Abstract
Parkinson’s disease is a neurodegenerative disorder characterised by nigrostriatal dopaminergic degeneration, and intracellular α-synuclein aggregation. Current pharmacological treatments are solely symptomatic so there is a need to identify agents that can slow or stop dopaminergic degeneration. One proposed class of therapeutics are neurotrophic factors which promote the survival of nigrostriatal dopaminergic neurons. However, neurotrophic factors need to be delivered directly to the brain. An alternative approach may be to identify pharmacological agents which can reach the brain to stimulate neurotrophic factor expression and/or their signalling pathways in dopaminergic neurons. BMP2 is a neurotrophic factor that is expressed in the human substantia nigra; exogenous BMP2 administration protects against dopaminergic degeneration in in vitro models of PD. In this study, we investigated the neurotrophic potential of two FDA-approved drugs, quinacrine and niclosamide, that are modulators of BMP2 signalling. We report that quinacrine and niclosamide, like BMP2, significantly increased neurite length, as a readout of neurotrophic action, in SH-SY5Y cells and dopaminergic neurons in primary cultures of rat ventral mesencephalon. We also show that these effects of quinacrine and niclosamide require the activation of BMP-Smad signalling. Finally, we demonstrate that quinacrine and niclosamide are neuroprotective against degeneration induced by the neurotoxins, MPP+ and 6-OHDA, and by viral-mediated overexpression of α-synuclein in vitro. Collectively, this study identifies two drugs, that are safe for use in patients' to 'are approved for human use, that exert neurotrophic effects on dopaminergic neurons through modulation of BMP-Smad signalling. This rationalises the further study of drugs that target the BMP-Smad pathway as potential neuroprotective pharmacotherapy for Parkinson’s disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Parkinson’s disease (PD) is a common neurodegenerative disorder characterised by the progressive degeneration of midbrain dopaminergic neurons and their axons, with the accumulation of intracellular aggregates of α-synuclein in Lewy bodies and Lewy neurites [1]. In recent years, there has been considerable interest in developing neurotrophic factor therapy for PD. This involves the delivery of neurotrophic factor genes, or recombinant proteins, to the midbrain or striatum to protect midbrain dopaminergic neurons and their axons from degeneration [2,3,4]. Despite initial success in pre-clinical models and in open-label trials [5,6,7,8,9,10,11], randomised controlled trials of two dopaminergic neurotrophic factors, glial cell line–derived neurotrophic factor (GDNF) and neurturin, have failed to meet their primary end points [12,13,14,15,16,17]. Thus, there has been increasing interest in identifying other neurotrophic factors that are capable of protecting dopaminergic neurons from degeneration. The challenge surrounding neurotrophic factor therapy, however, is that these proteins require direct administration to the brain, as they are rapidly metabolised in vivo and are unable to cross the blood brain barrier in adequate doses [18]. One proposed strategy to overcome this problem may be the identification of small molecule drugs or biologics that are capable of crossing the blood brain barrier and can selectively activate the receptors or downstream targets that mediate the action of specific neurotrophic factors.
Bone morphogenetic proteins (BMPs) are a group of neurotrophic factors that, like GDNF and neurturin, are members of the transforming growth factor (TGF)-β superfamily (for reviews see [18, 19]). We have recently shown, using gene co-expression analysis of the human substantia nigra (SN), that the BMP receptors (BMPRs) and BMP2 ligand exhibit correlated patterns of expression with multiple markers of dopaminergic neurons in the human SN, indicating a potential functional role for BMP-Smad signalling in dopaminergic neuron biology [20]. Moreover, we reported that recombinant human (rh)BMP2 promoted neurite growth, as a single cell readout of neurotrophic action, in SH-SY5Y cells and dopaminergic neurons treated with the neurotoxins 6-hydroxydopamine (6-OHDA) and 1-methyl-4-phenylpyridinium (MPP+), and in cells overexpressing α-synuclein [20]. These data suggest that clinically approved drugs that modulate BMP2 expression and/or the BMP2 signalling pathway may be a novel class of compounds for protecting dopaminergic neurons in PD.
In a recent study, Ghebes et al. carried out a screen of 1280 commercially available FDA-approved compounds in primary human tendon-derived cells to identify those capable of modulating the BMP2 signalling pathway, which is also important for local tendon and ligament repair [21]. In studies using a BMP reporter cell line, they reported that two drugs, niclosamide and quinacrine, modulated the BMP2 pathway [21]. Niclosamide is an anthelmintic agent that has been used clinically to treat tape worm infections [22], while quinacrine has a long history of clinical use in the treatment of malaria [23]. Given that niclosamide and quinacrine were found to modulate BMP2 signalling in primary tendon cells [21], and as rhBMP2 protects dopaminergic neurons in primary cultures of rat ventral mesencephalon (VM) from neurotoxin- and α-synuclein-induced degeneration [20], we hypothesised that these two drugs may protect against neurotoxin- and α-synuclein-induced dopaminergic degeneration in cellular models of PD.
Materials and Methods
Cell Culture
Human SH-SY5Y cells (ATCC; CRL-2266) and embryonic day (E) 14 rat VM cells were cultured as previously described [20, 24], under license with full ethical approval. Briefly, SH-SY5Y cells were maintained in Dulbecco’s Modified Eagle Medium F-12 (DMEM/F-12), supplemented with 10% foetal calf serum (FCS), 100 nM L-Glutamine, 100 U/ml penicillin and 10 μg/ml streptomycin (all from Sigma). E14 VM cells were cultured in DMEM/F-12 containing 100 nM L-glutamine, 6 mg/ml D-Glucose, 100 U/ml penicillin, 10 μg/ml streptomycin, with 2% B-27 supplement, and 1% FCS. All cell cultures were maintained at 37 °C and 5% CO2 for the duration of the experiment. Where indicated, cultures were treated with 5 μl dH20 (Vehicle), 0–10 nM niclosamide (Sigma) or quinacrine (Sigma), 0–200 ng/ml rhBMP2 (Gibco), 5 μM 6-OHDA (Sigma) or 5 μM MPP+ (Sigma) at varying times depending on the experiment and as indicated in the figure legends. Where specified, cells were pre-treated with 1 μg/ml dorsomorphin (Sigma), a small molecular inhibitor of BMP-Smad signalling [25] for 30 min prior to the addition of rhBMP2, niclosamide or quinacrine.
ELISA for Total and Phosphorylated-Smad 1
Activation of the phosphorylated-Smad (phospho-Smad) pathway by rhBMP2 was examined using the SMAD1 (total/phospho) Human InstantOne™ ELISA Kit (Invitrogen). In brief, SH-SY5Y cells were plated at a density of 5.0 × 104 cells per well of a 24-well plate and treated with or without 1 μg/ml dorsomorphin for 30 min prior to the addition of 50 ng/ml rhBMP2 for 2 h. Cells were then lysed and protein extracts were prepared for the ELISA as per the manufacturer’s instructions.
Transfection of SH-SY5Y Cells
SH-SY5Y cells were transfected using the TransIT-X2® Dynamic Delivery System (Mirus Bio) according to the manufacturer’s instructions. Briefly, SH-SY5Y cells were plated at a density of 5.0 × 104 cells per well of a 24-well plate. The TransIT-X2:DNA complex was prepared according to the manufacturer’s specifications using serum-free medium, TransIT-X2 reagent and 250 ng of the Smad binding element (SBE)-green fluorescent protein (GFP) reporter (Qiagen) before being added to the wells as per the manufacturer’s instructions.
Virus Preparation
An α-synuclein and GFP expression plasmid were generously donated by Dr Eilis Dowd (National University of Ireland, Galway) and Professor Deniz Kirik (Lund University, Sweden). Adeno-associated viral (AAV) vectors were then constructed by Vector Biosystems Inc, Philadelphia, USA. Briefly, AAV2-inverted terminal repeats that coded for the human wild-type α-synuclein or GFP were packaged using AAV6 capsid proteins producing AAV2/6 viral vectors. Transgene expression was driven by a synapsin-1 promoter and enhanced using a woodchuck hepatitis virus post-transcriptional regulatory element (WPRE). The final viral titres for AAV2/6-α-synuclein (AAV-αSyn) and AAV2/6-GFP (AAV-GFP) were 5.2 × 1013gc/ml and 5.0 × 1013gc/ml respectively.
Transduction of E14 VM Cultures
E14 VM cultures were plated at a density of 1.0 × 105 per well of a 24-well plate and transduced with 4 ul AAV-GFP or AAV-αSyn to achieve a multiplicity of infection (MOI) of 2.0 × 105. Cultures were then treated daily with 10 nM quinacrine or 10 nM niclosamide that commenced either at the time of infection (combined treatment) or treatment commenced 5 days after viral infection (delayed treatment). In both cases, the experimental end point was at 10 days in vitro (DIV).
Immunocytochemistry
SH-SY5Y cells or E14 VM cultures were fixed in 4% paraformaldehyde (PFA) and processed for immunocytochemistry as previously described [20]. Where indicated, cells were incubated for 16 h at 4 °C in the following antibodies: tyrosine hydroxylase (TH) (1:2000, Merck Millipore), phospho-Smad 1/5/8 (1:200; Cell Signalling) and α-synuclein (1:2000, Merck Millipore) and diluted in 1% bovine serum albumin (BSA) in 10 mM phosphate-buffered saline (PBS). Following 3 x 5 min washes in PBS with Triton X-100 (PBS-T), cells were incubated in the appropriate Alexa Fluor 488- or 594-conjugated secondary antibodies (1:500; Invitrogen) diluted in 1% BSA in 10 mM PBS, for 2 h at room temperature in the dark. Following 3 × 5 min washes in PBS-T, cells were counterstained with DAPI (1:3000; in 10 mM PBS; Sigma) and imaged using an Olympus IX71 microscope.
Analysis of Cellular Morphology and Nuclear Intensity
To assess cellular morphology or nuclear staining intensity, 5 randomly selected cells were measured in no less than 15 randomly selected fields per N, yielding at least 225 neurites or cell bodies analysed per experiment. To verify the number of TH-positive (TH+) cells and total cells, TH and DAPI counts were completed in 15 randomly selected fields and the numbers of TH+ cells were expressed as percentages of the total DAPI-stained cells. For experiments involving AAV vectors, 5 randomly selected cells were measured in no less than 6 randomly selected fields per N, yielding at least 90 neurites or cell bodies analysed per experiment. To verify the number of TH+ cells and total cells, TH and DAPI counts were completed in 6 randomly selected fields and the numbers of TH+ cells were expressed as percentages of the total DAPI-stained cells. All experiments were repeated at least 3 times and all analyses were performed using Image J software.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism 8 (©2020 GraphPad Software, CA USA). A one-way or two-way ANOVA with post hoc Dunnett, Holm-Sidak or uncorrected Fisher’s LSD test, or unpaired Student’s t-tests, were used as appropriate to determine significant differences between groups. Results were deemed to be significant when p < 0.05. All data are presented as mean ± SEM.
Results
Quinacrine and Niclosamide Promote Neurite Growth in SH-SY5Y Cells and in Primary Midbrain Dopaminergic Neurons
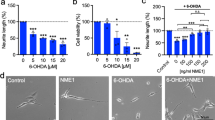
We first showed that SH-SY5Y cells are a useful screening tool for small molecule drug discovery, using rhBMP2 as our positive control (Supplementary Fig. 1). Given that rhBMP2 is known to promote neurite growth in a number of neuronal cell types, including dopaminergic neurons [20, 26,27,28], and that quinacrine and niclosamide have been shown to upregulate BMP2 expression in tendon tissue [21], we next tested the ability of both compounds to promote neurite growth as a readout of neurotrophic action at an individual cellular level. To do this, we performed a dose-response experiment in which SH-SY5Y cells were treated daily with increasing concentrations of either quinacrine or niclosamide (up to 10 nM), or with 50 ng/ml rhBMP2 as a positive control, for 72 h. Comparable results were found for both compounds whereby a concentration of 1 or 10 nM niclosamide (Fig. 1a, c) or quinacrine (Fig. 1d, f) significantly increased neurite length relative to untreated controls, and to a similar extent to that induced by rhBMP2. These effects were not secondary to any effects of these drugs on cell number as we found that the numbers of cells in cultures treated with niclosamide (Fig. 1b, c) or with quinacrine (Fig. 1e, f) were not significantly different from the controls. Collectively, these data show that quinacrine and niclosamide promote neurite growth without any effects on cell number in SH-SY5Y cell cultures.
Quinacrine and niclosamide promote neurite growth in SH-SY5Y cells and in primary midbrain dopaminergic neurons. a Total neurite length, b total cell number and c representative photomicrographs of SH-SY5Y cells following treatment with Niclosamide at the concentrations indicated for 72 h. d Total neurite length, e total cell number and f representative photomicrographs of SH-SY5Y cells following treatment with quinacrine at the concentrations indicated for 72 h. Scale bar = 50 μM. All data are presented as mean ± SEM from at least three experiments (*p < 0.05, **p < 0.01, ***p < 0.001 vs. control or as indicated. One-way ANOVA with Tukey’s post hoc test). g Total neurite length and h numbers of TH+ neurons as a percentage of the untreated control following treatment with Niclosamide at the concentrations indicated for 72 h. i Total neurite length and j numbers of TH+ neurons as a percentage of the untreated control following treatment with quinacrine at the concentrations indicated for 72 h. k, l Representative photomicrographs of E14 VM cultures following 10 nM treatment of k niclosamide and l quinacrine for 72 h. Scale bar = 50 μM. All data are presented as mean ± SEM from at least three experiments (*p < 0.05, **p < 0.01, ***p < 0.001 vs. control. One-way repeated measures ANOVA with Dunnett’s post hoc test or unpaired student’s t test)
Although SH-SY5Y cells are a useful screening tool, they do not recapitulate all features of dopaminergic neurons. Therefore, we next performed a dose-response experiment in which we added increasing concentrations (0–10 nM) of quinacrine and niclosamide to primary cultures of the E14 rat VM for 72 h and then analysed the neurite length and numbers of TH+ dopaminergic neurons in these cultures. We found that 0.1–10 nM concentrations of either niclosamide (Fig. 1g, k) or quinacrine (Fig. 1i, l) induced a significant increase in neurite length in dopaminergic neurons relative to untreated controls, and at a comparable level to neurite growth induced by rhBMP2. Given that a 10 nM concentration of niclosamide or quinacrine exerted the maximal increase in neurite length, we also examined TH+ cell survival at this concentration and found that both compounds did not increase the numbers of dopaminergic neurons in these cultures (Fig. 1h, j, k, l). Collectively, these data show that quinacrine and niclosamide are well tolerated by dopaminergic neurons and are capable of eliciting phenotypic changes that are similar to those induced by the neurotrophic factor BMP2. Coupled with previous work showing that these drugs modulate BMP2 expression in tendon cells [21], collectively, these data suggested that the effects of these drugs may be mediated through the canonical BMP-Smad signalling pathway.
Quinacrine and niclosamide activate BMP-Smad-dependent transcription in SH-SY5Y cells and increase phospho-Smad levels in primary dopaminergic neurons which are required for their neurite growth-promoting effects.
To test the hypothesis that quinacrine or niclosamide promote neurite growth through activation of the canonical BMP-Smad signalling, we next used a BMP-Smad reporter construct in which a BMP responsive element (BRE) drives the expression of GFP. Therefore, an increase in intracellular GFP expression is indicative of increased BMP-Smad-dependent transcription (Fig. 2a). SH-SY5Y cells were transiently transfected with 250 ng of the reporter plasmid. At 24-h post-transfection, 10 nM of niclosamide or quinacrine, or 50 ng/ml rhBMP2 (as a positive control), was added to these cultures for a further 24 h. Treatment with 10 nM quinacrine or niclosamide significantly increased GFP expression compared to untreated controls, to a similar extent as rhBMP2 treatment (Fig. 2b, c). These data show that quinacrine and niclosamide stimulate a BMP-Smad-dependent transcriptional response in SH-SY5Y cells.
Quinacrine and niclosamide activate BMP-Smad-dependent transcription in SH-SY5Y cells and increase phospho-Smad levels in primary dopaminergic neurons which are required for their neurite growth-promoting effects. a Schema showing SBE-GFP reporter system. b SBE-driven GFP expression expressed as a percentage of that in vehicle cells and c representative photomicrographs of SH-SY5Y cells following 24-h treatment of 10 nM niclosamide or quinacrine or 50 ng/ml rhBMP2. Scale bar = 10 μM. d Phospho-Smad activation and e, f representative photomicrographs of E14 VM cultures, immunocytochemically stained for e TH and f phospho-Smad, following 24-h treatment of 10 nM niclosamide or quinacrine with or without 1 μg/ml dorsomorphin. g Total neurite length of TH+ neurons in E14 VM cultures following 24-h treatment of 10 nM niclosamide or quinacrine with or without 1 μg/ml dorsomorphin. Scale bar = 50 μM. All data are presented as mean ± SEM from at least three experiments (*p < 0.05, **p < 0.01 vs. control or as indicated. One-way repeated measures ANOVA with b Dunnett’s post hoc test or d, g uncorrected Fisher’s LSD test)
Given that quinacrine and niclosamide activate BMP-Smad-dependent transcription in SH-SY5Y cells, we next examined the relevance of this finding to primary cultures of dopaminergic neurons. We could not perform the same reporter assay as the transfection efficiency of primary cultures of the E14 rat VM is very low, which is compounded by them being mixed cultures, so we therefore used a modified approach. As phospho-Smad levels are indicative of BMP-Smad pathway activation, we immunocytochemically double-stained these cultures for TH and phospho-Smad1/5/8 which allowed us to analyse phospho-Smad1/5/8 levels specifically in TH+ dopaminergic neurons. We found that treatment with 10 nM quinacrine or 10 nM niclosamide for 24 h led to significant increases in phospho-Smad-1/5/8 levels in TH+ neurons, which was completely prevented by pre-treatment with the BMPR inhibitor, dorsomorphin (Fig. 2d, f). Similarly, quinacrine- and niclosamide-induced increases in neurite growth were prevented by pre-treatment with dorsomorphin (Fig. 2e, g). Collectively, these data show that both quinacrine and niclosamide may mediate their phenotypic effects on midbrain dopaminergic neurons via the BMP-Smad signalling pathway. As BMPs have been shown to have neuroprotective effects on dopaminergic neurons [20, 26,27,28,29], this suggested that quinacrine and niclosamide may protect against dopaminergic degeneration, which is the pathological hallmark of PD.
Quinacrine and Niclosamide Protect Dopaminergic Neurons from MPP+ and 6-OHDA-Induced Degeneration
We next tested the hypothesis that quinacrine and niclosamide can protect dopaminergic neurons against degeneration induced by the neurotoxins, 6-OHDA and MPP+. To do this, primary cultures of E14 rat VM were treated with 10 nM of quinacrine or niclosamide or vehicle and cultured with or without 5 μM 6-OHDA or MPP+, for 72 h. For niclosamide, a two-way repeated measures ANOVA revealed a significant effect of both neurotoxins (F(1, 3) = 169.9, p = 0.0010) on neurite length, but no effect of niclosamide (F(1.234, 3.701) = 4.449, p = 0.1072) and no interaction between niclosamide and the neurotoxins (F(1.326, 3.978) = 0.5978, p = 0.5291). Furthermore, post hoc testing using an uncorrected Fisher’s LSD test revealed a significant reduction in neurite length of MPP+ (p = 0.0095) and 6-OHDA (p = 0.0018) versus the control (Fig. 3a, b), whereas no significant difference was found for MPP+ (p = 0.5321) and 6-OHDA (p = 0.1662) cultures co-treated with niclosamide. For quinacrine, a two-way repeated measures ANOVA revealed a significant interaction between quinacrine × neurotoxins (F(1.44, 4.32) = 10.52, p = 0.0247) and both neurotoxins (F(1, 3) = 161.6, p = 0.0011) on neurite length, but no effect of quinacrine alone (F(1.049, 3.146) = 2.081, p = 0.2429). Furthermore, post hoc testing using an uncorrected Fisher’s LSD test revealed a significant reduction in neurite length of MPP+ (p = 0.0008) and 6-OHDA (p = 0.0273) versus the control (Fig. 3c, e). While no significant difference was found for MPP+ (p = 0.7516) and 6-OHDA (p = 0.7713) cultures co-treated with quinacrine.
Quinacrine and niclosamide protect dopaminergic neurons from MPP+ and 6-OHDA-induced degeneration. a Representative photomicrographs and b total neurite length of E14 VM cultures after 72-h treatment with 10 nM niclosamide with or without 5 μM MPP+ or 6-OHDA. c Total neurite length and e representative photomicrographs of E14 VM cultures after 72-h treatment with 10 nM quinacrine with or without 5 μM MPP+ or 6-OHDA. d Numbers of TH+ neurons as a percentage of the control in E14 VM cultures after 72-h treatment with 10 nM quinacrine or niclosamide with or without 5 μM MPP+ or 6-OHDA. Scale bar = 50 μM. All data are presented as mean ± SEM from at least three experiments (*p < 0.05, **p < 0.01, ***p < 0.001 vs. control; two-way repeated measures ANOVA with uncorrected Fisher’s LSD post hoc test)
We next sought to determine whether quinacrine and niclosamide can protect TH+ neurons against the neurotoxins MPP+ or 6-OHDA. Here, a two-way repeated measures ANOVA revealed a significant effect of the neurotoxins (F(1, 2.001) = 23.03, p = 0.0408) and the drug compounds (F(1.183, 2.366) = 21.16, p = 0.0316) on TH+ neuron number. However, there was no significant interaction between the drug compounds and the neurotoxins on TH+ cell number (F(1.293, 2.585) = 1.22, p = 0.3863). In addition, post hoc testing using an uncorrected Fisher’s LSD test revealed a significant reduction in neurite length of MPP+ (p = 0.0097) and 6-OHDA (p = 0.0263) versus the control (Fig. 3d). While no significant difference was found for MPP+ (p = 0.1210) and 6-OHDA (p = 0.0.964) cultures co-treated with niclosamide and no significant difference was found for MPP+ (p = 0.1167) and 6-OHDA (p = 0.3655) cultures co-treated with quinacrine. Collectively, these data show that quinacrine and niclosamide can protect dopaminergic neurons against neurotoxin-induced degeneration.
Quinacrine and Niclosamide Protect Dopaminergic Neurons from AAV-α-Synuclein-Induced Axon Degeneration
As PD is characterised by the accumulation of α-synuclein, finally, we sought to assess whether quinacrine and niclosamide could also protect dopaminergic neurons against α-synuclein-induced degeneration in vitro. To do this, we used an AAV-αSyn expression vector to express α-synuclein in dopaminergic neurons in primary cultures of the E14 rat VM (Fig. 4a). Cultures were transduced with AAV-GFP (as the control) or AAV-αSyn of varying MOIs to establish an MOI that had a detrimental effect on neurite length after 5 DIV, as a readout of neurodegeneration at an individual neuron level (Supplementary Fig. 2). AAV-αSyn with an MOI of 2.0 × 105 led to significant reductions in neurite length compared to the control AAV-GFP group.
Quinacrine and niclosamide protect midbrain dopaminergic neurons against α-synuclein-induced axon degeneration. a Representative photomicrographs of E14 VM cultures after transduction with AAV2/6-GFP or AAV2/6-α-synuclein at an MOI of 2 × 105 and immunocytochemically stained for TH and α-synuclein, and counterstained with DAPI after 5 DIV. b Number and c neurite length of TH+ neurons in E14 VM cultures transduced with AAV-GFP or AAV-α-synuclein and cultured with or without 10 nM quinacrine or niclosamide daily for 10 DIV. d DA neuron number, e DA neurite length and f representative photomicrographs of E14 TH+ neurons infected with AAV-GFP or AAV-α-synuclein and cultured with or without 10 nM quinacrine or niclosamide after 5 DIV daily up to 10 DIV. Scale bar = 50 μm. All data are presented as mean ± SEM from at least three experiments (*p < 0.05, **p < 0.01 vs. control; two-way ANOVA with Holm-Sidak’s post hoc test). DA = dopaminergic
To assess whether treatment with quinacrine or niclosamide could protect against α-synuclein-induced degeneration, quinacrine or niclosamide was administered using two different treatment paradigms in this model. In the first experiment, primary cultures of the E14 rat VM were transduced with either AAV-GFP or AAV-aSyn vectors and treated daily thereafter for 10 DIV in a concurrent treatment paradigm. A two-way ANOVA revealed a significant α-synuclein × drug interaction on the numbers of TH+ dopaminergic neurons in these cultures (F(2, 10) = 5.388, p = 0.0258). Post hoc testing showed that α-synuclein resulted in a significant reduction in dopaminergic neuron number (p = 0.0015), that was not seen in cultures treated with quinacrine (p = 0.4409) or niclosamide (p = 0.4409) (Fig. 4b). We also examined dopaminergic neurite length, and found a significant effect of α-synuclein (F(1, 4) = 9.656, p = 0.0360) and drug treatment (F(2, 8) = 21.37, p = 0.0006), with no significant interaction (F(2, 8) = 1.861, p = 0.2170). Post hoc testing showed that α-synuclein resulted in a significant reduction in dopaminergic neurite length (p = 0.0315), that was not seen in cultures treated with quinacrine (p = 0.4922) or niclosamide (p = 0.4922) (Fig. 4c). These data show that concurrent treatment with quinacrine or niclosamide prevents α-synuclein-induced reductions in dopaminergic neuron number and neurite length.
Experiment 2 involved a delayed drug treatment paradigm. Primary cultures of the E14 rat VM were transduced with the AAV-GFP or AAV-aSyn vectors and left for 5 days. We then treated the cultures with quinacrine or niclosamide on day 5 and daily thereafter for an additional 5 DIV (10 days total). A two-way ANOVA showed a significant α-synuclein × drug interaction on the numbers of TH+ dopaminergic neurons in these cultures (F(2, 10) = 6.420, p = 0.0161). Post hoc testing revealed that α-synuclein resulted in a significant reduction in dopaminergic neuron number (p = 0.0016), that was not seen in cultures treated with quinacrine (p = 0.5259) or niclosamide (p = 0.7871) (Fig. 4d). An examination of dopaminergic neurite length revealed a significant effect of α-synuclein (F(1, 4) = 13.83, p = 0.0205) and drug treatment (F(2, 8) = 43.65, p < 0.0001) with no significant interaction (F(2, 8) = 4.103, p = 0.0594) (Fig. 4e, f). Post hoc testing showed that α-synuclein resulted in a significant reduction in dopaminergic neurite length (p = 0.0040), that was not seen in cultures treated with quinacrine (p = 0.1278) or niclosamide (p = 0.4256). These data show that quinacrine or niclosamide, when given to neurons with an established α-synuclein load, can prevent α-synuclein-induced reductions in dopaminergic neuron neurite length.
Discussion
Neurotrophic factor therapy has attracted significant interest as a potential disease-modifying approach for the treatment of PD. However, the application of neurotrophic factors in a clinical setting currently requires direct administration to the brain via invasive surgery and has had limited success to date [12,13,14,15,16,17, 30]. BMP2 has been shown to have potent neurotrophic effects on dopaminergic neurons [20, 26, 28, 29]. Similar to other members of the BMP family, BMP2 exerts its effects via the canonical Smad signalling pathway [31, 32]. Therefore, the identification of small molecule compounds, that are known to cross the blood brain barrier and that could modulate the expression of BMP2 and/or BMP-Smad signalling in dopaminergic neurons, has potential therapeutic relevance for PD. The clinical application of such agents through non-surgical routes would be hugely advantageous from a safety perspective and allow wider application to patients. The FDA-approved small molecule drugs, quinacrine and niclosamide, have been shown to upregulate BMP2 expression in a dose-dependent manner in a BMP2 reporter cell line and in human tendon-derived tissue [21]. Given the substantial evidence in support of BMP2 as a neurotrophic factor for dopaminergic neurons, this study investigated the effect of quinacrine and niclosamide on dopaminergic neurons using SH-SY5Y cells and primary cultures of the E14 rat VM.
Firstly, SH-SY5Y cells were verified as an appropriate tool to screen drug compounds for neurotrophic ability and for activation of the canonical Smad signalling pathway. SH-SY5Y cells were shown to express high levels of BMP2’s receptors, BMPR1B and BMPR2, as well as Smad-1, Smad-5 and Smad-8, the effector molecules involved in initiating Smad signalling [33]. In addition, treatment of SH-SY5Y cells with rhBMP2 resulted in the activation of the Smad pathway, as shown by increased phospho-Smad activation, as well as increased neurite length, and both of these effects were supressed by co-treatment with dorsomorphin. This finding is supported by several studies showing that rhBMP2 preferentially binds to BMPRI receptors [34,35,36] to activate canonical Smad signalling in C2C12 cells [37], SH-SY5Y cells [28, 38] and primary cultures of dopaminergic neurons [31]. In this study, both quinacrine and niclosamide were also found to promote neurite growth in SH-SY5Y cells and in cultured dopaminergic neurons, to the same extent as rhBMP2. Neither of these two compounds affected cell viability, rationalising their safe application to dopaminergic neurons. In contrast to this finding, exposure to 5 μM quinacrine induced the cell death of human tendon cells after 7 DIV [21]; however, the concentration of quinacrine used on dopaminergic neurons in our study was significantly lower, at 10 nM. In addition, quinacrine and niclosamide were also found to activate the Smad signalling pathway, as evidenced by upregulated Smad-dependent transcription in SH-SY5Y cells, and increased phospho-Smad levels in cultured dopaminergic neurons. This effect on Smad signalling was not seen in human tendon cells [21]; however, in that study, the cells were exposed to quinacrine or niclosamide for 2 h, in contrast to the 24-h timepoint used in the present study. Moreover, the effects of both quinacrine and niclosamide on neurite length and on phospho-Smad levels were blocked by dorsomorphin, showing that they were mediated through activation of the canonical Smad signalling pathway. This is consistent with findings showing that BMP neurotrophic effects were prevented by the use of dorsomorphin, which demonstrated their dependence on the canonical Smad signalling pathway to elicit neurotrophic action [28, 31].
We have recently shown that rhBMP2 promotes TH+ cell survival and neurite length, and that it protects against the selective dopaminergic neurotoxins MPP+ and 6-OHDA in primary cultures of the E14 VM [20]. In this study, quinacrine and niclosamide also demonstrated significant neuroprotection against MPP+- and 6-OHDA-induced neurite degeneration and cell death. In support of this finding, quinacrine has previously been shown to protect striatal dopamine levels in vivo against 6-OHDA and MPTP striatal lesioning in Wistar rats and C57BL mice, respectively [39]. In the MPTP study, adult male mice were administered one of four different doses of quinacrine via intraperitoneal (i.p.) injection 30 min prior to the administration of 30 mg/kg MPTP daily for 5 days. Mice were sacrificed on day 5 and striata were collected for HPLC analyses. For the 6-OHDA study, Wistar rats were administered one of four different doses of quinacrine via i.p. injection 30 min prior to a unilaterally 6-OHDA lesion to the right striatum and were treated daily thereafter for 4 days. On day 5, animals were sacrificed and striata were collected for HPLC analyses. In both studies, they found MPTP and 6-OHDA significantly reduced striatal dopamine and glutathione (GSH), which was attenuated by co-treatment with quinacrine in a dose-dependent fashion [39]. In addition, niclosamide fully prevented the degeneration induced by MPP+ and 6-OHDA on neurite length, and was protective against the effect of MPP+, but not 6-OHDA, on TH+ cell survival. These findings demonstrate that quinacrine and niclosamide can protect midbrain dopaminergic neurons against neurotoxin-induced dopaminergic degeneration, which is the clinical hallmark of PD.
We have previously shown that rhBMP2 is protective against axon degeneration induced by the overexpression of both wild-type and mutant A53T α-synuclein plasmids in E14 VM cultures [20]. Therefore, in this experiment, E14 VM cultures were transduced with an AAV-αSyn vector at an MOI that induced axon degeneration in dopaminergic neurons. The approach to this study was twofold: to investigate if quinacrine and niclosamide could protect dopaminergic nerve cells and their terminals from α-synuclein, and if so, if these compounds could restore dopaminergic nerve terminals after degeneration had already occurred. Concurrent treatment with quinacrine or niclosamide at the time of AAV-αSyn transduction protected TH+ neurons against α-synuclein-induced axon degeneration and promoted dopaminergic cell survival. In addition, treatment with quinacrine or niclosamide 5 days after the AAV-αSyn infection, when degeneration had already occurred, also protected TH+ neurons against α-synuclein-induced axon degeneration and promoted dopaminergic cell survival. These findings are particularly noteworthy as axon degeneration is thought to be a central early pathological event that precedes symptom onset and initiates nigrostriatal neuron loss in PD [40, 41]. Thus, the advent of new therapeutics that can substantially restore dopaminergic nerve terminals within the nigrostriatal pathway is important.
In addition to its effects on BMP signalling in dopaminergic neurons, it is worth noting that quinacrine has also been shown to be a potent non-selective inhibitor of phospholipase A2 (PLA2) [39, 42]. PLA2 is an important enzyme in the inflammatory response, and within the brain, PLA2 regulates the conversion of arachidonic acid into proinflammatory mediators and its reabsorption into the membrane [43]. When PLA2 activity becomes disrupted, abnormally high numbers of proinflammatory mediators are produced, resulting in oxidative stress and neuroinflammation analogous to neurological diseases, such as PD [43]. PLA2 is also known to play a role in regulating the synaptic release of dopamine within the adrenal medulla [44]. A study demonstrated that rats that had received a unilateral intranigral injection of PLA2 displayed significantly increased apomorphine-induced rotational behaviour, suggesting that PLA2 can cause severe inhibition of dopamine release within the nigrostriatal pathway [45]. In support of this, another study observed that mice deficient in group IV cytosolic PLA2 were resistant to the degenerative effects of the dopaminergic neurotoxin MPTP [46]. These studies support the hypothesis that inhibitors of PLA2, like quinacrine, could be a promising lead for the treatment of neurological disorders. Interestingly, it has been shown that TGF-β signalling in mesangial cells, leading to the activation of Smad proteins, attenuates PLA2 signalling [47]. Consequently, further work is needed to clarify whether a relationship exists between BMP-Smad signalling and PLA2 activity in dopaminergic neurons, which may further elucidate the mechanism of action of quinacrine.
Similarly, niclosamide is also known to modulate several cell signalling cascades, including Wnt/β-catenin, mTOR, JAK/STAT3 and NF-κB, and can cause mitochondrial depolarisation through the uncoupling of oxidative phosphorylation [22, 48, 49]. This is interesting as PINK1, a mitochondrial serine/threonine-protein kinase, regulates mitochondrial homeostasis and is activated by mitochondrial depolarisation [50]. Furthermore, point mutations in the PINK1 gene are responsible for autosomal recessive forms of early-onset PD [51]. Niclosamide treatment has also been shown to indirectly activate endogenous PINK1 signalling in cultured cortical neurons [52]. These findings suggest a possible therapeutic role for niclosamide to slow the progression of PD neuropathology through the activation of PINK1, as well as BMP signalling.
In summary, this study has shown that the FDA-approved drugs quinacrine and niclosamide elicit positive phenotypic changes in both SH-SY5Y cells and primary cultures of dopaminergic neurons. Specifically, these drugs confer neurotrophic effects on SH-SY5Y cells and cultured dopaminergic neurons by stimulating the canonical BMP-Smad signalling pathway. Furthermore, quinacrine and niclosamide have been shown to be protective against MPP+- and 6-OHDA-induced dopaminergic degeneration and can protect and restore dopaminergic axon degeneration caused by AAV-αSyn. Further work is now needed to examine the effects of quinacrine and niclosamide in in vivo models of PD, such as the AAV-αSyn rat model, and to further explore the mechanism of action of these compounds. This study adds considerable evidence to the potential use of these small molecule drugs to protect dopaminergic neurons and justifies their further investigation as potential therapeutic agents for neuroprotection in PD.
Data Availability
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
References
Lees AJ, Hardy J, Revesz T (2009) Parkinson's disease. Lancet (London, England) 373(9680):2055–2066
Kelly MJ, O'Keeffe GW, Sullivan AM (2015) Viral vector delivery of neurotrophic factors for Parkinson's disease therapy. Expert Rev Mol Med 17:e8
Paul G, Sullivan AM (2019) Trophic factors for Parkinson's disease: where are we and where do we go from here? Eur J Neurosci 49(4):440–452
Sullivan AM, Toulouse A (2011) Neurotrophic factors for the treatment of Parkinson's disease. Cytokine Growth Factor Rev 22(3):157–165
Choi-Lundberg DL, Lin Q, Chang YN, Chiang YL, Hay CM, Mohajeri H et al (1997) Dopaminergic neurons protected from degeneration by GDNF gene therapy. Science (New York, NY) 275(5301):838–841
Kordower JH, Emborg ME, Bloch J, Ma SY, Chu Y, Leventhal L et al (2000) Neurodegeneration prevented by lentiviral vector delivery of GDNF in primate models of Parkinson's disease. Science (New York, NY) 290(5492):767–773
Kordower JH, Herzog CD, Dass B, Bakay RA, Stansell J 3rd, Gasmi M et al (2006) Delivery of neurturin by AAV2 (CERE-120)-mediated gene transfer provides structural and functional neuroprotection and neurorestoration in MPTP-treated monkeys. Ann Neurol 60(6):706–715
Ramaswamy S, McBride JL, Herzog CD, Brandon E, Gasmi M, Bartus RT et al (2007) Neurturin gene therapy improves motor function and prevents death of striatal neurons in a 3-nitropropionic acid rat model of Huntington's disease. Neurobiol Dis 26(2):375–384
Su X, Kells AP, Huang EJ, Lee HS, Hadaczek P, Beyer J, Bringas J, Pivirotto P et al (2009) Safety evaluation of AAV2-GDNF gene transfer into the dopaminergic nigrostriatal pathway in aged and parkinsonian rhesus monkeys. Hum Gene Ther 20(12):1627–1640
Gill SS, Patel NK, Hotton GR, O'Sullivan K, McCarter R, Bunnage M, Brooks DJ, Svendsen CN et al (2003) Direct brain infusion of glial cell line-derived neurotrophic factor in Parkinson disease. Nat Med 9(5):589–595
Slevin JT, Gerhardt GA, Smith CD, Gash DM, Kryscio R, Young B (2005) Improvement of bilateral motor functions in patients with Parkinson disease through the unilateral intraputaminal infusion of glial cell line—derived neurotrophic factor. J Neurosurg 102(2):216–222
Lang AE, Gill S, Patel NK, Lozano A, Nutt JG, Penn R, Brooks DJ, Hotton G et al (2006) Randomized controlled trial of intraputamenal glial cell line-derived neurotrophic factor infusion in Parkinson disease. Ann Neurol 59(3):459–466
Patel NK, Bunnage M, Plaha P, Svendsen CN, Heywood P, Gill SS (2005) Intraputamenal infusion of glial cell line–derived neurotrophic factor in PD: a two-year outcome study. Ann Neurol 57(2):298–302
Olanow CW, Goetz CG, Kordower JH, Stoessl AJ, Sossi V, Brin MF, Shannon KM, Nauert GM et al (2003) A double-blind controlled trial of bilateral fetal nigral transplantation in Parkinson's disease. Ann Neurol 54(3):403–414
Warren Olanow C, Bartus RT, Baumann TL, Factor S, Boulis N, Stacy M, Turner DA, Marks W et al (2015) Gene delivery of neurturin to putamen and substantia nigra in Parkinson disease: a double-blind, randomized, controlled trial. Ann Neurol 78(2):248–257
Whone A, Luz M, Boca M, Woolley M, Mooney L, Dharia S, Broadfoot J, Cronin D et al (2019) Randomized trial of intermittent intraputamenal glial cell line-derived neurotrophic factor in Parkinson's disease. Brain. 142(3):512–525
Whone AL, Boca M, Luz M, Woolley M, Mooney L, Dharia S, Broadfoot J, Cronin D et al (2019) Extended Treatment with glial cell line-derived neurotrophic factor in parkinson's disease. J Parkinsons Dis 9:301–313
O'Keeffe GW, Hegarty S, Sullivan A (2017) Targeting bone morphogenetic protein signalling in midbrain dopaminergic neurons as a therapeutic approach in Parkinson's disease. Neuronal Signaling
Goulding SR, Sullivan AM, O'Keeffe GW, Collins LM (2020) The potential of bone morphogenetic protein 2 as a neurotrophic factor for Parkinson's disease. Neural Regen Res 15(8):1432–1436
Goulding SR, Sullivan AM, O'Keeffe GW, Collins LM (2019) Gene co-expression analysis of the human substantia nigra identifies BMP2 as a neurotrophic factor that can promote neurite growth in cells overexpressing wild-type or A53T α-synuclein. Parkinsonism Relat Disord 64:194–201
Ghebes CA, van Lente J, Post JN, Saris DB, Fernandes H (2017) High-throughput screening assay identifies small molecules capable of modulating the BMP-2 and TGF-beta1 Signaling pathway. SLAS Discov 22(1):40–50
Chen W, Mook RA Jr, Premont RT, Wang J (2018) Niclosamide: beyond an antihelminthic drug. Cell Signal 41:89–96
Ehsanian R, Van Waes C, Feller SM (2011) Beyond DNA binding - a review of the potential mechanisms mediating quinacrine's therapeutic activities in parasitic infections, inflammation, and cancers. Cell Commun Signal 9:13
Hegarty SV, Sullivan AM, Keeffe GW (2016) Protocol for evaluation of neurotrophic strategies in Parkinson’s disease-related dopaminergic and sympathetic neurons in vitro. 2016
Yu PB, Hong CC, Sachidanandan C, Babitt JL, Deng DY, Hoyng SA, Lin HY, Bloch KD et al (2008) Dorsomorphin inhibits BMP signals required for embryogenesis and iron metabolism. Nat Chem Biol 4(1):33–41
Jordan J, Bottner M, Schluesener HJ, Unsicker K, Krieglstein K (1997) Bone morphogenetic proteins: neurotrophic roles for midbrain dopaminergic neurons and implications of astroglial cells. Eur J Neurosci 9(8):1699–1709
Reiriz J, Espejo M, Ventura F, Ambrosio S, Alberch J (1999) Bone morphogenetic protein-2 promotes dissociated effects on the number and differentiation of cultured ventral mesencephalic dopaminergic neurons. J Neurobiol 38(2):161–170
Hegarty SV, Sullivan AM, O'Keeffe GW (2013) BMP2 and GDF5 induce neuronal differentiation through a Smad dependant pathway in a model of human midbrain dopaminergic neurons. Mol Cell Neurosci 56:263–271
Krieglstein K, Suter-Crazzolara C, Hotten G, Pohl J, Unsicker K (1995) Trophic and protective effects of growth/differentiation factor 5, a member of the transforming growth factor-beta superfamily, on midbrain dopaminergic neurons. J Neurosci Res 42(5):724–732
Nutt JG, Burchiel KJ, Comella CL, Jankovic J, Lang AE, Laws ER Jr et al (2003) Randomized, double-blind trial of glial cell line-derived neurotrophic factor (GDNF) in PD. Neurology. 60(1):69–73
Hegarty SV, Collins LM, Gavin AM, Roche SL, Wyatt SL, Sullivan AM, O’Keeffe GW (2014) Canonical BMP-Smad signalling promotes neurite growth in rat midbrain dopaminergic neurons. NeuroMolecular Med 16(2):473–489
Weiss A, Attisano L (2013) The TGFbeta superfamily signaling pathway. Wiley Interdiscip Rev Dev Biol 2(1):47–63
Wrana JL, Attisano L (2000) The Smad pathway. Cytokine Growth Factor Rev 11(1-2):5–13
Weber D, Kotzsch A, Nickel J, Harth S, Seher A, Mueller U, Sebald W, Mueller TD (2007) A silent H-bond can be mutationally activated for high-affinity interaction of BMP-2 and activin type IIB receptor. BMC Struct Biol 7:6
Mueller TD, Nickel J (2012) Promiscuity and specificity in BMP receptor activation. FEBS Lett 586(14):1846–1859
Wang RN, Green J, Wang Z, Deng Y, Qiao M, Peabody M, Zhang Q, Ye J et al (2014) Bone Morphogenetic Protein (BMP) signaling in development and human diseases. Genes Dis 1(1):87–105
Heinecke K, Seher A, Schmitz W, Mueller TD, Sebald W, Nickel J (2009) Receptor oligomerization and beyond: a case study in bone morphogenetic proteins. BMC Biol 7:59
Hegarty SV, Sullivan AM, O'Keeffe GW (2017) Endocytosis contributes to BMP2-induced Smad signalling and neuronal growth. Neurosci Lett 643:32–37
Tariq M, Khan HA, Al Moutaery K, Al DS (2001) Protective effect of quinacrine on striatal dopamine levels in 6-OHDA and MPTP models of Parkinsonism in rodents. Brain Res Bull 54(1):77–82
Kordower JH, Olanow CW, Dodiya HB, Chu Y, Beach TG, Adler CH, Halliday GM, Bartus RT (2013) Disease duration and the integrity of the nigrostriatal system in Parkinson’s disease. Brain. 136(8):2419–2431
O'Keeffe GW, Sullivan AM (2018) Evidence for dopaminergic axonal degeneration as an early pathological process in Parkinson's disease. Parkinsonism Relat Disord 56:9–15 (56:9-15)
Talk AC, Muzzio IA, Matzel LD (1997) Phospholipases and arachidonic acid contribute independently to sensory transduction and associative neuronal facilitation in Hermissenda type B photoreceptors. Brain Res 751(2):196–205
Farooqui AA, Ong WY, Horrocks LA (2006) Inhibitors of brain phospholipase A2 activity: their neuropharmacological effects and therapeutic importance for the treatment of neurologic disorders. Pharmacol Rev 58(3):591–620
Kudo I, Matsuzawa A, Imai K, Murakami M, Inoue K (1996) Function of type II phospholipase A2 in dopamine secretion by rat neuronal PC12 cells. J Lipid Mediat Cell Signal 14(1-3):25–31
Brunner J, Gattaz WF (1995) Intracerebral injection of phospholipase A2 inhibits dopamine-mediated behavior in rats: possible implications for schizophrenia. Eur Arch Psychiatry Clin Neurosci 246(1):13–16
Klivenyi P, Beal MF, Ferrante RJ, Andreassen OA, Wermer M, Chin MR, Bonventre JV (1998) Mice deficient in group IV cytosolic phospholipase A2 are resistant to MPTP neurotoxicity. J Neurochem 71(6):2634–2637
Xin C, Ren S, Kleuser B, Shabahang S, Eberhardt W, Radeke H, Schäfer-Korting M, Pfeilschifter J et al (2004) Sphingosine 1-phosphate cross-activates the Smad signaling cascade and mimics transforming growth factor-beta-induced cell responses. J Biol Chem 279(34):35255–35262
Kadri H, Lambourne OA, Mehellou Y (2018) Niclosamide, a drug with many (re)purposes. ChemMedChem. 13(11):1088–1091
Alasadi A, Chen M, Swapna GVT, Tao H, Guo J, Collantes J, Fadhil N, Montelione GT et al (2018) Effect of mitochondrial uncouplers niclosamide ethanolamine (NEN) and oxyclozanide on hepatic metastasis of colon cancer. Cell Death Dis 9(2):215
Kazlauskaite A, Muqit MM (2015) PINK1 and Parkin – mitochondrial interplay between phosphorylation and ubiquitylation in Parkinson's disease. FEBS J 282(2):215–223
Valente EM, Abou-Sleiman PM, Caputo V, Muqit MMK, Harvey K, Gispert S et al (2004) Hereditary early-onset Parkinson's disease caused by mutations in PINK1. Science (New York, NY) 304(5674):1158–1160
Barini E, Miccoli A, Tinarelli F, Mulholland K, Kadri H, Khanim F, Stojanovski L, Read KD et al (2018) The anthelmintic drug niclosamide and its analogues activate the parkinson's disease associated protein kinase pinK1. Chembiochem 19(5):425–429
Funding
This publication has emanated from research conducted with the financial support of a RISAM PhD scholarship from Munster Technological University (R00094948) and a research grant from Science Foundation Ireland (SFI) under the grant numbers 15/CDA/3498 (G.O’K.).
Author information
Authors and Affiliations
Contributions
SG performed the experiments, analysed the data and co-wrote the manuscript. ML, AS, LC and GOK co-wrote the manuscript. SG, LC and GOK designed the experiments. LC and GOK supervised the study.
Corresponding authors
Ethics declarations
Ethics Approval
Animal tissue was obtained under license with full ethical approval.
Consent to Participate
N/A.
Consent for Publication
N/A.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
Fig. 1. SH-SY5Y cells as a tool to study drugs affecting BMP-Smad signalling. Representative photomicrographs showing immunocytochemical staining for (a) BMPR1B, (b) BMPR2 and (c) Smad1/5/8 expression in SH-SY5Y cells. (d) Representative photomicrographs of immunocytochemical staining of phospho-Smad1/5/8 and (e) intensity of phospho-Smad1/5/8 as measured using ELISA in SH-SY5Y cells after treatment with 50 ng/ml rhBMP2 with and without 1 μg/ml dorsomorphin for 2 h. (f) Total neurite length and (h) representative photomicrographs of SH-SY5Y cells after treatment with 10, 50 or 200 ng/ml rhBMP2 for 24 h. (g) Total neurite length and (i) representative photomicrographs of SH-SY5Y cells after treatment with 1 μg/ml dorsomorphin with and without 50 ng/ml rhBMP2 for 24 h. All data are presented as mean ± SEM from at least three experiments. (*p < 0.05, **p < 0.01, ***p < 0.001 vs. control; +++p < 0.001 vs rhBMP2. One-way ANOVA with Tukey’s post-hoc test). Fig. 2. AAV-α-synuclein affects neurite length of cultured dopaminergic neurons in a concentration-dependent manner. (a) Graph and (b) representative photomicrographs of TH+ neurons transduced with AAV2/6-GFP or AAV2/6-α-synuclein at the MOI’s indicated for 5 DIV. Scale bar = 50 μm. All data are presented as mean ± SEM from at least three experiments. (**p < 0.01 vs. control; Unpaired Student’s t test). (DOCX 528 kb)
Rights and permissions
About this article
Cite this article
Goulding, S.R., Lévesque, M., Sullivan, A.M. et al. Quinacrine and Niclosamide Promote Neurite Growth in Midbrain Dopaminergic Neurons Through the Canonical BMP-Smad Pathway and Protect Against Neurotoxin and α-Synuclein-Induced Neurodegeneration. Mol Neurobiol 58, 3405–3416 (2021). https://doi.org/10.1007/s12035-021-02351-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-021-02351-8