Abstract
This study aimed to evaluate the various neuro-otological symptoms experienced by patients with COVID-19 disease. This is a retrospective study conducted from September 2020 to August 2021. Patients with positive RTPCR tests for COVID-19, aged between 18 and 60 years were included in the study. The patients were assessed for neuro-otological symptoms, the type, frequency, and character of these symptoms, their relation with age, gender and COVID-19 disease. Of the 286 patients, 64 (22.3%) had neuro-otological symptoms. The mean age of the patients was 36.3 ± 8.1 years. The frequency of neuro-otological symptoms was higher in females than males and was more frequent in the age group of 18–30 years as compared with other age groups. Of these 64 patients, 29 had vertigo (10.1%), 21 (7.3%) tinnitus, 16 (5.5%) experienced hearing loss. Like many viral diseases, apart from its typical prodromal symptoms, COVID-19 can also cause symptoms like tinnitus, hearing loss, and vertigo.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In the realm of infectious diseases, a pandemic is the worst-case scenario. Several pandemics have emerged in the last two decades like Zika Virus, Middle East Respiratory Syndrome Coronavirus (MERS-CoV), Swine flu, Severe Acute Respiratory Syndrome (SARS) [1], COVID-19 disease is one such pandemic that barrelled through 180 countries to become a global pandemic and a public health emergency of international concern.
COVID-19 diseases, also called Severe Acute Respiratory Syndrome (SARS), was first identified in Wuhan, China. The onset of transmission is believed to have possibly started with bats, spread to cats and then to humans in China, followed by its spread across all borders [2, 3]. SARS characteristically manifests as a respiratory disease, generally 5 days after infection (1–14 days), with symptoms like dry cough, fever, headache, body aches and shortness of breath. Other symptoms can include: loss of taste or smell, headache, chest pain, conjunctivitis, nausea, vomiting, diarrhoea and rashes. It spreads through respiratory droplets and aerosols from coughs and sneezes [4,5,6,7]. The severity of COVID-19 symptoms can range from very mild to severe. Some people may have only a few symptoms, and some people may have no symptoms at all, also called asymptomatic and some people may experience worsened symptoms, such as worsened shortness of breath and pneumonia [4,5,6,7]. SARS-CoV-2 infection can also affect extrapulmonary, sensory, and neural systems, manifesting as sudden onset olfactory and/or gustatory dysfunction [8, 9], otologic symptoms [10,11,12], and neurological complications [13, 14]. It has been shown that the pathogenicity of SARS-CoV-2 is carried out by a surface receptor, angiotensin-converting enzyme 2, or ACE2 [15]. The ACE2 receptor is present on many cell of organs such as the heart, kidney, lung, central and peripheral nervous system and acts as a functional receptor for the virus [16].
While the virus is well-known for its trademark effects on respiratory function, neuro-otological damage has been reported to affect a considerable proportion of cases manifesting as vertigo, tinnitus, hearing loss [17]. The study aims is to describe uncharacteristic but not uncommon neuro-otologic manifestations of SARS-CoV-2 infection like vertigo, tinnitus, hearing loss and to study the prevalence and possible mechanisms of the onset of these symptoms.
Materials and Method
The retrospective study was conducted in the Department of ENT and HNS, GMC and SMGS hospital, Jammu from September 2020 to August 2021. Verbal consent was obtained from patients or their attendants. The study was performed in accordance with the principles of the Declaration of Helsinki. This study was approved by the Ethics Committee of our college.
Inclusion Criteria
The study group included 286 patients with a positive nasopharyngeal swab for COVID-19.
Exclusion Criteria
Hospitalization in Intensive Care Unit due to severe COVID-19 disease.
History of psychiatric or neurological disorders.
History of acoustic trauma.
History of prolonged noise exposure.
Presence of known audiological pathologies before the diagnosis of covid-19.
History of previous ear surgery.
History of treatment with chloroquine or hydroxychloroquine for their reported ototoxic effects.
Patients who refused to take part in the study.
Patients who were isolated in their homes were questioned by teleconsultation and patients who were admitted were questioned and examined by ENT doctors on COVID ward duty.
We used Dizziness Questionnaire provided by the House Ear Clinic [18].
Dizziness Questionnaire



Statistical Analysis
Microsoft Excel was used to evaluate the data. Statistics for all data were presented as mean ± standard deviation, number and percentages.
Results
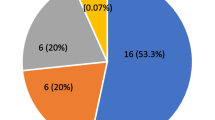
There were neuro-otological symptoms in 64 (22.3%) of 286 patients. Of these 64 patients, 29 had vertigo (10.1%), 21 (7.3%) tinnitus, 16 (5.5%) experienced hearing loss (Fig. 1).
Demographics
Forty-five (70.3%) of the 64 patients with neuro-otological symptoms were females and 19 (29.6%) were males, with a male:female ratio of 2.3:1. These symptoms were seen at a higher frequency in females compared to males (Fig. 2).
The average age of the respondents was 36.3 ± 8.1 years. When patient with neuro-otological symptoms were divided into 5 age groups, it was revealed that the symptoms were mostly observed in the 18–30 years age group (Fig. 3).
Tinnitus
Twenty-one (7.4%) patients, 12 females and 7 males complained of tinnitus. The characteristics of tinnitus as described by the patient were as follows and shown in Figs. 4 and 5:
-
3 (14.2%) patients had tinnitus in both ears.
-
18(85.7%) patients had tinnitus in one ear.
-
17 (80%) had episodic.
-
4 (19%) had persistent.
-
11 (52.3%) had soft tinnitus (Heard only in a quiet situation).
-
7 (33.3%) had moderately loud tinnitus (Heard only in an ordinary situation).
-
3 (14.2%) had loud tinnitus (Heard and noticed in all situations).
Equilibrium Disorder
Ninety-two patients (32.1%) reported dizziness and out of these, 29 (31.5%) patients had vertigo according to the dizziness questionnaire used, out of which, 22 females and 7 males complained of vertigo. The features associated with vertigo as described by the patient were as follows and shown in Figs. 6 and 7:
-
13 (44.8%) experienced vertigo during the COVID-19 disease isolation.
-
16 (55.2%) patients experienced after 14 days of isolation.
-
23 (79.3%) had associated mild imbalance which was more significant during initial attacks.
-
22 (75.8%) had associated nausea and vomiting.
-
11 (37.9%) vertigo was triggered by a change in head position.
In 13 (44.8%) patients, initially, the duration of an acute attack of vertigo lasted for 3 h and then the intensity was gradually improved with consecutive episodes and in 16 (55.1%) patients it was limited to minutes.
Our patients did not show any other neurologic signs and symptoms (dysarthria, dysphagia, weakness, sensory loss, vision loss, loss of consciousness or facial droop) based on the questionnaire.
Hearing Loss
Sixteen (5.5%) patients, 11 females and 5 males experienced reduced hearing after the COVID-19 diagnosis. The characteristics of hearing loss as described by the patient were as follows:
-
5 (31%) patients had reduced hearing in both ears.
-
11(68.7) patients had reduced hearing in one ear.
Nine (14.6%) reported both vertigo and tinnitus, 11 patients (17.1%) reported both vertigo and hearing loss, 13 patients (20.3%) experienced both tinnitus and hearing loss and four patients (6.25%) reported all the three, after COVID-19 diagnosis.
Discussion
Coronaviruses (CoVs; family Coronaviridae, subfamily Coronavirinae) are enveloped, positive-stranded, RNA viruses having large genomes that are susceptible to recombination and mutations [19,20,21]. The mode of transmission of COVID-19 is through direct contact or respiratory droplets from an infected individual and indirect contact through fomites can also act as a vehicle of transmission [22]. As soon as the virus enters the body, the nuclear envelope is derived from the host cell’s membrane. S-protein spikes (glycoprotein spikes embedded within that membrane) allow the cell to attach to a new cell and infect it [23]. S-proteins interact with the cell surface receptor, angiotensin-converting enzyme 2, or ACE2 [15]. The ACE2 receptor is present on cells of organs such as the heart, kidney, lungs, central and peripheral nervous system. S-Proteins contain an S1/S2 activation cleavage site that is activated by furin (serine endoprotease). Furin using its enzymatic activity causes irreversible cleavage of precursor proteins to enter their biologically active state. This furin autocleavage helps the S-protein’s subunits to separate, and allow for the virus to enter host cells [15]. The interaction of the glycoproteins of SARS-CoV-2 to the ACE2 receptor cause subsequent cycles of viral replication, eventually leading to neuronal damage if neural tissues are involved [24]. Some viral infections are known to cause hearing loss like cytomegalovirus, rubella and measles precede sensorineural hearing loss [25, 26]. Most of them typically damaging inner ear cells [27]; however, some viruses can also affect the auditory brainstem [27]. Some studies have reported the role of some viral infectons in vertigo pathogenesis as Epstein-Barr virus, cytomegalo virus, rubella virus, adenoviruses, influenza virus and herpes simplex virus [28].
It is often postulated that infection with neurotropic viruses can cause vestibular neuritis which occurs due to chronic inflammation of nerve. Also, vestibular nerve degeneration with sparing of the peripheral receptor structure has been suggested. Some authors opinionate that the superior vestibular nerve is more commonly involved in this disease as compared to the inferior one; this may be attributed to the longer and narrower bony canal traversed by the superior nerve, making it more susceptible to compressive swelling [29]. Direct vestibular nerve infection by the virus is a possible mechanism as proposed by some studies [30]. Milionis et al. linked vestibular neuritis to the increase in levels of Plasma fibrinogen and C-reactive protein (CRP) as a result of the inflammatory event of the viral infection [31]. Bumm et al. supported the immunological theory in vestibular neuritis pathogenesis. They detected the presence of T4 T-helper and T8 T-suppressor cells by specific monoclonal antibodies in inner ear diseases (e.g., vestibular neuritis, sudden hearing loss, Bell’s palsy, and Meniere’s disease) [32].
Coronavirus related neuro-otological symptoms, such as tinnitus and balance disorders, have been described [10, 23, 33,34,35,36,37], as neurotrophic and neuroinvasive capabilities are typical of some coronaviruses [38]. Moreover, a novel and interesting link between COVID-19 and newly diagnosed hearing loss, vertigo and tinnitus have been reported [12]. Some reports linked vertigo to post-covid infection [39, 40]. This association is not elucidated sufficiently and the published studies about it are few. The mechanism of vestibular neuritis in SARS-COV-2 is probably by viral or post-viral inflammatory disorder [41].
Demographics
Our study results are congruent with the previous study in regards with the demographics; neuro-otological symptoms were common in the young adult age group and in females.
Hearing Loss
Our results show 4.8% of the included patients had hearing loss, which is congruent with the previous studies. Hearing loss has been reported in previous studies [16, 23, 33, 38, 42,43,44]. Sriwijitalai and Wiwanitkitb first reported neurosensory hearing loss in one patient (1.22%) out of 82 patients [38]. Elibol et al. studied 155 patients, out of which 89 (57.4%) patients had otolaryngological symptoms. Of these 89 patients, 2 (1.2%) tinnitus, 1 (0.6%) had Bell’s palsy, 1 (0.6%) had sudden hearing loss problems [33]. These studies also provide no evidence considering the severity (i.e., slight to profound) and the type of the hearing loss (i.e., sensory-neural hearing loss (SNHL), conductive hearing loss (CHL), or mixed hearing loss), or the potential pathology involved. A study by Mustafa et al. reported high-frequency pure tone thresholds and TEOAE amplitudes were significantly worse in COVID-19 patients [43]. Regarding the potential type of hearing loss, few case series [37, 45] and case reports [34, 45,46,47,48] have reported SNHL as the type of HL in COVID-19 patients and theorized that it results from the direct impact of SARS-CoV-2 on the organ of Corti, stria vascularis, and/or spiral ganglion [43, 49].
Vertigo
Dizziness is an umbrella term in medicine that is classified using patients’ history into vertigo, disequilibrium, presyncope, and lightheadedness [50, 51]. Vertigo is a false sensation of whirling or rotation originating from the vestibular system, the main causes of vertigo are benign paroxysmal positional vertigo, Ménière’s disease, vestibular neuritis, and labyrinthitis [52] Disequilibrium is feeling off-balance or wobbly, the main causes are Parkinson’s disease and diabetic neuropathy. Presyncope is the feeling of losing consciousness or blacking out. Lightheadedness is feeling disconnected from the environment, is commonly associated with psychogenic or psychiatric origins such as anxiety, depression, and somatoform disorders [53].The inner ear structures are susceptible to ischemia and vascular damage, which eventually leads to auditory and balance dysfunction [41]. It has been studied that vasculitis also is one of the clinical manifestations of COVID-19 [54] which can be a possible mechanism of auditory and balance dysfunction.
Vestibular neuritis or acute peripheral vestibulopathy is a viral or post-viral inflammatory disease, which involves the vestibular part of the eighth cranial nerve [55]. The disease is clinically diagnosed with vertigo and develops acutely over minutes to hours, accompanied by nausea and vomiting, possibly due to irritation of the emetic tracts which are in proximity with the vestibular nerve/nuclei [56].
In our study, vertigo was present in 9.4% of cases which is similar to the percentage of vertigo reported in some studies [11, 16, 43, 58] but some studies have results incongruent with ours [42, 57, 58]. Various case reports [37, 39, 59,60,61,62,63] have been published describing acute vertigo attacks in COVID-19 patients. Viola et al. did a multicentre study that included 15 hospitals using an online 10-item close-ended questionnaire developed to identify the presence of tinnitus and balance disorders in patients with COVID-19. Thirty-four patients (18.4%) reported equilibrium disorders after the COVID-19 diagnosis. Of these, 32 patients reported dizziness (94.1%) and 2 (5.9%) reported acute vertigo attacks. Forty-three patients (23.2%) reported tinnitus; 14 (7.6%) reported both tinnitus and equilibrium disorders [42]. Some authors have presented results different from ours like Panda et al., who evaluated 145 patients with mild COVID-19 to report that 96 patients (66.2%) had ENT manifestations, out of which 7 (1%) patients accounted for vertigo [57]. Picciotti et al. studied eight patients (4 F, 4 M, aged between 44 and 69 years) with COVID-19 infections complaining of vertigo and concluded the cause to be benign paroxysmal positional vertigo (BPPV). He has stated that vestibular evaluation showed involvement of posterior semicircular canals in five patients and horizontal in three. They hypothesized that BPPV in COVID-19 infections can be related to drugs, prolonged bed rest and direct damage by a viral infection on the peripheral vestibular system and in particular on the otolithic membrane due to the cytopathic effect of the virus and to the inflammatory response [58]. Mao et al. studied 214 hospitalized patients with laboratory confirmed diagnosis of severe acute respiratory syndrome coronavirus-2 infection, 78 patients (36.4%) had neurologic manifestations, out of which 36 (16.8%) had dizziness however, they have not described the type or cause of dizziness [14]. Han et al. has reported a case study of a 47-year-old man who came to the emergency department with a chief complaint of fever, cough, stuffy nose, nausea and vertigo. [59]. Karam et al. reported a case study of 60-year-old diabetic and hypertensive female patient with a 9-days history of COVID-19 symptoms who presented to the emergency department with acute vertigo attack without nausea or vomiting [60].
Tinnitus
In our study, tinnitus was present in 7.4% of cases which corresponds to the percentage reported in few studies [11, 16, 23] but some studies have results incongruent with ours [7, 33, 42, 57, 64]. Elibol et al., Lechien et al. and Kardas et al. have reported tinnitus in 1.2, 0.3 and 2% while Viola et al. has reported 23.2% cases complaining of tinnitus. Tinnitus in patients with COVID-19 has been attributed to SARS-CoV-2 on the auditory system and also to the anxiety caused by the unpredictability of the disease [65].
Limitations of the Study
Our study has several limitations which may have affected the outcome of the study. It has a small population size as compared with some other studies which have done multicentric research over a larger geographical area. We have not assessed the type of hearing loss and have not compared the severity of COVID-19 with neuro-otological symptoms. Also, the patients who were home isolated during the COVID-19 infection could not be assessed and evaluated clinically.
Conclusion
COVID-19 is a viral infection that affects respiratory, gastrointestinal, cardiac and nervous systems varying from person to person. There is no doubt it primarily affects the respiratory tract but this study along with many other studies prove the diverse manifestations of COVID-19, among which include vertigo, tinnitus and hearing loss. This is the first study from our region focussing only on neuro-otological symptoms of COVID-19 and unlike many other studies; we have not used online reporting forms. We have used the teleconsultation facility of our department for home isolated patients and the hospitalized patients were assessed by ENT surgeons on COVID duty.
References
Piret J, Boivin G (2021) Pandemics throughout history. Front Microbiol 15(11):631736. https://doi.org/10.3389/fmicb.2020.631736
Guan WJ, Ni ZY, Hu Y et al (2020) Clinical characteristics of coronavirus disease 2019 in China. N Eng J Med 382:1708–1720
Yuki K, Fujiogi M, Koutsogiannaki S (2020) COVID-19 pathophysiology: a review. Clin Immunol 215:108427.
Krishnan A, Hamilton JP, Alqahtani SA et al (2021) A narrative review of coronavirus disease 2019 (COVID-19): clinical, epidemiological characteristics, and systemic manifestations. Intern Emerg Med 16(4):815–830
Singhal T (2020) A review of Coronavirus Disease-2019 (COVID-19). Indian J Pediatr 87(4):281–286
Wu Z, McGoogan JM (2020) Characteristics of, and important lessons from the Coronavirus Disease 2019 (COVID-19) Outbreak in China: summary of a report of 72314 cases from the Chinese center for disease control, and prevention. JAMA 25(3):278–280
Lechien JR, Chiesa-Estomba CM, Place S, Van Laethem Y, Cabaraux P et al (2020) Clinical and epidemiological characteristics of 1420 European patients with mild-to-moderate Coronavirus Disease. J Intern Med 288(3):335–344
Agyeman AA, Chin KL, Landersdorfer CB, Liew D, Ofori-Asenso R (2020) Smell and taste dysfunction in patients with COVID-19: a systematic review and meta-analysis. Mayo Clin proceed 95:1621–1631
Rocke J, Hopkins C, Philpott C, Kumar N (2020) Is loss of sense of smell a diagnostic marker in COVID-19: a systematic review and meta analysis. Clin Otolaryngol 45:914–922
Almufarrij I, Uus K, Munro KJ (2020) Does coronavirus affect the audiovestibular system? A rapid systematic review. Int J Audiol 59:487–491
Saniasiaya J, Kulasegarah J (2020) Auditory cinchonism in COVID Era. Ear Nose Throat J 99:597–598
Maharaj S, Bello Alvarez M, Mungul S, Hari K (2020) Otologic dysfunction in patients with COVID-19: a systematic review. Laryngoscope Investiga Otolaryngol 5:1192–1196
Abboud H, Abboud FZ, Kharbouch H, Arkha Y, El Abbadi N, El Ouahabi A (2020) COVID-19 and SARS-Cov-2 infection: pathophysiology and clinical effects on the nervous system. World Neurosurg 140:49–53
Mao L, Jin H, Wang M et al (2020) Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan. China JAMA Neurol 77:683–690
Sandoiu A (2020) Why does SARS-CoV-2 Spread So Easily? Brighton, Medical News Today. Available online at: https://www.medicalnewstoday.com/articles/why-does-sars-cov-2-spread-so-easily#Spike-protein-on-the-new-coronavirus. Accessed April 12, 2020.
Özçelik KM, Eğilmez OK, Özçelik MA, Güven M (2020) Otolaryngological manifestations of hospitalised patients with confirmed COVID-19 infection. Eur Arch Otorhinolaryngol 278(5):1675–1685
Harapan BN, Yoo HJ (2021) Neurological symptoms, manifestations, and complications associated with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-and coronavirus disease 19 (COVID-19). J Neurol 23:1–13
PATIENT QUESTIONNAIRES Dizziness/Vertigo (2011) Otolaryngologic Clin North Am 44(2):497–507
Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W et al (2020) A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 579:270–273. https://doi.org/10.1038/s41586-020-2012-7
Li JY, You Z, Wang Q, Zhou ZJ, Qiu Y, Luo R et al (2020) The epidemic of 2019-novel-coronavirus (2019-nCoV) pneumonia and insights for emerging infectious diseases in the future. Microbes Infect 22:80–85. https://doi.org/10.1016/j.micinf.2020.02.002
Forni D, Cagliani R, Clerici M, Sironi M (2017) Molecular evolution of human coronavirus genomes. Trends Microbiol 25:35–48. https://doi.org/10.1016/j.tim.2016.09.001
Fiani B, Covarrubias C, Desai A, Sekhon M, Jarrah R (2020) A contemporary review of neurological sequelae of COVID-19. Front Neurol 11:640. https://doi.org/10.3389/fneur.2020.00640
Dougherty P (2020) How Furin and ACE2 Interact with the Spike on SARS-CoV-2 Assay Genie. Available online at: https://www.assaygenie.com/how-furin-and-ace2-interact-with-the-spike-on-sarscov2.
Baig AM, Khaleeq A, Ali U, Syeda H (2020) Evidence of the COVID-19 virus targeting the CNS: tissue distribution, host–virus interaction, and proposed neurotropic mechanisms. ACS Chem Neurosci 11:995–998. https://doi.org/10.1021/acschemneuro.0c00122
Young YH (2020) Contemporary review of the causes and differential diagnosis of sudden sensorineural hearing loss. Int J Audiol 59(4):243–253. https://doi.org/10.1080/14992027.2019.1689432
Cohen BE, Durstenfeld A, Roehm PC (2014) Viral causes of hearing loss: a review for hearing health professionals. Trends Hear 18:1–17. https://doi.org/10.1177/2331216514541361
Abramovich S, Prasher DK (1986) Electrocochleography and brain-stem potentials in Ramsay Hunt syndrome. Arch Otolaryngol Head Neck Surg 112(9):925–928. https://doi.org/10.1001/archotol.1986.03780090021002
Hirata T, Sekitani T, Okinaka Y, Matsuda Y (1989) Seroviro-logical study of vestibular neuronitis. Acta Otolaryngol Suppl 468:371–373
Flint PW (2021) Cummings otolaryngology, 7th edition. Mosby/Elsevier, Philadelphia, pp 2320–35
Greco A, Macri GF, Gallo A, Fusconi M, De Virgilio A, Pagliuca G et al (2014) Is vestibular neuritis an immune related vestibular neuropathy inducing vertigo? J Immunol Res 2014:459048
Milionis HJ, Mittari V, Exarchakos G, Kalaitzidis R, Skevas AT, Elisaf MS (2003) Lipoprotein (a) and acute-phase response in patients with vestibular neuronitis. Eur J Clin Investig 33(12):1045–1050
Bumm P, Schlimok G (1986) Lymphocyte subpopulations and HLA-DR determinations in diseases of the inner ear and Bell’s palsy. HNO 34(12):525–527
Elibol E (2020) Otolaryngological symptoms in COVID-19. Eur Arch Otorhinolaryngol 27(4):1233–1236. https://doi.org/10.1007/s00405-020-06319-7
Malayala SV, Raza A (2020) A case of COVID-19-induced vestibular neuritis. Cureus 12(6):e8918.
Munro KJ, Uus K, Almufarrij I, Chaudhuri N, Yioe V (2020) Persistent self-reported changes in hearing and tinnitus in post-hospitalisation COVID-19 cases. Int J Audiol 1:1–2
Freni F, Meduri A, Gazia F, Nicastro V, Galletti C, Aragona P (2020) Symptomatology in head and neck district in coronavirus disease (COVID-19): a possible neuroinvasive action of SARS-CoV-2. Am J Otolaryngol 41(5):102612. https://doi.org/10.1016/j.amjoto.2020.102612
Karimi-Galougahi M, Naeini AS, Raad N, Mikaniki N, Ghorbani J (2020) Vertigo and hearing loss during the COVID-19 pandemic-is there an association? Acta Otorhinolaryngol Ital. https://doi.org/10.14639/0392-100X-N0820
Sriwijitalai W, Wiwanitkit V (2020) Hearing loss and COVID-19: a note. Am J Otolaryngol 41(3):102473. https://doi.org/10.1016/j.amjoto.2020.102473
Malayala SV, Raza A (2020) A case of COVID-19-induced vestibular neuritis. Cureus 12(6):8918
Vanaparthy R, Malayala SV, Balla M (2020) COVID-19-induced vestibular neuritis, hemi-facial spasms and Raynaud’s phenomenon: a case report. Cureus 12(11):11752
Lee H, Sohn SI, Cho YW, Lee SR, Ahn BH, Park BR et al (2006) Cerebellar infarction presenting isolated vertigo: frequency and vascular topographical patterns. Neurology 67(7):1178–1183
Viola P, Ralli M, Pisani D, Malanga D, Sculco D, Messina L et al (2021) Tinnitus and equilibrium disorders in COVID-19 patients: preliminary results. Eur Arch Otorhinolaryngol 278(10):3725–3730. https://doi.org/10.1007/s00405-020-06440-7
Mustafa MWM (2020) Audiological profile of asymptomatic Covid-19 PCR-positive cases. Am J Otolaryngol 42(3):102483. https://doi.org/10.1016/j.amjoto.2020.102483
Zahran, M, Ghazy R, Ahmed O et al (2021) Atypical otolaryngologic manifestations of COVID-19: a review. Egypt J Otolaryngol 37(5). https://doi.org/10.1186/s43163-021-00075-z
Abdel Rhman S, Abdel Wahid A (2020) COVID-19 and sudden sensorineural hearing loss, a case report. Otolaryngol Case Rep 16:100198.
Degen C, Lenarz T, Willenborg K (2020) Acute profound sensorineural hearing loss after COVID-19 pneumonia. Mayo Clin Proceed 95:1801–1803
Koumpa FS, Forde CT, Manjaly JG (2020) Sudden irreversible hearing loss post-COVID-19. BMJ Case Rep 13:e238419.
Lang B, Hintze J, Conlon B (2020) Coronavirus disease 2019 and sudden sensorineural hearing loss. J Laryngol Otol, pp 1–3.
Uranaka T, Kashio A, Ueha R (2020) Expression of ACE2, TMPRSS2, and furin in mouse ear tissue. bioRxiv.
Post R, Dickerson L (2010) Dizziness: a diagnostic approach. Am Fam Physician 82:361–369
Muncie HL, Sirmans SM, James E (2017) Dizziness: approach to evaluation and management. Am Fam Physician 95(3):154–162
Gelfand AE (2016) Essentials of audiology, 4th edn. Thieme, New York
Kim SK, Kim YB, Park IS, Hong SJ, Kim H, Hong SM (2016) Clinical analysis of dizzy patients with high levels of depression and anxiety. J Audiol Otol 20:174–178
Roncati L, Ligabue G, Fabbiani L et al (2020) Type 3 hypersensitivity in COVID-19 vasculitis. Clin Immunol 217:108487.
Hegemann SCA, Wenzel A (2017) Diagnosis and treatment of vestibular neuritis/neuronitis or peripheral vestibulopathy (PVP)? Open questions and possible answers. Otol Neurotol 38:626–631
Yates BJ, Catanzaro MF, Miller DJ, McCall AA (2014) Integration of vestibular and emetic gastrointestinal signals that produce nausea and vomiting: potential contributions to motion sickness. Exp Brain Res 232:2455–2469
Panda S, Mohamed A, Sikka K et al (2021) Otolaryngologic manifestation and long-term outcome in mild COVID-19: experience from a Tertiary Care Centre in India. Indian J Otolaryngol Head Neck Surg 73:72–77. https://doi.org/10.1007/s12070-020-02217-w
Picciotti PM, Passali GC, Sergi B, De Corso E (2021) Benign Paroxysmal Positional Vertigo (BPPV) in COVID-19. Audiol Res 11:418–422. https://doi.org/10.3390/audiolres11030039
Han W, Quan B, Guo Y, Zhang J, Lu Y, Feng G, Wu Q, Fang F, Cheng L, Jiao N, Li X, Chen Q (2020) The course of clinical diagnosis and treatment of a case infected with coronavirus disease 2019. J Med Virol 92(5):461–463. https://doi.org/10.1002/jmv.25711
Motawea KR, Monib FA (2021) New onset vertigo after COVID-19 infection. A case report. Indian J Otolaryngol Head Neck Surg 4:1–3. https://doi.org/10.1007/s12070-021-02715-5
Fadakar N, Ghaemmaghami S, Masoompour SM et al (2020) A first case of acute cerebellitis associated with coronavirus disease (COVID-19): a case report and literature review. Cerebellum 19:911–914
Liu C, Zhou J, Xia L, Cheng X, Lu D (2020) 18F-FDG PET/CT and serial chest CT findings in a COVID-19 patient with dynamic clinical characteristics in different period. Clin Nucl Med 45:495–496
Chern A, Famuyide AO, Moonis G, Lalwani AK (2021) Bilateral sudden sensorineural hearing loss and intralabyrinthine hemorrhage in a patient with COVID-19. Otol Neurot 42:e10–e14
Karadaş Ö, Öztürk B, Sonkaya AR (2020) A prospective clinical study of detailed neurological manifestations in patients with COVID-19. Neurol Sci 41:1991–1995
Beukes E, Baguley D, Jacquemin L et al (2020) Changes in tinnitus experiences during the COVID-19 Pandemic. Front Public Health 8:592878.
Funding
No funding was received for conducting this study. The authors declare that they have no financial interest.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, analysis and the first draft of the manuscript was written by Dr. Faizah Deva and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
None.
Ethics Approval
Approval was obtained from the ethics committee of our college. The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Deva, F.A.L., Gupta, A., Manhas, M. et al. Neuro-otological Symptoms: An Atypical Aspect of COVID-19 Disease. Indian J Otolaryngol Head Neck Surg 74 (Suppl 2), 3273–3282 (2022). https://doi.org/10.1007/s12070-022-03088-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12070-022-03088-z