Abstract
To see the radiological anatomy of inner ear malformations in hearing impaired children. This study was a prospective observational study done at a tertiary care teaching hospital in north India. The included were children of 1–7 years with first time detected severe to profound SNHL. The evaluation of hearing was done by brainstem evoked response audiometry. Those with history of chronic suppurative otitis media and conductive hearing loss were excluded. The all included were undergone high resolution computed tomography of the tympano-mastoid region. A written informed consent was taken. The institutional ethics committee approved the study protocol. We screened 130 children of 1–7 years of age with complaints of severe to profound hearing impairment (SNHL) by BERA. We excluded 38 and 14 children who had conductive hearing loss with or without CSOM and did not give consent, respectively. Among recruited 78 children, 47 (60.3%) had severe (71–90 dB) and 31 (39.7%) had profound (> 90 dB) SNHL. The majority of children (60/78, 76.9%) had a bilateral hearing impairment, only 18 (23.1%) had unilateral hearing impairment. The mean ± SD of age was 3.9 ± 1.3 years (95% CI = 3.6–4.2). The incidence of severe to profound SNHL was higher, though not statistically significant among the girls. Only 7.7% children with severe to profound SNHL were detected within first 2 years of life. Most of the children (92.3%) with severe to profound SNHL were detected after 2 years of age. Among 78 with severe to profound SNHL, 16 (20.5%) children had structural anomalies in their internal ears. Only 2 (6.9%) ears had single anomaly, others (27, 93.1%) had multiple anomalies. A maximum number of anomalies found in one ear were 5. The most common anomalies were found in the vestibule. The total number of anomalies were 115. Five children (6.4%) with severe to profound SNHL had craniofacial dysmorphism. Approximately 20% of children with severe to profound SNHL have congenital anomalies in their internal ears. It is crucial to know these anomalies before planning of CI. HRCT of tympano-mastoid is the first modality to know these anomalies. MRI should also be included to know the details of vestibulocochlear nerve. Since, the neurocognitive outcome would be the best if CI will be done as early as possible preferably around 1 year of age. Therefore, children must be screened at the appropriate age, firstly by OAE followed by BERA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The congenital sensorineural hearing loss (SNHL) is a standout amongst the most widely recognized birth defects with an occurrence of roughly 1:1000 live births [1]. The most inner ear malformations occur during the first trimester of the pregnancy when the development of the membranous labyrinth interrupted due to either a result of inborn genetic error or a teratogenic exposure [2].The normal auditory responses from the brain are either delayed or non-existent in deaf children, indicating that brain maturation is highly dependent upon appropriate stimulation [3]. Imaging of cases of congenital SNHL is often performed in an endeavor to decide a hidden pathology. High Resolution Computed Tomography (HRCT) of the temporal bone is a first line imaging investigation of a child suffering from sensorineural hearing loss (SNHL). Since this determines suitability of cochlear implant (CI) and other vital information about anatomy of internal auditory canal (IAC). Therefore, HRCT of the temporal bone is mandatory before CI in children with SNHL. CI must be implanted in cochlear lumen for the maximum result [4]. The IAC is viewed as stenotic if its distance across is littler than 3 mm. The normal breadth fluctuates from 4 to 8 mm [5, 6]. Around 20% of children with SNHL have anatomical defects in IAC [2]. Since, there is a lack of clinical and exploratory examinations to analyze the anatomical and functional aspects of the IAC in children, especially in India. Additionally, there is high inter-individual variability and anatomical variations, therefore further studies are required to dodge misinterpretations and enhance the nature of clinical outcomes [7]. The present study is aimed to see the radiological anatomy of inner ear malformation in hearing impaired children and its correlation with hearing loss.
Methods
This study was a prospective observational study done at Department of Otorhinolaryngology and Head and Neck Surgery of King George’s Medical University, Lucknow, a tertiary care teaching hospital of north India. The study was conducted between July 2014 and June 2016. The included were children of 1–7 years with first time detected severe to profound SNHL. The evaluation of hearing was done by brain stem evoked response audiometry (BERA).Those with a history of chronic suppurative otitis media (CSOM) and conductive hearing loss were excluded. The all included were undergone high resolution computed tomography (HRCT) of the tympano-mastoid region (Philips Brilliance® 64 CT scanner). To minimise motion artefacts, children were sedated with an oral preparation of triclofos at a dose of 20 mg/kg. All the HRCT scans were examined and reported by radiologist and otorhinolaryngologist independently without knowing clinical history of the cases. Whenever there was a different opinion about findings of HRCT scan, the consensus was made by the majority. We recruited 78 children (156 ears) with severe to profound SNHL. A written informed consent was taken from parents or legal guardian before enrollment in the study. The institutional ethics committee approved the study protocol.
The demographic, anthropometric, clinical and investigative data were recorded on pre-formed questionnaires. An endeavour was made to discover the etiology of deafness by the method of a careful history. Etiology factors including congenital factors, prematurity, difficult delivery along with hypoxic ischemic encephalopathy, infectious, neonatal jaundice with kernicterus, ototoxicity and injury. These data were entered into MS excel and analysed by Epi-Info 7.0 software.
Results
We screened 130 children of 1–7 years of age with complaint of hearing impairment (SNHL) by BERA. We excluded 38 and 14 children who had the conductive hearing loss with or without CSOM and did not give consent, respectively. The study flow has been given in Fig. 1. The base line characteristics of the children are given in Table 1. The mean ± SD of age was 3.9 ± 1.3 years (95% CI = 3.6–4.2), median = 3.9 years, IQR = 1.7 (1st quartile 3.1 and 3rd quartile 4.7). The incidence of severe to profound SNHL was higher though not statistically significant among the girls. The TORCH titre, estimated by ELISA method was done only in 23 (29.5%) mothers of recruited children, among them only 3 (13.0%) were positives. The age distribution of recruited children is given in Table 2. Only 7.7% children with severe to profound SNHL were detected within first 2 years of life. Most of the children (32.1 + 26.9 = 59.0%) with severe to profound SNHL were detected between 3 and 5 years of age (Table 2). Among recruited 78 children, 47 (60.3%) had severe (71–90 dB) and 31 (39.7%) had profound (> 90 dB) SNHL. The majority of children (60/78, 76.9%) had the bilateral hearing impairment, only 18 (23.1%) had the unilateral hearing impairment (Table 3).
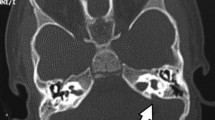
Among 78 with severe to profound SNHL, 16 (20.5%) children had structural anomalies in their internal ears. Bilateral and unilateral abnormalities were found in 13 and 3 children respectfully. So, we studied 29 abnormal ears. Only 2 (6.9%) ears had the single anomaly, others (27, 93.1%) had multiple anomalies. A maximum number of anomalies were found in one ear was 5. The most common anomalies were found in the vestibule. The details of anomalies found in 29 ears are given in Table 4. A total number of anomalies were 115 and their distribution is given in Table 5. We found five children (6.4%) with severe to profound SNHL had craniofacial dysmorphism. They have not categorized any particular syndrome because of lack of complete resemblance to a syndrome. All they had anomalies in their internal ear.
Discussion
The present study found a total number of 115 anomalies in 29 ears of 16 patients by HRCT of tympano-mastoid of 78 children with severe to profound SNHL. Since MRI is better than HRCT only to visualise vestibulocochlear nerves [8], therefore HRCT should not underestimate for the study of temporal bone [9, 10]. We found 20.5% children with severe to profound SNHL had anomalies in their internal ears. A similar incidence was reported by Haider et al. (20%) [11], Jackler et al. (20%) [12] and Sujata de et al. (22%) [13]. There is a range of reporting of anomalies by various other researchers from 14.3% [8] to 42% [14] detected by HRCT of tympano-mastoid. The variations in reporting may be due different age group and severity of hearing impairment in children and types of computed tomography along with improving imaging technologies.
The most common anomalies were related to vestibules. We found 27 ears out of 29 (93.1%) had anomalies in the vestibule. Similar findings were reported by other researchers too. Agarwal et al. [8] reported 87.1% and Sujata et al. 76% [13]. The cochlear anomalies, second most common anomalies with an incidence of 79.3% in the present study were similar to reported by Sujata de et al. (76%) [13]. Vestibular aqueduct dilation was found 55.2% in our study among anomalous ears. Similar incidence is reported by other researchers too including Haider et al. 50% [11], Sujata de et al. 32% [13], Bamiou et al. 30.3% [15].
Since, there is the variable rate of reporting of various congenital anomalies in children with SNHL. This is attributed to various factors in the studies including different sample sizes, imaging technologies and the better understanding of the anatomy of the internal ear in SNHL. Apart from these, there is a need for studies based on larger samples with correlation to other factors including intrauterine infections, genetic constituents etc.
Only 7.7% children with severe to profound SNHL were detected within first 2 years of life. Most of the children (59.0%) with severe to profound SNHL were detected between 3 and 5 years of age and others even later. In other words, a large number of patients (92.3%) are detected severe to profound SNHL after 2 years of age. Since the best result of CI is obtained when this happens within first 2 years of life. Inability to identify early to the children with hearing impairment may result in lifelong deficits in speech and language acquisition, poor scholastic performance and personal-social and behaviour problems [16]. Therefore, early detection of hearing impairment is vital. For this, two steps can be seriously considered. One is to educate the community about when to suspect and benefits of early detection of hearing impairment, so that care seeking behaviour of the parents and care taker can be appropriate. Second is for clinicians, especially ENT surgeons and Pedatricians. They must consider a routine hearing evaluation in every child. If not feasible in resource limited countries like ours it should be mandatory in at least high-risk groups [17] including family history of SNHL, premature birth, hyperbilirubinemia requiring exchange transfusion, bacterial meningitis, perinatal asphyxia, mechanical ventilation for ≥ 5 days, delayed milestones, abnormal phenotypic appearance and mother has been exposed to radiation, teratogenic drugs and TORCH infections.
This study has some limitations. The present study did not include MRI that can detect the abnormalities of vestibulocochlear nerve and vestibular fluid. Absence of vestibulocochlear nerve is a contraindication for CI surgery because this nerve is required to conduct the cochlear implant impulses [18, 19].
Conclusion
Approximately 20% of children with severe to profound SNHL have congenital anomalies in their internal ears. It is crucial to know these anomalies before planning of CI. HRCT of tympano-mastoid region is the first modality to know these anomalies. MRI should also be included to know details of the vestibulocochlear nerve. Since the neurocognitive outcome would be the best if CI will be done as early as possible preferably around 1 year of age. Therefore, children must be screened at an appropriate age, firstly by OAE followed by BERA.
References
St Martin MB, Hirsch BE (2008) Imaging of hearing loss. Otolaryngol Clin North Am 41(1):157–178
Jackler RK (2008) Congenital malformation of inner ear. In: Gleeson M (ed) Scott- Brown’s Otolaryngology, head and neck surgery, vol 3, 7th edn. Hodder Arnold, London, pp 2727–2739
Eggermont JJ, Ponton CW, Don M, Waring MD, Kwong B (1997) Maturational delays in cortical evoked potentials in cochlear implant users. Acta Otolaryngol 117(2):161–163
Gleeson TG, Lacy PD, Bresnihan M, Gaffney R, Brennan P, Viani L (2003) High resolution computed tomography and magnetic resonance imaging in the pre-operative assessment of cochlear implant patients. J Laryngol Otol 117(9):692–695
Guirado RC (1992) Malformations du conduit auditif interne. Revue de laryngologie, otologie, rhinologie, Bordeaux
Valvassori GE, Pierce RH (1964) The Normal Internal Auditory Canal. Am J Roentgenol Radium Ther Nucl Med 92:1232–1241
Chakeres DW (1984) CT of ear structures: a tailored approach. Radiol Clin North Am 22(1):3–14
Agarwal SK, Singh S, Ghuman SS, Sharma S, Lahiri AK (2014) Radiological assessment of the indian children with congenital sensorineural hearing loss. Int J Otolaryngol. 2014:808759
Woolley AL, Oser AB, Lusk RP, Bahadori RS (1997) Preoperative temporal bone computed tomography scan and its use in evaluating the pediatric cochlear implant candidate. Laryngoscope 107(8):1100–1106
Seidman DA, Chute PM, Parisier S (1994) Temporal bone imaging for cochlear implantation. Laryngoscope 104(5):562–565
Haider AG, Hani MB, Mohammed AH (2011) CT scan value of temporal bone in assessment of congenital deafness. Fac Med Baghdad 4:367–370
Jackler RK, Luxford WM, House WF (1987) Congenital malformations of the inner ear: a classification based on embryogenesis. Laryngoscope 97:2–14
De Sujata, Archbold S, Clarke R (2008) Investigation and management of deaf child. In: Gleeson M (ed) Scott-brown’s otolaryngology, head and neck surgery, vol 1, 7th edn. Hodder Arnold, London, pp 845–859
Simons JP, Mandell DL, Arjmand EM (2006) Computed tomography and magnetic resonance imaging in pediatric unilateral and asymmetric sensorineural hearing loss. Arch Otolaryngol Head Neck Surg 132(2):186–192
Bamiou DE, Phelps P, Sirimannaet T (2000) Temporal bone computed tomography findings in bilateral sensorineural hearing loss. Arch Dis Child 82:257–260
Stevenson J, McCann D, Watkin P, Worsfold S, Kennedy C (2010) The relationship between language development and behaviour problems in children with hearing loss. J Child Psychol Psychiatry 51:77–83
Joint Guidelines on Infant Hearing (2007) Year 2007 position statement: principles and guidelines for early hearing detection and intervention programs. Pediatrics 120:898–921
Lenarz TH, Hartrampf R, Battmer R, Bertram B, Lesinski A (1996) Cochlear implantation in very young children. Laryngorhinootologie 75(12):719–726
Gray RF, Ray J, Baguley DM, Vanat Z, Begg J, Phelps PD (1998) Cochlear implant failure due to unexpected absence of the eighth nerve—a cautionary tale. J Laryngol Otol 112(7):646–649
Author information
Authors and Affiliations
Contributions
SK, BNM, SPA, KKY conceptualized, designed the study. SK, BNM collected the data. SK and KKY analysed the data. SK and KKY wrote the manuscript. The final version of the manuscript was approved by all authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Kumar, S., Majhi, B.N., Yadav, K.K. et al. Radiological Anatomy of Inner Ear Malformation in Hearing Impaired Children and it’s Correlation with Hearing Loss: A Hospital Based Observational Study. Indian J Otolaryngol Head Neck Surg 70, 278–283 (2018). https://doi.org/10.1007/s12070-017-1238-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12070-017-1238-7