Abstract
Exercise can act as a disease-modifying agent in Parkinson’s disease (PD), and we have previously demonstrated that voluntary exercise in running wheels during 2 weeks normalizes striatopallidal dopaminergic signaling and prevents the development of l-DOPA-induced dyskinesia (LID) in C57BL/6 mice. We now tested whether LID in Swiss albino mice could be attenuated by treadmill-controlled exercise alone or in combination with the reference antidyskinetic drug amantadine. The daily intraperitoneal (i.p.) treatment with three different doses of l-DOPA/benserazide (30/12.5, 50/25, or 70/35 mg/kg) during 3 weeks induced increasing levels of LID scores in hemiparkinsonian Swiss albino mice previously lesioned with a unilateral intrastriatal injection of 6-hydroxydopamine (6-OHDA, 10 μg). Then, we addressed the antidyskinetic effects of treadmill-controlled exercise by comparing LID, induced by l-DOPA/benserazide (50/25 mg/kg, i.p.) during 4 weeks, in sedentary and daily exercised mice. Exercise reduced LID and improved motor skills of dyskinetic mice, as indicated by decreased contralateral bias, increase in maximal load test, and latency to fall in rotarod. The antidyskinetic effect of amantadine (60 mg/kg, i.p.) was only observed in sedentary mice, indicating the absence of synergistic antidyskinetic effect of the combination of treadmill exercise plus amantadine. Finally, Western blot analysis unraveled an ability of exercise to increase the striatal immunocontent of glial cell-derived neurotrophic factor (GDNF), apart from normalizing striatal levels of tyrosine hydroxylase. These findings show that controlled treadmill exercise attenuates LID and provide the first indication that the antidyskinetic effects of treadmill exercise may involve increased striatal GDNF levels.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The development of l-DOPA-induced dyskinesia (LID), also known as abnormal involuntary movements (AIMs), constitutes a major limitation in the long-term pharmacological treatment of Parkinson’s disease (PD) patients. About 90% of PD patients develop LID within a decade of onset of l-DOPA treatment, representing an important socioeconomic cause of distress [1]. The exact molecular mechanisms associated with LID development remain unknown. The prime candidates are alterations in dopaminergic neurotransmission, described in animal models of PD, such as dysregulation in presynaptic control of vesicular dopamine (DA) loading and DA release, as well as decreased DA reuptake by DA transporter (DAT) [1, 2].
Previous findings indicated that exercise prevents the loss of dopaminergic neurons and alterations in dopaminergic neurotransmission in the nigrostriatal pathway in rodent models of PD [3,4,5,6]. Our group previously reported that voluntary exercise in running wheels during 2 weeks normalized striatopallidal dopaminergic signaling and attenuated the development of LID in 6-hydroxydopamine (6-OHDA)-hemiparkinsonian C57BL/6 mice treated with l-DOPA/benserazide (25/12.5 mg/kg, intraperitoneal (i.p.)) [7]. Interestingly, Klemann et al. [8] recently investigated the molecular mechanisms underlying the beneficial effects of forced physical exercise in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyrimidine (MPTP)-treated mice through analyses of the RNA sequencing data. The authors reported that controlled treadmill exercise during 4 weeks in MPTP-treated mice induced opposite effects compared to previously reported effects of l-DOPA on the expression of mRNAs in the substantia nigra and the ventromedial striatum [8].
However, it remains to be determined if a controlled schedule of imposed exercise (such as in a treadmill) also affords antidyskinetic effects. In addition, it is unknown if exercise can synergize with amantadine treatment, LID’s current reference therapeutics, to alleviate LID. Finally, there is still no evidence to link the known ability of exercise to stimulate the production of neurotrophic factors, such as brain-derived neurotrophic factor (BDNF) [9] and glial cell-derived neurotrophic factor (GDNF) [10] with the control of dyskinesia. GDNF emerges as a particularly promising candidate since it exerts a pivotal role in the development and maintenance of spinal motor neurons and midbrain dopaminergic neurons [11, 12]. Moreover, GDNF downregulation has been implicated in PD pathophysiology [12].
Therefore, in the present study, we first carried out a dose-response curve of the development of LID in 6-OHDA-hemiparkinsonian Swiss albino mice to investigate the putative antidyskinetic effects of treadmill-controlled exercise alone or in combination with amantadine treatment and we gauged the possible correlation with striatal GDNF levels.
Methods and Materials
Subjects and 6-OHDA-Induced Hemiparkinsonism
A total of 62 adult male Swiss albino mice (8–10 weeks old, 30–35 g) from the Animal Facility of Federal University of Santa Catarina (UFSC, Florianópolis, Brazil) were used. All experimental protocols were approved by the Institutional Animal Care and Use Committee (IACUC) of UFSC (protocol PP00357).
The experimental designs are illustrated in Figs. 1a and 2a. Mice were housed in collective cages with controlled environment: 12/12-h light/dark cycle (lights on at 7 am) and room temperature of 23 ± 1 °C with ad libitum access to food and water. The animals were anesthetized with an i.p. injection of ketamine (90 mg/kg) and xylazine (10 mg/kg). All drugs were purchased from Sigma-Aldrich and were freshly diluted in saline (0.9% NaCl). The injection volume used was 0.1 mL/10 g body weight. 6-OHDA (10 μg in 1 μL of 0.1% sodium metabisulfite diluted in 0.9% NaCl) was injected in the right dorsolateral striatum using a stereotaxic apparatus with the following coordinates (in mm): AP 0.5, ML 2, and DV 3 [13]. The hemiparkinsonian state of 6-OHDA-lesioned mice was confirmed using two different criteria: (i) net rotations following a challenge with R(−)-apomorphine and (ii) side bias in spontaneous forelimb use in the cylinder task.
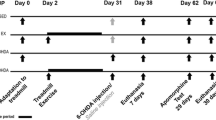
Experimental design (a). All mice received a single intrastriatal administration of 6-OHDA (10 μg) and experienced a typical hemiparkinsonian phenotype, as indicated by an increase in asymmetry score (contralateral bias) in the cylinder test (***P < 0.001, Student’s t test) (b). 6-OHDA-treated mice also showed increased contralateral rotations (4 weeks) measured 30 min after a challenge with apomorphine (APO, 0.5 mg/kg, subcutaneous (s.c.)) (***P < 0.001 vs. ipsilateral, Student’s t test) (c). Mice were then separated into four groups with intraperitoneal treatments with either saline or three different doses of l-DOPA/benserazide (30/12.5 mg/kg, 50/25 mg/kg, or 70/35 mg/kg) during 3 weeks, and the AIM score was repeatedly evaluated during the period of treatment (*P < 0.05 all tested l-DOPA/benserazide doses vs. saline; &P < 0.05 l-DOPA/benserazide 70/35 mg/kg vs. other two doses; #P < 0.05 l-DOPA/benserazide 50/25 mg/kg vs. day 1 of the same dose, ANOVA with repeated measures followed by Newman-Keuls post hoc tests). Data are expressed as mean ± SEM (n = 8–12 animals/group). AIMs, abnormal involuntary movements; R(−)-APO R(−)-apomorphine; s.c., subcutaneous; i.p., intraperitoneal; 6-OHDA, 6-hydroxydopamine; vs., versus; wks, weeks
Experimental design (a). After a single intrastriatal administration of 6-OHDA (10 μg), mice displayed increased counterclockwise rotations after 4 weeks of surgery (data not shown). Mice were then separated in exercise (treadmill) or sedentary groups, and both groups were treated with l-DOPA/benserazide (50/25 mg/kg, i.p.) during 4 weeks (a). Exercised mice showed a reduced AIM score from 14th, 21th, and 28th days of treatment (***P < 0.001 vs. sedentary; ###P < 0.001 vs. exercised on 1st day; ANOVA with repeated measures followed by Newman-Keuls post hoc tests) (b). Amantadine administration (60 mg/kg, i.p.) decreased AIM score only in sedentary group (&&&P < 0.001, Student’s t test) (b). Exercise also improved motor skills of mice, as indicated by a significant decrease in contralateral bias (c), increase in maximal load test (d), and latency to fall in rotarod (e) (***P < 0.001 vs. basal; ###P < 0.001 vs. 6-OHDA sedentary at the end of the protocol, ANOVA with repeated measures followed by Newman-Keuls post hoc tests). Data are expressed as mean ± SEM (n = 8–12 animals/group). AIMs, abnormal involuntary movements; OHDA, 6-hydroxydopamine; s.c., subcutaneous; i.p., intraperitoneal; vs., versus; wks, weeks
Behavioral Tasks
Animals were habituated for 1 h in a sound- and light-attenuated (12 lx) room before behavioral tasks, which were carried out during the light phase of the cycle between 10 am and 4 pm. All behaviors were monitored through a video camera, and the images were analyzed with the AnyMaze® video tracking (Stoelting Co., Wood Dale, IL, USA) by an experienced experimenter who was not blind to the experimental groups.
R(−)-Apomorphine Challenge
Mice were challenged with the non-selective DA receptor agonist R(−)-apomorphine (0.5 mg/kg, subcutaneous (s.c.)) in plastic tubes (19-cm diameter, 22 cm high). Net rotations were counted during drug-free habituation (10 min) and after treatment with R(−)-apomorphine (30 min) diluted in saline. After 4 weeks of recovery from 6-OHDA surgery (Fig. 1a), mice with an insufficient number of rotations (< 2 counterclockwise rotations/min) [14] were discarded (around 20% of mice).
Cylinder Task
Bradykinesia and motor impairments were assessed in individual glass cylinders (12-cm diameter, 15 cm high), after 6-OHDA surgery, at 4 weeks in experiment 1, and at three different times in experiment 2: (i) before 6-OHDA lesion, (ii) 4 weeks after 6-OHDA surgery, and (iii) 4 weeks after l-DOPA/benserazide treatment (40 min after the last injection). In this task, we examined side bias in spontaneous forelimb use during explorative activity, which corresponds to the extent of a 6-OHDA lesion in rodents, by counting the number of wall contacts made with the forepaws. A measure of forelimb use asymmetry was obtained by expressing the number of touches performed by the paw contralateral to the lesion (left paw) as a percentage of the total number of touches recorded for each session [15].
Rotarod
The rotarod was used to evaluate the motor coordination of the animals [16]. The accelerating rotarod apparatus (Insight Scientific Equipments, Ribeirão Preto, SP, Brazil) consists of a grooved metal roller (6 cm in diameter) and separated 9-cm-wide compartments elevated at 16 cm. The spindle speed was increased from 6 to 20 rpm over a maximal period of 300 s, and the time spent on the accelerating rotarod and the corresponding rpm were determined. At baseline (performed 40 min before treatment), mice that were able to perform the task within 30–120 s were chosen for the experiment. Rotarod performance was assessed at three different times: (i) before 6-OHDA lesion, (ii) 4 weeks after 6-OHDA surgery, and (iii) 4 weeks after l-DOPA/benserazide treatment (40 min after the last injection).
LID and Exercise
LID was determined based on the evaluation of abnormal involuntary movements (AIM score), which were assessed in individual glass cylinders (12-cm diameter, 15-cm high). Mice were daily treated with a single i.p. injection of l-DOPA methyl ester hydrochloride plus benserazide hydrochloride, a peripheral DOPA decarboxylase inhibitor. In experiment 1, we performed a dose-response curve of the development of LID upon daily i.p. treatment with the three doses of l-DOPA/benserazide (30/12.5 mg/kg; 50/25 mg/kg, or 70/35 mg/kg) during 3 weeks. The assessment of LID was made at 1, 4, 7, 14, and 21 days (Fig. 1a). In experiment 2, a selected dose of l-DOPA/benserazide (50/25 mg/kg, i.p.) was daily administered during 30 days and LID was assessed at 1, 7, 14, 21, 28, and 30 days. The AIM score was manually assessed during 120 min after l-DOPA/benserazide administration. AIM score was classified into orofacial, forelimb, axial or dystonic, and locomotive, and the severity was scored for 1 min every 10 min using a 0–4 scale, as previously described [17, 18].
In experiment 2, the exercised group ran on a horizontal treadmill with individual lanes (26 × 8.5 × 12 cm) [14]. The animals were divided into two groups: exercised (n = 12) and sedentary controls (n = 12). Firstly, mice were habituated to the treadmill (16 m/min, 10 min) during 4 days. After resting 1 day, the mice were subject to an incremental exercise load test with the treadmill at 3% grade; mice started running at 16 m/min, and the speed increased 2 m/min every 3 min. We next determined the maximal speed of the exercised mice by submitting them to a progressive running exercise program (4 weeks, 5 times/week, with time-out on weekends), where the time and speed were adjusted every week. The mice in the exercised group began running at a speed of 20 m/min (60% obtained in incremental exercise load test) in the first and second weeks and 23 m/min in the third and fourth weeks. The velocity was increased in 5 m/min every week, and the total training time started in 30, 35, 40, and 45 min every week, respectively [14].
In addition, after the 30 days of DOPA/benserazide (50/25 mg/kg, i.p.) treatment without or with the exercise protocol, we tested the only approved antidyskinetic drug amantadine hydrochloride (60 mg/kg, i.p.) [7, 13], which was administered 90 min before l-DOPA/benserazide (Fig. 2a).
Western Blotting
Forty minutes after the last l-DOPA/benserazide injection, mice were killed by cervical dislocation, and the striatum was dissected, placed in liquid nitrogen, and stored at − 80 °C until analysis. Striata samples were mechanically homogenized in 400–500 μL of 50 mM Tris base pH 7.0 with 1 mM EDTA, 100 mM NaF, 2 mM Na3VO4, 1% Triton X-100, 10% glycerol, and a protein inhibitor cocktail from Sigma-Aldrich (Germany). Lysates were centrifuged (1000×g, 10 min, 4 °C) to obtain the supernatants, followed by a 1/1 (v/v) dilution in 100 mM Tris base pH 6.8 with 4 mM EDTA, 8% SDS, 20% glycerol, and 8% β-mercaptoethanol. The samples (60 μg protein/track, bovine serum albumin as protein standard) were separated in a 10% SDS-PAGE gel. Proteins were then transferred to nitrocellulose membrane using a semidry blotting apparatus (1.2 mA/cm2; 1.5 h). Non-specific interactions were blocked with 5% fat-free dry milk in TBS (10 mM Tris, 150 mM NaCl, pH 7.5). Immunodetection was carried out using different polyclonal primary antibodies, namely mouse anti-TH (1:1000, Santa Cruz®), anti-DAT (1:1000, Santa Cruz®), or anti-GDNF (1:1000, Santa Cruz®). The antibodies were diluted in TBS with 0.05% Tween-20 (TBS-T, pH 7.5), containing bovine serum albumin (0.5%). After all steps of blocking and incubation, the membranes were washed 3× (for 5 min each) with TBS-T. Reactions were developed by ECL. Optical density of the Western blot bands was normalized by β-actin and plotted in arbitrary units (au).
Statistical Analysis
Data are presented as mean ± standard error of mean (SEM). The contralateral bias was transformed in percentage to allow parametric statistical analysis. Data from the cylinder task and the challenge with R(−)-apomorphine in experiment 1 were analyzed using Student’s t test. The rest of data were compared using analysis of variance (ANOVA) with subsequent Newman-Keuls post hoc tests when appropriate. The accepted level of significance was P < 0.05.
Results
Dose-Response Curve of LID in 6-OHDA-Lesioned Swiss Albino Mice
In experiment 1, 6-OHDA-lesioned mice exhibited a higher number of contralateral than ipsilateral rotations following the R(−)-apomorphine challenge (P < 0.001), without significant changes over the 4 weeks of evaluation (P = 0.06) (Fig. 1c). 6-OHDA-lesioned mice also displayed a marked contralateral bias in spontaneous forelimb use in the cylinder task (P < 0.001) (Fig. 1b).
Regarding the dose-response curve of LID, it was observed that the two higher tested doses of l-DOPA (50 mg/kg and 70 mg/kg) induced higher AIM score in comparison with the lowest tested dose of l-DOPA (30 mg/kg) (F(3, 21) = 76.29, P < 0.05) (Fig. 1d). Moreover, the AIM score was stable up to 21 days of daily treatment with l-DOPA (for both doses 50 and 70 mg/kg) (Fig. 1d). Based on these results, the dose of 50 mg/kg of l-DOPA was selected for the next experiments aiming to investigate the antidyskinetic effects of treadmill exercise in 6-OHDA-lesioned Swiss albino mice (Video 1).
Treadmill Exercise Reduced LID and Improved Motor Impairments in Hemiparkinsonian Mice
Animals exercised in the treadmill showed a significant reduction of the AIM score in comparison to the sedentary group at 14, 21, and 28 days of l-DOPA/benserazide treatment (50/25 mg/kg, i.p.) (P < 0.05, Fig. 2b). After 30 days of l-DOPA treatment and treadmill exercise, the antidyskinetic effects of a single amantadine dose (60 mg/kg, i.p.) were probed in the sedentary and exercised groups. Amantadine reduced AIM score in the sedentary group (Fig. 2b), but not in the exercised group (Fig. 2b). Thus, there was no synergistic antidyskinetic effect of the combination of treadmill exercise plus amantadine.
Treadmill-controlled exercise also alleviated 6-OHDA-induced motor impairments, since exercised animals displayed a reduced contralateral bias in the cylinder task after 4 weeks of l-DOPA treatment in comparison to sedentary group (F(1, 17) = 67.21, P < 0.001) (Fig. 2c). Exercised animals also presented a recovery of 6-OHDA-induced decrease in the incremental exercise load test (F(1, 17) = 83.59, P < 0.001) (Fig. 2d) and in the latency to fall off the rotarod (F(1, 17) = 75.92, P < 0.001) (Fig. 2e).
Effects of Treadmill Exercise on Striatal Neurochemical Changes in Dyskinetic Mice
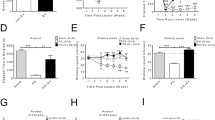
The 6-OHDA-lesioned striatum (right hemisphere) of dyskinetic mice displayed a marked decrease (about 45%) of TH immunocontent in comparison to the non-injured striatum (control) (F(1, 20) = 20.23, P < 0.001), which was reversed by treadmill exercise (F1(1, 20) = 28.65, P < 0.05) (Fig. 3a). Moreover, it was observed a significant increase in DAT immunocontent in the 6-OHDA-lesioned striatum of the sedentary group, which was not normalized by treadmill exercise (F(1, 20) = 11.54, P < 0.05) (Fig. 3b). Remarkably, treadmill exercise induced a significant increase in GDNF immunocontent in the 6-OHDA-lesioned striatum in comparison with the non-lesioned striatum of dyskinetic mice (F(1, 20) = 9.05, P < 0.05) (Fig. 3c).
Quantification of TH, DAT, and GDNF immunocontent in the non-lesioned striatum (control) and lesioned striatum (6-OHDA) of hemiparkinsonian mice treated with l-DOPA. Four weeks after a single administration of 6-OHDA (10 μg injected in the right dorsolateral striatum), mice were separated in exercise (treadmill) or sedentary groups, and both groups were treated with l-DOPA/benserazide (50/25 mg/kg, i.p.) during 4 weeks. Exercise reversed the loss of TH immunocontent in the lesioned striatum (6-OHDA) (a), while being devoid of effects on the increased striatal DAT density observed in the lesioned striatum (6-OHDA) in comparison to the non-lesioned striatum (control) of dyskinetic mice (b). Exercise increased the GDNF immunocontent in 6-OHDA-lesioned striatum of dyskinetic mice (c). *P < 0.05 vs. control (hemisphere contralateral to injury); #P < 0.05 vs. control sedentary; &P < 0.05 vs. 6-OHDA sedentary (two-way ANOVA followed by Newman-Keuls post hoc tests). Data are expressed as mean ± SEM (n = 6 animals/group). DAT, dopamine transporter; GDNF, glial cell-derived neurotrophic factor; kDa, kilodalton; TH, tyrosine hydroxylase; vs., versus; wks, weeks
Discussion
The present study provides the first evidence that a schedule of controlled exercise in a treadmill attenuates LID and improves motor impairments in Swiss albino mice previously lesioned with a unilateral intrastriatal injection of 6-OHDA. The current model of LID in Swiss albino mice was validated by the observed antidyskinetic effect of amantadine, the only approved drug to clinically manage dyskinesia [19]. Finally, whereas controlled exercise leads to contradictory effects on different dopaminergic markers, it increased the levels of GDNF in the striatum, unraveling a new neurochemical candidate mechanism to understand the antidyskinetic effects of controlled exercise.
The controlled exercise schedule in a treadmill reduced AIM score and improved motor impairments of 6-OHDA-induced hemiparkinsonian mice treated with l-DOPA, as indicated by a significant decrease in contralateral bias, increase in maximal load test, and latency to fall of the rotarod. These results corroborate our previous findings showing that voluntary exercise in running wheels during 2 weeks prevented the development of LID in 6-OHDA-hemiparkinsonian C57BL/6 mice [7]. Therefore, both controlled exercise and voluntary exercise [7] seem of potential interest to control dyskinesia. This prompts the hypothesis that exercise may represent a good adjuvant therapy for l-DOPA treatment in PD. Although it must be conceded that the current study was not designed to directly compare the efficacy of exercise and amantadine, it is remarkable that the antidyskinetic effects of treadmill exercise seemed more evident than that afforded by the single tested dose of amantadine. However, it remains to be determined whether the antidyskinetic effects of amantadine in the present model of LID can be improved by chronic administration and higher doses.
Our preclinical study is in agreement with the conclusions of a clinical study reporting a decrease of dyskinesia of PD patients after intensive rehabilitation treatment of 4 weeks of physiotherapy, 5 days a week [20]. This particular exercise protocol also decreased dyskinesia and improved the rating of the patients in the Unified Parkinson’s Disease Rating Scale (UPDRS), during 6 months [21], in agreement with the converging evidence from clinical and experimental studies that physical exercise can act as a disease-modifying agent in PD [22]. Indeed, previous studies have demonstrated that exercise alleviates motor impairments of rats and mice lesioned with 6-OHDA or MPTP [3,4,5,6, 23]. Therefore, it is possible that the improvement in the rotarod test and in contralateral bias in the cylinder test observed in the exercised group may also encompass the ability of exercise to alleviate motor complications.
The exact mechanisms engaged by exercise to control dyskinesia remain unknown. Preclinical studies have indicated that exercise prevents the loss of dopaminergic neurons as well as alterations in dopaminergic neurotransmission in the nigrostriatal pathway in rodent models of PD [3,4,5,6]. We have also previously reported that the antidyskinetic effects of voluntary exercise were associated with the normalization of striatopallidal dopaminergic signaling, namely the hyperphosphorylation of DARPP-32 (DA and cAMP-regulated phosphoprotein 32-kDa protein) and c-Fos expression [7]. Interestingly, it was recently demonstrated that treadmill exercise during 4 weeks modulates some l-DOPA-regulated molecular pathways in the substantia nigra and ventromedial striatum of the MPTP mouse model of PD, particularly signaling involving DA, neuropeptides, and endocannabinoids [8].
In the present study, the evaluation of striatal dopaminergic markers indicated that treadmill exercise reversed the loss of TH immunocontent, while being devoid of effects on the increased DAT density, observed in the 6-OHDA-lesioned striatum in comparison to the non-lesioned striatum of dyskinetic mice. These results corroborate recent findings described by Huang et al. [24] showing increased striatal DAT levels in rats after 4 weeks of 6-OHDA injection in medial forebrain bundle (MFB) and the treatment with a high l-DOPA dose (60 mg/kg) during 30 days. On the other hand, contrasting with these findings, Petzinger et al. [25] reported that treadmill exercise during 28 days actually decreased the expression of DAT and TH in the striatum of mice lesioned with MPTP. Marked differences in the exercise program and neurotoxin models of PD used in these studies may represent confounding factors, but the reasons for these discrepant observations are not known and cast reasonable doubts on this mainstream hypothesis that exercise directly controls the striatal dopaminergic system to normalize motor function.
In this context, our pioneering observation that treadmill exercise increases selectively the levels of GDNF in the 6-OHDA-lesioned striatum of dyskinetic mice provides a novel mechanistic insight. Cohen et al. [10] demonstrated that preinjury forced limb use can prevent the behavioral and neurochemical deficits to the subsequent administration of 6-OHDA and that this may be due in part to neuroprotective effects of GDNF. In fact, over the last years, GDNF has been implicated in the survival of dopaminergic and noradrenergic neurons [12]. For instance, a single administration of GDNF (4.5 μg) into the striatum of mice protected DA terminals against the loss of TH immunoreactivity induced by intrastriatal of 6-OHDA (0.5 μg) [26]. Moreover, a single intrastriatal injection of GDNF (10 or 100 μg) 4 weeks after the unilateral intrastriatal administration of 6-OHDA in Sprague-Dawley rats induced a recovery of the nigrostriatal DA system, as indicated by decreased apomorphine-induced rotations as well as higher density of TH-positive DA fibers in the striatum and higher number of DA cell bodies in the substantia nigra [27]. Of high importance, the intraventricular administration of GDNF (5 μg) improved MPTP-induced disability, reversed DA cell loss in the substantia nigra, and also diminished l-DOPA-induced dyskinesia, which may relate to the ability of GDNF to partly restore nigral dopaminergic transmission or to modify the activity of striatal output pathways [28]. In this context, the present observation that exercise induced a significant increase of striatal GDNF immunocontent provides the first demonstration in animal models of LID that exercise causes a parallel increase of both motor function and striatal GDNF levels. However, further research is needed to clarify the best exercise program (controlled vs. voluntary, intensity, and duration) to afford antidyskinetic effects as well as to ascertain the proposed role of GDNF in this beneficial impact of exercise, and this constitutes a very interesting field that requires additional research.
In conclusion, the present findings provide the first evidence that controlled exercise in treadmill attenuates LID in 6-OHDA-lesioned mice. Moreover, although the origin of LID remains unknown, the current results indicate that the observed antidyskinetic effects of treadmill exercise can be associated, at least in part, to an increase in striatal GDNF levels.
References
Cenci MA (2017) Molecular mechanisms of L-DOPA-induced dyskinesia. Handb Behav Neurosci 24:857–871
Carta M, Bezard E (2011) Contribution of pre-synaptic mechanisms to L-DOPA-induced dyskinesia. Neuroscience 198:245–251
Aguiar AS, Lopes SC, Tristão FSM, Rial D, de Oliveira G, da Cunha C, Raisman-Vozari R, Prediger RD (2016) Exercise improves cognitive impairment and iopamine metabolism in MPTP-treated mice. Neurotox Res 29:118–125
Aguiar AS Jr, Araújo AL, da-Cunha TR et al (2009) Physical exercise improves motor and short-term social memory deficits in reserpinized rats. Brain Res Bull 79:452–457
Fisher BE, Petzinger GM, Nixon K, Hogg E, Bremmer S, Meshul CK, Jakowec MW (2004) Exercise-induced behavioral recovery and neuroplasticity in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-lesioned mouse basal ganglia. J Neurosci Res 77:378–390
Toy WA, Petzinger GM, Leyshon BJ, Akopian GK, Walsh JP, Hoffman MV, Vučković MG, Jakowec MW (2014) Treadmill exercise reverses dendritic spine loss in direct and indirect striatal medium spiny neurons in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) mouse model of Parkinson’s disease. Neurobiol Dis 63:201–209
Aguiar AS, Moreira ELG, Hoeller AA et al (2013) Exercise attenuates levodopa-induced dyskinesia in 6-hydroxydopamine-lesioned mice. Neuroscience 243:46–53
Klemann CJHM, Xicoy H, Poelmans G, Bloem BR, Martens GJM, Visser JE (2018) Physical exercise modulates L-DOPA-regulated molecular pathways in the MPTP mouse model of Parkinson’s disease. Mol Neurobiol 55:5639–5657
Wu S-Y, Wang T-F, Yu L, Jen CJ, Chuang JI, Wu FS, Wu CW, Kuo YM (2011) Running exercise protects the substantia nigra dopaminergic neurons against inflammation-induced degeneration via the activation of BDNF signaling pathway. Brain Behav Immun 25:135–146
Cohen AD, Tillerson JL, Smith AD, Schallert T, Zigmond MJ (2003) Neuroprotective effects of prior limb use in 6-hydroxydopamine-treated rats: possible role of GDNF. J Neurochem 85:299–305
Sopova K, Gatsiou K, Stellos K, Laske C (2014) Dysregulation of neurotrophic and haematopoietic growth factors in Alzheimer’s disease: from pathophysiology to novel treatment strategies. Curr Alzheimer Res 11:27–39
d’Anglemont de Tassigny X, Pascual A, Lopez-Barneo J (2015) GDNF-based therapies, GDNF-producing interneurons, and trophic support of the dopaminergic nigrostriatal pathway. Implications for Parkinson’s disease. Front Neuroanat 9:10
da Conceição FSL, Ngo-Abdalla S, Houzel J-C, Rehen SK (2010) Murine model for Parkinson’s disease: from 6-OH dopamine lesion to behavioral test. J Vis Exp 35:1376
Nishimura F, Yoshikawa M, Kanda S, Nonaka M, Yokota H, Shiroi A, Nakase H, Hirabayashi H et al (2003) Potential use of embryonic stem cells for the treatment of mouse parkinsonian models: improved behavior by transplantation of in vitro differentiated dopaminergic neurons from embryonic stem cells. Stem Cells 21:171–180
Pavón N, Martín AB, Mendialdua A, Moratalla R (2006) ERK phosphorylation and FosB expression are associated with L-DOPA-induced dyskinesia in hemiparkinsonian mice. Biol Psychiatry 59:64–74
Jiang C, Wan X, Jankovic J, Christian ST, Pristupa ZB, Niznik HB, Sundsmo JS, le W (2004) Dopaminergic properties and experimental anti-parkinsonian effects of IPX750 in rodent models of Parkinson disease. Clin Neuropharmacol 27:63–73
Cenci MA (2007) Dopamine dysregulation of movement control in L-DOPA-induced dyskinesia. Trends Neurosci 30:236–243
Santini E, Valjent E, Usiello A, Carta M, Borgkvist A, Girault JA, Herve D, Greengard P et al (2007) Critical involvement of cAMP/DARPP-32 and extracellular signal-regulated protein kinase signaling in L-DOPA-induced dyskinesia. J Neurosci 27:6995–7005
Wolf E, Seppi K, Katzenschlager R, Hochschorner G, Ransmayr G, Schwingenschuh P, Ott E, Kloiber I et al (2010) Long-term antidyskinetic efficacy of amantadine in Parkinson’s disease. Mov Disord 25:1357–1363
Frazzitta G, Bertotti G, Morelli M, Riboldazzi G, Pelosin E, Balbi P, Boveri N, Comi C et al (2012) Rehabilitation improves dyskinesias in parkinsonian patients: a pilot study comparing two different rehabilitative treatments. NeuroRehabilitation 30:295–301
Frazzitta G, Morelli M, Bertotti G et al (2012) Intensive rehabilitation treatment in parkinsonian patients with dyskinesias: a preliminary study with 6-month followup. Parkinsons Dis 2012:4–7
Mantri S, Fullard ME, Duda JE, Morley JF (2018) Physical activity in early Parkinson disease. J Parkinsons Dis 8:107–111
Aguiar ASJ, Duzzioni M, Remor AP et al (2016) Moderate-intensity physical exercise protects against experimental 6-hydroxydopamine-induced hemiparkinsonism through Nrf2-antioxidant response element pathway. Neurochem Res 41:64–72
Huang L, Deng M, Zhang S, Lu S, Gui X, Fang Y (2017) β-Asarone and levodopa coadministration increases striatal levels of dopamine and levodopa and improves behavioral competence in Parkinson’s rat by enhancing dopa decarboxylase activity. Biomed Pharmacother 94:666–678
Petzinger GM, Walsh JP, Akopian G, Hogg E, Abernathy A, Arevalo P, Turnquist P, Vuckovic M et al (2007) Effects of treadmill exercise on dopaminergic transmission in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-lesioned mouse model of basal ganglia injury. J Neurosci 27:5291–5300
Lindgren N, Leak RK, Carlson KM, Smith AD, Zigmond MJ (2008) Activation of the extracellular signal-regulated kinases 1 and 2 by glial cell line-derived neurotrophic factor and its relation to neuroprotection in a mouse model of Parkinson’s disease. J Neurosci Res 86:2039–2049
Aoi M, Date I, Tomita S, Ohmoto T (2000) The effects of intrastriatal single injection of GDNF on the nigrostriatal dopaminergic system in hemiparkinsonian rats: behavioral and histological studies using two different dosages. Neurosci Res 36:319–325
Iravani MM, Costa S, Jackson MJ, Tel BC, Cannizzaro C, Pearce RKB, Jenner P (2001) GDNF reverses priming for dyskinesia in MPTP-treated, L-DOPA-primed common marmosets. Eur J Neurosci 13:597–608
Funding
This work was supported by grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Universal 408676/2016-7), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES-FCT), Programa de Apoio aos Núcleos de Excelência (PRONEX-Project NENASC), and Fundação de Apoio à Pesquisa do Estado de Santa Catarina (FAPESC). MS received scholarships from CNPq, and AES received scholarship from FAPESC/CAPES. ASA and RDP are supported by research fellowships from CNPq.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All experimental protocols were approved by the Institutional Animal Care and Use Committee (IACUC) of UFSC (protocol PP00357).
Conflict of Interest
The authors declare that they have no conflict of interest.
Electronic Supplementary Material
ESM 1
(WMV 7410 kb)
Rights and permissions
About this article
Cite this article
Speck, A.E., Schamne, M.G., S. Aguiar, A. et al. Treadmill Exercise Attenuates l-DOPA-Induced Dyskinesia and Increases Striatal Levels of Glial Cell-Derived Neurotrophic Factor (GDNF) in Hemiparkinsonian Mice. Mol Neurobiol 56, 2944–2951 (2019). https://doi.org/10.1007/s12035-018-1278-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-1278-3