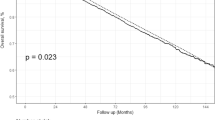
Abstract
Breast cancer is the second most diagnosed malignancy in American women with a lifetime occurrence of 1 in 8 women in the United States. There has been a dearth of research focusing on regional differences in breast cancer mortality with respect to race in the US. It is crucial to identify regions that are lagging to uplift the outreach of breast cancer care to certain races. Data for this study were obtained from the 2016–2018 Nationwide Inpatient Sample. In-hospital mortality, race and hospital regions for the patients with the primary diagnosis of Malignant Neoplasms of Breast were studied. Baseline characteristics of participants were summarized using descriptive statistics. The patient population was stratified as per race, hospital region, gender, therapy received and family history. Logistic regression was performed to derive the odds ratio while adjusting for different variables. 99, 543 patients with metastatic breast cancer were identified. African Americans (AAs) were found to have the highest reported deaths at 5.54%, followed by Asians and Pacific Islanders at 4.80% and Caucasians 4.09% (p < 0.0001). The odds of dying were significantly higher in the AA population when compared to Caucasian population (OR 1.391 (1.286–1.504)), and the odds were consistently higher across all regions of the US. In terms of regional disparities with respect to race, AA’s had highest mortality in the south whereas all other races had the highest mortality in the west. It was seen that races identifying as “others” had significantly higher odds of dying in the Northeast. It is crucial to identify racial differences in the various regions across the US in order to implement appropriate outreach strategies to tackle these disparities.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Breast cancer is the second most diagnosed malignancy in American women, with 1 in every 8 women being diagnosed with it during their lifetime [1]. It is anticipated that 300,590 new cases of breast cancer and 43,700 deaths owing to breast cancer will occur in 2023 [2]. Incidence rates continue to rise in the United States. Even though breast cancer mortality has been reported to have declined in the past decade, the decline has been slower when compared to the 1990s and the 2000s.
Racial disparities in breast cancer mortality continue to persist despite the advent and implementation of screening modalities by national agencies. African American (AA) females are less likely to be diagnosed with breast cancer and yet bear a 40% higher burden of mortality of this malignancy in comparison to white women [1].
Many studies have postulated that the racial disparities among breast cancer patients and their mortality trends vary due to different receptor expressions and stages of disease at the time of diagnosis [3,4,5,6]. Furthermore, socioeconomic variables and lifestyle are also plausible explanations for this discrepancy [7,8,9,10]. Even in clinical trials, there remains an underrepresentation of all ethnic minorities [11,12,13,14].
A wealth of published data exists on different factors that impact breast cancer mortality, such as race, age [15,16,17,18,19] and socioeconomic status [20, 21]. There is limited research on regional differences studying the relationship between race and regional variations [22]. In addition, only a few studies have been published so far that have studied a large population to highlight racial disparities in breast cancer [23].
Using data from the 2016–2018 Nationwide Inpatient Sample (NIS), our study assessed the racial and regional differences in all-cause mortality in 99,543 patients diagnosed with metastatic breast cancer.
Methods
Data for this study were obtained from the 2016–2018 Nationwide Inpatient Sample (NIS). The NIS is maintained as part of the Health Care Utilization Project (HCUP) of the Agency for Healthcare Research and Quality (AHRQ). It is the largest all-payer inpatient care database in the US and comprises a 20% stratified random sample of all US hospital discharges. Each discharge in the NIS is de-identified, so all discharges are considered independent. The unit for this analysis was each discharge rather than each individual.
Study population
We identified all patients with a primary diagnosis of Malignant Neoplasms of Breast by querying the database using the International Classification of Disease-Clinical Modification, 10th revision (ICD-10 CM) code “C50”.
Study variables
The ICD 10 CM codes were used to find other variables, which are mentioned in Table 1. In-hospital mortality, race, and hospital regions were studied using HCUP variables named “DIED,” “RACE,” and “HOSP_REGION,” respectively.
Aims and objectives
Our study’s main aim was to study racial disparity in all-cause mortality in patients with metastatic neoplasms of breasts. Our secondary goal was to evaluate the racial and gender gap in all-cause mortality in patients with malignant neoplasms of breasts in different regions of the USA.
Statistical analysis
We used SAS 9.4 (SAS Institute Inc, Cary, North Carolina) for data analysis. Categorical variables and continuous variables were assessed by the Rao–Scott χ2 test and the Pooled t test, respectively. A two-tailed alpha of < 0.05 was required for statistical significance. The baseline characteristics of participants were summarized using descriptive statistics. Logistic regression was performed to derive the odds ratio while adjusting for different variables (age, race, hospital region, family history of cancer, immunotherapy). The modified Charlson Comorbidity Index (CCI), a validated weighted comorbidities disease severity score composed of 17 comorbid conditions, was calculated for all participants.
Results
We identified 99,543 patients with metastatic breast cancer, of which ~ 99% were females. The mean age of the study population as documented in Table 2, was 63.33 years. 67% of the population was Caucasians, 16.91% African Americans, 9.14% Hispanics, and 3.28% Asians and Pacific Islanders. Almost 38% of the study population is from the Southern region, followed by 22% from the Northeast, 21% from the Midwest, and 19% from the West.
As demonstrated in Table 2, in terms of all-cause mortality, the older population (65.02 ± 14.23 vs. 63.25 ± 14.23, p < 0.0001) and though insignificantly, males were more likely to die (5.08% vs. 4.43%, p = 0.2885). African Americans had the highest mortality (5.54%), followed by Asian and Pacific Islanders (4.80%), Others (4.65%), Hispanics (4.57%), Native Americans (4.53%), and Caucasians (4.09%) (p < 0.0001). Highest mortality was noted in the West region (5.10%), followed by South (4.56%), Midwest (4.06%), and the least in the Northeast (4.03%) (p < 0.0001).
When evaluating all-cause mortality in breast cancer cases in different regions, it is seen that a significantly younger population had a higher mortality in the South (~ 64 years) compared to other regions (p < 0.0001), as shown in Table 3. Overall, Caucasians were noted to have the most number of deaths in all the regions, followed by African Americans in Northeast, South, and Midwest, and Hispanics in West. It was also noted that African Americans had significantly higher deaths in the South (~ 30%), while Hispanics (16%), and Asians and Pacific Islanders (10.5%) had significantly higher deaths in the West compared to other regions (p < 0.0001). There was no statistical difference in Charlson Comorbidity Index in different regions. (p = 0.5377). Cases with a positive family history of any primary malignancy had more deaths in the Midwest. (~ 7%, p = 0.0005).
As seen in Table 4, the odds ratio was calculated for all-cause mortality and different variables. Being male does not decrease the odds of dying from metastatic breast cancer compared to females (0.858 (0.655–1.125)). But after adjusting for other variables, being female significantly increased the odds of dying from breast cancer. Males had higher odds of dying in the South (1.136 (1.044–1.236)) and West (1.281 (1.164–1.408)) when compared to the Northeast, while females had no significant difference in odds of dying from metastatic breast cancer in any particular region. Native American males had no significant odds of dying, while African American (1.446 (1.336–1.564)), Hispanics (1.165 (1.046–1.298)), Asian and Pacific Islander (1.191 (1.005–1.411)), and Other (1.213 (1.021–1.441)) race males had higher odds of dying from metastatic breast cancer when compared to Caucasian males. Native American and Hispanic females had significantly decreased odds of dying compared to Caucasian females. Of note, we have specified the states that belong to the different regions specified in Table 5.
Without adjusting all the other variables, African Americans (OR 1.391 (1.286–1.504)) had significantly higher odds of dying when compared to Caucasians. The odds of all-cause mortality for African Americans remain high in all the regions compared to Caucasians. Hispanics have significantly higher odds of all-cause mortality compared to Caucasians in the Southern region (OR 1.421 (1.209–1.671)), and no statistical difference in other regions. Native Americans (OR < 0.001 (< 0.001–< 0.001)) had significantly lower odds of dying in the Northeast, with no statistical difference in other regions. Asian and Pacific Islanders had no statistical difference in odds of dying from breast cancer in all the regions when compared to Caucasians. Races other than the above had significantly higher odds of dying in the Northeast (OR 1.646 (1.238–2.189)) and the West (OR 1.503 (1.059–2.133)), with lower odds in the Midwest (OR 0.302 (0.096–0.943)) and no statistical difference in odds of all-cause mortality in the South when compared to Caucasians.
Discussion
A large magnitude of studies have postulated the possible causes behind the racial disparities noted among AA’s and Caucasians. These studies have attributed the differences to the gaps in access to health care, insurance coverage [24, 25], higher prevalence of a more aggressive form of breast cancer [26, 27], as well as delayed diagnosis in AA’s [3, 24, 26, 28, 29]. AA’s have also been noted to be diagnosed with breast cancer at later stages of their disease which has been postulated as another cause for the disparities observed [30]. Some have attributed it to lack of use of adjuvant therapy [31, 32]. Social determinants in terms of marital status and stressors in life significantly contribute to this incongruity with higher mortality being seen in AA’s that are unmarried [7]. Mammography screening rates being lower in AA’s is another plausible explanation [33,34,35]. Financial hardships among certain racial groups are also an important consideration [36]. Racial disparities in molecular testing are also prevalent [37] and the gap widens between urban and rural groups [38]. Differences in mortality rates in AA’s as compared to Caucasian women have been shown to be statistically insignificant when adjusted for Medicaid enrollment and poverty [21, 39]. The presence of more comorbidities has been demonstrated to be associated with higher all-cause mortality seen in AA’s [40]. Furthermore, the minimal inclusion of certain races in clinical trials and the limited representation of these races among decision-makers and policy-makers broadens the divide that already exists and hinders the reach of new policies to the minorities that are often overlooked [11, 41]. Possible avenues of finding solutions to this problem are ensuring there is equal representation in leadership roles, in the medical community, and positions crucial to decision-making in trials.
Regional disparities in breast cancer mortality within the realm of San Francisco, Georgia, and North Carolina have previously been explored [42,43,44]. Regional and racial disparities in ten cities of the US with the highest AA population have also been documented [22]. The divide between metropolitan and rural regions has been highlighted as well. However, there has been a scarcity in studies examining the regional variation with respect to race across the entirety of the country. Several studies have attempted to identify potential reasons for the variances in the incidence of breast cancer diagnoses across various states. The rural and urban area divide has been hypothesized to be an important factor, with the highest risk being found in the urbanized areas [44].
To our knowledge, there are limited reports stratifying regional differences in breast cancer mortality in the US according to sociodemographic characteristics, such as race/ethnicity, age, and treatment/management [1]. Our study contributes to the limited literature, showing that the South of the country bore the major burden of breast cancer deaths when it came to the younger population and racial minorities; African Americans followed by Hispanics.
In our study, a large population of 99,543 individuals, mostly females and AA’s was studied. Given that AAs made up 12.4% of the US population in 2020, the 16.91% population of AAs included in the study is a good representation of the group. With the exception of the southern region, which made up 38% of the overall population, the whole population under study was distributed similarly across the various US regions.
According to our study's noteworthy findings, the populations with the higher odds of all-cause mortality were men, older people, African Americans, and the population belonging to the West. It was seen that AAs had higher odds of dying in all regions when compared to Caucasians. (OR 1.391, 1.286–1.504) This was consistent with the information seen in the American Cancer Society’s breast cancer statistics [1], the only data source that breaks down breast cancer fatalities by race and area from 2016 through 2020. According to the data, except for Washington, AAs consistently had a higher death rate than Caucasians in every state in the US. It's important to note that the data presented here was stated in terms of death rates rather than odds ratios. Another study conducted by Sighoko et al. revealed that in 10 different US cities, non-Hispanic black women had a higher rate of breast cancer mortality than non-Hispanic white women [22]. Our study showed that AA females non-significantly (1.502 (0.793–2.844) had higher odds, and Hispanics had significantly lower odds of all-cause mortality than Caucasian females [22].
It is interesting to note that regional differences in racial disparities owing to non-cancer diagnoses such as cardiovascular diseases, has also been studied and should be factored in while talking about all-cause mortality owing to metastatic breast cancer. Although the racial disparities due to cardiovascular disease are on the decline in all regions of the country [45], some studies have noted that the southern part of the country is still lagging when it comes to mortality reduction [46]. Some studies have also alluded to the rural–urban divide in cardiovascular deaths [47].
A similar study to ours was conducted over a population of 218,879 based on SEER data from 11 different regions from the years 1990 to 2001 [48]. This study had similar findings to ours, highlighting that AAs consistently had the poorest survival in most regions. They did not find significant variations in breast cancer survival among the various regions except for Detroit, which had higher mortality than other regions. They attributed it to the presence of a greater number of young black women and the region having the second highest proportion of stage 4 and unstaged cancer. Contrarily in our study, being from the South and West increased the odds of all-cause mortality compared to the Northeast. To get a better understanding of the patterns in breast cancer mortality, it was crucial to conduct a study looking at the most recent trends over the past few years.
Our study had limitations associated with administrative claims data, which contains codes produced for billing and documentation purposes. Being a retrospective study, we can only report an association between races and all-cause mortality in metastatic breast cancer patients. In-depth research is required to check whether racial differences occur in different regions. Since ICD-10-CM codes were used to identify all the diagnoses and associated comorbid conditions, the possibility of coding errors cannot be overlooked. Various codes, like Family History of Primary Malignancy, are non-specific and do not point toward breast, ovarian, or endometrial cancer specifically. We require a more thorough investigation into how factors other than race, such as SES, urbanization, use of all forms of therapy, tumor grade, and hormone receptor status, affect regional disparities, similar to the study conducted on the Californian population [44]. This would help identify possible causes behind these differences and better address confounding variables. In addition, the presence of major cancer institutes in the region and their impact on the breast cancer disparities is another factor that should be explored and weighed in future studies.
Conclusion
It is crucial to identify regional disparities with respect to race in diagnosis, treatment, and mortality before we devise a paradigm to tackle this dissonance. Encouragement of guideline-based screening for all, along with ensuring continuity of care, access to health insurance that targets regions with significant disparities in cancer and health-related outcomes, and a diversified patient representation in clinical trials, are crucial to eliminate these disparities.
References
Giaquinto AN, Sung H, Miller KD, et al. Breast cancer statistics, 2022. CA Cancer J Clin. 2022;72(6):524–41. https://doi.org/10.3322/CAAC.21754.
Siegel Mph RL, Miller KD, Sandeep N, et al. Cancer statistics, 2023. CA Cancer J Clin. 2023;73(1):17–48. https://doi.org/10.3322/CAAC.21763.
Iqbal J, Ginsburg O, Rochon PA, Sun P, Narod SA. Differences in breast cancer stage at diagnosis and cancer-specific survival by race and ethnicity in the United States. JAMA. 2015;313(2):165–73. https://doi.org/10.1001/JAMA.2014.17322.
Rauscher GH, Silva A, Pauls H, Frasor J, Bonini MG, Hoskins K. Racial disparity in survival from estrogen and progesterone receptor positive breast cancer: implications for reducing breast cancer mortality disparities. Breast Cancer Res Treat. 2017;163(2):321. https://doi.org/10.1007/S10549-017-4166-Z.
Kim G, Pastoriza JM, Qin J, et al. Racial disparity in distant recurrence-free survival in patients with localized breast cancer: a pooled analysis of National Surgical Adjuvant Breast and Bowel Project trials. Cancer. 2022;128(14):2728–35. https://doi.org/10.1002/CNCR.34241.
Chang CS, Kitamura E, Johnson J, Bollag R, Hawthorn L. Genomic analysis of racial differences in triple negative breast cancer. Genomics. 2019;111(6):1529–42. https://doi.org/10.1016/J.YGENO.2018.10.010.
Babatunde OA, Eberth JM, Felder T, et al. Social determinants of racial disparities in breast cancer mortality among black and white women. J Racial Ethn Health Disparities. 2021;8(1):147. https://doi.org/10.1007/S40615-020-00766-Y.
Yu XQ. Socioeconomic disparities in breast cancer survival: relation to stage at diagnosis, treatment and race. BMC Cancer. 2009;9(1):364. https://doi.org/10.1186/1471-2407-9-364/COMMENTS.
Coughlin SS. Social determinants of breast cancer risk, stage, and survival. Breast Cancer Res Treat. 2019;177(3):537–48. https://doi.org/10.1007/S10549-019-05340-7.
Hardy D, Du DY. Socioeconomic and racial disparities in cancer stage at diagnosis, tumor size, and clinical outcomes in a large cohort of women with breast cancer, 2007–2016. J Racial Ethn Health Disparities. 2021;8(4):990–1001. https://doi.org/10.1007/S40615-020-00855-Y.
Duma N, Aguilera JV, Paludo J, et al. Representation of minorities and women in oncology clinical trials: review of the past 14 years. J Oncol Pract. 2018;14(1):e1–10. https://doi.org/10.1200/JOP.2017.025288.
Khandwala P, Desai D, Das DG, Desai A (2020) Racial disparities in cancer clinical trials. 38(Suppl 29):97–97.https://doi.org/10.1200/JCO.2020.38.29_SUPPL.97
Aldrighetti CM, Niemierko A, van Allen E, Willers H, Kamran SC. Racial and ethnic disparities among participants in precision oncology clinical studies. JAMA Netw Open. 2021;4(11):e2133205–e2133205. https://doi.org/10.1001/JAMANETWORKOPEN.2021.33205.
Grette KV, White AL, Awad EK, et al. Not immune to inequity: minority under-representation in immunotherapy trials for breast and gynecologic cancers. Int J Gynecol Cancer. 2021. https://doi.org/10.1136/IJGC-2021-002557.
San Miguel Y, Gomez SL, Murphy JD, et al. Age-related differences in breast cancer mortality according to race/ethnicity, insurance, and socioeconomic status. BMC Cancer. 2020;20(1):1–9. https://doi.org/10.1186/S12885-020-6696-8/TABLES/3.
Lorona NC, Malone KE, Li CI. Racial/ethnic disparities in risk of breast cancer mortality by molecular subtype and stage at diagnosis. Breast Cancer Res Treat. 2021;190(3):549–58. https://doi.org/10.1007/S10549-021-06311-7.
Ooi SL, Martinez ME, Li CI. Disparities in breast cancer characteristics and outcomes by race/ethnicity. Breast Cancer Res Treat. 2011;127(3):729–38. https://doi.org/10.1007/S10549-010-1191-6.
Jatoi I, Sung H, Jemal A. The emergence of the racial disparity in U.S. breast-cancer mortality. N Engl J Med. 2022;386(25):2349–52. https://doi.org/10.1056/NEJMP2200244/SUPPL_FILE/NEJMP2200244_DISCLOSURES.PDF.
Whitman S, Orsi J, Hurlbert M. The racial disparity in breast cancer mortality in the 25 largest cities in the United States. Cancer Epidemiol. 2012;36(2):e147–51. https://doi.org/10.1016/J.CANEP.2011.10.012.
Singh GK, Jemal A. Socioeconomic and racial/ethnic disparities in cancer mortality, incidence, and survival in the United States, 1950–2014: over six decades of changing patterns and widening inequalities. J Environ Public Health. 2017. https://doi.org/10.1155/2017/2819372.
Bradley CJ, Given CW, Roberts C. Race, socioeconomic status, and breast cancer treatment and survival. J Natl Cancer Inst. 2002;94(7):490–6. https://doi.org/10.1093/JNCI/94.7.490.
Sighoko D, Hunt BR, Irizarry B, Watson K, Ansell D, Murphy AM. Disparity in breast cancer mortality by age and geography in 10 racially diverse US cities. Cancer Epidemiol. 2018;53:178–83. https://doi.org/10.1016/J.CANEP.2018.02.003.
Eley JW, Hill HA, Greenberg RS, et al. Racial differences in survival from breast cancer: results of the National Cancer Institute Black/White cancer survival study. JAMA. 1994;272(12):947–54. https://doi.org/10.1001/JAMA.1994.03520120057031.
Ko NY, Hong S, Winn RA, Calip GS. Association of insurance status and racial disparities with the detection of early-stage breast cancer. JAMA Oncol. 2020;6(3):385–92. https://doi.org/10.1001/JAMAONCOL.2019.5672.
Spada NG, Geramita EM, Zamanian M, van Londen GJ, Sun Z, Sabik LM. Changes in disparities in stage of breast cancer diagnosis in pennsylvania after the Affordable Care Act. J Womens Health. 2021;30(3):324–31. https://doi.org/10.1089/JWH.2020.8478.
Carey LA, Perou CM, Livasy CA, et al. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA. 2006;295(21):2492–502. https://doi.org/10.1001/JAMA.295.21.2492.
Shoemaker ML, White MC, Wu M, Weir HK, Romieu I. Differences in breast cancer incidence among young women aged 20–49 years by stage and tumor characteristics, age, race, and ethnicity, 2004–2013. Breast Cancer Res Treat. 2018;169(3):595–606. https://doi.org/10.1007/S10549-018-4699-9.
Chlebowski RT, Chen Z, Anderson GL, et al. Ethnicity and breast cancer: factors influencing differences in incidence and outcome. J Natl Cancer Inst. 2005;97(6):439–47. https://doi.org/10.1093/JNCI/DJI064.
Nahleh Z, Otoukesh S, Mirshahidi HR, et al. Disparities in breast cancer: a multi-institutional comparative analysis focusing on American Hispanics. Cancer Med. 2018;7(6):2710–7. https://doi.org/10.1002/CAM4.1509.
Williams F, Thompson E. Disparities in breast cancer stage at diagnosis: importance of race, poverty, and age. J Health Dispar Res Pract. 2017;10(3):34.
Bickell NA, Wang JJ, Oluwole S, et al. Missed opportunities: racial disparities in adjuvant breast cancer treatment. J Clin Oncol. 2006;24(9):1357–62. https://doi.org/10.1200/JCO.2005.04.5799.
Reeder-Hayes KE, Meyer AM, Dusetzina S, Liu H, Wheeler SB. Racial disparities in initiation of adjuvant endocrine therapy of early breast cancer. Breast Cancer Res Treat. 2014;145(3):743–51. https://doi.org/10.1007/S10549-014-2957-Z.
McCarthy EP, Burns RB, Coughlin SS, et al. Mammography use helps to explain differences in breast cancer stage at diagnosis between older black and white women. Ann Intern Med. 1998;128(9):729–36. https://doi.org/10.7326/0003-4819-128-9-199805010-00005.
Teysir J, Gegechkori N, Wisnivesky JP, Lin JJ. Racial disparities in surveillance mammography among older breast cancer survivors. Breast Cancer Res Treat. 2019;176(2):461–7. https://doi.org/10.1007/S10549-019-05250-8.
Miller BC, Bowers JM, Payne JB, Moyer A. Barriers to mammography screening among racial and ethnic minority women. Soc Sci Med. 2019;239:112494. https://doi.org/10.1016/J.SOCSCIMED.2019.112494.
Gerend MA, Pai M. Social determinants of Black-White disparities in breast cancer mortality: a review. Cancer Epidemiol Biomark Prev. 2008;17(11):2913–23. https://doi.org/10.1158/1055-9965.EPI-07-0633.
Choi JJ, Fikre T, Fischman A, Buck AK, Ko NY. The role of race and insurance status in access to genetic counseling and testing among high-risk breast cancer patients. Oncologist. 2022;27(10):832. https://doi.org/10.1093/ONCOLO/OYAC132.
Zahnd WE, Ranganathan R, Adams SA, Babatunde OA. Sociodemographic disparities in molecular testing for breast cancer. Cancer Causes Control. 2022;33(6):843–59. https://doi.org/10.1007/S10552-022-01575-W.
DeGuzman PB, Cohn WF, Camacho F, Edwards BL, Sturz VN, Schroen AT. Impact of urban neighborhood disadvantage on late stage breast cancer diagnosis in Virginia. J Urban Health. 2017;94(2):199. https://doi.org/10.1007/S11524-017-0142-5.
Tammemagi CM, Nerenz D, Neslund-Dudas C, Feldkamp C, Nathanson D. Comorbidity and survival disparities among Black and White patients with breast cancer. JAMA. 2005;294(14):1765–72. https://doi.org/10.1001/JAMA.294.14.1765.
Murthy VH, Krumholz HM, Gross CP. Participation in cancer clinical trials: race-, sex-, and age-based disparities. JAMA. 2004;291(22):2720–6. https://doi.org/10.1001/JAMA.291.22.2720.
Moubadder L, Collin LJ, Nash R, et al. Drivers of racial, regional, and socioeconomic disparities in late-stage breast cancer mortality. Cancer. 2022;128(18):3370–82. https://doi.org/10.1002/CNCR.34391.
Weber JJ, Kachare SD, Vohra NA, Fitzgerald TF, Wong JH. Regional disparities in breast cancer outcomes and the process of care. AM Surg. 2014;80(7):669–74. https://doi.org/10.1177/000313481408000721.
Parise CA, Caggiano V. Regional variation in disparities in breast cancer specific mortality due to race/ethnicity, socioeconomic status, and urbanization. J Racial Ethn Health Disparities. 2017;4(4):706–17. https://doi.org/10.1007/S40615-016-0274-4.
Kyalwazi AN, Loccoh EC, Brewer LC, et al. Disparities in cardiovascular mortality between Black and White adults in the United States, 1999 to 2019. Circulation. 2022;146(3):211–28. https://doi.org/10.1161/CIRCULATIONAHA.122.060199.
Singh GK, Azuine RE, Siahpush M, Williams SD. Widening geographical disparities in cardiovascular disease mortality in the United States, 1969–2011. Int J MCH AIDS. 2015;3(2):134. https://doi.org/10.21106/ijma.46.
Cross SH, Mehra MR, Bhatt DL, et al. Rural-urban differences in cardiovascular mortality in the US, 1999–2017. JAMA. 2020;323(18):1852–4. https://doi.org/10.1001/JAMA.2020.2047.
Grann V, Troxel AB, Zojwalla N, Hershman D, Glied SA, Jacobson JS. Regional and racial disparities in breast cancer-specific mortality. Soc Sci Med. 2006;62(2):337–47. https://doi.org/10.1016/J.SOCSCIMED.2005.06.038.
Acknowledgements
This data has previously been presented as an Abstract at ASCO Annual Meeting 2022.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by DD. The first draft of the manuscript was written by AK and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Informed consent
Informed consent was not obtained as the data were obtained from NIS and was anonymized.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Khosla, A., Desai, D., Singhal, S. et al. Racial and regional disparities in deaths in breast cancer. Med Oncol 40, 210 (2023). https://doi.org/10.1007/s12032-023-02083-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-023-02083-w