface-acoustic-wave detection. Sens Actuator B-Chem 320:128345. https://doi.org/10.1016/j.snb.2020.128345
Umesha S, Manukumar HM (2018) Advanced molecular diagnostic techniques for detection of food-borne pathogens: current applications and future challenges. Crit Rev Food Sci Nutr 58:84–104. https://doi.org/10.1080/10408398.2015.1126701
Wan JZ, Ai JJ, Zhang YH, Geng XH, Gao Q, Cheng ZL (2016) Signal-off impedimetric immunosensor for the detection of Escherichia coli O157:H7. Sci Rep 6:19806. https://doi.org/10.1038/srep19806
Wang F, Lu C-H, Willner I (2014) From cascaded catalytic nucleic acids to enzyme–DNA nanostructures: controlling reactivity, sensing, logic operations, and assembly of complex structures. Chem Rev 114:2881–2941. https://doi.org/10.1021/cr400354z
Wang HX, Zhao YW, Bie ST, Suo TC, Jia GC, Liu BS, Ye RP, Li Z (2019a) Development of an electrochemical biosensor for rapid and effective detection of pathogenic Escherichia coli in licorice extract. Appl Sci Basel 9:295. https://doi.org/10.3390/app9020295
Wang WW, Han X, Chu LQ (2019b) Polyadenine-mediated immobilization of aptamers on a gold substrate for the direct detection of bacterial pathogens. Anal Sci 35:967–972. https://doi.org/10.2116/analsci.19P110
Wang KY, Bu SJ, Ju CJ, Han Y, Ma CY, Liu WS, Li ZY, Li CT, Wan JY (2019c) Disposable syringe-based visual immunotest for pathogenic bacteria based on thecatalase mimicking activity of platinum nanoparticle-concanavalin A hybrid nanoflowers. Microchim Acta 186:57. https://doi.org/10.1007/s00604-018-3133-7
Wang CH, Wu JJ, Lee GB (2019d) Screening of highly-specific aptamers and their applications in paper-based microfluidic chips for rapid diagnosis of multiple bacteria. Sens Actuator B-Chem 284:395–402. https://doi.org/10.1016/j.snb.2018.12.112
Wang Z, Yao X, Zhang Y, Wang R, Ji Y, Sun J, Zhang D, Wang J (2020a) Functional nanozyme mediated multi-readout and label-free lateral flow immunoassay for rapid detection of Escherichia coli O157:H7. Food Chem 329:127224. https://doi.org/10.1016/j.foodchem.2020.127224
Wang CN, Gao X, Wang S, Liu YQ (2020b) A smartphone-integrated paper sensing system for fluorescent and colorimetric dual-channel detection of foodborne pathogenic bacteria. Anal Bioanal Chem 412:611–620. https://doi.org/10.1007/s00216-019-02208-z
Wang L, Shen X, Wang T, Chen P, Qi N, Yin B-C, Ye B-C (2020c) A lateral flow strip combined with Cas9 nickase-triggered amplification reaction for dual food-borne pathogen detection. Biosens Bioelectron 165:112364. https://doi.org/10.1016/j.bios.2020.112364
Wen YL, Wang LL, Xu L, Li LY, Ren SZ, Cao CM, Jia NQ, Aldalbahi A, Song SP, Shi JY, Xia JY, Liu G, Zuo XL (2016) Electrochemical detection of PCR amplicons of Escherichia coli genome based on DNA nanostructural probes and polyHRP enzyme. Analyst 141:5304–5310. https://doi.org/10.1039/c6an01435f
Wilson D, Materon EM, Ibanez-Redin G, Faria RC, Correa DS, Oliveira ON (2019) Electrical detection of pathogenic bacteria in food samples using information visualization methods with a sensor based on magnetic nanoparticles functionalized with antimicrobial peptides. Talanta 194:611–618. https://doi.org/10.1016/j.talanta.2018.10.089
Wu G, Dai Z, Tang X, Lin Z, Lo PK, Meyyappan M, Lai KWC (2017) Graphene field-effect transistors for the sensitive and selective detection of Escherichia coli using pyrene-tagged DNA aptamer. Adv Healthc Mater 6:1700736. https://doi.org/10.1002/adhm.201700736
Wu RR, Ma Y, Pan JM, Lee SH, Liu JX, Zhu HJ, Gu RX, Shea KJ, Pan GQ (2018) Efficient capture, rapid killing and ultrasensitive detection of bacteria by a nano-decorated multi-functional electrode sensor. Biosens Bioelectron 101:52–59. https://doi.org/10.1016/j.bios.2017.10.003
Xu M, Wang RH, Li YB (2016a) Rapid detection of Escherichia coli O157:H7 and Salmonella typhimurium in foods using an electrochemical immunosensor based on screen-printed interdigitated microelectrode and immunomagnetic separation. Talanta 148:200–208. https://doi.org/10.1016/j.talanta.2015.10.082
Xu M, Wang RH, Li YB (2016b) An electrochemical biosensor for rapid detection of E. coli O157:H7 with highly efficient bifunctional glucose oxidase-polydopamine nanocomposites and Prussian blue modified screen-printed interdigitated electrodes. Analyst 141:5441–5449. https://doi.org/10.1039/c6an00873a
Yang Z, Liu Y, Lei C, Sun XC, Zhou Y (2016a) Ultrasensitive detection and quantification of E-coli O157:H7 using a giant magnetoimpedance sensor in an open-surface microfluidic cavity covered with an antibody-modified gold surface. Microchim Acta 183:1831–1837. https://doi.org/10.1007/s00604-016-1818-3
Yang MH, Jeong SW, Chang SJ, Kim KH, Jang M, Kim CH, Bae NH, Sim GS, Kang T, Lee SJ, Choi BG, Lee KG (2016b) Flexible and disposable sensing platforms based on newspaper. ACS Appl Mater Interfaces 8:34978–34984. https://doi.org/10.1021/acsami.6b10298
Yang T, Huang H, Zhu F, Lin Q, Zhang L, Liu J (2016c) Recent progresses in nanobiosensing for food safety analysis. Sensors 16:1118. https://doi.org/10.3390/s16071118
Yao L, Wang L, Huang FC, Cai GZ, Xi XG, Lin JH (2018) A microfluidic impedance biosensor based on immunomagnetic separation and urease catalysis for continuous-flow detection of E-coli O157:H7. Sens Actuator B-Chem 259:1013–1021. https://doi.org/10.1016/j.snb.2017.12.110
Ye WW, Chen T, Mao YJ, Tian F, Sun PL, Yang M (2017) The effect of pore size in an ultrasensitive DNA sandwich-hybridization assay for the Escherichia coli O157:H7 gene based on the use of a nanoporous alumina membrane. Microchim Acta 184:4835–4844. https://doi.org/10.1007/s00604-017-2530-7
You ZH, Qiu QM, Chen HY, Feng YY, Wang X, Wang YX, Ying YB (2020a) Laser-induced noble metal nanoparticle-graphene composites enabled flexible biosensor for pathogen detection. Biosens Bioelectron 150:111896. https://doi.org/10.1016/j.bios.2019.111896
You S-M, Luo K, Jung J-Y, Jeong K-B, Lee E-S, Oh M-H, Kim Y-R (2020b) Gold nanoparticle-coated starch magnetic beads for the separation, concentration, and SERS-based detection of E. coli O157:H7. ACS Appl Mater Interfaces 12:18292–18300. https://doi.org/10.1021/acsami.0c00418
YuhanaAriffin E, Heng LY, Tan LL, Abd Karim NH, Hasbullah SA (2020) A highly sensitive impedimetric DNA biosensor based on hollow silica microspheres for label-free determination of E. coli. Sensors (Basel, Switzerland) 20:1279. https://doi.org/10.3390/s20051279
Zaouri N, Cui ZF, Peinetti AS, Lu Y, Hong PY (2019) DNAzyme-based biosensor as a rapid and accurate verification tool to complement simultaneous enzyme-based media for E. coli detection. Environ Sci Wat Res Technol 5:2260–2268. https://doi.org/10.1039/c9ew00795d
Zhang XN, Shen JZ, Ma HL, Jiang YX, Huang CY, Han E, Yao BS, He YY (2016) Optimized dendrimer-encapsulated gold nanoparticles and enhanced carbon nanotube nanoprobes for amplified electrochemical immunoassay of E. coli in dairy product based on enzymatically induced deposition of polyaniline. Biosens Bioelectron 80:666–673. https://doi.org/10.1016/j.bios.2016.02.043
Zhang XN, Jiang YX, Huang CY, Shen JZ, Dong XY, Chen GW, Zhang W (2017) Functionalized nanocomposites with the optimal graphene oxide/Au ratio for amplified immunoassay of E. coli to estimate quality deterioration in dairy product. Biosens Bioelectron 89:913–918. https://doi.org/10.1016/j.bios.2016.09.098
Zhang J, Oueslati R, Cheng C, Zhao L, Chen J, Almeida R, Wu J (2018a) Rapid, highly sensitive detection of Gram-negative bacteria with lipopolysaccharide based disposable aptasensor. Biosens Bioelectron 112:48–53. https://doi.org/10.1016/j.bios.2018.04.034
Zhang JL, Wang JJ, Zhang XQ, He FJ (2018b) Rapid detection of Escherichia coli based on 16S rDNA nanogap network electrochemical biosensor. Biosens Bioelectron 118:9–15. https://doi.org/10.1016/j.bios.2018.07.041
Zhang Y, Zhu LQ, He PG, Zi FT, Hu XZ, Wang QJ (2019a) Sensitive assay of Escherichia coli in food samples by microchip capillary electrophoresis based on specific aptamer binding strategy. Talanta 197:284–290. https://doi.org/10.1016/j.talanta.2019.01.040
Zhang X, Xie GM, Gou D, Luo P, Yao Y, Chen H (2019b) A novel enzyme-free electrochemical biosensor for rapid detection of Pseudomonas aeruginosa based on high catalytic Cu-ZrMOF and conductive Super P. Biosens Bioelectron 142:111486. https://doi.org/10.1016/j.bios.2019.111486
Zhang XZ, Wang XC, Yang QQ, Jiang XY, Li Y, Zhao J, Qu KM (2020) Conductometric sensor for viable Escherichia coli and Staphylococcus aureus based on magnetic analyte separation via aptamer. Microchim Acta 187:43. https://doi.org/10.1007/s00604-019-3880-0
Zhao YX, Chen F, Li Q, Wang LH, Fan CH (2015) Isothermal amplification of nucleic acids. Chem Rev 115:12491–12545. https://doi.org/10.1021/acs.chemrev.5b00428
Zhao Y-W, Wang H-X, Jia G-C, Li Z (2018) Application of aptamer-based biosensor for rapid detection of pathogenic Escherichia coli. Sensors 18:2518. https://doi.org/10.3390/s18082518
Zhao YC, Sarkar A, Wang XF (2020a) Peptide nucleic acid based tension sensor for cellular force imaging with strong DNase resistance. Biosens Bioelectron 150:111959. https://doi.org/10.1016/j.bios.2019.111959
Zhao YA, Zeng DX, Yan C, Chen W, Ren JL, Jiang Y, Jiang LY, Xue F, Ji DJ, Tang F, Zhou MQ, Dai JJ (2020b) Rapid and accurate detection of Escherichia coli O157:H7 in beef using microfluidic wax-printed paper-based ELISA. Analyst 145:3106–3115. https://doi.org/10.1039/d0an00224k
Zheng LB, Wan Y, Qi P, Sun Y, Zhang D, Yu LM (2017) Lectin functionalized ZnO nanoarrays as a 3D nano-biointerface for bacterial detection. Talanta 167:600–606. https://doi.org/10.1016/j.talanta.2017.03.007
Zheng L, Cai G, Wang S, Liao M, Li Y, Lin J (2019) A microfluidic colorimetric biosensor for rapid detection of Escherichia coli O157:H7 using gold nanoparticle aggregation and smart phone imaging. Biosens Bioelectron 124–125:143–149. https://doi.org/10.1016/j.bios.2018.10.006
Zhong JL, Zhao XH (2018) Isothermal amplification technologies for the detection of foodborne pathogens. Food Anal Methods 11:1543–1560. https://doi.org/10.1007/s12161-018-1177-2
Zhong M, Yang L, Yang H, Cheng C, Deng WF, Tan YM, Xie QJ, Yao SZ (2019) An electrochemical immunobiosensor for ultrasensitive detection of Escherichia coil O157:H7 using CdS quantum dots-encapsulated metal-organic frameworks as signal-amplifying tags. Biosens Bioelectron 126:493–500. https://doi.org/10.1016/j.bios.2018.11.001
Zhou Y, Marar A, Kner P, Ramasamy RP (2017) Charge-directed immobilization of bacteriophage on nanostructured electrode for whole-cell electrochemical biosensors. Anal Chem 89:5735–5742. https://doi.org/10.1021/acs.analchem.6b03751
Zhou C, Zou HM, Li M, Sun CJ, Ren DX, Li YX (2018a) Fiber optic surface plasmon resonance sensor for detection of E. coli O157:H7 based on antimicrobial peptides and AgNPs-rGO. Biosens Bioelectron 117:347–353. https://doi.org/10.1016/j.bios.2018.06.005
Zhou X, Zhang PB, Lv FT, Liu LB, Wang S (2018b) Photoelectrochemical strategy for discrimination of microbial pathogens using conjugated polymers. Chem Asian J 13:3469–3473. https://doi.org/10.1002/asia.201800783
Zhou YY, Fang WN, Lai KQ, Zhu YH, Bian XJ, Shen JL, Li Q, Wang LH, Zhang WJ, Yan J (2019) Terminal deoxynucleotidyl transferase (TdT)-catalyzed homo-nucleotides-constituted ssDNA: Inducing tunable-size nanogap for core-shell plasmonic metal nanostructure and acting as Raman reporters for detection of Escherichia coli O157:H7. Biosens Bioelectron 141:111419. https://doi.org/10.1016/j.bios.2019.111419
Zhu Y, Jović M, Lesch A, Tissières Lovey L, Prudent M, Pick H, Girault HH (2018) Immuno-affinity amperometric detection of bacterial infections. Angew Chem Int Ed 57:14942–14946. https://doi.org/10.1002/anie.201808666
Zou Y, Liang J, She Z, Kraatz H-B (2019) Gold nanoparticles-based multifunctional nanoconjugates for highly sensitive and enzyme-free detection of E.coli K12. Talanta 193:15–22. https://doi.org/10.1016/j.talanta.2018.09.068
Funding
This work was financially supported by the National Key Research and Development Project of China (No. 2019YFC1605305), Hunan Provincial Natural Science Foundation of China (No. 2019JJ50998), National Natural Science Foundation of China (No. 31972173), Scientific Research Fund of Hunan Provincial Education Department (18B165), and Program for Science & Technology Innovation Platform/Talents of Hunan Province (2019TP1029, 2018XK2006).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Conflict of Interest
Ying Huang declares that she has no conflict of interest. Zhipeng Su declares that he has no conflict of interest. Wang Li declares that he has no conflict of interest. Jiali Ren declares that she has no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Ying Huang and Zhipeng Su contributed equally to this work.
Rights and permissions
About this article
Cite this article
Huang, Y., Su, Z., Li, W. et al. Recent Progresses on Biosensors for Escherichia coli Detection. Food Anal. Methods 15, 338–366 (2022). https://doi.org/10.1007/s12161-021-02129-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-021-02129-7