Abstract
Napier grass (Pennisetum purpureum Schumach) has been identified as a potential energy crop in Thailand. However, information regarding the biomass production characteristics of napier grass in response to K and N fertilization is required to guide management decisions for biofuel feedstock. Our objective was to determine the effects of K and N rates on biomass yield, developmental morphology, nutrient content and removal. This experiment was conducted during 2016 and 2017 at Kamphaeng Saen (KPS), Thailand. The experimental design was a split plot arranged in a randomized complete block (RCB) with four replications. Three K fertilization rates (0, 250, and 500 kg ha−1) were used for the main plots and four N fertilization rates (0, 250, 500, and 1000 kg ha−1) were arranged as subplots. With increasing N fertilization rate, most growth parameters increased, except leaf to stem ratio and dry matter concentration decreased. Total dry matter yield (TDMY) increased from 16.6 to 43.0 t ha−1 and from 15.2 to 41.6 t ha−1 in 2016 and 2017, respectively, as N rate increased from 0 to 1000 kg ha−1. However, growth and TDMY were not affected by K fertilizer. Increased TDMY resulted from an increase in the proportion of elongating tillers leading to advancing mean stage by count (MSC) from 2.35 to 2.45. Therefore, although napier grass demonstrated large biomass production under multiple harvest regimes, its higher removal rates (285.0–1615. kg K ha−1 and 86.7–422.2 kg N ha−1) were varied with fertilization rates indicating a potential for higher fertilizer requirements and production costs over time.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The perennial C4 grasses have recently been investigated for their potential production of lignocellulosic biomass. In the USA, switchgrass (Panicum virgatum), a perennial warm-season grass native to North America, has been identified as a model biofuel crop [1], while miscanthus species (Miscanthus x giganteus) have potential for a promising cellulosic feedstock under European conditions [2]. Both species of C4 perennial grasses, when grown in temperate climates, provide remarkably high annual biomass and cellulose content with low agricultural inputs.
For Thailand, napier grass or elephant grass (Pennisetum purpureum Schumach.) has been classified by the Ministry of Energy as a dedicated energy crop [3]. Napier grass is a C4 perennial bunchgrass with tall canopy, vigorous tillering, and large leaf area, which are taken as fundamental factors for high productivity. This grass species originated from tropical Africa, and has been introduced to all tropical and sub-tropical regions of the world to be widely used as a forage source for ruminant animals [4].
However, its productivity and nutritive value as forage are limited under drought stress and cold conditions. To solve these problems, interspecific hybrids of the perennial Pennisetum purpureum Schumach (napier grass) x annual Pennisetum americanum (L.) Leeke (pearl millet) with higher forage yield and quality were developed [5, 6]. These hybrids were widely distributed to farmers in subtropical and tropical countries, including Thailand [7].
Because of its relatively higher herbage yield and higher protein content, one of the napier grass hybrids, ‘Pakchong 1’ has recently been developed and promoted by the Department of Livestock Development in Thailand. ‘Pakchong 1’ has produced biomass yields exceeding 500 t ha−1 (six cutting times per year). Due to its high nutritive values and digestibility [8], there has been particular interest in using napier grass for conversion into biomethane with anaerobic digestion [9]. There has also been increasing interest in the production of bioenergy via direct combustion of napier grass [10]. This grass is highly productive in limited areas and maximum biomass yield has been achieved by increasing fertilization rates, especially K and N fertilization [11, 12]. To evaluate if napier grass is an economically viable grass for the production of biofuel feedstock for a particular area, information regarding the optimum rates of fertilizer application is required.
Potassium is one of the most required essential macronutrients by grasses. It plays a key role in improving grass growth and development because it is involved in improving metabolic and physiologic functions, such as cation–anion balance, enzyme activation, and stomatal mechanism [11]. Plants normally contain 2–5% K of dry weight for optimal growth and development, varied with species, growth stage, and plant’s organ [13]. However, napier grass has been shown to be a luxury consumer of K, ranging from 31 to 67 g K kg−1 [14]. For N, it is always a limiting factor in the production of tropical grasses, including napier grass. It can be responsive to high N application rates up to 2000 kg N ha−1 for both total dry matter yield and N content. Novo et al. [13] reported that increasing K application rates in combination with low N application rate increased dry biomass yield of napier grass. These effects are more pronounced when harvest frequency is increased [15]. Both K and N are taken up easily and are accumulated abundantly in the above-ground parts of forage crops [16]. The high nutrient concentrations accumulated in harvested biomass suggest that significant fertilizer input will be needed because large quantities of nutrients are removed from the soil. For example, in Africa, napier grass yields were reduced rapidly in the second year after harvesting [17]. Typically, high annual biomass yield with low mineral element contents, including K, Cl, N, and S, are desirable traits for the production of bioenergy and biofuels [18]. These biomass characteristics can be obtained with proper management practices, in particular, the use of optimal fertilizer applications and appropriate harvest management [7, 19].
In addition to optimal fertilization management, potential biomass productivity and chemical composition of perennial grasses depend on plant morphology and canopy structure changes occurring during growth and development [20, 21]. Crop phenology allowing long growing season with optimum leaf to stem ratio (LAI) and few proportion reproductive structures in canopy is associated with increased biomass production [22, 23]. Moreover, there are the relationships between morpho-agronomic traits and biomass quality within napier grass genotypes. To obtain high calorific value and large biomass yield for napier grass, the plant height and stalk diameter should be increased [24]. Waramit et al. [21] have demonstrated that the mean maturity of warm-season grasses, including switchgrass, indiangrass, and big blue stem, was increased with N fertilization. In contrast, the mean maturity stage of eastern gamagrass (Tripsacum dactyloides L.) decreased when N fertilization at 140 kg N ha−1 was applied.
In temperate zones, one-cut per year harvest after grass senescence has been recommended to attain the highest dry matter concentration, the minimum nutrient concentrations, and removal rates in harvested biomass [21, 25, 26]. However, multiple harvests per year to increase biomass yield of forage grasses and to provide higher income for farmers is a common practice in tropical zones, including Thailand [7]. Consequently, greater nutrient removal with increased harvest frequency could decrease inherent soil fertility. In Thailand, the maximum biomass yield of napier grass cv. Muaklek (36.9 t DM ha−1) occurred with high harvest frequency (2-month inter-cutting interval). However, higher harvest frequency increased N concentration in biomass from 1.2% with 12-month cutting to 1.8% with 2-month cutting. In the southeastern USA, biomass yield of napier grass cv. Merkeron and a breeding line UF1 were not different between one and two harvests per year in the first two years of the experiment [27]. However, two harvests per year removed twice as much N and P in the harvested biomass than one harvest per year. Knoll et al. [19] reported that napier grass produced a high biomass yield (11.2–36.3 t DM ha−1) without fertilizer application in the first three harvests that then sharply decreased (5.0 t DM ha−1) in the fourth harvest. This was likely because very large amounts of both N (374 kg N ha−1) and K (1,121 kg K ha−1) were removed from the soil over the first three harvests.
Sustainable biomass production systems depend on how the removal and concentration of nutrients in harvested biomass are affected by the fertilization rates. A higher rate of nutrient content in harvested biomass often negatively affects feedstock quality [26, 28] and increases fertilizer input costs [29, 30].
Therefore, optimal fertilization management is a key factor for the success of long-term biomass crop production. However, research on productivity, optimum nutrient concentrations, nutrient removal rates, and developmental morphology of napier grass as affected by the interaction of K and N fertilization remains limited. Therefore, the objective of this study was to determine the effects of K and N fertilizer rate on biomass yield, developmental morphology, nutrient content, and removal rates in hybrid napier grass for bioenergy production.
Materials and Methods
Site Description
Field experiments were carried out in 2016 and 2017 at Kamphaeng Saen (KPS) district, Nakhon Pathom province, Thailand (13° 59′ 38“ N, 99° 58’ 32” E). The studied soil was a silty clay loam, fine-silty, mixed, semiactive, isohyperthermic Typic Haplustalfs and showed the following values: pH 6.73, ECe 1.54 dS m−1, total N 0.15%, organic matter 2.43%, available P 110.6 mg kg−1 soil (Mehlich III), exchange K 229.7 mg kg−1 soil (Mehlich III), exchange Ca 3256.8 mg kg−1, and exchange Mg 541.7 mg kg−1 soil. Weather data were collected from a weather station located approximately 5 km from the research site (Table 1). Rainfall and temperature between two studied years (January 2016–December 2017) varied. In 2016, total rainfall was 789.7 mm less than the average long-term (25 years) rainfall (1228.0 mm). Rainfall amounts were especially great between July and October. The average temperature (28.9 °C) was slightly higher than the average long-term temperature (27.9 °C) except in early 2016. In 2017, the total rainfall of 770.4 mm was lower than the long-term average as well. Similar to 2016, rainfall was especially high between July and October. In this year, the average temperature was 28.5 °C, which was slightly higher than the average long-term temperature. As compared between both experimental years, total rainfall for 2017 was slightly lower than that for 2016, while the average temperature for both years was similar. The total rainfall of both studied years was, therefore, commonly dryer than the average long-term rainfall (Table 1).
Experimental Design and Field Agronomic Methods
The experiment was a randomized complete block design in a split-plot arrangement, with four replications. The main plot was three K application rates (0, 250, and 500 kg K ha−1 year−1 of potassium chloride). The subplot was four N application rates (0, 250, 500, and 1000 kg N ha−1 year1 of ammonium sulfate). Plant stock of hybrid napier grass (Pennisetum purpureum Schumach x P. americanum (L.) Leeke cv. Pakchong 1) was obtained from Nakhon Ratchasima Animal Nutrition Research and Development Center, Pakchong district, Nakhon Rachasima province, Thailand. To produce similar-sized plants for the experiment, napier grass clumps were cut into individual stems (two nodes per stem) and propagated in plastic pots (diameter 10 cm; height 15 cm). The potted plants were grown in a greenhouse and watered every day for one month. In August 2015, potted napier grass plants were transplanted manually into the experimental plots conventionally prepared using disk plows and cultivation furrow. The distance between rows was 1.3 m and plant spacing was 0.5 m. Individual subplots consisted of five rows, 8 m long, and 6.5 m wide. Each row had 18 plants (90 plants per subplot). To enable the uniform growth of the grass stand, a standardized cut (70 days after transplanting) at ground level was made on November 2015. The fertilization treatments were applied as six equal split applications at 2-month intervals after each cutting (at the standardized cut, and at the 1st, 2nd, 3rd, 4th, and 5th harvest). After each harvest and at the symptom of drought stress in both years (except the 4th harvest in 2016, and the 3rd and 4th harvest in 2017), all plots were immediately irrigated totalling 70 mm in 2016 and 50 mm in 2017 to ensure unrestricted grass growth. Weeds were controlled by hand hoeing at 25 days after planting.
Data Collection and Analytical Procedure
Plant height, total tiller number, and stalk diameter were measured on ten plants chosen at random before each harvest date. The above-ground biomass samples were harvested at 2-month intervals following the standardized cut. The six harvests occurred in January, March, May, July, September, and November for both years. At each harvest date, the whole plant was randomly hand-clipped at ground level from an area of 5.2 × 1.5 m (9 plants) within each subplot. After each harvest, the guard rows and remaining plants of the plots were cut and all cut materials were removed from the plots. After recording fresh weights in the field, a representative sub-sample of 15 tillers was randomly taken to be hand-separated into leaf and stem, and then dried at 65 °C in a forced-air oven for 72 h and then reweighed to determine dry matter concentration (g dry matter per kg fresh weight) and dry matter yield (t per ha).
Quantifying Morphological Development
At each harvest, the number of live tiller samples was determined, as a measure of morphological development, using the mnemonic scale developed by Moore et al. [31]. The individual grass tillers were classified into four primary growth stages including: vegetative (leaf development), stem elongation, reproductive (floral development), and seed development and ripening. Within each primary growth stage, secondary growth stages describing specific events were given numerical indices to quantify grass canopy development. The developmental stage of the grass tiller population was described by a mean stage by count (MSC), calculated using the following equation:
Where Si = growth stage (0 to 4.9), Ni = number of tillers in stage Si, C = total number of tillers.
Potassium and Nitrogen Analyses
The dried samples were ground sequentially using a Wiley mill (Model 4, Thomas Manufacturing, PA) to pass an 8-mm screen and reground to uniformity using a 1-mm screen. Potassium concentration in the dried ground material for each treatment was determined using atomic absorption spectrometry, flame photometry, and spectrophotometry according to the methods of AOAC [32]. For determining N analysis, dried ground material was quantified by the Kjeldahl procedure [33]. Average K and N concentration in harvested biomass for the six-cut system was calculated as a weighted average.
K and N removal (kg ha−1) were determined as:
Statistical Analyses
The data for each experimental year were separately analyzed because of the differences in the response of biomass yield and chemical composition to fertilizer application dates, harvest dates, irrigation dates, and variable climatic conditions from year to year (Table 1). Analysis of variance (ANOVA) was investigated to determine the main effects and interactions of K and N application rates. Differences between means were separated using the Duncan’s new multiple range test at the 0.05 probability level. The responses to K and N fertilization rates were tested using orthogonal polynomial contrasts. The relationships between TDMY and nutrient removal, and nutrient content and nutrient removal were evaluated using correlation analysis. All analyses were conducted in R version 3.3.2 using the nlme package [34].
Results
Growth Characteristics
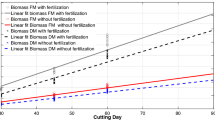
There was no K x N rate interaction for most of the growth characteristics in both years, except for LAI in 2017 (Table 2). Therefore, the effects of K and N rate on the growth traits were evaluated independently. The results showed that N fertilization affected all growth parameters, but K fertilization did not affect these traits for napier grass (Table 2). In 2016, as N fertilization rate increased from 0 to 1000 kg N ha−1, plant height increased in quadratic manner from 162.3 to 209.7 cm, while stalk diameter, total tiller number, and LAI increased in linear manner from 1.33 to 1.41 cm, 18.2 to 23.4 tiller m−2, and 1.06 to 3.42, respectively (Table 3). Similarly, the results in 2017 showed that plant height increased quadratically from 156.3 to 201.9 cm, whereas total tiller number increased linearly from 18.9 to 23.0 tiller m−2 with N fertilization rate increased, but there was a difference in stalk diameter among N fertilization rates in this year. However, there was a K x N rate for LAI in 2017 indicating that the linear increase in LAI as N rates increased from 250 to 1000 kg N ha−1 was less pronounced for napier grass with no K fertilization (1.6–2.5) than napier grass receiving K fertilization (1.6–3.4) (Fig. 1). Thus, this suggested that LAI of napier grass was more responsive to N fertilization at adequate K supply.
Total Dry Matter Yield (TDMY)
There was no K x N rate interaction for TDMY in either 2016 or 2017 (Table 2). Averaged over N fertilization rates, K fertilization did not affect TDMY for napier grass in both 2016 and 2017 (Table 2). As expected, increased N rate contributed greatly to TDMY in both years. When compared with the control treatment with no N fertilization, TDMY of napier grass supplied with N fertilizer at the rates of 250, 500, and 1000 kg N ha−1 increased by 70, 122, and 159% in 2016 and 66, 120, and 173% in 2017, respectively (Table 3). Maximum TDMY was 43.0 t ha−1 in 2016 and 41.6 t ha−1 in 2017 when applied with the highest N rate of 1000 kg N ha−1. Across K and N fertilization rates, TDMY in 2016 was 31.2 t ha−1, which was slightly greater than that in 2017 (28.9 t ha−1) (Table 3).
Dry Matter Concentration (DMC)
Dry matter concentration varied with N fertilization, but did not with K fertilization (Table 2). When N rate increased from 0 to 1000 kg N ha−1, DMC declined linearly from 22.2 to 20.7 g kg−1 in 2016 and from 21.5 to 20.7 g kg−1 in 2017 (Table 4).
Leaf to Stem Ratio (LSR)
There was no response of LSR to K fertilization in either experimental year (Table 2). However, LSR decreased in linear and quadratic manners as N application rate increased in 2016 and 2017, respectively. The application of N at 250, 500, and 1000 kg N ha−1 resulted in decreased LSR by 7.5%, 15.1%, and 20.4%, respectively, in 2016 (Table 4) and by 14.4%, 18.6%, and 22.7%, respectively, in 2017. No interaction effect of K x N rate was detected for LSR showing consistency of N effect among K fertilization rates (Table 1).
Tiller Development
No interaction effect of K x N rate was detected for the morphological indices of mean stage by count (MSC) in either year, indicating independence between the factors (Table 2). Additionally, there were no differences in the MSC among K fertilization rates. However, N fertilization had an effect on MSC for both years. In 2016, the MSC increased linearly from 2.4 (fourth node palpable) for the control plots to 2.5 (mid-elongation stage with 5 nodes) for plots receiving 1000 kg N ha−1 (Table 4). Similar to 2016, increasing N fertilization rates from 0 to 1000 kg N ha−1 increased MSC linearly from 2.3 (third node palpable) to 2.4 (fourth node palpable) in 2017. This suggested that napier grass receiving N fertilization advanced MSC during each two-month period of growth.
Figure 2 showed that napier grass stands receiving N fertilizer provided a larger proportion of tillers developing to the elongating stage than did those in the unfertilized plots. Averaged over six harvests, the elongating tiller number (ETN) increased linearly from 112,536 to 224,501 tiller ha−1 and from 114,103 to 206,553 tiller ha−1 for 2016 and 2017, respectively, as N fertilization rates increased (Table 4 and Fig. 2). When compared with elongating tillers, fewer tillers in vegetative stage (VTN) changed with N fertilization rates, as indicated by the proportion of tiller populations in either year. In 2016, the VTN significantly decreased in a linear manner from 37,607 to 18,234 tiller ha−1 as N fertilization rate increased. However, the tiller populations of vegetative and elongating stage were not affected by K fertilization.
N Content
There was no K x N interaction effect on average annual N content in harvested above-ground biomass (Table 1). Nitrogen content increased relatively as N fertilization rate increased in both years. Averaged over six harvest dates, N fertilization at 250, 500, and 1000 kg N ha−1 increased N content by 5.5, 16.4, and 34.3%, respectively, for 2016 and by 3.1, 17.0, and 39.1%, respectively, for 2017, when compared with biomass receiving no N fertilization (Table 5). However, N content showed no response to K fertilization rate (Table 2).
K Content
The interaction of K by N application was significant for K content in 2016; however, the interaction effect was not significant in 2017 (Table 2). In 2016, the interaction effect occurred because there was fluctuation in the shape of the response curves among N application rates (Fig. 3). This indicated that the K content in harvested biomass receiving 250 kg K ha−1 increased to the same level (44.0 and 44.9 g K kg−1), across 250 and 500 kg N ha−1, which was greater than the biomass receiving 1000 kg N ha−1 (37.3 g K kg−1). However, K content in harvested biomass receiving 250 kg K ha−1 in combination with 0 and 1000 kg N ha−1 was 39.6 and 37.3 g K kg−1, respectively, which was consistent with that of biomass receiving no K fertilizer (41.7 and 35.0 3 g K kg−1, respectively). In contrast to 2016, the increase in K content as increased K application rate across four N fertilization rates was at the same rate in 2017. In this year, the K content increased in linear manner from 24.5 to 33.3 g kg−1 as K fertilization rates increased from 0 to 500 kg K ha−1. However, the K content declined linearly from 34.3 to 25.7 g kg−1, as N fertilization rate increased from 0 to 1000 kg N ha− 1 (Table 5).
Total Annual Nitrogen Removal
There was no K x N interaction effect on total annual N removal in either year (Table 2). Additionally, there was no difference in total annual N removal among K fertilization rates. This is likely due to there being no response of the annual dry matter yield to K fertilization and that of N content in harvested biomass (Table 2). Unlike K fertilization, N fertilization affected N removal in the harvested biomass for both years. Biomass N removal increased linearly from 127.2 to 422.2 kg ha−1 in 2016 and from 106.9 to 394.9 kg ha−1 in 2017, as N fertilizer rate increased from 0 to 1000 kg N ha−1 (Table 5).
Total Annual K Removal
There was no K and N fertilization rate interaction for total annual K removal in harvested biomass (Table 2). Increasing N application rate from 0 to 250, 500, and 1000 kg N ha−1 increased K removal quadratically from 795.5 to 1159.7 (45.8%), 1513.8 (90.3%), and 1615.1 (103.0%) kg ha−1, respectively, in 2016, and from 546.7 to 769.7 (41.0%), 1002.7 (83%) and 1089.9 (99%) kg ha−1, respectively, in 2017 (Table 5). Similar to N fertilization, K removal increased linearly from 1093.9 to 1167.0 (6.7%) and to 1525.2 (39.4%) kg ha−1, respectively, in 2016, and from 942.8 to 1044.6 (10.8%) and to 1282.7 (36.1%) kg ha−1, respectively, in 2017, as K fertilizer rate increased from 0 to 250 and 500 kg K ha−1.
Discussion
The positive effect of N fertilization on TDMY in the current study is similar to that determined in previous studies on other C4 perennial grasses [29, 35, 36]. These studies suggested that TDMY for warm-season grasses peaked at 500 kg N ha−1 or less, while maximum dry matter yield of napier grass in this study occurred at the greatest N fertilization rate of 1000 kg N ha−1 for both years (Table 3). The increased biomass yield of warm-season grasses in response to N fertilization was attributed to an increase in stem development [21]. For napier grass, a greater stem development was reflected in a decreased leaf to stem ratio (Table 3) and in an increased proportion of elongating tillers (Fig. 2). However, TDMY of napier grass did not reach a plateau at the highest rate of 1000 kg N ha−1 (Table 3). It is expected that N fertilization at higher rates than 1000 kg N ha−1 could lead to further increases in total dry matter yields. A raised biomass yield in response to N fertilization in this study is consistent with earlier studies reported that biomass yield of napier grass increased with N fertilizer rates up to 1600 kg N ha−1 producing a maximum yield of 57.95 t ha−1 [37]. In addition, they demonstrated that the dry matter production of napier grass increased in a quadratic manner as N rates increased. An increase in dry matter concentration is important for the complete determination of calorific value [38]. Munalula and Meincken [39] reported that the highest calorific value was significantly correlated to the highest dry matter density.
In contrast, Novo et al. and Almeida et al. [13, 40] demonstrated that the response of biomass yield was inversely proportional to the increment of N application rate. Additionally, Stida et al. [41] showed that dry matter production decreased in a linear manner with increasing N application rate from 400 to 1600 kg N ha−1 (within an K application rate of 500 kg K ha−1). In their studies, the maximum dry matter production of three napier grass genotypes ranged from 50.7 to 55.1 t ha−1 and occurred at the lowest application rate of 400 kg N ha−1 in combination with 500 kg K ha−1. However, a significant relationship was not observed at the lower application rate of 200 kg K ha−1. This indicates that biomass yield response to N fertilization was associated with the K application rate. However, there was no K fertilization effect and no K x N rate interaction effect on TDMY of napier grass in the present study. This is likely because soil K at the experimental site (229.7 mg K kg−1) might have met the K requirement of napier grass. Consistent with previous studies, Jungers et al. and Shield et al. [36, 42] suggested that biomass yield for both temperate and tropical grasses responded to N fertilization but did not respond to K fertilization. In addition, Dampney [43] and Rahman et al. [44] found that K fertilizer application did not affect total annual DM yield of napier grass when K content in the soil was greater than 120 mg kg−1. Similar to other different C4 grasses, K did not affect herbage yield and N content, even when applied in combination with high rates of N and P [45, 46]. However, although there was no effect of K on biomass yield, luxury consumption of K gave high K concentration for napier grass (Table 5 and Fig. 3) comparable to the amounts reported by Valencia-Gica et al. [47] (37.4–41.9 g K kg−1) in a study comparing nutrient removal among tropical grasses. Although napier grass was usually adapted to a range of soil conditions, the maximum TDMY was produced in areas with high rainfall, over 1500 mm per year [48]. Norsuwan et al. [49] reported that yield responses for napier grass to increasing N fertilization rates were linear at 1.0 ETo (reference evapotranspiration) irrigation treatment, but curvilinear at 0.5 ETo irrigation treatment and rainfed condition. Therefore, the results in the present study showing quadratic response of TDMY to N fertilization rate in drier conditions than average years (Table 1) might not be applicable to other sites where different rainfall conditions prevail.
An increase in LAI plays a key role in increasing dry matter yield of napier grass [22]. Even with excessively high LAI causing high shading within the canopy of various plants, light penetration in the canopy of napier grass was improved by stem elongation, stem erection, and self-thinning. The maximum LAI of 3.42 for napier grass observed in this study (Fig.1) was less than the value reported by Kubota et al. [22]. This suggests that LAI of napier grass would be able to further increase up to 15.4.
An increase in N fertilization rate stimulated stem growth and the overall development for napier grass (Fig. 2), therefore decreasing LSR (Table 3). Leaf to stem ratio decreased with increasing N fertilization was because of enhanced stem elongation [50]. This agrees with previous observations of LSR response to N fertilization for Urochloa brizantha [51]. The change in the chemical constituents (e.g., lignocellulose and mineral contents) of warm-season grasses was highly correlated with the variation in LSR [52]. Grass stems contain greater fiber and lower element constituents than do leaves [53]. High lignocllulose and low element composition in biomass are desirable traits by contributing more energy to thermochemical conversion process [54]. Therefore, grass biomass with low LSR decreases the efficiency of biochemical conversion processes by reducing the availability of cellulose and hemicellulose contents [55].
Optimizing grass biomass yield and quality for biofuel production is affected by changes in morphology [52]. This study has indicated that MSC of napier grasses increased with increasing N fertilization rates in agreement with a study by Waramit et al. [21]. This suggested that the advanced MSC with N fertilization was likely because N application increased the density of elongating and reproductive tillers per unit area. Similarly, it is shown in our study that N fertilization increased stem development and the proportion of elongating tillers (Table 4 and Fig. 2).
Increasing N fertilization rates increased N concentration but decreased K concentration in harvested biomass, which was comparable to those observed earlier [56, 57]. Other previous studies suggested that N content for tropical grasses increased as N application rate increased, but the degree of increase depended on N rate and harvest frequency [15, 25, 58]. A decrease in the K concentration in plant tissues as a function of the N rates was caused by the dilution effect due to a greater dry biomass yield at higher N fertilization treatment [56]. However, high concentrations of elements, including N and K, in biomass feedstock are undesirable for biofuel production and negatively affect conversion and combustion systems [59]. To maximize dry matter yield of grasses, N application is therefore a key management practice but may reduce biomass quality by increasing the concentration of N.
Additionally, this study has shown that K removal rate positively responded to both K and N fertilization rates. This was due more to the response of TDMY than the K concentration in harvested biomass. Our result indicated that total annual K removal was positively correlated with TDMY (r = 0.937, P < 0.05 in 2016; r = 0.876, P < 0.05 in 2017), but was not correlated with biomass K content (r = 0.049; P = 0.7763 in 2016; r = 0.2565; P = 0.911 in 2017), whereas the increase in N removal rate as N fertilizer rate increased was a result of both greater TDMY and higher N content of biomass. Total annual N removal was positively correlated with N content (r = 0.809923, P < 0.05 in 2016; r = 0.90, P < 0.05 in 2017) and positively correlated with TDMY (r = 0.9486898, P < 0.05 in KPS and r = 0.973, P < 0.05 in 2017) (data not shown). These findings are in agreement with previous studies [25, 58, 60]. Moreover, the proportion of nutrients (K and N) in harvested biomass removed at the 60-day harvest was relative to fertilizer addition. Therefore, this result suggests that nutrient limitations occurred for both K and N in this experiment. Additionally, biomass K was removed by 305.0–466.8% and 256.5–417.8% of added K in 2016 and 2017, respectively. Although TDMY were not affected by K fertilization, the K removal rates were above the added fertilization rates. Consequently, K demand for napier grass may have been met even in the control plots receiving no K fertilizer for both experimental years. Unlike K removal rate, when napier grass was fertilized with 250, 500, and 1000 kg N ha−1, N removal rates in harvested biomass were less than the amount of N added (86.6, 62.4, and 42.2% of added N, respectively, in 2016, and 71.6, 53.6, and 39.5% of added N, respectively, in 2017). Biomass N removal rate below fertilization rates (<100%) coincided with the increased TDMY as N application rate increased (Tables 3 and 4) indicating that the soil N availability may have been in deficit even at the highest N rates.
The stand persistence and regrowth potential of perennial grasses are reduced when above-ground biomass is removed frequently without fertilization [58]. The current study has demonstrated that the increasing N fertilization rates in a multiple-harvest system resulted in relatively greater annual biomass production of napier grass when compared with the control treatment. Kering et al. [26] reported that although there was potential to increase C4-grass biomass through N fertilization and multiple harvests per year, higher nutrient concentrations and removal rates in a multiple-harvest system may be less sustainable for biomass energy production systems than the one-cut system in late season when nutrients have been remobilized to below-ground storage reserves. The greater nutrient concentrations in harvested biomass not only result in lower feedstock quality for bioenergy production but also involve a greater nutrient removal from soil [61]. This may lead to the corresponding increase in fertilizer requirements and costs associated with greater nutrient removal. However, the ability to increase biomass feedstock supply on a year-round basis for lignocellulosic biorefineries is likely to be able to justify using a multiple-harvest system and an optimal fertilization management.
Conclusions
This study increases the understanding of how different K and N fertilization rates affect biomass yield, morphological development, and nutrient removal for napier grass in a multiple-harvest system. Biomass yield of napier grass increased in a quadratic manner with increasing N fertilizer rates, but there was no difference among K fertilization rates. Greater N fertilization slightly increased mean stage count index for napier grass harvested at 60-day intervals resulting from an increase in the proportion of elongating tillers within the swards, but resulted in decreased leaf to stem mass ratio. Biomass K and N concentration and removal were greater as their fertilization rates increased. Excessive concentration of nutrients, especially K, in harvested biomass of napier grass must be considered when fertilizing napier grass for biofuel purposes. Finally, napier grass could be one of the best suited perennial grasses in tropical climates due to its desired morphological characteristics and relatively high biomass yield when fertilization is managed properly. However, future studies of this grass for biofuel purposes should aim at achieving minimum nutrient removal and maximum lignocellulose yields.
References
David K, Ragauskas AJ (2010) Switchgrass as an energy crop for biofuel production: a review of its ligno-cellulosic chemical properties. Energy Environ Sci 3:1182–1190 https://doi.org/10.1039/B926617H
Arnoult S, Brancourt-Hulmel M (2015) A review on Miscanthus biomass production and composition for bioenergy use: genotypic and environmental variability and implications for breeding. Bioenerg Res 8:502–526
Waramit N, Chaugool J (2014) Napier grass:a novel energy crop development and the current status in Thailand. J ISSAAS 20(1):139–150
Mwendia SW, Wanyoike M, Wahome RG, Mwangi DM (2006) Farmer’s perceptions on importance and constraints facing Napier grass production in Central Kenya. Livestock Research for Rural Development. http://www.lrrd.org/lrrd18/11/mwen18160.htm. Accessed 2 January 2020
Hanna WW, Chaparro C, Mathews B, Burns JC, Sollenberger LE, Carpenter JR (2004) Perennial Pennisetums. In: Moser LE, Byron LB, Sollenberger LE (eds) Warm-season (C4) grasses. ASA/CSSA/ SSSA, Madison, pp 503–535
Premaratne S, Premalal GGC (2006) Hybrid Napier (Pennisetum perpureum x Pennisetum americarnum) var. CO-3: a resourceful fodder grass for dairy development in Sri Lanka. J Agri Sci–Sri Lanka 2(1):22–33
Rengsirikul K, Ishii Y, Kangvansaichol K, Pripanapong P, Sripichitt P, Punsuvon V, Vaithanomsat P, Nakamanee G, Tudsri S (2011) Effects of inter-cutting interval on biomass yield, growth components and chemical composition of napiergrass (Pennisetum purpureum Schumach) cultivars as bioenergy crops in Thailand. Grassl Sci 57(3):135–141
Turano B, Tiwari UP, Jha R (2016) Growth and nutritional evaluation of napier grass hybrids as forage for ruminants. Tropical Grasslands-Forrajes Tropicales 4(3):168–178
Bangprasit P, Chavalparit O, Usapein P (2017) Life cycle assessment of Napier Pakchong 1 grass as a feedstock for anaerobic digestion to produce electricity: a case study in Thailand. 2017 International Conference on Green Energy and Applications (ICGEA), Singapore, pp 123–129 https://doi.org/10.1109/ICGEA.2017.7925468
Machado JC, Carneiro PCS, Costa J, Resende MDV, Silva FJ, Souza JE (2017) Bioenergetic potential and genetic diversity of elephantgrass via morpho-agronomic and biomass quality traits. Ind Crop Prod 95:485–492
Marschner H (2011) Marschner’s mineral nutrition of higher plants. Academic, Cambridge
Woodard KR, Sollenberger LE (2015) Production of biofuel crops in Florida: elephant grass. https://edis.ifas.ufl.edu/ag302. Accessed 1 December 2019
Novo AAC, Daher RF, Gravina DAG, Costa ES, Ogliari J, Araujo KC, Menezes BRDS, Poncinio NJ, Oliveira EDS, Silva VB (2016) Effect of nitrogen and potassium fertilization on morpho-agronomic traits of three elephant grass (Pennisetum purpureum Schum.) genotypes for biomass production. African J Biotec 15(43):2411–2423
Foster HL (1969) The effects of different management and fertilizer treatments on the soil and leaf nutrient status of an elephant grass ley. East Afr Agr Forestry J 34(4):468–475
Ferraris R (1980) Effect of harvest interval, nitrogen rates and application times on Pennisetum purpureum grown as an agroindustrial crop. Field Crops Res 3:109–120
Harada Y (1992) Composting and land application of animal wastes. Asian-Australasian Journal of Animal Sci 5(1):113–121
Boonman JG (1993) East Africa’s grasses and fodders: their ecology and husbandry. Springer, Dordrecht. https://springerlink.bibliotecabuap.elogim.com/book/10.1007%2F978-94-015-8224-7. Accessed 1 January 2020
Brosse N, Dufour A, Meng X, Sun Q, Ragauskas A (2012) Miscanthus: a fast-growing crop for biofuels and chemicals production. Biofuels Bioprod Biorefin 6(5):580–598
Knoll JE, Anderson WF, Malik R, Hubbard RK, Strickland TC (2013) Production of napier grass as a bioenergy feedstock under organic versus inorganic fertilization in the southeast USA. Bioenerg Res 6(3):974–983
Redfearn DD, Moore KJ, Vogel KP, Waller SS, Mitchell RB (1997) Canopy architecture and morphology of switchgrass populations differing in forage yield. Agron J 89(2):262–269
Waramit N, Moore KJ, Heaton E (2014) Nitrogen and harvest date affect developmental morphology and biomass yield of warm-season grasses. GCB Bioenerg 6(5):534–543
Kubota F, Matsuda Y, Agata W, Nada K (1994) The relationship between canopy structure and high productivity in napiergrass, Pennisetum purpureum Schumach. Field Crops Res 38:105–110
Porter J, Kirsch MMN, Streibig J, Felby C (2007) Choosing crops as energy feedstocks. Nat Biotechnol 25:716–717
Rocha JR, Machado JC, Carneiro PC, Carneiro JD, Resende MD, Pereira AV, Carneiro JE (2017) Elephant grass ecotypes for bioenergy production via direct combustion of biomass. Ind Crop Prod 95:27–32
Guretzky JA, Biermacher JT, Cook BJ, Kering MK, Mosali J (2011) Switchgrass for forage and bioenergy: harvest and nitrogen rate effects on biomass yields and nutrient composition. Plant Soil 339(1–2):69–81
Kering MK, Butler TJ, Biermacher JT, Guretzky JA (2012) Biomass yield and nutrient removal rates of perennial grasses under nitrogen fertilization. Bioeng Res 5(1):61–70
Na CI, Sollenberger LE, Erickson JE, Woodard KR, Vendramini JM, Silveira ML (2015) Management of perennial warm-season bioenergy grasses. I. Biomass harvested, nutrient removal, and persistence responses of elephantgrass and energycane to harvest frequency and timing. Bioenerg Res 8(2):581–589
Monti A, Virgilio N, Venturi G (2008) Mineral composition and ash content of six major energy crops. Biomass Bioenergy 32(3):216–223
Haque M, Epplin FM, Taliaferro CM (2009) Nitrogen and harvest frequency effect on yield and cost for four perennial grasses. Agron J 101(6):1463–1469
Aravindhakshan SC, Epplin FM, Taliaferro CM (2011) Switchgrass, bermudagrass, flaccidgrass, and lovegrass biomass yield response to nitrogen for single and double harvest. Biomass Bioenergy 35(1):308–319
Moore KJ, Moser LE, Vogel KP, Waller SS, Johnson BE, Pedersen JF (1991) Describing and quantifying growth stages of perennial forage grasses. Agron J 83(6):1073–1077
AOAC (2003) Official methods of analysis of AOAC international, vol I, 17th edn. AOAC, Washington, DC
Kirk PL (1950) Kjeldahl method for total nitrogen. Anal Chem 22(2):354–358
R Core Team (2016) R:A language and environment for statistical computing version 3.3.2. https://www.R-project.org/. Accessed 19 Jan 2018
Kering MK, Butler TJ, Biermacher JT, Guretzky JA (2012) Biomass yield and nutrient removal rates of perennial grasses under nitrogen ferilization. Bioenerg Res 5:61–70
Jungers JM, Sheaffer CC, Lamb JA (2015) The effect of nitrogen, phosphorus, and potassium fertilizers on prairie biomass yield, ethanol yield, and nutrient harvest. Bioenerg Res 8(1):279–291
Oliveira EDS, Daher RF, Ponciano NJ, Gravina GDA, Santana JADA, Gottardo RD, Menezes BRDS, Souza PMD, Souza CLMD, Siva VBD, Rocha ADS, Novo AAC (2015) Variation of morpho-agronomic and biomass quality traits in elephant grass for energy purposes according to nitrogen levels. Am J Plant Sci 6(11):1685–1696
Rocha JR et al (2017) Bioenergetic potential and genetic diversity of elephantgrass via morpho-agronomic and biomass quality traits. Ind Crop Prod 95:485–492
Munalula F, Meincken M (2009) An evaluation of south African fuelwood with regards to calorific value and environmental impact. Biomass Bioenergy 33:415–420
Almeida BDO, Daher RF, Novo AAC, Gravina GDA, Vivas M, Moraes CRDO, Menezes BRDS, Furlani EP, Araujo MDSBD, Silva VBD (2016) Effect of nitrogen and potassium fertilizations on elephant grass genotypes used for energy purposes in northern Rio de Janeiro state, Brazil. Afr J Agri Res 11(49):5043–5049
Stida WF, Almeida BDO, Daher RF, Moraes CDO, Vidal AKF, Freitas RS, Tardin FD, Menezes BRDS, Furlani EP, Vivas M (2018) Effect of nitrogen and potassium fertilization doses on elephant-grass genotypes for energy purposes. J Agri Sci 10(10):446–459
Shield IF, Barraclough TJP, Riche AB, Yates NE (2012) The yield response of the energy crops switchgrass and reed canary grass to fertiliser applications when grown on a low productivity sandy soil. Biomass Bioenergy 42:86–96
Dampney PMR (1992) The effect of timing and rate of potash application on the yield and herbage composition of grass grown for silage. Grass Forage Sci 47(3):280–289
Rahman MM, Ishii Y, Niimi M, Kawamura O (2010) Interactive effects of nitrogen and potassium fertilization on oxalate content in napiergrass (Pennisetum purpureum). Asian-Australasian J Animal Sci 23(6):719–723
Rehm GW, Moline WJ, Schwartz EJ (1972) Response of a seeded mixture of warm-season prairie grasses to fertilization. J Range Management 25(6):452–456
Hall KE, George JR, Riedl RR (1982) Herbage dry matter yields of switchgrass, big bluestem, and Indiangrass with N fertilization. Agron J 74(1):47–51
Valencia-Gica RB, Yost RS, Porter G (2012) Biomass production and nutrient removal by tropical grasses subsurface drip-irrigated with dairy effluent. Grass Forage Sci 67(3):337–349
Kabirizi J, Muyekho F, Mulaa M, Msangi R, Pallangyo B, Kawube G, Zziwa E, Mugerwa S, Ajanga S, Lukwago G, Wamalwa NIE, Kariuki I, Mwesigwa R, Nannyeenya NW, Atuhairwe A, Awalla J, Namazzi C, Nampijja Z (2015) Napier grass feed resource: production, constraints and implications for smallholder farmers in Eastern and Central Africa. The Eastern African Agricultural Productivity Project (EAAPP): Naivasha, Kenya. https://bit.ly/2XwMER2. Accessed 1 January 2020
Norsuwan T, Marohn C, Jintrawet A (2014) Effects of irrigation treatments and nitrogen applications on Napier grass planted in dry season as energy crop at Chiang Mai province. Khon Kaen Agr J 42(SUPPL 2):1–7 https://ag2.kku.ac.th/kaj/PDF.cfm?filename=01%2037_57.pdf&id=1414&keeptrack=38. Accessed 2 Jan 2020
Alves FGS, Carneiro MSS, Edvan RL, Candido MJD, Furtado RN, Pereira ES, Neto LBM, Mota RRM, Nascimento KS (2018) Agronomic and nutritional responses of Carajas elephant grass fertilized with protected and non-protected urea. Semina Ciênc Agrár 39:2181–2189
Bonfim-Silva EM, Xavier Correa de Morais LPV, Pacheco AB, da Silva TJA, de Freitas Sousa HH, do Espirito Santo ES (2018) Interaction between nitrogen and potassium in fertilization of the piata grass (Urochloa brizantha): productive and phytometric characteristics. Australian J Crop Sci 12:1122–1228
Mulkey VR, Owens VN, Lee DK (2006) Management of switchgrass-dominated conservation reserve program lands for biomass production in South Dakota. Crop Sci 46(2):712–720
Madakadze IC, Radiotis T, Li J, Goel K, Smith DL (1999) Kraft pulping characteristics and pulp properties of warm season grasses. Bioresour Technol 69(1):75–85
Hayn M, Steiner W, Klinger R, Steinmuller H, Sinner M, Esterbauer H (1993) Basic research and pilot studies on the enzymatic conversion of lignocellulosics. In: Saddler JN (ed) Bioconversion of forest and agricultural plant residues. CAB, Wallingford, pp 33–72
Sun Y, Cheng J (2002) Hydrolysis of lignocellulosic materials for ethanol production: a review. Bioresour Technol 83(1):1–11
de Morais Léa PVXC, Bonfim-Silva EM, Pacheco AB, Abreu JGD, da Silva TJ, Polizel AC (2016) Nitrogen and potassium in the cultivation of Piatã grass in Brazilian Cerrado soil. Revista Brasileira de Engenharia Agrícola e Ambiental, 20 (11): 984–989
Zhang F, Niu J, Zhang W, Xinping C, Chunjian L, Yuan L, Xie J (2010) Potassium nutrition of crops under varied regimes of nitrogen supply. Plant Soil 335:21–34
Seepaul R, Macoon B, Reddy KR, Evans WB (2014) Harvest frequency and nitrogen effects on yield, chemical characteristics, and nutrient removal of switchgrass. Agron J 106(5):1805–1816
Meincken M, Tyhoda L (2014) Biomass quality. In: Seifert T (ed) Bioenergy from wood: sustainable production in the tropics. Springer, Dordrecht, pp 169–184
Pedroso GM, Hutmacher RB, Putnam D, Wright SD, Six J, van Kessel C, Linquist BA (2013) Yield and nitrogen management of irrigated switchgrass systems in diverse ecoregions. Agron J 105(2):311–320
Knoll JE, Anderson WF, Strickland TC, Hubbard RK, Malik R (2012) Low-input production of biomass from perennial grasses in the coastal plain of Georgia, USA. Bioenerg Res 5(1):206–214
Funding
This research was supported financially by Kasetsart University Graduate School.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dokbua, B., Waramit, N., Chaugool, J. et al. Biomass Productivity, Developmental Morphology, and Nutrient Removal Rate of Hybrid Napier Grass (Pennisetum purpureum x Pennisetum americanum) in Response to Potassium and Nitrogen Fertilization in a Multiple-Harvest System. Bioenerg. Res. 14, 1106–1117 (2021). https://doi.org/10.1007/s12155-020-10212-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12155-020-10212-w