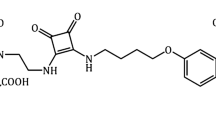
Abstract
Objective
This study aimed to assess the potential of 68Ga-DOTA-FAPI-04 PET/CT for the detection of the radioiodine-refractory differentiated thyroid cancer (RR-DTC) lesions.
Methods
We analyzed the 68Ga-DOTA-FAPI-04 PET/CT imaging data of 24 RR-DTC patients (7 men and 17 women; 49.6 ± 10.5 year). Clinical data were collected including history, last post-therapeutic radioiodine whole body scan, contemporary CT, thyroglobulin, and antithyroglobulin. Target lesions were selected and measured by the RECIST 1.1. The mean growth rates of the target lesions in the past 6 months were recorded. Tumor uptake of lesions were quantified by SUVmax and the tumor-to-background ratios. The correlation between SUVmax and target lesion growth rate and thyroglobulin was analyzed.
Results
On patient-based analysis, positive metastases were detected in 87.5% (21/24) patients. Except for the lymph node (LN) metastasis of 3 patients (patient 6, 12 and 17#) and the lung metastasis of another 3 patients (patient 9, 13 and 21#), most of the lesions were positive on 68Ga-DOTA-FAPI-04 PET/CT images, including LN metastasis and distant metastasis such as lung, bone and pleura. There were altogether 33 target lesions including 30 lung metastases and 3 LN metastases with the mean SUVmax and the growth rate were 4.25 and 6.51%, respectively. SUVmax was statistically associated with the growth rates of the target lesions (p = 0.047). No statistically significant correlation was found between the SUVmax and the serum thyroglobulin levels (p = 0.139).
Conclusions
68Ga-DOTA-FAPI-04 PET/CT has a promising detection rate for RR-DTC metastasis. The FAPI uptake of the tumor may provide a potential therapeutic target for RR-DTC.
Trial registry
NIH Clinical Trials.gov (NCT04499365).
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Thyroid cancer is the most common endocrine malignancy with a growing incidence, ranking ninth in the global incidence of cancer [1]. The majority of thyroid cancers are differentiated thyroid cancer (DTC). Optimistic prognosis of DTC is commonly achieved because of its adequate management strategies, such as surgery, radioiodine (131I) treatment, and levothyroxine therapy. However, radioiodine-refractory DTC (RR-DTC) have become the main cause of disease-specific death with a 10-year survival rate as low as 10% [2], which represents a very difficult management situation.
Cancer guidelines of the American Thyroid Association recommend the use of 18F-FDG PET/CT for the detection of RR-DTC lesions [3]. However, 18F-FDG is neither a tumor specific tracer nor a therapeutic one. Novel tracers with a specific target are urgently needed for diagnosis and therapy.
68Ga-DOTA-FAPI-04 is a novel radiotracer targeting fibroblast activation protein (FAP), which is highly expressed in cancer-associated fibroblasts (CAFs) of many epithelial carcinomas [4,5,6]. 68Ga-DOTA-FAPI-04 serves as an alternative to 18F-FDG for the assessment of malignant tumors and adds important diagnostic value in the context of challenging 18F-FDG PET/CT cancer subtypes in previous studies [7, 8]. The uptake of 68Ga-DOTA-FAPI-04 was observed in many various tumor entities like thyroid cancer [4, 9,10,11,12,13]. Unlike 18F-FDG, radiolabeled FAP inhibitors (FAPI) makes FAP-imaging and therapy possible. This study first reported 68Ga-DOTA-FAPI-04 PET/CT imaging in a cohort of RR-DTC patients.
Materials and methods
Patients
This study was approved by the Clinical Research Ethics Committee of the First Affiliated Hospital of Fujian Medical University and conducted in accordance with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards (ID 2019-XJS-1130). This study was also registered online at NIH ClinicalTrials.gov (NCT04499365). Informed written consent was obtained from all of these enrolled patients.
RR-DTC is classified in patients with appropriate TSH stimulation and iodine preparation in four basic ways according to 2015 American Thyroid Association Management Guidelines [3]. From December 2020 to November 2021, 24 RR-DTC patients were recruited from the First Affiliated Hospital of Fujian Medical University. The eligibility criteria were as follows: (i) adult patients (aged 18 years or older); (ii) a negative pregnancy test; (iii) clinically acceptable renal and hepatic function; (iv) RR-DTC patients with structural lesions, treated by 131I more than twice.
Radiopharmaceutical preparation
Good-manufacturing-practice (GMP) grade precursors DOTA-FAPI-04 were purchased from Jiangsu Huayi Technology Co. (Jiangsu, China). 68GaCl3 was eluted from a [68Ge]/[68Ga] generator (JSC Isotope, Obninsk, Russia) using 5 mL of 0.1 M hydrochloride acid. Radiolabeling was performed manually in a hot cell as previously reported [14, 15]. Radiochemical purity of the final product was over 95%. The median specific activity was 1.67 (range 1.25–2.76) MBq/ug.
PET/CT imaging
The injection activity of 68Ga-DOTA-FAPI-04 was calculated according to the patient’s weight (1.85–2.22 MBq/kg). Before imaging, all the subjects were asked to urinate completely. The emission scan using a hybrid PET/CT scanner (Biograph mCT 64, SIEMENS, Germany) began 30 min after intravenous injection. The patient was supine on the examination bed. CT scan was performed from the head to upper thighs. The following parameters for the CT scan were used: tube voltage of 110 kV, current of 120 mA, and slice thickness of 3 mm, reconstruction interval 1 mm with a sharp reconstruction kernelD. A PET scan was immediately performed after the CT scan in 3D acquisition mode with 6–8 bed positions and 3 min/position. TOF mode/reconstruction is utilized. All the obtained data were transferred to the MultiModality Workstation (syngoTM MI, SIEMENS, Germany); data were reconstructed using the ordered subset expectation maximization algorithm (two iterations and 21 subsets); CT data were used for attenuation correction; and the reconstructed images were then co-registered and displayed. An additional chest CT was performed with the patient in the supine position at full inspiration, without intravenous contrast medium. The CT acquisition protocol of chest CT 6 months ago was similar to that of FAPI PET/CT imaging.
Image analysis
Studies were analyzed by a group of two experienced nuclear medicine physicians (W Miao and S Chen). Any difference in opinion was resolved by consensus. All metastases were confirmed by pathological examination, medical imaging including post-therapeutic radioiodine whole body scan, 131I single photon emission computed tomography/computed tomography (SPECT/CT), high-resolution ultrasonography (US) and CT, serum thyroglobulin (TG) test, and clinical follow-up of at least 6 months as well (Table 1). In addition to elevated TG level, a diagnosis of metastatic lymph node (LN) was established in terms of at least two of the following signs: microcalcification, cystic aspect, peripheral vascularization, and hyperechogenicity in US examination [16, 17]. Because of ethical concerns, biopsies and histopathology were not available for most of the analyzed metastases except for lung metastasis in six patients, pleura metastasis in one patient and bone metastasis in three patients (Table 1).
RR-DTC metastases were coded as positive if the activity exceeded that of the adjacent background tissues. The positive lesions found on FAPI imaging were divided into target lesions and non-target lesions according to the Response Evaluation Criteria in Solid Tumors (RECIST) 1.1 [18]. The target lesions including lung metastases which were greater than 10 mm by CT scan and malignant LNs which were greater than 15 mm in short axis when assessed by CT scan (CT scan slice thickness recommended to be no greater than 5 mm).
The region of interest (ROI) was drawn manually on the entire target lesions on PET cross-sections. The maximum standardized uptake value (SUVmax) and the average standardized uptake value (SUVmean) was calculated for the quantitative analysis. The target-to-background ratio (TBR) was calculated by dividing the lesion SUVmax by the SUVmean of the ascending aorta. The normal organs were evaluated with a 1-cm-diameter (for the small organs [parotid gland and submaxillary gland]) to 2-cm-diameter (liver, spleen, bone marrow and kidney) sphere placed inside the organ parenchyma [9].
The size of well-defined metastatic lesions such as lung and LNs was measured. Lung metastases were measured by the longest diameter in the axial plane. The size of the metastatic LN was determined by the short diameter of the CT cross-section. The mean growth rates of target lesions were defined as follows: [(lesion size in FAPI imaging-lesion size 6 months ago)/lesion size 6 months ago] × 100%. The clinical data of the patients were recorded in detail for subsequent analysis. Serum thyroid stimulating hormone (TSH), TG, and antithyroglobulin (TGAb) levels were measured before 68Ga-DOTA-FAPI-04 PET/CT imaging by an electrochemiluminescent immunoassay on a Cobas analyzer (CCM + cobas8000, Roche Ltd., Basel, Switzerland). The correlation between SUVmax and growth rates of the target lesions was analyzed. Meanwhile, the relationship between SUVmax and TG was studied in patients with negative TGAb.
Immunohistochemistry of FAP expression
Immunohistochemical staining of FAP was performed on the tumor tissue obtained from biopsy specimens in one patient with pleural metastasis. An antibody against FAP (ab207178, Abcam) was used.
Statistical analysis
Statistical analysis was performed using SPSS Statistics 23 (IBM Inc., Armonk, NY, USA). All quantitative data were expressed as mean ± standard deviation or as medians with ranges. Kruskal–Wallis test was used to compare median values. The correlation between the two variables was performed by Spearman correlation analysis. A p value of less than 0.05 was set as indicating a statistical significance.
Results
Patients’ characteristics
We analyzed the data for 24 RR-DTC patients (7 men and 17 women; 49.6 ± 10.5 year; 25–66 years) in our center from December 2020 to November 2021. Majority patients (22/24) had papillary thyroid cancer (PTC) histotype. Among these patients, the metastatic tissues of 10 patients did not ever concentrate 131I, 131I is concentrated in some lesions of 3 patients but not in others, the tumor tissue loses the ability to concentrate 131I after previous evidence of 131I-avid disease (in the absence of stable iodine contamination) in 3 patients, and metastatic disease in other 8 patients progressed despite significant concentration of 131I. The enrolled patients received an average of 3.87 times of 131I treatment, and the average activity was 24.24 GBq. Patient 2# has been taking donafenib tosilate tablets 0.3 g bid for 28 months, patients 8# and 19# have been taking apatinib mesylate tablets for over 28 months and 34 months, respectively. Patient 1# had been treated with donafenib tosilate tablets 0.3 g bid 1 week and stopped because of a severe allergic reaction. The rest subjects only received levothyroxine therapy after the last 131I treatment. TSH level of all patients enrolled was less than 0.01 mIU/L. Except of 1 patient who had high TGAb (2854 IU/mL), the mean TG level of the rest patients was 791.7 ng/mL. BRAFV600E mutations were detected in 4/24 patients. Three patients had positive BRAFV600E mutation, while only one patient had negative (Table 1).
According to pathological results, clinical information and follow-up, 6 patients had bone metastasis, including patient 14# with only bone metastasis. Majority of enrolled patients (23/24) had lung metastasis. There were 3 cases of pleural metastasis. Metastases involving both LN and distant organs were revealed in 12 patients. The detailed clinical characteristics of the enrolled patients are summarized in Table 1.
Detection of patient-based RR-DTC metastasis
Positive metastases were detected in 87.5% (21/24) patients. Majority of the patients (23/24) were complicated with lung metastasis, among which 6 patients had negative FAPI imaging of lung metastasis. Twelve patients had LN metastasis, of which 3 patients had no abnormal FAPI uptake of lymph node metastasis. The typical positive image of 68Ga-DOTA-FAPI-04 PET/CT and detailed illustration of the enrolled patients are presented in Fig. 1 and Table 2.
Detection of all-lesion-based RR-DTC metastasis
Most of the lesions were positive on 68Ga-DOTA-FAPI-04 PET/CT images, including LN metastasis and distant metastasis such as lung, bone and pleura (Figs. 2, 3). A total of 118 well-defined metastatic lesions were included in the analysis, including 11 LN lesions, 80 lung lesions and 27 bone lesions. Median size of metastatic LN and lung lesions measured 0.9 cm (range 0.8–2.3 cm) and 1.0 cm (range 0.6–2.7 cm), respectively. The mean SUVmax of LN, lung, and bone metastasis was 7.06 ± 0.39, 6.39 ± 0.91 and 4.01 ± 0.48 (p = 0.000), respectively. The mean TBR of LN, lung, and bone metastasis was 7.96 ± 1.57, 4.29 ± 3.07 and 3.35 ± 1.90 (p = 0.000), respectively. SUVmax was statistically associated with the metastatic organs (p = 0.000). While SUVmax was not statistically associated with the serum TG levels before the PET scan (p = 0.139).
A 43-year-old female with left pleura metastasis was referred to 68Ga-DOTA-FAPI-04 PET/CT imaging (a axial CT; b axial 68Ga-DOTA-FAPI-04 PET; c axial fusion image) which revealed multiple intense FAPI-avid nodules of the left pleura (as indicated by the arrow). A 62-year-old female with lung metastasis underwent 68Ga-DOTA-FAPI-04 PET/CT imaging (d axial CT; e axial 68Ga-DOTA-FAPI-04 PET; f axial fusion image) which revealed multiple nodules with intermediate to high uptake of FAPI of bilateral lungs (as pointed by the arrow). A 59-year-old female with lymph node metastasis underwent 68Ga-DOTA-FAPI-04 PET/CT imaging (g axial CT; h axial 68Ga-DOTA-FAPI-04 PET; i axial fusion image) which revealed the strong FAPI-avid lymph nodes in the mediastinum (as shown by the arrow)
A 52-year-old male with bone metastasis was referred to 68Ga-DOTA-FAPI-04 PET/CT imaging (a, d, g, axial CT; b, e, h, axial 68Ga-DOTA-FAPI-04 PET; c, f, i, axial fusion image) which revealed the skull and vertebrae lesions with mild-to-moderate uptake of FAPI, including mixed (a), osteolytic (d), and osteogenic (g) metastatic lesions (as pointed by the arrow)
All bone metastasis were clearly displayed (Fig. 3). A total of 27 bone metastatic lesions were found, including 3 in skulls, 2 in sternum, 2 in clavicles, 1 in rib, 11 in vertebrae, 6 in pelvis and 2 in femurs. Osteolysis, osteogenesis and mixed type of bone metastasis were 14, 4 and 9, respectively. The mean SUVmax of osteolysis, osteogenesis and mixed bone metastasis was 4.16 ± 0.52, 2.14 ± 0.39 and 4.15 ± 0.31, respectively. The mean TBR of osteolysis, osteogenesis and mixed bone metastasis was 3.88 ± 0.64, 1.77 ± 0.32 and 3.24 ± 0.17, respectively.
Detection of target lesion-based RR-DTC metastasis
There were altogether 33 target lesions including 30 lung metastases and 3 LN metastases in 11 patients for the detection of the mean growth rates. In patient 4# and 24#, there was no follow-up CT images in the past 6 months. The mean SUVmax and the growth rate of the target lesions was 4.25 and 6.51%, respectively. Spearman’s rank correlation test revealed a positive correlation between the growth rates of the target lesions and SUVmax (r = 0.348, p = 0.047) (Fig. 4).
A 59-year-old female with lung metastasis underwent 68Ga-DOTA-FAPI-04 PET/CT imaging comparing with her chest CT scan 6 months ago. In axial CT image 6 months ago, the metastatic pulmonary nodule (white arrow) measured 13.7 mm (a axial CT) and 10 mm (d axial CT), respectively. In 68Ga-DOTA-FAPI-04 PET/CT imaging, the target lesions measured 16.1 mm (b axial CT) and 11 mm (e axial CT) with SUVmax of 12.0 (c axial fusion image) and 4.1 (f axial fusion image), respectively
Detection of non-target lesion-based RR-DTC metastasis
Metastatic lung lesions with a diameter smaller than 1 cm were defined as non-target lesions. All patients with lung metastasis had non-target lesions. SUVmax, fluctuated between 0.7 and 4.4, was hard to match non-target lesions correctly because of the patients’ respiratory movement. The total number is difficult to determine and meaningless.
Immunohistochemistry
One pleural metastatic tumor sample obtained from patient 6# was assessed for FAP expression by immunohistochemistry. Stromal cells around the tumor had prominent FAP expression. 68Ga-DOTA-FAPI-04 PET showed high uptake (SUVmax = 10.3) in the pleural metastasis of this patient [19].
Organ distribution of 68Ga-DOTA-FAPI-04 PET/CT imaging in RR-DTC patients
68Ga-DOTA-FAPI-04 PET imaging of 24 patients with metastatic RR-DTC resulted in no adverse events. Except for the accumulation in the kidney and bladder due to the main excretion pathway, most patients (23/24) had only mild 68Ga-DOTA-FAPI-04 uptake in the renal parenchyma. Patient 8# had moderate uptake in renal parenchyma (SUVmean, 2.61) who had proteinuria 2 + (Fig. 1). The mean SUVmean of parotid gland and the submaxillary gland were 2.97 ± 0.47 and 2.90 ± 0.39, respectively. The mean SUVmean of liver, spleen and bone marrow were 1.74 ± 0.13, 1.59 ± 0.11 and 0.49 ± 0.03, respectively.
Discussion
In our study, the results proved moderate-to-high 68Ga-DOTA-FAPI-04 avid in majority metastatic and recurrent RR-DTC lesions. LN and distant metastasis such as lung, pleura, and bone could be clearly detected by 68Ga-DOTA-FAPI-04 PET with statistical difference in the uptake degree. Osteolytic, osteogenic or mixed bone metastases can be clearly demonstrated. Meanwhile, the lesions with higher SUVmax progress faster in a short term, suggesting that the expressions of FAP are related to the progression of the disease (Fig. 4), which is in line with previous studies [20]. To our knowledge, this study was the first attempt of FAPI-04 PET imaging in 24 RR-DTC patients.
There was no obvious FAPI-04 uptake in metastatic lesions of 3 patients (patient 7#, 18# and 19#). We found that the number of lesions in these patients was less than 10 and the diameter of lesions was less than 1 cm. Two patients with relative low TG level (13.1 ng/ml and 10.4 ng/ml) indicated that the tumor load in these two patients were lower than the others, which may be related to the lower uptake of FAPI. This needs to be confirmed by further research. Except moderate FAPI-avid lung metastases were found in patient 4# with low TG level (3.74 ng/ml), other patients with intense FAPI-avid lesions had high TG levels (range 57.8—> 5000 ng/ml). This finding is consistent with the results of the case previously reported by Fu [21]. There was no statistical significance between SUVmax of metastatic lesions and TG level in this study. The results showed that there was no correlation between the level of TG and the expression of FAP.
The management of RR-DTC metastatic lesions has caused a dilemma. Surgery is not eligible for multiple metastases. 131I cannot be concentrated by RR-DTC metastasis or effectively kill tumor cells [22]. In the last decade, TKIs such as sorafenib and lenvatinib have been approved for the treatment of progressive RR-DTC based on phase III clinical trials [23, 24]. However, TKI is not a specific targeted drug for RR-DTC, because it mainly targets vascular endothelial growth factor receptor (VEGFR) [24], which plays an anti-tumor effect mainly by inhibiting tumor neovascularization. And the relatively high incidence of side effects such as hand-foot syndrome, diarrhea, and hypertension also limit the clinical application of TKIs [25]. Personalized therapy is highly desirable for these patients. In recent years, targeted radionuclide therapy (TRT) in neuroendocrine tumor and prostate cancer have shown good effect [26,27,28]. But there are no suitable radioactive drugs for TRT in RR-DTC patients. Unlike 18F-FDG, DOTA-FAPI can also be labeled with various therapeutic radionuclides such as 90Y, 177Lu and 225Ac. Tadashi Watabe et al. have successfully proved the concept that α-therapy targeting FAP in the cancer stroma is effective [29]. Ballal S et al. have reported 177Lu-DOTA.SA.FAPi radionuclide therapy in an end-stage breast cancer patient [30]. Therefore, FAP may be a promising target for theranostics. FAPI is not specific for de-differentiated thyroid cancer. However, in this study, intermediate to high uptakes of 68Ga-DOTA-FAPI-04 were observed in RR-DTC lesions, and strong FAP expression in stromal cells was confirmed by immunohistochemistry in such lesions [19]. Based on previous study and our findings, TRT based on FAPI may be presented as an alternative to RR-DTC [21, 29, 30].
In this study, mild-to-moderate 68Ga-DOTA-FAPI-04 uptake was found in the liver, spleen, renal and bone marrow, which were consistent with those of previous studies [6, 9]. We noticed that the intense uptake of 68Ga-DOTA-FAPI-04 in parotid gland and submaxillary gland had been found in 7 patients. This may be owing to the atrophy of the secretory parenchyma and salivary gland fibrosis which caused by several times of 131I treatment [31, 32]. It reminds us to pay attention to parotid gland protection in the future FAP TRT.
There are some limitations in this study. The main limitation of this study is that most of the lesions have not been confirmed by pathology. After all, it is unethical to perform pathological biopsies on all suspected lesions with abnormally high uptake of 68Ga-DOTA-FAPI-04 to validate PET/CT results. The second limitation is that these RR-DTC patients did not undergo 18F-FDG PET/CT examination simultaneously, so it remains unknown the added value of FAPI PET/CT compared to FDG PET, which needs further research.
Conclusion
In this study, intermediate to high uptakes of 68Ga-DOTA-FAPI-04 were observed in RR-DTC lesions. 68Ga-DOTA-FAPI-04 PET/CT is a promising modality for diagnosing and opening up new application for radioligand therapy in RR-DTC.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021. https://doi.org/10.3322/caac.21660.
Durante C, Haddy N, Baudin E, Leboulleux S, Hartl D, Travagli JP, Caillou B, Ricard M, Lumbroso JD, De Vathaire F, Schlumberger M. Long-term outcome of 444 patients with distant metastases from papillary and follicular thyroid carcinoma: benefits and limits of radioiodine therapy. J Clin Endocrinol Metab. 2006;91(8):2892–9.
Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, Pacini F, Randolph GW, Sawka AM, Schlumberger M, Schuff KG, Sherman SI, Sosa JA, Steward DL, Tuttle RM, Wartofsky L. 2015 American thyroid association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the american thyroid association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid. 2016;26(1):1–133.
Garin-Chesa P, Old LJ, Rettig WJ. Cell surface glycoprotein of reactive stromal fibroblasts as a potential antibody target in human epithelial cancers. Proc Natl Acad Sci USA. 1990;87(18):7235–9.
Hamson EJ, Keane FM, Tholen S, Schilling O, Gorrell MD. Understanding fibroblast activation protein (FAP): substrates, activities, expression and targeting for cancer therapy. Proteomics Clin Appl. 2014;8(5–6):454–63.
Lindner T, Loktev A, Altmann A, Giesel F, Kratochwil C, Debus J, Jäger D, Mier W, Haberkorn U. Development of quinoline-based theranostic ligands for the targeting of fibroblast activation protein. J Nucl Med. 2018;59(9):1415–22.
Chen H, Pang Y, Wu J, Zhao L, Hao B, Wu J, Wei J, Wu S, Zhao L, Luo Z, Lin X, Xie C, Sun L, Lin Q, Wu H. Comparison of [(68)Ga]Ga-DOTA-FAPI-04 and [(18)F] FDG PET/CT for the diagnosis of primary and metastatic lesions in patients with various types of cancer. Eur J Nucl Med Mol Imaging. 2020;47(8):1820–32.
Chen H, Zhao L, Ruan D, Pang Y, Hao B, Dai Y, Wu X, Guo W, Fan C, Wu J, Huang W, Lin Q, Sun L, Wu H. Usefulness of [(68)Ga]Ga-DOTA-FAPI-04 PET/CT in patients presenting with inconclusive [(18)F]FDG PET/CT findings. Eur J Nucl Med Mol Imaging. 2021;48(1):73–86.
Giesel FL, Kratochwil C, Lindner T, Marschalek MM, Loktev A, Lehnert W, Debus J, Jäger D, Flechsig P, Altmann A, Mier W, Haberkorn U. (68)Ga-FAPI PET/CT: biodistribution and preliminary dosimetry estimate of 2 DOTA-containing FAP-targeting agents in patients with various cancers. J Nucl Med. 2019;60(3):386–92.
Kratochwil C, Flechsig P, Lindner T, Abderrahim L, Altmann A, Mier W, Adeberg S, Rathke H, Röhrich M, Winter H, Plinkert PK, Marme F, Lang M, Kauczor HU, Jäger D, Debus J, Haberkorn U, Giesel FL. (68)Ga-FAPI PET/CT: tracer uptake in 28 different kinds of cancer. J Nucl Med. 2019;60(6):801–5.
Koerber SA, Staudinger F, Kratochwil C, Adeberg S, Haefner MF, Ungerechts G, Rathke H, Winter E, Lindner T, Syed M, Bhatti IA, Herfarth K, Choyke PL, Jaeger D, Haberkorn U, Debus J, Giesel FL. The role of (68)Ga-FAPI PET/CT for patients with malignancies of the lower gastrointestinal tract: first clinical experience. J Nucl Med. 2020;61(9):1331–6.
Röhrich M, Loktev A, Wefers AK, Altmann A, Paech D, Adeberg S, Windisch P, Hielscher T, Flechsig P, Floca R, Leitz D, Schuster JP, Huber PE, Debus J, von Deimling A, Lindner T, Haberkorn U. IDH-wildtype glioblastomas and grade III/IV IDH-mutant gliomas show elevated tracer uptake in fibroblast activation protein-specific PET/CT. Eur J Nucl Med Mol Imaging. 2019;46(12):2569–80.
Shi X, Xing H, Yang X, Li F, Yao S, Zhang H, Zhao H, Hacker M, Huo L, Li X. Fibroblast imaging of hepatic carcinoma with (68)Ga-FAPI-04 PET/CT: a pilot study in patients with suspected hepatic nodules. Eur J Nucl Med Mol Imaging. 2021;48(1):196–203.
Rettig WJ, Garin-Chesa P, Healey JH, Su SL, Ozer HL, Schwab M, Albino AP, Old LJ. Regulation and heteromeric structure of the fibroblast activation protein in normal and transformed cells of mesenchymal and neuroectodermal origin. Cancer Res. 1993;53(14):3327–35.
Liu B, Zhang Z, Wang H, Yao S. Preclinical evaluation of a dual sstr2 and integrin α(v)β(3)-targeted heterodimer [(68)Ga]-NOTA-3PEG(4)-TATE-RGD. Bioorg Med Chem. 2019;27(21): 115094.
Leboulleux S, Girard E, Rose M, Travagli JP, Sabbah N, Caillou B, Hartl DM, Lassau N, Baudin E, Schlumberger M. Ultrasound criteria of malignancy for cervical lymph nodes in patients followed up for differentiated thyroid cancer. J Clin Endocrinol Metab. 2007;92(9):3590–4.
Leenhardt L, Erdogan MF, Hegedus L, Mandel SJ, Paschke R, Rago T, Russ G. 2013 European thyroid association guidelines for cervical ultrasound scan and ultrasound-guided techniques in the postoperative management of patients with thyroid cancer. Eur Thyroid J. 2013;2(3):147–59.
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, Kaplan R, Lacombe D, Verweij J. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228–47.
Chen Y, Zheng S, Zhang J, Yao S, Miao W. Pleural metastasis of papillary thyroid cancer depicted by 68Ga-FAPI PET/CT. Clin Nucl Med. 2022. https://doi.org/10.1097/RLU.0000000000004038.
Liu F, Qi L, Liu B, Liu J, Zhang H, Che D, Cao J, Shen J, Geng J, Bi Y, Ye L, Pan B, Yu Y. Fibroblast activation protein overexpression and clinical implications in solid tumors: a meta-analysis. PLoS One. 2015;10(3): e0116683.
Fu H, Fu J, Huang J, Su X, Chen H. 68Ga-FAPI PET/CT in thyroid cancer with thyroglobulin elevation and negative iodine scintigraphy. Clin Nucl Med. 2021;46(5):427–30.
Van Nostrand D. Radioiodine refractory differentiated thyroid cancer: time to update the classifications. Thyroid. 2018;28(9):1083–93.
Schlumberger M, Tahara M, Wirth LJ, Robinson B, Brose MS, Elisei R, Habra MA, Newbold K, Shah MH, Hoff AO, Gianoukakis AG, Kiyota N, Taylor MH, Kim SB, Krzyzanowska MK, Dutcus CE, de las Heras B, Zhu J, Sherman SI. Lenvatinib versus placebo in radioiodine-refractory thyroid cancer. N Engl J Med. 2015;372(7):621–30.
Brose MS, Nutting CM, Jarzab B, Elisei R, Siena S, Bastholt L, de la Fouchardiere C, Pacini F, Paschke R, Shong YK, Sherman SI, Smit JW, Chung J, Kappeler C, Peña C, Molnár I, Schlumberger MJ. Sorafenib in radioactive iodine-refractory, locally advanced or metastatic differentiated thyroid cancer: a randomised, double-blind, phase 3 trial. Lancet. 2014;384(9940):319–28.
Cheng L, Fu H, Jin Y, Sa R, Chen L. Clinicopathological features predict outcomes in patients with radioiodine-refractory differentiated thyroid cancer treated with sorafenib: a real-world study. Oncologist. 2020;25(4):e668–78.
Sorbye H, Kong G, Grozinsky-Glasberg S. PRRT in high-grade gastroenteropancreatic neuroendocrine neoplasms (WHO G3). Endocr Relat Cancer. 2020;27(3):R67-r77.
Violet J, Sandhu S, Iravani A, Ferdinandus J, Thang SP, Kong G, Kumar AR, Akhurst T, Pattison DA, Beaulieu A, Mooi J, Tran B, Guo C, Kalff V, Murphy DG, Jackson P, Eu P, Scalzo M, Williams S, Hicks RJ, Hofman MS. Long-term follow-up and outcomes of retreatment in an expanded 50-patient single-center phase II prospective trial of (177)Lu-PSMA-617 theranostics in metastatic castration-resistant prostate cancer. J Nucl Med. 2020;61(6):857–65.
Brabander T, van der Zwan WA, Teunissen JJM, Kam BLR, Feelders RA, de Herder WW, van Eijck CHJ, Franssen GJH, Krenning EP, Kwekkeboom DJ. Long-term efficacy, survival, and safety of [(177)Lu-DOTA(0), Tyr(3)]octreotate in patients with gastroenteropancreatic and bronchial neuroendocrine tumors. Clin Cancer Res. 2017;23(16):4617–24.
Watabe T, Liu Y, Kaneda-Nakashima K, Shirakami Y, Lindner T, Ooe K, Toyoshima A, Nagata K, Shimosegawa E, Haberkorn U, Kratochwil C, Shinohara A, Giesel F, Hatazawa J. theranostics targeting fibroblast activation protein in the tumor stroma: (64)Cu- and (225)Ac-labeled FAPI-04 in pancreatic cancer xenograft mouse models. J Nucl Med. 2020;61(4):563–9.
Ballal S, Yadav MP, Kramer V, Moon ES, Roesch F, Tripathi M, Mallick S, ArunRaj ST, Bal C. A theranostic approach of [(68)Ga]Ga-DOTA.SA.FAPi PET/CT-guided [(177)Lu]Lu-DOTA.SA.FAPi radionuclide therapy in an end-stage breast cancer patient: new frontier in targeted radionuclide therapy. Eur J Nucl Med Mol Imaging. 2021;48(3):942–4.
Ahn BC. Reduction of salivary gland damage during radioiodine therapy for differentiated thyroid cancers. Nucl Med Mol Imaging. 2020;54(3):126–7.
Klein Hesselink EN, Brouwers AH, de Jong JR, van der Horst-Schrivers AN, Coppes RP, Lefrandt JD, Jager PL, Vissink A, Links TP. Effects of radioiodine treatment on salivary gland function in patients with differentiated thyroid carcinoma: a prospective study. J Nucl Med. 2016;57(11):1685–91.
Funding
This study was funded in part by the National Natural Science Foundation of China (NSFC) 81971651, 82171982. Natural Science Foundation of Fujian 2019J01454, 2020J05249. Fujian Provincial Health Commission Science and Technology and Programme 2020GGA045.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was approved by the institutional review board of our hospital and written informed consent for publication of this report was obtained from the patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, Y., Zheng, S., Zhang, J. et al. 68Ga-DOTA-FAPI-04 PET/CT imaging in radioiodine-refractory differentiated thyroid cancer (RR-DTC) patients. Ann Nucl Med 36, 610–622 (2022). https://doi.org/10.1007/s12149-022-01742-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-022-01742-8