Abstract
Stress is an overwhelming problem associated with neuronal damage leading to anxiety and depression. The compound 3, 4, 5-trimethoxycinnamic acid (TMCA) has shown anti-stress effects; however, its derivatives remained unknown for their anxiolytic properties. Here, therefore, we investigated derivatives of TMCA (dTMCA) for their anxiolytic effects using immobilization and electric shock-induced stress in rats. Derivatives of TMCA ameliorated anxiety in mice and rats revealed by extended period of time spent in the open arms of elevated plus maze. Stress-mediated repression of tyrosine hydroxylase (TH) protein expression in the amygdala regions of rat brain and dopamine levels in the PC12 cells was restored by two selected derivatives (TMCA#5 and TMCA#9). Unlike TH expression, stress-induced protein expression of phospho-extracellular signal-regulated kinase (pERK) was unaffected by both derivatives in rats. Given the preferential inhibitory activity of dTMCA on dopamine and serotonin receptors, serotonergic road map of cellular signaling could be their target for anxiolytic effects. Thus, dTMCA would be promising agents to prevent neuronal damage associated with rampant stressful conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Stress is the major causative factor for abnormal behaviors such as anger, anxiety, and depression. Many stress models exist in experimental animals, among which immobilization [1] and electric shock stress [2] are most commonly used and easy to implement. Immobilization stress has been used extensively and accepted widely for studying physical and psychological alterations during stress.
Stress is associated with an increase in tyrosine hydroxylase expression in neurons of ventral tegmental area (VTA) and amygdala regions of the brain as a mechanism of adaptation. Tyrosine hydroxylase (TH) is considered to be the rate-limiting enzyme in the biosynthesis of catecholamine at central and peripheral nervous system [2]. Studies indicated that TH activity in the brain and adrenal gland was modulated during immobilization and electroconvulsive shock stressors [2]. Etievant et al. reported that forced swim test showed marked reduction in TH activity in the brain [3]. Given the detailed knowledge of catecholamine signaling pathways in the brain, many investigators have examined the regulation of TH activity within specific catecholamine cell body regions of the brain and the areas to which they ramify in an attempt to understand the function of catecholamine systems in the central nervous system [4].
Mitogen-activated protein kinase (MAPK) pathway is an intracellular signaling cascade implicated in learning, memory, immune system, and stress [5]. Extracellular signal-regulated protein kinases 1 and 2 (ERK1/2) has been documented in response to ischemia, visceral pain, and electric shock stress. Shen et al. demonstrated that restraint stress activates ERK-MAPK pathway [6] thereby ERK1/2 phosphorylation has been hypothesized as an intracellular signaling mechanism that mediate antidepressant efficacy; however, evidences were largely limited to studies in naive rodents or in vitro models [7] in spite of few reports obtained from postmortem studies in depressed suicide subjects [8]. It is also uncertain whether ERK1/2 is involved in reversing depression during antidepressant therapy or its phosphorylation is uniformly regulated in cortico-limbic circuits [9].
Studies so far have begun to identify key molecules from natural products to develop therapeutic agents against stress-related diseases [2]. 3, 4, 5-trimethoxycinnamic acid (TMCA) is one of the constituents of Onji (roots of Polygala tenuifolia), an important herb used in oriental medicine for sedative and anxiolytic effects [1]. The anxiolytic effects of TMCA have been mediated through serotonin (5-HT) subtype 1A receptor (5-HT1A) and gamma-aminobutyric acid type A (GABAA) receptors [10] thereby exploring the crucial role of serotonergic signaling in stress-related conditions [11]. In stressed mice, TMCA augmented restoration of repressed nuclear receptor related-1 (Nurr1) protein expression [2], which is closely linked with the development of dopaminergic neuron in the midbrain. Previously, we synthesized and evaluated derivatives of TMCA (dTMCA) for anti-narcotic actions [12]. However, their therapeutic potential under stressful conditions remained unknown. Amygdala is associated with anxiety-like behavior induced by restraint stress [13]. TH and phosphorylated ERK (pERK) may play a role in amygdala of corticosterone treated rats [14]. We have been reported that TMCA regulates stress-induced anxiety and has an agonistic activity on 5-HT1A [2, 15]. Therefore, we aimed to study the effects of dTMCA on anxiety induced by stress. Furthermore, we tested whether dTMCA regulates TH and pERK expression in amygdala in stressed rats.
Materials and Methods
Animals
C57BL/6 (male, 20–22 g) at 8 weeks old and Sprague–Dawley rats (SD rats; male, 260–280 g) at 7 weeks old were obtained from Daehan Biolink (Eumsung, Korea). All rodents were kept on 12 h light-dark cycle at a constant temperature of 24 ± 3 °C in animal facility room. The rodents were housed in plastic cages, fed on rodent chow, provided water ad libitum, and allowed to adapt the new environment for 1 week before the experiment. The study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. All experiments based on the guidelines approved by the Animal Care and Committee of Ewha Womans University and Chungbuk National University (CBNUA-436-12-02). The schedule of animal experiments and information on experiments group is described in Fig. S1.
Chemicals
8-Hydroxy-2-(di-n-propylamino) tetralin hydro-bromide (8-OH-DPAT), haloperidol, clozapine, apomorphine hydrochloride, R(+)-8-OH DPAT hydrobromide, and WAY-100635 were obtained from Sigma (Sigma Aldrich, St. Louis, USA); Radioligands, [3H]Spiperone, [3H]YM-09151-2, [3H]Ketanserin, and [3H]LSD were purchased from PerkinElmer (PerkinElmer Life and Analytical Sciences, Boston, USA); [3H]8-OH DPAT and [3H]Mersulergin were obtained from Amersham Biosciences (Buckinghamshire, UK); cloned human D2L, D4.2, 5-HT subtype 6 receptor (5-HT6), 5-HT subtype 7 receptor (5-HT7), and rat D3 receptors were obtained from PerkinElmer; human 5-HT1A, 5-HT subtype 2A receptor (5-HT2A), and 5-HT subtype 2C receptor (5-HT2C) were purchased from Euroscreen (Brussels, Belgium); cloned human D3 receptor membranes were obtained from Korea Research Institute of Chemical Technology.
Derivatives of 3, 4, 5-Trimethoxycinnamic Acid
dTMCA was supplied from Department of Chemistry, Yonsei University in Korea and synthesized as previously described [12]. Briefly, a stirred solution (1.0 g, 4.2 mmol) of TMCA in dry dichloromethane (30 mL) was added to a mixture of 1-ethyl (dimethylaminopropyl) carbodiimide (EDCI, 0.97 g, 5.0 mmol), 1-hydroxybenzotriazole (HOBt, 0.68 g, 5.0 mmol), and triethylamine (TEA, 0.70 mL, 5.0 mmol) and then the reaction mixture was stirred at room temperature for 30 min. Several amines were then added into the reaction mixture and the resulting mixture was stirred at room temperature for 24 h. The reaction mixture was washed with brine (20 mL) and water (20 mL). The organic layer was separated, dried over anhydrous MgSO4, filtered, and the solvent was removed under reduced pressure. The residue was purified by flash column chromatography (silica gel, ethyl acetate/hexanes = 2:1, v/v) and chemical structures (Fig. 1) were determined by NMR spectroscopy. dTMCA was dissolved in DMSO and diluted with chromophore in 10%. According to our previous report, 8-OH-DPAT and dTMCA show similar cellular response, such as pERK 1/2 elevation in cortical neuronal cells [12]. Therefore, we suggested that dTMCA has an agonistic activity on 5-HT1A.
Structure of TMCA and dTMCA. a 3,4,5-Trimethoxycinnamic acid (TMCA). b 1-Pyrrolidin-1-yl-3-(3,4,5-trimethoxy-phenyl)-propenone (dTMCA#1). c 1-Morpholin-4-yl-3-(3,4,5-trimethoxy-phenyl)-propenone (dTMCA#2). d N-Allyl-3-(3,4,5-trimethoxy-phenyl)-acrylamide (dTMCA#3). e N-Furan-2-ylmethyl-3-(3,4,5-trimethoxy-phenyl)-acrylamide (dTMCA#4). f N-Decyl-3-(3,4,5-trimethoxy-phenyl)-acrylamide (dTMCA#5). g 1-(4-Benzyl-piperidin-1-yl)-3-(3,4,5-trimethoxy-phenyl)-propenone (dTMCA#6). h N-(Tetrahdro-furan-2-ylmethyl)-3-(3,4,5-trimethoxy-phenyl)-acrylamide (dTMCA#7). i N-(4-Ethyl-phenyl)-3-(3,4,5-trimethoxy-phenyl)-acrylamide (dTMCA#8). j N-Methyl-3-(3,4,5-trimethoxy-phenyl)-acrylamide (dTMCA#9). k 1-[4-(3,5-Dimethoxy-phenyl)-piperazin-1-yl]-3-(3,4,5-trimethoxy-phenyl)-propenone (dTMCA#10). dTMCA, derivatives of TMCA
Measurement of Binding Affinity on Receptors and Transporter
The binding affinity of TMCA and dTMCA on receptors and transporter was measured as described previously [12, 15, 16]. In brief, aliquots of frozen membrane from target receptors and transporter expressed CHO-K1 cell line (PerkinElmer Life and Analytical Sciences, Waltham, MA, USA), radioisotope labeled ligands, such as [3H]8-OH-DPAT (PerkinElmer), and appropriate concentrations of TMCA and dTMCA were added to reaction buffer. Reaction mixtures were filtered and radioactivity retained in the filter was counted using MicroBeta Plus (Wallac, Finland). The binding affinity (IC50) of TMCA and dTMCA was calculated by non-linear regression analysis (GraphPad Prism Program, San Diego, CA, USA).
Immobilization Stress and Electric Shock
For immobilization stress and electric shock, the head of individual mice was inserted into well ventilated 50 ml conical tubes with a small hole on the tube without forward or backward movement, and then they received inescapable 2 mA electric shocks from a 300-volt shock source. Experiments were started 1 h after drug administration and each stimulation lasted 0.1 s, with an inter-shock interval of 9.9 s for 2 min. This procedure was repeated for 7 consecutive days. Acute immobilization stress was performed in mice using 50 ml conical tubes with a small hole on the tube without forward or backward movement. They were immobilized for 6 h in the animal homeroom in 1 day. And EPM experiment was started immediately after immobilization. For rats, immobilization stress was performed using a plastic rodent restrainer (Decapi-cone, Braintree, MA, USA) that fits with rats and they were immobilized for 2 h in the animal homeroom each day for 7 days. Next, rats received uncontrollable 5 mA electric shocks with 300-volt source delivered to the grid floor by a constant current shocker with cycle timer. Experiments were started 1 h after drug administration and stimulation elapsed 1 s with an inter-shock interval of 19 s for 5 min. The control group was placed in their original cages undisturbed in the same experimental room. This procedure was repeated for 7 consecutive days. dTMCA (5 mg/kg) or 8-OH-DPAT (DPAT, 0.1 mg/kg) was administered to animal intraperitoneally 1 h before immobilization stress for 7 consecutive days.
Anxiety Measurement on EPM
The elevated plus maze (EPM) test is a suitable rodent model of anxiety used extensively to discover novel anxiolytic agents and investigate psychological and neurochemical basis of anxiety [17]. The EPM was made of black plexiglass with an elevation of 50 cm from the floor. The apparatus consists of four arms (mouse: 40 × 8 cm, rat: 50 × 10 cm) positioned at right angles to each other. The two walls of the arms are 20 cm in height (closed arms) whereas the other arms are without walls (open arms). The illumination at the center was adjusted to 50 lux. Each animal was initially placed on the central platform and allowed to explore the arms for 5 min for mice and for 10 min for rats. The time of entry in the open areas was recorded. A home video system was used to track mice and rats and measured the amount of time spent stationary. Animals were sacrificed 3 h after the EPM test [14]. After decapitation, the brain was taken out from the skull and amygdala were isolated.
Western Blotting Analysis
Amygdala tissues were homogenized with homogenization buffer (0.25 M sucrose, 10 mM Tris-cl pH 7.4, 0.5 mM EDTA, 1 mM PMSF, 1 mM Na3Vo4) and centrifuged twice at 13,500 rpm for 15 min at 4 °C. Protein concentrations were determined using the protein assay kit (PIERCE Biotechnology, Inc, Rockford, IL). 4× sample buffer was added to protein and boiled for 4 min at 100 °C. Equal amount of proteins were separated by 10% polyacrylamide gels and they were electrophoretically transferred to polyvinylidene fluoride (PVDF) membranes. Transfer blots were blocked in 3% skim milk in TBS-Tween 0.1% for 1 h and incubated with antibodies specific to total ERK1/2 and pERK1/2 (ERK1/2, 1:1000; pERK1/2, 1:2000; Santa Cruz Biotechnology, CA, USA), TH (1:1000; Santa Cruz Biotechnology, CA, USA), TH (1:1000; abcam, MA, USA), GAPDH (1:5000; Cell Signaling Technology, Danvers, USA), and GAPDH (1:1000; Lab frontier, Seoul, Korea) at 4 °C overnight. Blots were washed three times for 30 min in 0.1% TBST, and incubated with an appropriate horseradish peroxidase-conjugated secondary antibody (Zymed Laboratiories, South San Francisco, USA). Bound antibodies were visualized following chemiluminescence detection (Amersham Pharmacia Biotechnology, CA, USA). Scion Image was used for analysis of immunoblot intensity.
PC12 Cells Culture
PC12 cells, the pheochromocytoma of rat adrenal medulla, were purchased from ATCC (Manassas, VA, USA). The cells were cultured in Roswell Park Memorial Institute (RPMI) 1640 cell culture media supplemented with horse serum (10%, Gibco, MA, USA), fetal bovine serum (FBS; 10%, Gibco, Carlsbad, CA, USA), and antibiotic-antimycotic (1%, Gibco, Waltham, MA, USA), and maintained in a humidified incubator at 37 °C and 5% CO2. The number of seeded cells was 3×106 cells per well in poly-L-lysine coated 6 well plates at passage number 6. The seeded cells were treated with either 2 μM drugs (dTMCA #5 or dTMCA #9) or 200 ng/mL corticosterone or both. The dose of corticosterone was chosen according to a previous study [18]. The cells were harvested after 2 h.
Dopamine Measurement
The harvested cells were sonicated with perchloric acid (0.1 M) and centrifuged at 12,000 RPM for 15 min at 4 °C. The supernatants were filtered with 0.22 μm PVDF syringe filter (Chromdisc, Hwaseong-si, Gyeonggi-do, Korea). The filtered samples were injected 20 μL into the HPLC system (Shiseido, Tokyo, Japan). The C-18 ODS column (EiCOMPAK SC-50DS, 2.1 Φ × 150 mm, EiCOM, Tokyo, Japan) was used as the HPLC column, and the samples mobile phase [17% methanol, 190 mg/L sodium 1-octanesulfonate (SOS), 5 mg/L EDTA·2Na in 0.1 M acetic acid-citric acid buffer (pH 3.5)] at a flow rate of 0.23 mL/min.
Statistical Analysis
The data were expressed as mean ± standard error (S.E.). The normal distributions of the residuals were tested using the Shapiro–Wilk test, and data were analyzed with one-way analysis of variance (ANOVA) followed by Holm–Sidak post hoc t-test using SigmaPlot 12 software (Systat Software, San Jose, CA, USA).
Results
dTMCA#5 Had Highly Inhibitory Activities on Dopamine and Serotonin Receptor and Serotonin Transporter
The inhibitory activities of TMCA and dTMCA on dopamine and serotonin receptor and serotonin transporter are shown in Tables 1 and 2. Among the drugs, dTMCA#5 had the highest inhibitory activity on both receptor or dopamine and serotonin and serotonin transporter as evidenced by binding affinity test results.
TMCA Derivatives Ameliorated Stress-Induced Anxiety in Mice and Rats
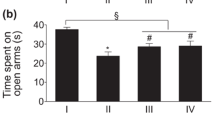
Previously, we determined the anxiolytic effects of TMCA in mice [2]. Here, we evaluated if dTMCA exhibits anti-stress effects that could be measurable on reduced time and frequency spent in the open arms of elevated plus maze (EPM). We observed that mice spent less time in the open arms when they get stressed by immobilization and electric shock; however, this was improved by dTMCA at a dosage of 5 mg/kg as it can be seen from the extended period of time spent in the open arms of EPM [F(10,55) = 3.372, p = 0.002 (Fig. 2)]. Next, we were also interested to determine whether stressed rats show similar behavioral patterns like that of mice when they are treated with dTMCA. In this case, TMCA#5 and TMCA#9 were randomly selected for the experiment based on the observation that all dTMCA portray similar patterns of stress relief in mice that were staying longer in the open arms of EPM. We found that stressed rats pretreated with TMCA#5 and TMCA#9 at 5 mg/kg showed an extended period of time spent in the open arms of EPM and this was equated with unstressed groups [F(3,20) = 7.388, p = 0.002 (Fig. 3a); F(10,55) = 10.438, p < 0.001 (Fig. 3b)] and with rats that were treated with the anti-depressant drug (8-OH-DPAT) used at a concentration of 0.1 mg/kg [F(3,20) = 5.43, p = 0.007 (Fig. 3c)].
Inhibitory effects of dTMCA on stress-induced anxiety in mice. Time in open arms was measured for 5 min by the elevated plus maze test using mice stressed by immobilization and electric shock for 7 consecutive days. The dTMCA was administrated at 5 mg/kg to the mice before measurement of the time in open arms. Data are expressed as mean ± S.E. (n = 6) and were analyzed using one-way ANOVA followed by Newman-Keul’s multiple comparison post hoc t-test (#p < 0.05 vs. control group, *p < 0.05 vs. stress control group). dTMCA, derivatives of TMCA
Inhibitory effects of TMCA#5, TMCA#9, and DPAT on stress-induced anxiety in rats. Time in open arms was measured for 10 min by the elevated plus maze test using mice stressed by immobilization and electric shock 7 consecutive days. The a TMCA#5 or b TMCA#9 was administrated at 5 mg/kg to the rats before measurement of the time in open arms. Data are expressed as mean ± S.E. (n = 6) and were analyzed using one-way ANOVA followed by Newman-Keul’s multiple comparison post hoc t-test (#p < 0.05 vs. control group, *p < 0.05 vs. stress control group). c Time in open arms was measured for 5 min by the elevated plus maze test using rat stressed by immobilization and electric shock 7 consecutive days. The DPAT was administrated at 0.1 mg/kg to the rats before measurement of the time in open arms. Data are expressed as mean ± S.E. (n =6) and were analyzed using one-way ANOVA followed by Newman-Keul’s multiple comparison post hoc t-test (#p < 0.05 vs. control group, **p < 0.01 vs. stress control group). dTMCA, derivatives of TMCA
Stress Mediated Downregulation of Tyrosine Hydroxylase Protein Expression in the Amygdala Was Restored During Pretreatment with dTMCA
Tyrosine hydroxylase is the rate-limiting enzyme for the biosynthesis of catecholamines and its expression is associated with working memory and the response to chronic stress [19]. Here, our intention was to evaluate if acute stress reduced the expression of TH in the amygdala of rats and whether this reduction was restored by pretreatment with dTMCA. We found that acute stress attenuated the expression of TH in the amygdala region of the brain; however, this was recovered when rats get treated with TMCA#5 and TMCA#9 derivatives at 5 mg/kg dosage [F(3,20) = 10.744, p < 0.001 (Fig. 4a); F(3,20) = 9.656, p < 0.001 (Fig. 4b)]. It is well known that stress modulates the expression of ERK [20]; hence, we evaluated pERK protein expression in the amygdala region of the brain from rats pretreated with dTMCA and then exposed to stress. In our results, pERK was not significantly changed by stress, and there was also no appreciable change in protein expression by dTMCA#5 and dTMCA#9 pretreatment (Fig. S2). Selective 5-HT1A agonist (8-OH-DPAT), hereafter named as DPAT, produced anxiolytic effects in rats [21]. Therefore, we used DPAT at 0.1 mg/kg for a mere reason of comparing its effect with the effect of dTMCA#5 and dTMCA#9 on TH and pERK expression. DPAT restored stress attenuated TH expression [F(3,20) = 7.903, p = 0.001 (Fig. 5)] like that of dTMCA but pERK expression (Fig. S2) induced by stress.
Inhibitory effects of TMCA#5 and TMCA#9 on stress-induced expression change of TH and pERK/ERK in rat amygdala. Change in TH expression caused by stress and a TMCA#5 or b TMCA#9 was detected by Western blotting using specific antibodies and normalized to the relative amplification of GAPDH in the rat amygdala. Data are expressed as the mean ± S.E. (n = 6) and were analyzed using one-way ANOVA followed by Newman-Keul’s multiple comparison post hoc t-test (#p < 0.05 vs. each control group, *p < 0.05 vs. each stress control group). dTMCA, derivatives of TMCA. TH, tyrosine hydroxylase
Inhibitory effects of DPAT on stress-induced anxiety in rat and expression change of TH and pERK/ERK in rat amygdala. Change in a TH and b pERK expression caused by stress and DPAT were detected by Western blotting using specific antibodies and normalized to the relative amplification of a GAPDH and b ERK in the rat amygdala. Data are expressed as the mean ± S.E. (n = 6) and were analyzed using one-way ANOVA followed by Newman-Keul’s multiple comparison post hoc t-test (#p < 0.05 vs. each control group, *p < 0.05 vs. each stress control group)
Stress Hormone-Induced Downregulation of Dopamine Concentration in the PC12 Cells Was Reversed by Pretreatment of dTMCA
HPLC analysis showed that the dopamine concentration of the PC12 cells was significantly decreased compared to the control group when exposed to 200 ng/mL of corticosterone for 2 h. However, dTMCA#5 and dTMCA#9 significantly inhibited the decrease of dopamine concentrations induced by corticosterone in the PC12 cells [F(5,30) = 6.96, p < 0.001 (Fig. 6)].
Effects of TMCA#5 and TMCA#9 on the corticosterone-induced dopamine reduction in PC12 cells. Dopamine levels in PC12 cells were measured by using HPLC system. Data are expressed as the mean ± S.E. (n = 6) and were analyzed using one-way ANOVA followed by Holm–Sidak post hoc t-test (##p < 0.01 vs. control group; *p < 0.05 vs. corticosterone control group). dTMCA, derivatives of TMCA
dTMCA Had Blood–Brain Barrier Permeability and CNS Activity
In the development of drugs that act on the CNS, it is important to confirm whether CNS activity and blood–brain barrier (BBB) permeability are possible. In our results, TMCA has no BBB permeability; however, dTMCA has BBB permeability and CNS activity (Table 3, Fig. 7, Fig. S3). In addition, we have been reported that TMCA has anti-anxiety activity at high dose of 50 mg/kg [2]. Therefore, our results suggest that dTMCA can penetrate the BBB and act directly in the CNS.
Predicting CNS distribution of dTMCA. The logBB values for for the TMCA and dTMCA plotted against their corresponding log(PS*fu,brain) values. LogPS, the permeability-surface area product. LogBB, concentration of drug in the brain divided by concentration in the blood. fu, brain, free fraction in brain tissue homogenate. dTMCA, derivatives of TMCA
An Antagonist of 5-HT1A Interrupted Inhibitory Effects of dTMCA on Stress-Induced Anxiety Through Regulation of Tyrosine-Hydroxylase
WAY-100635 acts as an antagonist of the 5-HT1A. We evaluated whether pretreatment with WAY-100635 inhibited the anxiolytic effect of dTMCA in acutely stressed mice [F(5,24) = 7.251, p < 0.001 (Fig. 8a)]. We also evaluated whether WAY-100635 suppresses TH expression restored by dTMCA pretreatment in the amygdala of acutely stressed mice [F(5,24) = 9.917, p < 0.001 (Fig. 8b)].
Inhibitory effects of antagonist of 5-HT1A on stress-induce anxiety suppressed by dTMCA in mice and expression change of TH in mice amygdala. Time in open arms was measured for 5 min by the elevated plus maze test using mice stressed by immobilization for 6 h. The dTMCA was administrated at 5 mg/kg to the mice before measurement of the time in open arms. WAY-100635 was administrated at 0.03 mg/kg to the mice 1 h before immobilization stress. Data are expressed as mean ± S.E. (n = 5) and were analyzed using one-way ANOVA followed by Holm–Sidak post hoc t-test (*p < 0.05 vs. control group, #p < 0.05 vs. stress control group, †p < 0.05 vs. dTMCA #5+Stress group, and $p < 0.05 vs. dTMCA #9+Stress group). TH, tyrosine hydroxylase. dTMCA, derivatives of TMCA
Discussion
Stress is known to cause physiological alterations in the central nervous system, especially in the amygdala and frontal cortex of brain associated with behavioral changes, although the molecular mechanisms underlying the pathophysiology of stress are complex and less known. Central serotonergic systems were primary targets of stress and stress hormones, particularly 5-hydroxytryptamine (5-HT) receptor sensitivity and its modification by either acute, repeated or chronic stress [19]. Most antidepressant drugs increase the levels of monoamines serotonin and/or noradrenaline (NA), suggesting that biochemical imbalances within the 5-HT/NA systems may unravel the pathogenesis of stress [22].
It has been reported that TMCA acted on 5-HT1A and showed anxiolytic effect via the activation of serotonergic system [1, 2, 10]. However, whether dTMCA possessed similar anxiolytic effect and this occurred through serotonin or dopamine receptor binding remained unknown. The present study, therefore, explored the anxiolytic effects of dTMCA in stress models. We found that immobilized mice and rats exposed to consecutive electric shock induced anxiety since they spent a lesser time in the open arms of EPM compared to unexposed groups. However, pretreatment with dTMCA (5 mg/kg) showed extended period of stay in the open arms of EPM, suggesting their anxiolytic effects at this low concentration when compared with our previous report pertaining to the anti-stress and anti-depression effects of TMCA at 50 mg/kg concentration used [2]. Because of high BBB permeability which is predicted by in silico simulation, dTMCA may have a high potency in comparison with that of TMCA [23]. According to our previous report, dTMCA has high binding properties to 5-HT1A [12]. In this study, all dTMCAs have high binding properties to only 5-HT1A evidenced by IC50 values (8-OH-DPAT displacement assay, Table 2). In addition, all dTMCAs have significant anxiolytic activities, except dTMCA1 that also showed a trend in increase of open arm time in EPM test. Importantly, we found that pretreatment with 5HT1A antagonist WAY-100635 inhibited dTMCAs reversed time in open arm on stressed mice. Therefore, we suggest that 5-HT1A binding affinity is associated with anxiety behaviors. It can be speculated that dTMCA might possess more potent anti-stress effects by preferential binding to 5-HT1A subunit and this may follow serotonin signaling pathway in a similar manner like that of the specific 5-HT1A agonist, 8-OH DPAT. This notion can also be further strengthened by our earlier finding that dTMCA at concentrations of 20 mg/kg showed anti-narcotic effects via 5-HT1A binding [12]. A recent report also indicated that the activation of 5-HT1A in the posteroventral part of the medial nucleus of the amygdala can promote a reduction in fear and/or anxiety [24], suggesting compounds that can bind and activate 5-HT1A could have paramount importance to alleviate anxiety associated with stressful situations.
Because stress is associated with reduced expression of TH in the brain tissue [9], we further investigated the effects of dTMCA on stress-induced TH protein expression in the amygdala regions of the brain. We showed that stress is accompanied by attenuated expression of TH protein in the amygdala; however, this was significantly restored by dTMCA#5 and dTMCA#9. We also demonstrated that corticosterone induced decrease of dopamine concentration in PC12 cells was reversed by dTMCA#5 and dTMCA#9. It has been reported that 5-HT1A agonists such as 8-OH-DPAT increases dopamine release [3, 25, 26]. Therefore, we speculate that 5-HT1A activation may increase turnover of dopamine and consequently upregulate TH expression, similarly to a previous report [27]. 5-HT1A regulates dopamine synthesis via the tyrosine hydroxylase activity [28]. However, the mechanism underlying effects of 5-HT1A on tyrosine hydroxylase expression was not clearly understood yet. It is well known that 5-HT1A modulates anxiety-like behavior and cognitive function through CREB phosphorylation [29, 30]. CREB has an important role in the transcriptional regulation of tyrosine hydroxylase [31]. Therefore, we assume that dTMCA derivates may regulate TH transcription via the modulation of 5-HT1A-inudced CREB activity. Protective effects of dTMCA on the TH expression may play a role in dopamine neurotransmission. It is well known that the physiological and behavioral responses to stress are mediated by corticotropin-releasing factor (CRF) and dynorphin, which are abundant in the central nucleus of the amygdala; hence, significant interactions between amygdala norepinephrine (NE) with CRF were observed [32], indicating that TH expression restored by dTMCA during stress might have critical roles for the biosynthesis of NE to mediate behavioral response to stress. Interestingly, dTMCA may regulate TH and dopamine levels depending on whether animals or cells were stressed and exposed to corticosterone or not.
Considering the role of 8-OH-DPAT, a selective 5-HT1A agonist, for the activation of somatodendritic and/or postsynaptic 5-HT1A following exposure to restraint stress [33], 8-OH-DPAT treatment was used in rats for a mere reason of comparing its effect with dTMCA. Stress reduced the time spent in the open arms of EPM, but this was markedly extended by 8-OH-DPAT in a manner similar to the effect of dTMCA. However, the time in the open arms EPM was less than dTMCA [14]. We also found that stress-attenuated TH protein expression was recovered in the amygdala of stressed rats after 8-OH-DPAT treatment, suggesting its anti-stress effects might have been mediated through 5-HT1A activation. This is intriguing because 5-HT1A-dependent signaling cascades could be attenuated following exposure to uncontrollable stressors leading to stress-induced behavioral deficits [34].
In conclusion, our results demonstrated that dTMCA has shown anti-stress effects revealed by extended period of time spent in the EPM. Derivatives of TMCA, TMCA#5 (N-decyl-3-(3,4,5-trimethoxy-phenyl)-acrylamide) and TMCA#9 (N-methyl-3-(3,4,5-trimethoxy-phenyl)-acrylamide), restored stress-attenuated expression of TH protein in the amygdala region of the brain. We suggested that dTMCA may target 5HT1A subunits of serotonin receptor for their intracellular signaling pathway.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Jung JC, Moon S, Min D, Park WK, Jung M, Oh S (2013) Synthesis and evaluation of a series of 3,4,5-trimethoxycinnamic acid derivatives as potential antinarcotic agents. Chem Biol Drug Des 81(3):389–398. https://doi.org/10.1111/cbdd.12087
Leem YH, Oh S (2015) 3,4,5-Trimethoxycinnamin acid ameliorates restraint stress-induced anxiety and depression. Neurosci Lett 585:54–59
Etievant A, Betry C, Haddjeri N (2010) Partial dopamine D2/serotonin 5-HT1A receptor agonists as new therapeutic agents. Open Neuropsychopharmacol J 3
Mravec B, Vargovic P, Filipcik P, Novak M, Kvetnansky R (2015) Effect of a single and repeated stress exposure on gene expression of catecholamine biosynthetic enzymes in brainstem catecholaminergic cell groups in rats. Eur J Neurosci 42(2):1872–1886. https://doi.org/10.1111/ejn.12955
Krzyzowska M, Swiatek W, Fijalkowska B, Niemialtowski M, Schollenberger A (2014) The role of map kinases in immune response. Med J Cell Biol 2(3):125–138. https://doi.org/10.2478/v10052-010-0007-5
Shen CP, Tsimberg Y, Salvadore C, Meller E (2004) Activation of Erk and JNK MAPK pathways by acute swim stress in rat brain regions. BMC Neurosci 5:36. https://doi.org/10.1186/1471-2202-5-36
Sananbenesi F, Fischer A, Schrick C, Spiess J, Radulovic J (2003) Mitogen-activated protein kinase signaling in the hippocampus and its modulation by corticotropin-releasing factor receptor 2: a possible link between stress and fear memory. J Neurosci 23(36):11436–11443. https://doi.org/10.1523/jneurosci.23-36-11436.2003
Meltser I, Tahera Y, Canlon B (2009) Glucocorticoid receptor and mitogen-activated protein kinase activity after restraint stress and acoustic trauma. J Neurotrauma 26(10):1835–1845. https://doi.org/10.1089/neu.2008.0874
Gilad GM, McCarty R (1981) Difference in choline acetyltransferase but similarities in catecholamine biosynthetic enzymes in brains of two rat strains differing in their response to stress. Brain Res 206(1):239–243. https://doi.org/10.1016/0006-8993(81)90124-4
Lee CI, Han JY, Hong JT, Oh KW (2013) 3,4,5-Trimethoxycinnamic acid (TMCA), one of the constituents of Polygalae Radix enhances pentobarbital-induced sleeping behaviors via GABAAergic systems in mice. Arch Pharm Res 36(10):1244–1251. https://doi.org/10.1007/s12272-013-0167-6
Gupta D, Prabhakar V, Radhakrishnan M (2016) 5HT3 receptors: target for new antidepressant drugs. Neurosci Biobehav Rev 64:311–325
Jung JC, Moon S, Min D, Park WK, Jung M, Oh S (2013) Synthesis and evaluation of a series of 3,4,5-trimethoxycinnamic acid derivatives as potential antinarcotic agents. Chem Biol Drug Des 81(3):389–398
Moreno-Martínez S, Tendilla-Beltrán H, Sandoval V, Flores G, Terrón JA (2022) Chronic restraint stress induces anxiety-like behavior and remodeling of dendritic spines in the central nucleus of the amygdala. Behav Brain Res 416:113523. https://doi.org/10.1016/j.bbr.2021.113523
Lim H, Jang S, Lee Y, Moon S, Kim J, Oh S (2012) Enhancement of anxiety and modulation of TH and pERK expressions in amygdala by repeated injections of corticosterone. Biomol Ther (Seoul) 20(4):418–424. https://doi.org/10.4062/biomolther.2012.20.4.418
Jung JC, Min D, Kim H, Jang S, Lee Y, Park W, Khan IA, Moon HI, Jung M, Oh S (2010) Design, synthesis, and biological evaluation of 3,4,5-trimethoxyphenyl acrylamides as antinarcotic agents. J Enzyme Inhib Med Chem 25(1):38–43. https://doi.org/10.3109/14756360902932784
Moon SH (2012) Antinarcotic and anxiolytic effects of 3,4,5-trimethoxycinnamic acid and its analogues. The Graduate School Ewha Womans Universrity, Seoul, Republic of Korea
Dawson GR, Tricklebank MD (1995) Use of the elevated plus maze in the search for novel anxiolytic agents. Trends Pharmacol Sci 16(2):33–36
Yun J, Koike H, Ibi D, Toth E, Mizoguchi H, Nitta A, Yoneyama M, Ogita K, Yoneda Y, Nabeshima T, Nagai T, Yamada K (2010) Chronic restraint stress impairs neurogenesis and hippocampus-dependent fear memory in mice: possible involvement of a brain-specific transcription factor Npas4. J Neurochem 114(6):1840–1851. https://doi.org/10.1111/j.1471-4159.2010.06893.x
de Lores R, Arnaiz G, Antonelli MC (2016) In search of concomitant alterations of dopaminergic and neurotensinergic systems in stress conditions. Neurochem Res 41(1-2):423–430
Costa AP, Lopes MW, Rieger DK, Barbosa SG, Goncalves FM, Xikota JC, Walz R, Leal RB (2016) Differential activation of mitogen-activated protein kinases, ERK 1/2, p38(MAPK) and JNK p54/p46 during postnatal development of rat hippocampus. Neurochem Res 41(5):1160–1169
Ferreira R, Nobre MJ (2014) Conditioned fear in low- and high-anxious rats is differentially regulated by cortical subcortical and midbrain 5-HT(1A) receptors. Neuroscience 268:159–168
Hjorth S (2016) Looking back (and in)to the future: a personal reflection on 'Serotonin autoreceptor function and antidepressant drug action' (Hjorth et al., 2000). J Psychopharmacol 10:1129–1136
Zhao Z, Bai Y, Xie J, Chen X, He X, Sun Y, Bai Y, Zhang Y, Wu S, Zheng X (2019) Excavating precursors from the traditional Chinese herb Polygala tenuifolia and Gastrodia elata: synthesis, anticonvulsant activity evaluation of 3,4,5-trimethoxycinnamic acid (TMCA) ester derivatives. Bioorg Chem 88:102832. https://doi.org/10.1016/j.bioorg.2019.03.006
De Paula BB, Leite-Panissi CR (2016) Distinct effect of 5-HT1A and 5-HT2A receptors in the medial nucleus of the amygdala on tonic immobility behavior. Brain Res 15:152–158
Vollenweider FX, Vontobel P, Hell D, Leenders KL (1999) 5-HT modulation of dopamine release in basal ganglia in psilocybin-induced psychosis in man--a PET study with [11C]raclopride. Neuropsychopharmacology 20(5):424–433. https://doi.org/10.1016/s0893-133x(98)00108-0
Lorrain DS, Matuszewich L, Hull EM (1998) 8-OH-DPAT influences extracellular levels of serotonin and dopamine in the medial preoptic area of male rats. Brain Res 790(1-2):217–223. https://doi.org/10.1016/s0006-8993(98)00065-1
Blum M, McEwen BS, Roberts JL (1987) Transcriptional analysis of tyrosine hydroxylase gene expression in the tuberoinfundibular dopaminergic neurons of the rat arcuate nucleus after estrogen treatment. J Biol Chem 262(2):817–821
Johnson EA, Tsai CE, Shahan YH, Azzaro AJ (1993) Serotonin 5-HT1A receptors mediate inhibition of tyrosine hydroxylation in rat striatum. J Pharmacol Exp Ther 266(1):133–141
Cai CY, Wu HY, Luo CX, Zhu DY, Zhang Y, Zhou QG, Zhang J (2019) Extracellular regulated protein kinaseis critical for the role of 5-HT1a receptor in modulating nNOS expression and anxiety-related behaviors. Behav Brain Res 357:88–97. https://doi.org/10.1016/j.bbr.2017.12.017
Qiu Y, Wang Y, Wang X, Wang C, Xia ZY (2018) Role of the hippocampal 5-HT1A receptor-mediated cAMP/PKA signalling pathway in sevoflurane-induced cognitivedysfunction in aged rats. J Int Med Res 46(3):1073–1085. https://doi.org/10.1177/0300060517744037
Lewis-Tuffin LJ, Quinn PG, Chikaraishi DM (2004) Tyrosine hydroxylase transcription depends primarily on cAMP response element activity, regardless of the type of inducing stimulus. Mol Cell Neurosci 25(3):536–547. https://doi.org/10.1016/j.mcn.2003.10.010
Kravets JL, Reyes BA, Unterwald EM, Van Bockstaele EJ (2015) Direct targeting of peptidergic amygdalar neurons by noradrenergic afferents: linking stress-integrative circuitry. Brain Struct Funct 220(1):541–558
Samad N, Batool F, Haleem DJ (2007) Neurochemical and behavioral effects of 8-OH-DPAT following exposure to restraint stress in rats. Pharmacol Rep 59(2):173–180
Goto T, Toyoda A (2015) A mouse model of subchronic and mild social defeat stress for understanding stress-induced behavioral and physiological deficits. J Vis Exp 24(105):52973
Funding
This work was supported by the Ministry of Food and Drug Safety (20182MFDS422, 20182MFDS425, 22214MFDS252), a National Research Foundation of Korea (NRF) grant funded by the Korea government, Ministry of Science (MEST; MRC 2010-0029355) (MISIT; No. MRC, 2017R1A5A2015541), and “Regional Innovation Strategy (RIS)” through the NRF funded by the Ministry of Education (MOE) (2021RIS-001).
Author information
Authors and Affiliations
Contributions
Material preparation, data collection, analysis, and writing — review and editing were performed by Eunchong Hong. Material preparation, data collection, analysis, and writhing — original draft were performed by Hyun Kyu Min. Data curation, visualization, writhing — original draft, and writing — review and editing were performed by Heena Lim and Sun Mi Gu. Material preparation, data collection, and analysis were performed by Abdulaziz Jabborov, Taddesse Yayeh, Mijin Kim, Woo-Kyu Park, and Jae-Chul Jung. Conceptualization, data curation, project administration, supervision, resources, and writing — review and editing were performed by Jaesuk Yun and Seikwan Oh. All authors proofread the final manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
The study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. All experiments based on the guidelines approved by the Animal Care and Committee of Ewha Womans University and Chungbuk National University (CBNUA-436-12-02).
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PDF 206 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hong, E., Min, H.K., Lim, H. et al. Derivatives of 3, 4, 5-Trimethoxycinnamic Acid Ameliorate Stress-Induced Anxiety in Mice and Rats. Mol Neurobiol 60, 2737–2748 (2023). https://doi.org/10.1007/s12035-023-03240-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03240-y