Abstract
Deoxynivalenol (DON) is a protein synthesis inhibitor produced by the Fusarium species, which frequently contaminates grains used for human or animal consumption, thereby necessitating their effective monitoring and control. Antibody-based immunoassays provide efficient and cheap methods for screening this mycotoxin. To achieve high sensitivity and avoid enzymatic reaction, a new highly sensitive monoclonal antibody (MAb) and a fluorescent probe consisting of amino-functionalized core/multishell quantum dots (QDs), CdSe/CdS/Cd0.5Zn0.5S/ZnS, bio conjugated to 3-HS-DON-BSA were prepared; then, a sensitive and reliable direct competitive fluorescent-labeled immunosorbent assay (dcFLISA) was developed. Under optimized conditions, the 50% inhibition concentration (IC50) value of the dcFLISA was 5.6 μg/kg with limit of detection of 12.2 μg/kg in maize samples. The recoveries ranged from 77.3 to 84.3% with coefficients of variation (CV%) values below 10%. Good correlation between the dcFLISA and HPLC-MS/MS results in the naturally contaminated maize demonstrated that the dcFLISA was a rapid and sensitive with high-performance and reliable method for the detection of DON. The potential of this method to function as a simple and efficient strategy for small molecule on-site screening might be applied in the future.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The food contamination by mycotoxins is a serious problem all over the world and represents an important risk factor for human and animal health, as up to 70% of the world’s crop production may be contaminated (Streit et al. 2013). Deoxynivalenol (DON) is a common mycotoxin produced by some Fusarium species such as Fusarium graminearum and Fusarium culmorum (Li et al. 2015). Corn is an important raw material for animals, which often contaminated with DON in European Union (EU), Asian-Pacific region, and USA (Binder et al. 2007). One study showed that about 30% of 50 random samples of whole corn were contaminated with DON with the levels ranging from 340 to 1072 μg/kg in California (Kroutgreenberg et al. 2013). Consumption of those DON-contaminated corn by daily cattle could lead to serious illnesses and immunosuppressive effects, and it was ultimately hazard to people’s health through the milk food chain. Therefore, in great demand for monitoring and controlling the toxic effect of DON, the maximum residue limit (MRL) for DON was set at 1750 μg/kg in unprocessed maize. The detection of DON in corn fed to dairy cattle is a key step for implementation of prevention and control strategies. Thus, many analysis techniques have been developed and applied in DON detection, including high-performance liquid chromatography with ultraviolet or mass spectrometric detection detector (Yang et al. 2013; Hickert et al. 2015) and immunoassay (Li et al. 2012; Kim et al. 2014; Song et al. 2014; Li et al. 2015).
Among these techniques, antibodies-based immunoassay methods, i.e., enzyme-linked immunosorbent assay (ELISA) (Li et al. 2012) and lateral flow immunoassay (LFA) (Kim et al. 2014; Song et al. 2014), have formed alternative trends to instrumental methods in large-scale monitoring programs due to its cheap, rapid, and sensitive. Conventional ELISA, mainly indirect competitive ELISA (icELISA), always requires laborious, time-consuming incubation and washing steps. Moreover, enzyme-based labels in ELISA always suffer from the instability due to their denaturation and degradation (Zhang et al. 2014). And LFA was mainly used as a convenient qualitative tool for on-site screening (Kim et al. 2014; Yu et al. 2015). Compared with icELISA and LFA, direct competitive fluorescent-labeled immunosorbent assay (dcFLISA) using fluorescence materials as immune-probes could be analyzed quantitatively with wide linear working range and overcome those disadvantages. Thus, a sensitive dcFLISA for rapid and quantitative detection of DON is necessary.
To improve the sensitivity of dcFLISA, antibody with high affinity and fluorescence probe are two restrictive factors. Our team has produced a monoclonal antibody (MAb) against DON with IC50 of 61.1 μg/L (Li et al. 2012), lacking sensitivity for development of a highly sensitive dcFLISA. Thus, a MAb with higher sensitivity against DON is urgently needed. As we all known, organic dyes and fluorescence were always used as fluorescein-tracers; however, these fluorophores might suffer from photo bleaching and low emission intensity, which seriously compromised sensitivity and stability of immunoassay (Yang et al. 2011). Recently, quantum dots (QDs) as fluorescent labels in bioassays have attracted extensive interests due to their advantageous features, including excellent photostability, broad excitation spectrum, narrow fluorescent emission spectrum, and larger Stoke’s shift compared with those organic fluorophores (Algar et al. 2011; Beloglazova et al. 2012; Garcia-Fernandez et al. 2014; Speranskaya et al. 2014; Zhang et al. 2014; Duan et al. 2015). Core/multishell QDs, such as CdTe/CdS/ZnS and CdSe/CdS/Cd0.5Zn0.5S/ZnS, have been the most intensively explored in recent years due to its high fluorescence quality and favorable biocompatibility (He et al. 2008; Wei et al. 2014; Ma et al. 2016). In addition, CdSe/CdS/Cd0.5Zn0.5S/ZnS has been synthesized successfully and applied in chemiluminescence resonance energy transfer and LFA for detection of small molecular in our recent study (Ma et al. 2016; Zhang et al. 2017). Moreover, the dcFLISA only needs one step for detection, which saves time and costs by avoiding enzymatic reaction compared with icELISA (Garcia-Fernandez et al. 2014; Speranskaya et al. 2014a; Zhang et al. 2014). Therefore, the aim of this work was to develop one-step dcFLISA based on core/multishell QDs as probes to quantitative analysis of DON in maize. And the practicality and reliability of the dcFLISA were further confirmed by HPLC-MS/MS.
Materials and Methods
Materials
DON, 3-acetyl-deoxynivalenol (3-Ac-DON) or 15-acetyl-deoxynivalenol (15-Ac-DON), T-2 toxin, HT-2 toxin, neosolaniol (NEO), nivalenol (NIV), keyhole limpet hemocyanin (KLH), bovine serum albumin (BSA), Freund’s incomplete adjuvant (FIA), Freund’s complete adjuvant (FCA), PEG1500, hypoxanthine aminopterin thymidine (HAT), and a mouse MAb isotyping kit were obtained from Sigma-Aldrich (St. Louis. MO, U.SA). Peroxidase-conjugated goat antimouse IgG was purchased from Jackson ImmunoResearch Laboratories, Inc. (West Grove, PA, USA). 1-[3-(Dimethylamino) propyl]-3 ethylcarbodiimide hydrochloride (EDC∙HCl), carbonyldiimidazole, O-carboxymethyl oxime (CMO), succinic anhydrideand (HS), 1-butaneboronic acid, and N-hydroxysuccinimide (NHS) were supplied by Aladdin Chemistry Co. Ltd. (Shanghai, China). 1,2-Distearoyl-sn-glycero-3-phosphoethanolamine-N-[amino(polyethylene glycol)-2000] (DSPE-PEG-NH2) was supplied by Xi’an Ruixai Biologica Technology Co., Ltd. (Xi’an, China). The DSPE-PEG-NH2-modified CdSe/CdS/Cd0.5Zn0.5S/ZnS were previously prepared by our group (Ma et al. 2016).
Apparatus
FLISA and icELISA were performed employing a Spectramax M5 microplate reader acquired from Molecular Devices (Sunnyvale, CA, USA) to measure fluorescence intensity (FI) and optical density (OD450) signals.
Synthesis of Haptens of 3-HS-DON and 8-CMO-DON
The 3-HS-DON (Fig. 1) was synthesized by protection of the C7- and C15-hydroxyls of DON with 1-butylboronic acid, esterification at the C3-position with succinic anhydride, and then removal of the boronate ester as described previously (Casale et al. 1988). Briefly, 34 mg of 1-butaneboronic acid and 10 mg DON were dissolved in 5 mL of pyridine, and the mixture was stirred for 6 h at 35 °C. Subsequently, the mixture was heated to 90 °C, added to 10 mg HS, and reacted for 3 h. Then, the reaction mixtures were evaporated to dryness under N2, and the residue dissolved in 0.1 mL of methanol. After the silica gel column separation (chloroform/methanol = 5:1 plus 1.5% acetic acid as developing solution), the pure DON-3-HS was obtained.
The 8-CMO-DON was synthesized by reaction of DON with CMO (Fig. 1) as described previously (Casale et al. 1988; Sanders et al. 2014). Briefly, 5 mg DON and 6 mg CMO were dissolved in 5 mL of pyridine and heated at 80 °C for 4 h. Pyridine was removed under N2; the residue was dissolved in 5 mL of distilled water (pH adjusted to 8 with sodium bicarbonate). Unchanged DON was removed from the water phase by ethyl acetate. The aqueous phase was adjusted to pH = 3 by the addition of 1 M HCl and extracted three times with 5 mL of ethyl acetate. The extract was dried under vacuum, and residue was the hapten 8-CMO-DON.
Preparation of Hapten-Carrier Protein Conjugates
The hapten 3-HS-DON and 8-CMO-DON were conjugated to carrier protein (Fig. 1) using the active ester method (An et al. 2016; Zhang et al. 2016; Dong et al. 2017; Kong et al. 2017c; Kong et al. 2017b). 3-HS-DON or 8-CMO-DON (2 mg), 8 mg of EDC, and 6 mg of NHS were dissolved in 0.2 mL of dimethylformamide and stirred for 12 h at room temperature. Then, the reaction solution was added into 10 mg KLH (or 20 mg of BSA) in 10 mL 0.01 M phosphate-buffered saline (PBS, pH 7.4), and the mixture was stirred at 4 °C overnight. The conjugates were purified by dialysis against PBS and stored at − 20 °C.
Preparation of the Anti-DON Monoclonal Antibody
Female Balb/c mice were immunized with the immunogens (3-HS-DON-KLH and 8-CMO-DON-KLH) as described previously (Zhang et al. 2016). Each mouse was immunized three times with 100 μg immunogens at 3-week intervals. Seven days after the third vaccination, the antiserum was collected and characterized using indirect ELISA as described previously (Jiang et al. 2012). The mouse that produced a high titer of antibodies was sacrificed for fusion.
Spleen cells were fused with SP2/0 using PEG1500, and HAT was added for the selection of fused cells (Kong et al. 2017a; Kong et al. 2017d). Culture supernatant was screened by indirect ELISA and icELISA (Lee et al. 2013; Peng et al. 2016b; Wang et al. 2016). The cells with high affinity for the free DON were subcloned three times by limiting dilution method. Ascites fluids were produced and purified by the ammonium sulfate precipitation. To evaluate the specificity of the MAb, a set of DON analogs were used to calculate the cross reactivity (CR) values (Sun et al. 2010; Sanders et al. 2014; Peng et al. 2016a).
Synthesis and Characterization of the Fluorescence Probe: 3-HS-DON-BSA-QDs Bioconjugate
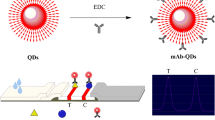
The highly luminescent core/multishell QDs, DSPE-PEG-NH2-modified CdSe/CdS/Cd0.5Zn0.5S/ZnS, were prepared as described in our recently reported (Speranskaya et al. 2014; Ma et al. 2016). Then, amino-functionalized of the QDs was conjugated with carboxyl groups from the antigen of 3-HS-DON-BSA by carbodiimide chemistry as described previously (Sun et al. 2010; Garcia-Fernandez et al. 2014; Speranskaya et al. 2014). Briefly, 0.4 mL of DSPE-PEG-NH2-modified CdSe/CdS/Cd0.5Zn0.5S/ZnS QDs, 0.5 mg of EDC, and 0.5 mg of NHS were added into 2 mL of morpholinoethanesulfonic acid (50 mM, pH 6.0). The mixture was reacted at room temperature for 30 min. An amount of 0.5 mg of 3-HS-DON-BSA was added and reacted for another 2 h. To terminate the reaction, 0.1 mL of 2% BSA was added for another 30 min. The bio conjugation of 3-HS-DON-BSA-QDs was purified by ultrafiltration using a 100 kDa membrane filter (Garcia-Fernandez et al. 2014). And the final volume was maintained at 1 mL. The characterization of the bio conjugate was carried out by fluorescence measurements and agarose gel electrophoresis.
Protocol of dcFLISA
The principle of dcFLISA was based on the competitive binding of free DON and bioconjugate of 3-HS-DON-BSA-QDs to immobilized anti-DON MAb (Fig. 2). The black costar 96-well microplates with high-binding surfaces were coated with 100 μL/well of 2-μg/mL MAb solution, and incubated at 4 °C for 8 h. Then, discarded the solution and blocked with 200 μL of 1% BSA in PBS (blocking buffer) at 37 °C for 1 h. After blocking buffer was discarded, 50 μL/well of standard solution (or sample extraction) and 50 μL/well of 3-HS-DON-BSA-QDs (diluted 400-fold in PBS) were added for 30 min at 37 °C. The plates were washed three times to remove unbound 3-HS-DON-BSA-QDs. The fluorescence of QDs from the 3-HS-DON-BSA-QDs recognized by anti-DON MAb was measured by Spectramax M5 microplate reader.
Analysis of Maize Samples
DON-free maize samples were supplied by the National Reference Laboratory for Veterinary Drug Residues (Beijing, China). The maize samples were finely milled using a knife mill Grindomix GM 200 (Retsch, Haan, Germany) and homogenized to obtain a representative samples. Then, 5 g of the maize flour was weighed into 50 mL polypropylene centrifuge tubes, and 25 mL of methanol/PBS (40:60, v/v) was added to each sample for extraction (Li et al. 2016). The mixture was vortexed for 3 min and centrifuged at 3000g for 5 min. The supernatant was diluted by 0.01 M PBS (pH 7.4) for analysis by the developed dcFLISA. A total of 20 blank samples were measured by the development of dcFLISA as a control. The limit of detection (LOD) was calculated using the following equation:
In addition, samples of naturally contaminated maize already detected by HPLC-MS/MS (Monbaliu et al. 2010) were analyzed by the developed dcFLISA.
Results and Discussion
Preparation of the Anti-DON Monoclonal Antibody
DON is a small molecule; it needs to be coupled with a carrier protein to elicit an immune response. There are three active sites of DON for coupling to carrier protein, C3, C15, and C8 in the previous references (Casale et al. 1988; Mills et al. 1990; Maragos and McCormick 2000; Li et al. 2012; Lee et al. 2013; Sanders et al. 2014). C7 was not selected for conjugation due to its low chemical activity. Although DON-15-hemiglutarate could be obtained by selectively removing the 3-Ac group from 3-Ac-DON-15-hemiglutarate (Sinha et al. 1995), unfortunately we failed. Thus, 3-HS-DON and 8-CMO-DON were synthesized as haptens for conjugating with carrier protein in this study. The molecular ion peaks (m/z) of 3-HS-DON and 8-CMO-DON were 395.1284 [m-H]− and 386.1026 [m-H]−, respectively, suggesting that both haptens were synthesized successfully. To elicit a better immune response, KLH was selected as the carrier protein for preparing immunogens and BSA was served as coating antigens (Goodrow and Hammock 1998). The haptens-to-BSA were identified by using matrix-assisted laser desorption time-of-flight mass spectrometry (MALDI-TOF-MS). The haptens-to-BSA molar ratios of 3-HS-DON-BSA and 8-CMO-DON-BSA were 9.5 and 6.7, respectively (Fig. 3).
The mice antisera were collected 7 days after the third immunization and characterized by indirect ELISA. Both 3-HS-DON-KLH and 8-CMO-DON-KLH received a better immunoreaction (Table 1). No. 1 from 3-HS-DON-KLH and No. 4 from 8-CMO-DON-KLH were sacrificed for fusion due to higher titer. After extensive screening and subclone, one cell line from No. 1 of 3-HS-DON-KLH, named 12E8, was obtained. The mouse MAb isotyping kit results showed that DON-12E8 had the heavy chain IgG2a and the light chain Kappa subtype. And the concentration of the purified MAb 12E8 by ammonium sulfate precipitation was 10 mg/mL.
The MAb 12E8 was evaluated by icELISA; the IC50 and CR% values and other antibodies from recent references are shown in Table 2. The IC50 value of MAb 12E8 was 2.95 μg/L using 3-HS-DON-BSA as coating antigen. The CR% towards 3-Ac-DON (IC50 value of 0.11 μg/L) and 15-Ac-DON (IC50 value of 12.76 μg/L) were 2681 and 23%, respectively (Table 2), while its CR% to other related mycotoxins, such as NIV, NEO, T-2 toxin, and HT-2 toxin, could be negligible. According to the results in Table 2, MAb 12E8 was the most sensitive than other antibodies (Maragos and McCormick 2000; Lee et al. 2013; Sanders et al. 2014) and displayed high affinity to 3-Ac-DON which was similar with those antibodies using 3-HS-DON as hapten (Lee et al. 2013; Sanders et al. 2014). To solve CR% of the present antibodies with the acetylated derivatives, 8-CMO-DON might be the best strategy. Although some positive cells were obtained from 8-CMO-DON-KLH using indirect ELISA, the antibodies secreted by those cells could not recognize the free-DON, which was consistent with the previous studies (Casale et al. 1988; Sanders et al. 2014). In addition, MAb 12E8 was not bound with the coating antigen of 8-CMO-DON-BSA by indirect ELISA, suggesting that C8 carbonyl of the DON was important in the reaction of antigen-antibody. It could be speculated that if the connecting sites were positioned on the C3 or C15, the obtained antibodies might have higher CR% values against 3-Ac-DON or 15-Ac-DON (Casale et al. 1988; Maragos and McCormick 2000; Maragos et al. 2006; Sanders et al. 2014), or else, anti-free-DON antibody could not be obtained using carbonyl C8 for coupling to carrier protein (Casale et al. 1988; Sanders et al. 2014). Based on the resulting immunological properties, MAb 12E8 was used in the subsequent studies.
Characterization of the 3-HS-DON-BSA-QDs Bioconjugate
Based on the results of icELISA (Table 2), 3-HS-DON-BSA was selected for conjugating with QDs because many carboxyl groups from the 3-HS-DON-BSA could conjugate with amino groups of DSPE-PEG-NH2-modified CdSe/CdS/Cd0.5Zn0.5S/ZnS QDs. Moreover, it has plenty of targets (DON, haptens-to-BSA molar ratios were 9.5) for antibody recognition. To characterize the purified bio conjugate, fluorescence measurements were performed to ensure that the bio conjugation was negligible effect on the fluorescence of QDs, and agarose gel electrophoresis was taken to verify the molecular weight of the QDs and the bio conjugate. As can be seen in Fig. 4, the bio conjugate of 3-HS-DON-BSA-QDs did not modify QD emission wavelength significantly (Fig. 4a), and 3-HS-DON-BSA-QDs swarmed slowly than QDs (Fig. 4b) under the UV-lamp due to the molecular weight of 3-HS-DON-BSA-QDs bigger than that of QDs or 3-HS-DON-BSA. Those results indicated that the 3-HS-DON-BSA-QDs were synthesized successfully. Therefore, it is possible to develop a fluorescent immunoassay with this fluorescence probe.
Development of the dcFLISA
The dcFLISA consists of a competitive fluorescent immunoassay where QDs are used as antigen labels. According to principle of the dcFLISA (Fig. 2), fluorescence signals of 3-HS-DON-BSA-QD binding on the immobilized anti-DON MAb were decreased with increasing concentration of DON in the standard solution or sample extraction. Standard curve of the dcFLISA with IC50 value of 5.59 μg/kg is shown in Fig. 5a (the fluorescence signal of the B0, without free DON, was about 150), which was similar with icELISA. In addition, the whole detection procedure could be completed within 1 h, avoiding the enzymatic reaction and saving 45 min compared with conventional icELISA.
a Standard curves of icELISA and dcFLISA for DON (the coating condition of dcFLISA: MAb 12E8 2 μg/mL or 50,000-fold diluted; fluorescent probe of 3-HS-DON-BSA-QDs: 400-fold diluted). b The specificity of the dcFLISA. c Calibration curves for DON detection in flour maize extract diluted by PBS using dcFLISA
To investigate selectivity of the dcFLISA, some related mycotoxins such as 3-Ac-DON, 15-Ac-DON, NIV, T-2 toxin, HT-2 toxin, and NEO were tested by the dcFLISA (Fig. 5b). Like icELISA, dcFLISA also recognized 3-Ac-DON and 15-Ac-DON, while its CR% to other related mycotoxins could be negligible.
Analysis of Spiked Samples
To demonstrate accuracy and precision of the developed dcFLISA, concentration of DON (50~200 μg/kg) in spiked maize was measured. According to the results in Fig. 5c, matrix-assisted standard curves were nearly indistinguishable from standard curve constructed in PBS when sample extraction was diluted 10 times, indicating that the extraction method was reasonable. The LOD was calculated that 0.244 μg/kg × dilution factor (50-fold) equal to 12.2 μg/kg in the maize, and recoveries ranged from 77.3 to 84.3% with coefficients of variation (CV%) values less than 10% (Table 3) in maize flour. These results indicate that the dcFLISA has good recovery from spiked samples with high precision. Moreover, some maize flour samples which were naturally contaminated by DON already detected by HPLC-MS/MS (Monbaliu et al. 2010) were analyzed by the developed dcFLISA. The amount of 3-Ac-DON and 15-Ac-DON was equivalent to DON by calculating the CR% (Table 4). The results exhibited good agreement with LC-MS/MS; however, the limitation of dcFLISA could not distinguish the sample contaminated by DON, 3-Ac-DON, or 15-Ac-DON and had a positive bias for naturally contaminated samples containing 3-Ac-DON due to relatively high CR% of 3-Ac-DON (2681%). However, 3-Ac-DON and 15-Ac-DON were far less frequently found and at lower levels than DON (Liu et al. 2016). Thus, the new dcFLISA developed here could be used for rapid and qualitative detection of DON contamination in maize.
Conclusions
A dcFLISA based on a new highly affinity monoclonal antibody and functionalized core/multishell CdSe/CdS/Cd0.5Zn0.5S/ZnS QDs as fluorescence probe was developed for quantitative analysis of DON in maize. The LOD was 12.2 μg/kg and recoveries ranged from 77.3 to 84.3% with CV% below 10%. Good correlation between the dcFLISA and HPLC-MS/MS results in the naturally contaminated maize samples indicated the reliability of dcFLISA.
References
Algar WR, Susumu K, Delehanty JB, Medintz IL (2011) Semiconductor quantum dots in bioanalysis: crossing the valley of death. Anal Chem 83(23):8826–8837
An L, Wang Y, Pan Y, Tao Y, Chen D, Liu Z, Yang W, Peng D, Yuan Z (2016) Development and validation of a sensitive indirect competitive enzyme-linked immunosorbent assay for the screening of florfenicol and thiamphenicol in edible animal tissue and feed. Food Anal Methods 9(9):2434–2443
Beloglazova NV, Speranskaya ES, De SS, Hens Z, Abe S, Goryacheva IY (2012) Quantum dot based rapid tests for zearalenone detection. Anal Bioanal Chem 403(10):3013–3024
Binder EM, Tan LM, Chin LJ, Handl J, Richard J (2007) Worldwide occurrence of mycotoxins in commodities, feeds and feed ingredients. Anim Feed Sci Tech 137(3):265–282
Casale WL, Pestka JJ, Hart LP (1988) Enzyme-linked immunosorbent-assay employing monoclonal-antibody specific for deoxynivalenol (vomitoxin) and several analogs. J Agric Food Chem 36(3):663–668
Dong G, Pan Y, Wang Y, Ahmed S, Liu Z, Peng D, Yuan Z (2017) Preparation of a broad-spectrum anti-zearalenone and its primary analogues antibody and its application in an indirect competitive enzyme-linked immunosorbent assay. Food Chem 247:8–15
Duan H, Chen XL, Xu W, Fu JH, Xiong YH, Wang A (2015) Quantum-dot submicrobead-based immunochromatographic assay for quantitative and sensitive detection of zearalenone. Talanta 132:126–131
Garcia-Fernandez J, Trapiella-Alfonso L, Costa-Fernandez JM, Pereiro R, Sanz-Medel A (2014) A quantum dot-based immunoassay for screening of tetracyclines in bovine muscle. J Agric Food Chem 62(7):1733–1740
Goodrow MH, Hammock BD (1998) Hapten design for compound-selective antibodies: Elisas for environmentally deleterious small molecules. Anal Chim Acta 376(1):83–91
He Y, Lu HT, Sai LM, Su YY, Hu M, Fan CH, Huang W, Wang LH (2008) Microwave synthesis of water-dispersed CdTe/CdS/ZnS core-shell-shell quantum dots with excellent photostability and biocompatibility. Adv Mater 20(18):3416–3421
Hickert S, Gerding J, Ncube E, Hubner F, Flett B, Cramer B, Humpf HU (2015) A new approach using micro HPLC-MS/MS for multi-mycotoxin analysis in maize samples. Mycotoxin Res 31(2):109–115
Jiang WX, Luo PJ, Wang X, Chen X, Zhao YF, Shi W, Wu XP, Wu YN, Shen JZ (2012) Development of an enzyme-linked immunosorbent assay for the detection of nitrofurantoin metabolite, 1-amino-hydantoin, in animal tissues. Food Control 23(1):20–25
Kim KY, Shim WB, Kim JS, Chung DH (2014) Development of a simultaneous lateral flow strip test for the rapid and simple detection of deoxynivalenol and zearalenone. J Food Sci 79(10):2048–2055
Kong D, Liu L, Song S, Zheng Q, Wu X, Kuang H (2017a) Rapid detection of tenuazonic acid in cereal and fruit juice using a lateral-flow immunochromatographic assay strip. Food Agr Immunol 2:1–11
Kong D, Xie Z, Liu L, Song S, Kuang H (2017b) Development of ic-ELISA and lateral-flow immunochromatographic assay strip for the detection of citrinin in cereals. Food Agr Immunol 28(3):414–426
Kong D, Xie Z, Liu L, Song S, Kuang H, Cui G, Xu C (2017c) Development of indirect competitive ELISA and lateral-flow immunochromatographic assay strip for the detection of sterigmatocystin in cereal products. Food Agr Immunol 28(2):260–273
Kong D, Xie Z, Liu L, Song S, Zheng Q, Kuang H (2017d) Development of an immunochromatographic assay for the detection of alternariol in cereal and fruit juice samples. Food Agr Immunol 2:1–12
Kroutgreenberg ND, Puschner B, Davidson MG, Depeters EJ (2013) Preliminary study to assess mycotoxin concentrations in whole corn in the California feed supply. J Dairy Sci 96(4):2705–2712
Lee HM, Song SO, Cha SH, Wee SB, Bischoff K, Park SW, Son SW, Kang HG, Cho MH (2013) Development of a monoclonal antibody against deoxynivalenol for magnetic nanoparticle-based extraction and an enzyme-linked immunosorbent assay. J Vet Sci 14(2):143–150
Li CL, Wen K, Mi TJ, Zhang XY, Zhang HY, Zhang SX, Shen JZ, Wang ZH (2016) A universal multi-wavelength fluorescence polarization immunoassay for multiplexed detection of mycotoxins in maize. Biosens Bioelectron 79:258–265
Liu Y, Lu Y, Wang L, Chang F, Yang L (2016) Occurrence of deoxynivalenol in wheat, Hebei Province, China. Food Chem 197 (Pt B) 197:1271–1274
Li Y, Liu G, Fu X, He J, Wang Z, Hou J, Cao X, Shi W, Zhang S (2015) High-sensitive chemiluminescent ELISA method investigation for the determination of deoxynivalenol in rice. Food Analy Methods 8(3):656–660
Li YS, Shi WM, Shen JZ, Zhang SX, Cheng LL, Wang ZH (2012) Development of a rapid competitive indirect ELISA procedure for the determination of deoxynivalenol in cereals. Food Agr Immunol 23(1):41–49
Ma M, Wen K, Beier RC, Eremin SA, Li CL, Zhang SX, Shen J, Wang ZH (2016) Chemiluminescence resonance energy transfer competitive immunoassay employing hapten-functionalized quantum dots for the detection of sulfamethazine. ACS Appl Mater Interfaces 8(28):17745–17750
Maragos CM, McCormick SP (2000) Monoclonal antibodies for the mycotoxins deoxynivalenol and 3-acetyl-deoxynivalenol. Food Agric Immunol 12(3):181–192
Maragos C, Busman M, Sugita-Konishi Y (2006) Production and characterization of a monoclonal antibody that cross-reacts with the mycotoxins nivalenol and 4-deoxynivalenol. Food Addit Contam 23(8):816–825
Mills EC, Alcock SM, Lee HA, Morgan MR (1990) An enzyme-linked immunosorbent assay for deoxynivalenol in wheat, utilizing novel hapten derivatization procedures. Food Agric Immunol 2(3):109–118
Monbaliu S, Van Poucke C, Detavernier C, Dumoulin F, Velde MVD, Schoeters E, Dyck SV, Averkieva O, Peteghem CV, De SS (2010) Occurrence of mycotoxins in feed as analyzed by a multi-mycotoxin LC-MS/MS method. J Agric Food Chem 58(1):66–71
Peng D, Chang F, Wang Y, Chen D, Liu Z, Zhou X, Feng L, Yuan Z (2016a) Development of a sensitive monoclonal-based enzyme-linked immunosorbent assay for monitoring T-2 toxin in food and feed. Food Addit Contam A 33(4):683–692
Peng D, Yang B, Pan Y, Wang Y, Chen D, Liu Z, Yang W, Tao Y, Yuan Z (2016b) Development and validation of a sensitive monoclonal antibody-based indirect competitive enzyme-linked immunosorbent assay for the determination of the aflatoxin M1 levels in milk. Toxicon 113:18–24
Sanders M, Guo Y, Iyer A, Garcia YR, Galvita A, Heyerick A, Deforce D, Risseeuw MDP, Calenbergh SV, Bracke M, Eremin S, Madder A, De SS (2014) An immunogen synthesis strategy for the development of specific anti-deoxynivalenol monoclonal antibodies. Food Addit Contam A 31(10):1751–1759
Sinha RC, Savard ME, Lau R (1995) Production of monoclonal-antibodies for deoxynivalenol and 15-acetyldeoxynivalenol by ELISA. J Agric Food Chem 43(6):1740–1744
Song S, Liu N, Zhao Z, Ediage EN, Wu S, Sun C, De SS, Wu A (2014) Multiplex lateral flow immunoassay for mycotoxin determination. Anal Chem 86(10):4995–5001
Speranskaya ES, Beloglazova NV, Lenain P, De SS, Wang ZH, Zhang SX, Hens Z, Knopp D, Niessner R, Potapkin DV, Goryacheva IY (2014) Polymer-coated fluorescent CdSe-based quantum dots for application in immunoassay. Biosens Bioelectron 53(4):225–231
Streit E, Naehrer K, Rodrigues I, Schatzmayr G (2013) Mycotoxin occurrence in feed and feed raw materials worldwide: long-term analysis with special focus on Europe and Asia. J Sci Food Agr 93(12):2892–2899
Sun MM, Du LY, Gao SQ, Bao YH, Wang SH (2010) Determination of 17 beta-oestradiol by fluorescence immunoassay with streptavidin-conjugated quantum dots as label. Steroids 75(6):400–403
Wang J, Wang Y, Pan Y, Feng L, Chen D, Liu Z, Peng D, Yuan Z (2016) Preparation of a broadly specific monoclonal antibody-based indirect competitive ELISA for the detection of benzodiazepines in edible animal tissues and feed. Food Anal Methods 9(12):1–13
Wei FD, Lin Y, Wu YZ, Sun X, Liu LP, Zhou P, Hu Q (2014) Double shell CdTe/CdS/ZnS quantum dots as a fluorescence probe for quetiapine determination in fumarate quetiapine tablets. Anal Methods 6(2):482–489
Yang D, Geng ZM, Yao JB, Zhang X, Zhang PP, Ma HX (2013) Simultaneous determination of deoxynivalenol, and 15-and 3-acetyldeoxynivalenol in cereals by HPLC-UV detection. World Mycotoxin J 6(2):117–125
Yang QH, Gong XQ, Song T, Yang JM, Zhu SJ, Li YH, Cui Y, Li YX, Zhang BB, Chang J (2011) Quantum dot-based immunochromatography test strip for rapid, quantitative and sensitive detection of alpha fetoprotein. Biosens Bioelectron 30(1):145–150
Yu Q, Li H, Li C, Zhang S, Shen J, Wang Z (2015) Gold nanoparticles-based lateral flow immunoassay with silver staining for simultaneous detection of fumonisin B1 and deoxynivalenol. Food Control 54:347–352
Zhang X, Wen K, Wang Z, Jiang H, Beier RC, Shen J (2016) An ultra-sensitive monoclonal antibody-based fluorescent microsphere immunochromatographic test strip assay for detecting aflatoxin M1 in milk. Food Control 60(2):588–595
Zhang X, Yu X, Wen K, Li C, Mujtaba MG, Jiang H, Shi W, Shen J, Wang Z (2017) Multiplex lateral flow immunoassays based on amorphous carbon nanoparticles for detecting three fusantium mycotoxins in maize. J Agric Food Chem 65(36):8063–8071
Zhang ZW, Li YY, Li PW, Zhang Q, Zhang W, Hu XF, Ding XX (2014) Monoclonal antibody-quantum dots CdTe conjugate-based fluoroimmunoassay for the determination of aflatoxin B1 in peanuts. Food Chem 146(1):314–319
Acknowledgements
This work was supported by grants from Natural Science Foundation of China (U1301214 and 31502114) and Special Fund for Agro-scientific Research in the Public Interest (201203040). And we are very thankful to Prof. Sarah De Saeger (Laboratory of Food Analysis, Faculty of Pharmaceutical Sciences, Ghent University) for providing naturally contaminated maize samples.
Author information
Authors and Affiliations
Contributions
Xiya Zhang and Xuezhi Yu contributed equally to this paper.
Corresponding author
Ethics declarations
Conflict of Interest
Xiya Zhang declares that he has no conflict of interest. Xuezhi Yu declares that she has no conflict of interest. Jianyi Wang declares that he has no conflict of interest. Qi wang declares that she has no conflict of interest. Hui Meng declares that he has no conflict of interest. Zhanhui Wang declares that he has no conflict of interest.
Ethical Approval
This article does not contain any studies with human subjects. All animal experiments described in this study were approved by the Animal Care Center of the China Agricultural University, Beijing, China, following all institutional and national guidelines for the care and use of laboratory animals.
Informed Consent
Not applicable.
Rights and permissions
About this article
Cite this article
Zhang, X., Yu, X., Wang, J. et al. One-Step Core/Multishell Quantum Dots-Based Fluoroimmunoassay for Screening of Deoxynivalenol in Maize. Food Anal. Methods 11, 2569–2578 (2018). https://doi.org/10.1007/s12161-018-1198-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-018-1198-x